Unit 7 Properties Of Bulk Matter Chapter 8 Change Of State Of Matter Short Answer Type Questions
Short Answer Questions on Change of State of Matter
Question 1. Tea gets cooled when sugar is added to it. Why?
Answer:
Tea gets cooled when sugar is added to it.
When sugar is added to tea, it melts. For this purpose, the required latent heat is collected by sugar from tea, thereby cooling the tea.
Question 2. The water equivalent of a calorimeter is 10 g and it contains 50 g of water at 15 °C. Some amount of ice, initially at -10°C is dropped in it and half of the ice melts till equilibrium is reached. What was the initial amount of ice that was dropped (when specific heat of ice = 0.5 cal · g-1 · °C-1, specific heat of water = 1.0 cal · g-1 · °C-1 and latent heat of melting of ice = 80 cal · g-1)?
- 10g
- 18 g
- 20 g
- 30 g
Answer:
Given
The water equivalent of a calorimeter is 10 g and it contains 50 g of water at 15 °C. Some amount of ice, initially at -10°C is dropped in it and half of the ice melts till equilibrium is reached.
Let, ice of m g is dropped in the calorimeter.
m x 0.5 x 10 + m/2 x 80 =60x 1 x 15
or, m = 20 g
The option 3 is correct.

Question 3. Steam at 100°C is passed into 20 g of water at 10°C. When water acquires a temperature of 80 °C, the mass of water present will be [Take specific heat of water = 1 cal · g-1 · °C-1 and latent heat of steam = 540 cal · g-1]
- 24 g
- 31.5 g
- 42.5 g
- 22.5 g
Answer:
Given
Steam at 100°C is passed into 20 g of water at 10°C.
Let m g of steam is passed into 20 g of water at 10° C.
Total heat released by the steam
= m x 540 + m( 100-80) =560/72 cal
Total heat absorbed by the water = 20(80-10) = 1400 cal
∴ 560m = 1400 or, m = 2.5 g
Now, mass of water present = 20 + 2.5 = 22.5 g.
The option 4 is correct.
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
Question 4. A piece of ice falls from a height h so that it melts completely, Only one-quarter of the heat produced is absorbed by the ice and all energy of ice gets converted into heat during its fall. The value of h is [latent heat of ice is 3.4 x 15 J/kg and g = 10N/kg]
- 544 km
- 136 km
- 68 km
- 34 km
Answer:
Given
A piece of ice falls from a height h so that it melts completely, Only one-quarter of the heat produced is absorbed by the ice and all energy of ice gets converted into heat during its fall.
Heat produced = mgh [ m = mass of ice]
According to the question,
1/4 (mgh) = mL (L = latent heat of fusion of ice]
or, \(h=\frac{4 L}{g}=\frac{4 \times 3.4 \times 10^5}{10}=13.6 \times 10^4 \mathrm{~m}=136 \mathrm{~km}\)
The option 2 is correct.
Latent Heat: Short Answer Questions
Question 5. A copper block of mass 2.5 kg is heated in a furnace to a temperature of 500°C and then placed on a large ice block. What is the maximum amount of ice that can melt? (specific heat of copper = 0.39 J · g-1 · K-1; heat of fusion of water = 335 J · g-1)
Answer:
Given
A copper block of mass 2.5 kg is heated in a furnace to a temperature of 500°C and then placed on a large ice block.
The ice block is at 0°C.
So the heat released by the copper block = 2500 x 0.39 x (500 – 0) = 25 x 39 x 500 J
The maximum amount m of ice would melt if the entire amount of heat released by the copper block is used up as latent heat of fusion of ice.
∴ m = \(\frac{25 \times 39 \times 500}{335}\) = 1455 g =1.455 kg
Unit 7 Properties Of Bulk Matter Chapter 8 Change Of State Of Matter Integer Type Question And Answers
In this type, the answer to each of the questions is a single-digit integer ranging from 0 to 9.
Practice Short Answer Questions on States of Matter
Question 1. A piece of ice (heat capacity =2100 J • kg-1 • °C-1 and latent heat = 3.36 x 105 J • kg-1) of mass m g is at -5°C at atmospheric pressure. It is given 420 J of heat so that the ice starts melting. Finally, when the ice-water mixture is in equilibrium, it is found that 1 g of ice has melted. Assuming there is no other heat exchange in the process, what will be the value of m?
