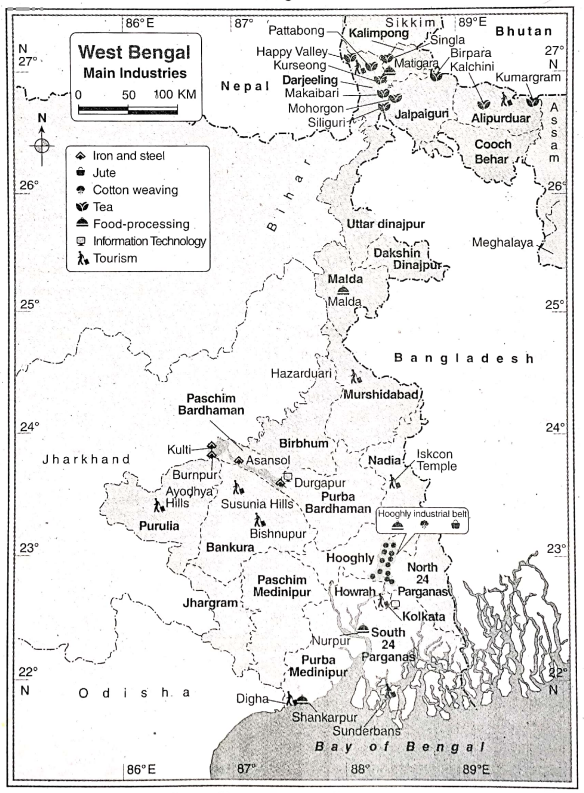
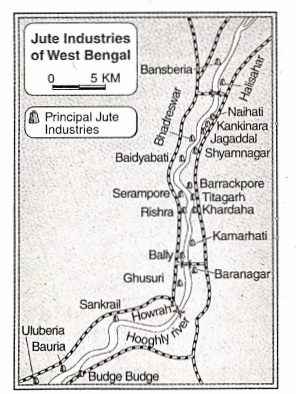
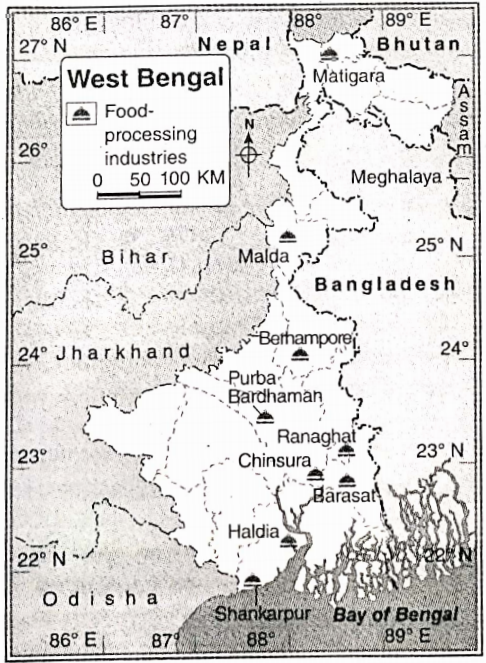
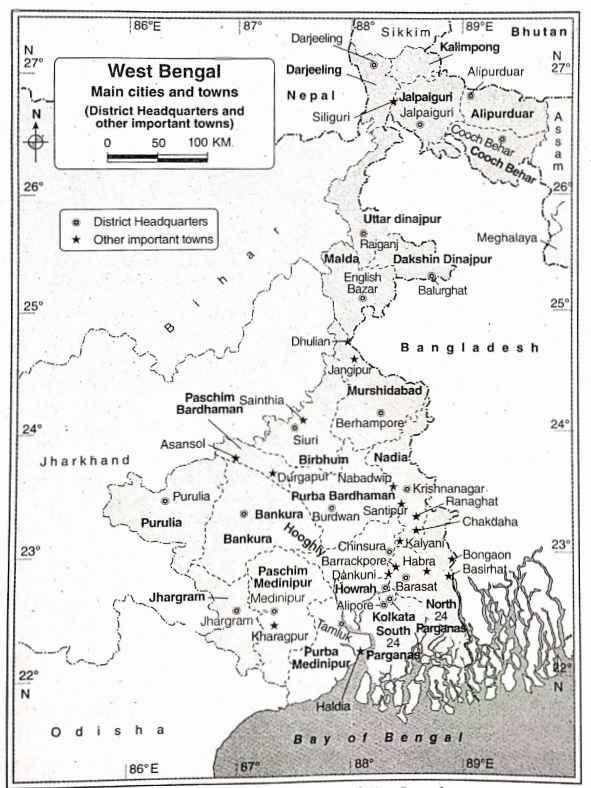
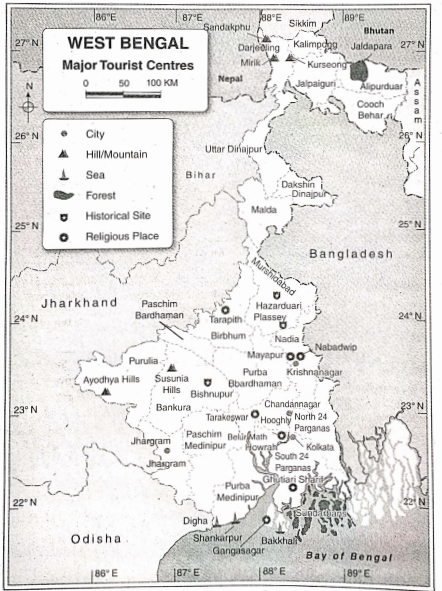
Some Basic Concepts Of Chemistry Introduction
Chemistry is an important branch of science. It deals with the source, composition, structure, and properties of matter with special reference to the physical and chemical changes that matter undergoes under different conditions.
With time, the greatest discoveries of chemistry have made human life more comfortable and have facilitated its advancement as well In the last few decades, a tremendous change in the field of chemistry has been observed.
It has become significantly vast and complex. For the convenience of research and a better understanding of the subject, it has been divided into several branches

Laws Of Chemical Combination, Atomic And Molecular Theory, Equivalent Weight
Importance And Scope Of Chemistry
Chemistry plays amajorrolein science and is often intertwined with different branches of science such as physics, biology, geology, etc. It has made many contributions to human civilization.
Principles of chemistry are found to be very useful in diverse areas such as weather patterns, biochemical processes, functioning of brains, operations of computers, etc.
Chemistry helps to fulfill human needs for food, health care products, and other materials required for improving the quality of life.
Read and Learn More WBCHSE Class 11 Chemistry
Some of the major contributions of chemistry are given below:
- Chemistry In Agriculture And Preservation Of Food
- Several chemical fertilizers like urea, ammonium sulfate, calcium nitrate, superphosphate of lime, etc. are used for better production of crops.
- By hydrogenation of edible oil, artificial fats (such as vanaspati) are prepared.
- It helps to protect crops from the harmful effects of insects and bacteria by the use of effective insecticides (such as gammexane, aldrin, parathion, etc.), herbicides, and fungicides.
- The use of preservatives {Example sodium benzoate, salicylic acid, potassium nitrate, sodium chloride, etc.) has helped to preserve food materials. like jam, jelly, butter, squashes, fish, offer, etc., for longer periods of time.
- Different chemical methods are available to indicate the presence of adulterants in order to ensure the supply of pure foodstuff.
Chemistry In Health Care And Sanitation
- Analgesics (For example aspirin, analgin, etc.) are used to give relief from different types of pain.
- Antipyretics (For example paracetamol, ibuprofen, etc.) are used to bring down body temperature during fever.
- Antiseptics such as Dettol, savlon, tincture of iodine, etc. are used to stop infection of wounds.
- Antibiotics (For example penicillin, cephalosporin, tetracycline, streptomycin, chloramphenicol) are used to curb infection and cure diseases like pneumonia, bronchitis, typhoid, tuberculosis, etc.
- Tranquilizers such as barbituric acid, veronal, valium, reserpine, etc. are prescribed to patients suffering from mental diseases to reduce their tension or anxiety.
- Anesthetics like chloroform, cocaine, novocaine, etc. are applied to patients to make surgical operations painless.
- Today dysentery and pneumonia have become curable by the use of penicillin and sulpha drugs.
- The widely used drug quinine has now been replaced by some more effective antimalarials like chloroquine, primaquine, etc.
- As a preventive measure against various types of diseases, vaccines are found to be used widely Example tetanus toxoid (for tetanus), TABC (for typhoid, paratyphoid A, B, and cholera), oral polio (for polio), etc.
- Nowadays life-saving drugs such as taxol and cisplatin are used in cancer therapy; Azidothymidine (AZT) is used for AIDS victims.
- Synthetic vitamins and tonics have significant contributions towards the better health of human beings.
- Bleaching powder, potassium permanganate, ozone gas, low concentration of chlorine, etc. are used for sterilization of water to make it suitable for drinking.
- Disinfectants like phenol and cresols are used to kill the micro-organisms present in drains, toilets, floors, etc.
Chemistry In Comforts, Pleasures And Luxuries
- The contribution of chemistry towards the betterment of human society is widely acknowledged by all and it has a profound influence on our daily life.
- Synthetic fibers such as terylene, nylon, rayon, dacron, orlon, etc., are used to prepare clothes that are more comfortable, durable, attractive, and easy to wash.
- Polythene is used for making toys, bottles, tubes, pipes, kitchen and domestic appliances, sheets for packing materials, and coated wires and cables.
- PVC is used for making rain-coats, hose pipes, conveyor belts, radio and TV components, insulating material for wires, cables, and other electrical goods, gramophone records, safety helmets, refrigerator components, bi-cycle, and motor-cycle mudguards, etc.
- Phenol-formaldehyde resin and bakelite are used for making combs, fountain pen barrels, electrical goods (switches and plugs), heater handles, telephone parts, cabinets for radio and television, etc. Films used in cameras are made of celluloid coated with suitable chemicals.
- Cosmetics such as cream, lipstick, sunscreen lotion, face powder, talcum powder, perfume, toothpaste, nail polish, shampoo, hair dye, etc., are all chemical substances.