Answer: 8
Question 2. 2 kg of ice at -20 °C is mixed with 5 kg of water 20 °C in an insulating vessel having a negligible heat capacity. Calculate the final mass (in kg) of water remaining in the container. It is given that the specific heat of water and ice are 1 kcal · kg-1 · °C-1 and 0.5 kcal · kg-1 · °C-1 respectively while the latent heat of fusion of ice is 80 kcal · kg-1.
Answer: 6
Question 3. In an industrial process, 10 kg of water per hour is to be heated from 20°C to 80°C. To do this steam at 150°C is passed from a boiler into a copper coil immersed in water. The steam condenses in the coil and is returned to the boiler as water at 90°C. How many kg of steam is required per hour? (specific heat of steam = 1 cal · g-1 · °C-1, steam = 540 cal · g-1)
Answer: 1
Question 4. A bullet of mass 10 x 10-3 kg moving with a speed of 20 m • s-1 hits an ice block (0°C) of 990 g kept at rest on a frictionless floor and gets embedded in it If ice takes 50% of KE lost by the system, the amount of ice melted (in grams) approximately is n x 10-3. Find the value of n.(J = 4.2 J • cal-1, latent heat of ice = 80 cal • g-1)
Answer: 3
Unit 7 Properties Of Bulk Matter Chapter 8 Change Of State Of Matter Comprehension Type Question And Answers
Read the following passages carefully and answer the questions at the end of them.
Question 1. An immersion heater, in an insulated vessel of negligible heat capacity brings 10 g of water to the boiling point from 16°C in 7 min. The water is replaced by 200 g of alcohol, which is heated from 16°C to the boiling point of 78 °C in 6 min 12 s, 30 g are vaporised in 5 min 6 s.
1. Power of heater is nearly
- 8.4 x 103 J · s-1
- 84 W
- 8.4 x 103 cal · s-1
- 20 W
Answer: 2. 84 W
2. The specific heat of alcohol is
- 0.6 J · kg-1 ·°C-1
- 0.6 cal · g-1 · °C-1
- 0.6 cal · kg-1· °C-1
- 0.6 J · °C-1
Answer: 2. 0.6 cal · g-1 · °C-1
3. The latent heat of vaporization of alcohol is
- 854 J · kg-1
- 854 x 103 J · kg-1
- 204 cal · g-1
- 204 cal · kg-1
Answer: 2. 854 x 103 J · kg-1
Real-Life Applications of Phase Changes: Short Answers
Question 2. In a physics practical examination, a student puts heat into a 500 g sample (solid) at the rate of 10 kJ · min-1, while recording its temperature as a function of time. He collects the data and plots the graph depicted. Assume no heat is lost.
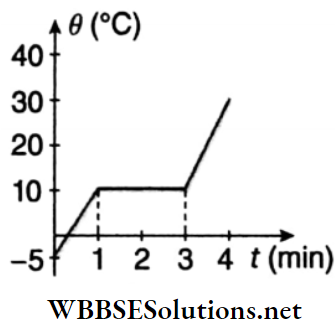
1. What is the latent heat of fusion?
- 35 kJ · kg-1
- 70 kJ · kg-1
- 25 kJ · kg-1
- 30 kJ · kg-1
Answer: 4. 30 kJ · kg-1
2. What is the specific heat of the substance in liquid phase?
- 1 kJ · kg-1 · K-1
- 1.5 kJ · kg-1 · K-1
- 6.67 kJ · kg-1 · K-1
- None of these
Answer: 1. 1 kJ · kg-1 · K-1
3. What is the specific heat of the substance in solid phase?
- \(\frac{4}{3} \mathrm{~kJ} \cdot \mathrm{kg}^{-1} \cdot \mathrm{K}^{-1}\)
- \(\frac{3}{4} \mathrm{~kJ} \cdot \mathrm{kg}^{-1} \cdot \mathrm{K}^{-1}\)
- \(\frac{40}{3} \mathrm{~kJ} \cdot \mathrm{kg}^{-1} \cdot \mathrm{K}^{-1}\)
- \(1 \mathrm{~kJ} \cdot \mathrm{kg}^{-1} \cdot \mathrm{K}^{-1}\)
Answer: 1. \(\frac{4}{3} \mathrm{~kJ} \cdot \mathrm{kg}^{-1} \cdot \mathrm{K}^{-1}\)