- Soaps and detergents used for cleaning clothes are chemical substances. Bio-degradable detergents are now in use to avoid environmental pollution.
- Paint, varnishes, and lacquer are applied on walls, wooden furniture, and metallic articles to make them more attractive, durable, and resistant to corrosion.
- Articles made of iron are electroplated by nickel, chromium, silver, gold, etc., so as to prevent them from rusting and to make them more attractive and durable.
- Ammonia, liquid sulfur dioxide CFC, etc. are used as refrigerants in refrigerators and air-conditioners.
- Cement, steel, iron, etc., are widely used m the construction of multi-storeyed buildings, dams, and bridges.
- LPG and natural gas having high calorific value are used as smokeless fuels for cooking. Compressed natural gas (CNG) is now used as a fuel in public vehicles in metropolitan cities.
Chemistry In Industry
- Chemistry plays an important role in the development and growth of a number of industries. Some important examples of manufacturing processes are
- Extraction of metals such as iron, aluminum, zinc, copper silver, gold, etc.
- Refining of petroleum to produce petroleum ether, gasoline (petrol), kerosene, diesel, paraffin oil, lubricating oil, solvent naphtha, liquid paraffin, petroleum jelly, paraffin wax, etc.
- Plastics such as polyethylene, PVC, bakelite, polyurethanes, Teflon, etc.
- Synthetic fibers such as nylon, terylene, rayon, etc.
- Paints, varnishes, lacquer, and synthetic dyes.
- Cement, glass, and ceramic materials.
The dark side of chemistry: Chemistry plays a pivotal role in our daily lives and luxuries. However improper use of chemistry has a negative impact on human society. In modern times, atomic energy is mainly used in chemical warfare.
Different chemical weapons and explosives like RDX are used for terrorist activities. Drugs like cocaine, LSD, and heroin have adverse effects on the youths.
Nature Of Matter
Anything which has mass occupies some space and can be felt by one or more of our senses is called matter.
Everything around us such as aspen, pencil, wood, water, milk, air, etc., and all living beings are composed of matter.
Classification of matter: Matter can be classified in two different ways—Physical classification and Chemical classification.
Physical classification of Matter: At ordinary temperature and pressure, matter can exist in three physical states viz., solid, liquid, and gas. The constituent particles of matter in the three states can be represented as shown in.
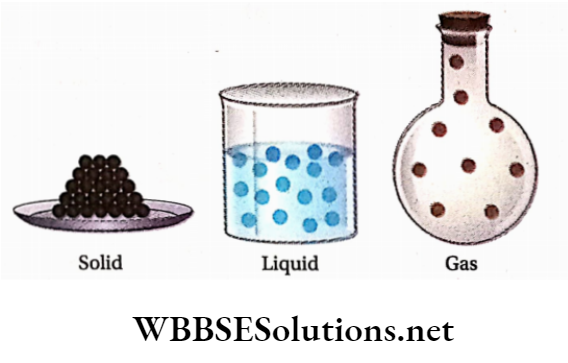
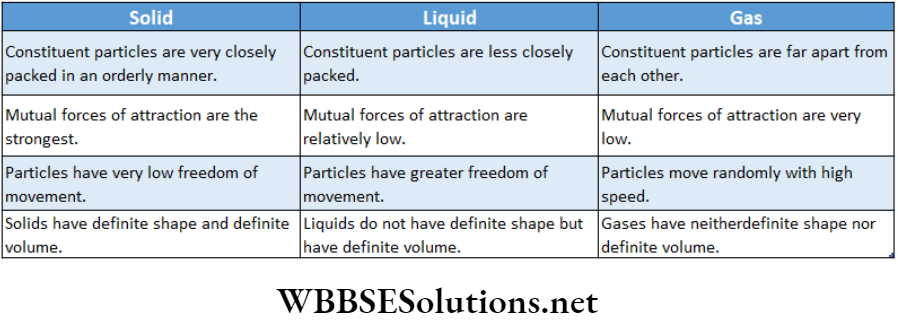
The essential points of differences between the three states of matter are given in the following table:
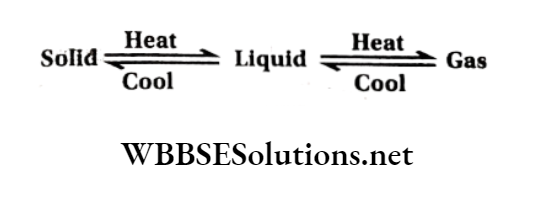
A given substance can be made to exist in the solid, liquid, or gaseous state by changing the conditions of temperature and pressure.
Chemical classification of Matter : On the basis of chemical composition, matter can be classified into two major categories such as
- Mixtures and
- Pure substances.
These can be further subdivided as follows:

A mixture is made up of two or more substances (present in any ratio) which are called its components. For example, a sugar solution consists of two components i.e., sugar and water.
In a homogeneous mixture, the components completely mix with each other and its composition remains uniform throughout.
The components of such a mixture cannot be seen even under a microscope. Some examples are air, glucose solution, seawater, petrol, etc.
In contrast to this, in heterogeneous mixtures, the composition is not uniform throughout, and sometimes the different components can be seen even by the naked eye.
For example, the mixtures of sugar and salt, sand, and iron filings are heterogeneous mixtures. The components of such mixtures can be separated by using physical methods such as filtration, crystallization, distillation, chromatography, etc.
Pure substances have characteristics different from that of the mixtures. They have fixed composition throughout the entire mass. Some examples are iron, copper, silver, gold, water, sucrose, etc.
Sucrose contains carbon, hydrogen, and oxygen in a fixed ratio and hence it has a fixed composition.
The constituents of pure substances cannot be separated by simple physical methods. Pure substances are further classified into elements and compounds.

Element: An element consists of only one type of particle. The constituent particles may be atoms or molecules.
Oxygen, nitrogen, sodium, copper, silver, etc., are some examples of elements. They all contain atoms of one type.
The smallest particles (having independent existence) present in metallic elements such as sodium, potassium, etc. are called atoms.
On the other hand, the smallest possible particles (having independent existence) of some other elements (such as oxygen, nitrogen, phosphorus, etc.) are called molecules, which consist of two or more atoms.
Thus, two atoms of oxygen and four atoms of phosphorus combine separately to form molecules of oxygen and phosphorus respectively.
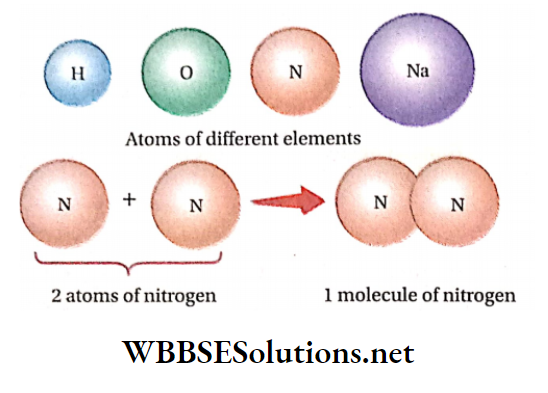
Two or more atoms of different elements combine together to form the molecule of a compound.
Examples of some compounds are water, carbon dioxide, ammonia, etc. The molecules of carbon dioxide and water are depicted in.
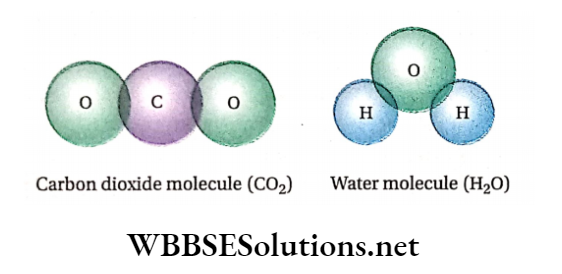
A carbon dioxide molecule consists of one carbon atom and two oxygen atoms. Similarly, a molecule of water is composed of two hydrogen atoms and one oxygen atom.
It is thus seen that the atoms of different elements are present in a compound in a fixed ratio and this ratio is the characteristic of a particular compound.
It is needless to mention that the properties of a compound are completely different from those of the constituent elements.
For example, hydrogen and oxygen are gaseous substances while the compound (water) formed by their combination is a liquid at ordinary temperature.
The constituents of a compound cannot be separated by physical methods. They can, however, be separated by chemical methods.
Physical Quantities
Characteristics of matter that can be examined as a measurable quantity are called physical quantities. Example length, mass, time, temperature, area, volume, velocity, acceleration, force, etc.
Units For Measurement Of Physical Quantities
A unit is defined as the standard of reference chosen for the measurement of any physical quantity.
Example: Suppose the length of a bench is 2 meters. Here length is the physical quantity andmetre is the unit length.
The numerical magnitude ‘2’ implies that the length of the bench is two times that of the value of 1 meter (which is the standard of reference chosen for the measurement of length).
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
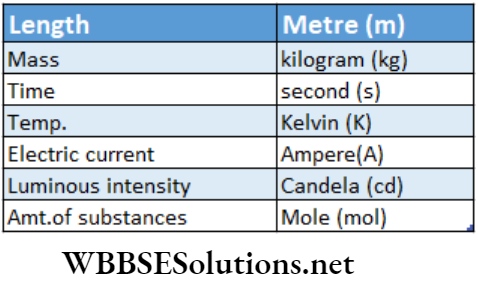
Fundamental Units Used In Different Systems
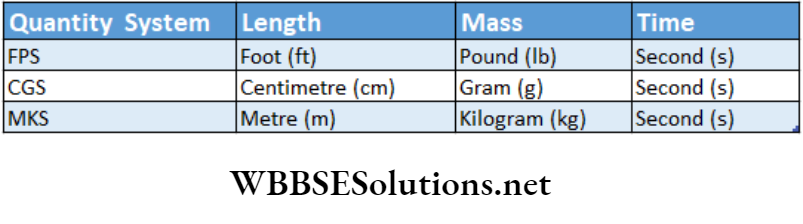
Basic physical quantities and their units in the SI system
Some Common Derived Units In CGS And SL System
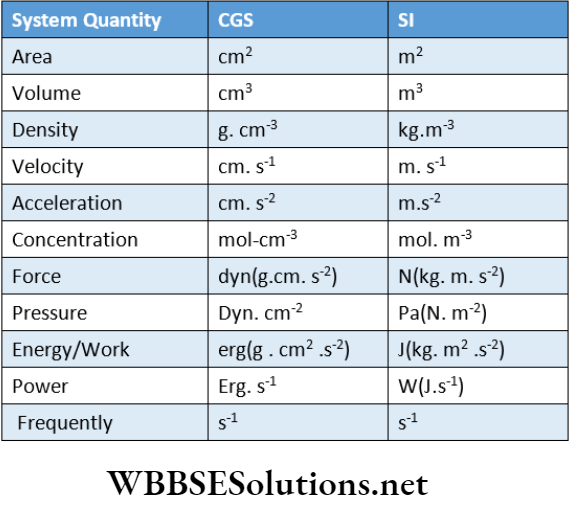
- The volume of liquids is commonly measured in a liter (L) but this is not a SI unit. 1 L = 1000 mL = 1000 cm3 = 1 dm3
- Wavelength is expressed in angstrom (A). 1A = 10-10 m
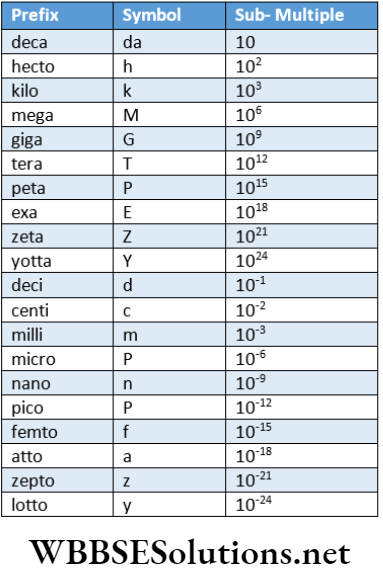
Some Commonly Used Prefixes In CGS And SI Systems
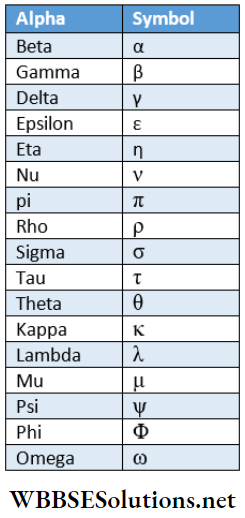
Frequently Used Greek Letters
Important points regarding the use of SI units:
- No dot (•) can be used in between the letters or at the end of the letters used for abbreviations of basic units. Thus, the symbol for centimeter is cm (it is neither c.m. nor cm.)
- Abbreviations of units do not have a plural ending. Thus, it is incorrect to write 5 ems or 12 gms. These should be 5 cm and 12 gm respectively.
- The abbreviations of units named after scientists start with capital letters and not with small letters. Some examples are Newton (N), Joule (J), Pascal (Pa), Ampere (A), etc. If the names are used in full instead of abbreviations then these start with small letters Example Newton, ampere, Pascal, etc.
- Abbreviations of other units such as meter (m), kilogram (kg), second (s), etc., start with small letters.
- The temperature in the kelvin scale should not be roprosonled with a degree (°). So, It Is proper to say 290K but not 2H- K.
- The derived units such as square meter and cubic centimeter are denoted ns m2 (but not sqm) and cm9 (but not cc) respectively.
- To indicate divisions, it is better to use inverse sign. However, the ‘/’ symbol can be used blit once only. One example is Kg-1.K-1 but not J/(Kg . K) or, J/Kg/K.
Conversion of physical quantities in different units involves the following steps:
1. Firstly, we have to determine a unit conversion factor, then
2. The given magnitude of the physical quantity in question, is multiplied by a suitable unit conversion factor such that all units are canceled out leaving behind only the required units.
This is illustrated by the following examples:
To express the length of a wooden pencil (say, 4 inches long) in cm:
We know, 1 inch = 2.54 cm
∴ \(\frac{1 \mathrm{inch}}{2.54 \mathrm{~cm}}=1=\frac{2.54 \mathrm{~cm}}{1 \mathrm{inch}}\)
Here, both the ratios \(\frac{1 \mathrm{inch}}{2.54 \mathrm{~cm}} \text { and } \frac{2.54 \mathrm{~cm}}{1 \text { inch }}\) and are equal to
‘1’ because the lengths 1 inch and 2.54 cm are exactly equal to each other. Either of these ratios is called unit conversion factor or simply unit factor.
The magnitude of any quantity will remain unchanged when it is multiplied by a suitable unit conversion factor.
Based on these rules, the length of the given wooden pencil can be expressed in cm as follows:
4 inch = 4 inch x 1 (unit factor)
⇒ \(=4 \mathrm{inch} \times \frac{2.54 \mathrm{~cm}}{\text { linch }} 4 \times 2.54 \mathrm{~cm}=10.16 \mathrm{~cm}\)
Here, the quantity inch’ is multiplied by a particular unit conversion factor so that the unit ‘inch’ gets canceled out.
To express the length of an iron rod (say, 30.48 cm long)in inches:
Here, 30.48 cm = 30.48 cm x 1 (unit conversion factor)
⇒ \(=30.48 \mathrm{~cm} \times \frac{1 \mathrm{inch}}{2.54 \mathrm{~cm}}=\frac{30.48}{2.54} \mathrm{inch}=12 \mathrm{inch}\)
In this case, the given length is multiplied by the particular unit conversion factor so that the unit ‘cm’ is canceled out from the numerator and the denominator.
To express a given volume (say, 51) of water in m³
Wo know, 1 L = 1000 cm³
Again, 1m = 100 cm \(\frac{1 \mathrm{~m}}{100 \mathrm{~cm}}=1=\frac{100 \mathrm{~cm}}{1 \mathrm{~m}}\)
⇒ \(\text { So, }\left(\frac{1 \mathrm{~m}}{100 \mathrm{~cm}}\right)^3 \approx 1^3=\left(\frac{100 \mathrm{~cm}}{1 \mathrm{~m}}\right)^3\)
⇒ \(\text { So, }\left(\frac{1 \mathrm{~m}}{100 \mathrm{~cm}}\right)^3=1^3=\left(\frac{100 \mathrm{~cm}}{1 \mathrm{~m}}\right)^3\)
⇒ \(\text { or, } \frac{1 \mathrm{~m}^3}{10^6 \mathrm{~cm}^3}=1=\frac{10^6 \mathrm{~cm}^3}{1 \mathrm{~m}^3}\)
Now, 5L =(5×1000)(5×1000)cm³=5000 cm³
=5000 cm³x1(unit factor)
⇒ \(=5000 \mathrm{~cm}^3 \times \frac{1 \mathrm{~m}^3}{10^6 \mathrm{~cm}^3}\)
⇒ \(=\frac{5000}{10^6} \mathrm{~m}^3=5 \times 10^{-3} \mathrm{~m}^3\)
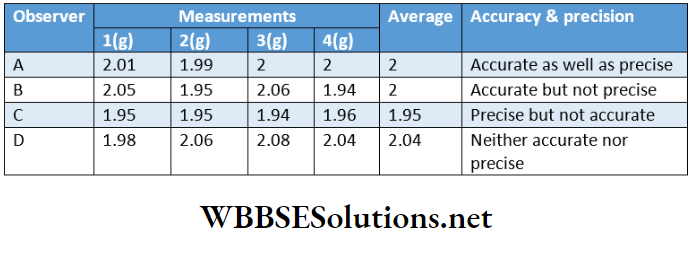
Accuracy And Precision
The accuracy of a measurement is the agreement of the measured value to the true value. As the difference between the measured value and the true value decreases, the accuracy of the measurement increases.
The degree of accuracy of any measurement depends upon
- The accuracy of the measuring device used and
- The skill of the operator. The difference between the measured value and the true value is called the absolute error.
Precision refers to the closeness of the results of various measurements for the same quantity.
Good precision does not necessarily mean good accuracy because various measurements may involve the same mistake repeatedly. This can be understood from the given illustration.
Let, the true value for a measurement be 2.00 g. Four different cases may arise when the actual measurements are carried out by different observers A, B, C, and D.
Significant Figures Definition
The total number of digits present in a number (starting from the first non-zero digit) including the last digit whose value is uncertain is called the number of significant figures.
Explanation: Suppose a student is asked to measure the length of a pencil with the aid of a meter scale (in which the closest distance between two successive marks is 0.1 cm). The student reports his experimental result as 15.4 cm.
Here the last digit (i.e., 4) of the reported result is not absolutely correct because there are two possibilities:
- The length of the pencil may be greater than 15.3 cm but slightly smaller than 15.4 cm or
- Its length may be much smaller than 15.5 cm but slightly greater than 15.4 cm.

From the above discussion, it can be stated that in the above-reported value (i.e., 15.4cm) there are three significant figures [the first two digits (1 and 5) are certain and the last digit (4) is uncertain].
Example: Suppose the mass of an object measured by an analytical balance is reported to be 12.4567 g. If the accuracy of the balance is 0.0001 g, the actual mass of the object will be (12.4567 ± 0.0001 )g i.e., the value lies between 12.4566g and 12.4568g.
Thus in the reported mass, the first five digits (1,2,4,5 and 6) are certain while the last digit (7) is uncertain. This means that there are significant figures in the reported mass.
Determination of the number of significant figures: The following rules are applied in determining the number of significant figures in a measured quantity.
All non-zero digits are significant.
Examples:
- There are two significant figures in the number 57.
- In 64.5 cm, there are three significant figures.
- In 0.4361g, there are four significant figures.
- 2. Zeros between two non-zero digits are significant.
Examples:
- There are four significant figures in the number 8005.
- 12.032 g has five significant figures.
- Zeros to the left of the first non-zero digit are not considered to be significant.
Examples:
- 0.53 mL has two significant figures (5 and 3).
- 0.0724 kg has three significant figures (7, 2 and 4).
- 0.009035 has four significant figures (9, 0, 3 and 5).
- If a number ends with one or more zeros and these zeros are to the right of the decimal point then these zeros become significant.
Examples:
- 4.0 has two significant (4 and 0).
- 2.500 has four significant (2, 5, 0, and 0).
- 0.040g has two significant (4 and 0).
- 0.4000 km has four significant (4, 0, 0 and 0).
5. If a number ends with one or more zeros but these zeros are not to the right of a decimal point, then these zeros may or may not be significant.
Examples: 10700 g may have three, four, or five significant figures. The ambiguity can be removed by expressing the value in an exponential form of the type N x 10n, where n = an integer and N = a number with a single non-zero digit to the left of the decimal point.
Now, the number, 10700 can be expressed (in scientific notations) in three different exponential forms, thereby indicating the presence of three, four, or five significant figures in the number.
10700 = 1.07 X 104 (Three significant)
= 1.070 X 104 (Four significant)
= 1.0700 X 104 (Five significant)
In these exponential terms, the significant figures of only the first factor {i.e., 1.07 1.070, or 1.0700) are to be counted (remembering that all zeros to the right of a decimal point are significant).
There are three significant in each of the numbers,1.54 x 10-2 and 1.54 x10-6 Similarly, there are four significant in Avogadro’s number 6.022 x 1023.
If a whole number ends with one or more zeros then these zeros are not considered while counting the number of significant. Thus there are only three significant 43700.
If however, the said number expresses the result of any experimental measurement, then such zeros are taken into consideration while counting the number of significant. Thus, if the measured distance between two places is 3200m (taking lm as the least measurable distance) then the number of significant figures in the measured distance is four.
Exactintegralnumbers such as the number of pencils in a dozen the number of grams in a kilogram or the number of centimeters in a meter do not have any uncertainty associated with them and hence these numbers have an infinite number of significant figures.
Examples:
- A number of pencils in a dozen = 12.0000. has an infinite number of significance.
- The number of grams in a kilogram = 1000.0000. number of significant.
Rules For Determination Of The Number Of Significant Figures In Final Results Involving Calculations
The observed results of various measurements may have different precisions. Thus, the results obtained at various stages of the calculation are to be rounded off because the final result cannot be more precise than that of the least precise measurement.
Rounding off: The following rules are employed foregrounding offa numbers to the desired number of significant.
1. If the digit, next to the last digit to be retained, is less than 5, the last digit to be retained is left unchanged and all other digits on its right are discarded.
Example: Suppose the result of a measurement is 2.73484. This can be rounded off to give—(a) 2.7348 (for reporting the result upto four decimal places) or, (b) 2.73 (for reporting the result upto two decimal places).
If the digit, next to the last digit to be retained, is greater than 5, the last digit to be retained is increased by 1 and all other digits on its right are discarded.
Example: Suppose the result of a measurement is 2.73687. This can be rounded off to give
2.7369 (for reporting the result upto four decimal places) or, (b) 2.74 (for reporting the result upto two decimal places).
3. If the digit, next to the last digit to be retained, is equal to 5, the last digit is kept unchanged if it is even, and is increased by 1 if it is odd.
Example: Suppose the result of a measurement is 12.63585. This be rounded off to given—(a) 2.6358 (for reporting the result upto four decimal places) or, (b) 2.64 (for reporting the result upto two decimal places).
5. Calculations involving addition and subtraction: The result of an addition or subtraction should be reported to the same number of decimal places as are present in the number having the least number of decimal places. The number of significant figures of different numbers does not play any role.
5. Calculations involving multiplication and division: The result of a multiplication or division should be reported to the same number of significant figures as possessed by the least precise term involved in the calculation.
6. Calculations involving multiple operations: If a calculation involves both multiplication and division, the result should be reported with the same number of significant figures as that of the least precise number involved, other than the integral number.
Leaving the integral number 4, the least precise number 0.62 has only two significant figures. So, the final result should be reported as 0.74 (two significant figures).’
Numerical Examples
Question 1. The density of a metallic substance is 7.2 g- cm-3. Find its density in the SI unit.
Answer:
Given:
The density of a metallic substance is 7.2 g- cm-3.
⇒ \(d=\frac{7.2 \mathrm{~g}}{1 \mathrm{~cm}^3}=\frac{\frac{7.2}{1000} \mathrm{~kg}}{\left(10^{-2}\right)^3 \mathrm{~m}^3}=\frac{7.2}{1000 \times 10^{-6}}\)
= 7.2×103=7200 kg.m-3
Density in the SI unit =7200 kg.m-3
Question 2. The wavelength of radiation is 643.5 nm. Find its wavelength in the SI unit.
Answer:
Given:
The wavelength of radiation is 643.5 nm.
We know, and = 10-9m
⇒ \(\text { Unit conversion factor }=\frac{10^{-9} \mathrm{~m}}{1 \mathrm{~nm}}\)
So, the wavelength of the radiation = 643.5 nm
⇒ \(=643.5 \mathrm{~nm} \times \frac{10^{-9} \mathrm{~m}}{1 \mathrm{~nm}}=6.435 \times 10^{-7} \mathrm{~m}\)
[Here, we choose the conversion factor that has nm in the denominator]
Question 3. In diamond, the average distance between two carbon atoms is 1.54A. Express the distance between two C-atoms in the SI unit.
Answer:
Given:
In diamond, the average distance between two carbon atoms is 1.54A.
1A = 10-7m
⇒ \(\text { Unit conversion factor }=\frac{10^{-10} \mathrm{~m}}{1}\) [Here, we choose the conversion factor that has Ain the denominator]
So, the average distance between two carbon atoms
⇒ \(=1.54=1.54\times \frac{10^{-10} \mathrm{~m}}{1}=1.54 \times 10^{-10} \mathrm{~m}\)
Question 4. The atomic mass of nitrogen is 14.00674u. Find out the mass of one nitrogen atom (up to 3 significant figures).
Answer:
Given:
The atomic mass of nitrogen is 14.00674u.
Mass of 6.022 x 1023 no. of-atoms = 14.00674g
⇒ \(\text { Mass of } 1 \mathrm{~N} \text {-atom }=\frac{14.00674}{6.022 \times 10^{23}} \mathrm{~g}=2.3259 \times 10^{-23} \mathrm{~g}\)
=2.32 x 10-23g
Question 5. If the density of water is Ig/mL, then find the number of H-atoms in 45 mL water (up to 3 significant figures).
Answer:
Given:
The density of water is Ig/mL
Mass of 45 mL water = 45 g [ density of water= Ig/mL]
Number of water molecules in 18 g of water = 6.022 x 1023
Number of-atoms in 18 g ofwater= 2 x 6.022 x 1023
[ 1 molecule of H2O contains 2 H-atoms]
Number of atoms in 45g of water
⇒ \(=\frac{2 \times 6.022 \times 10^{23} \times 45}{18}=3.011 \times 10^{24}=3.01 \times 10^{24}\)
[After after rounding up to 3 significant figures]
Question 6. The dimension of an iron block is 4.6in X 3.0in X 1.9in and the density of iron is 7.87g/cm3. Find out the mass of the iron block. [Given 1 in = 2.54 cm]
Answer:
Given:
The dimension of an iron block is 4.6in X 3.0in X 1.9in and the density of iron is 7.87g/cm3.
Mass of the iron block
⇒ \(\begin{aligned} =\left(4.6 \mathrm{in} \times \frac{2.54 \mathrm{~cm}}{1 \mathrm{in}}\right) \times(3.0 \mathrm{in} & \left.\times \frac{2.54 \mathrm{~cm}}{1 \mathrm{in}}\right) \\ & \times\left(1.9 \mathrm{in} \frac{2.54 \mathrm{~cm}}{1 \mathrm{in}}\right) \times\left(\frac{7.87}{1 \mathrm{~cm}^3}\right) \end{aligned}\)
= 3.381 x 103g = 3.4 X 103g [Afterrounding off]
Mass of the iron block = 3.4 X 103g
Question 8. Express 2.64 km distance in inches. [Given 1km = 1000m, lm – 1.094 yd, 1 yd = 36 in]
Answer:
= 1.0397 x 105in = 1.04 X 105in
[After rounding off up to 3 significant figures]
Laws Of Chemical Combination
Two or more substances react to form new substances. Such chemical reactions take place according to certain laws called the laws of chemical combination. These are
- Law of conservation of mass
- Law of constant proportions
- Law of multiple proportions
- Law of reciprocal proportions
Gay Lussac’s law of gaseous volumes. The first four laws deal with mass relationships while the fifth deals with the volumes of the reacting gases and products involved in the reaction.
Law Of Conservation Of Mass
Postulated by: French chemist, A. Lavoisier 1774.
Law Of Conservation Of Mass Statement
In any physical or chemical change, the total mass of the reactants is equal to that of the products,
Law Of Conservation Of Mass Explanation: Suppose two substances A and B react together to form two new substances C and D. According to the law, the sum of the masses of A and B will be equal to the sum of the masses of C and D.
Thus there will be no increase or decrease in the total mass of matter during a chemical reaction or a physical change.
So, the law can alternatively be stated as—Matter can neither be created nor be destroyed. Hence, the law is also known as the law of indestructibility matter.
Law Of Conservation Of Mass Example: Aqueous solutions of sodium chloride and silver nitrate are taken in two separate conical flasks and the flasks are weighed together in a balance.
Then, the contents of the flasks are mixed together. Consequently, a curdy white precipitate is found to be formed due to the following chemical reaction.
⇒ \(\mathrm{NaCl}+\mathrm{AgNO}_3 \rightarrow \mathrm{AgCl} \downarrow+\mathrm{NaNO}_3\)
The flasks along with the contents are again weighed together and it is noticed that there is no change in mass. This justifies the law of conservation of mass.
Law Of Conservation Of Mass Limitations:
Modification of the law of conservation of mass: According to Einstein’s theory of relativity, mass and energy are interconvertible. Mass (m) gets converted into energy (E) according to Einstein’s equation, E = me2 (where c = velocity of light).
In ordinary chemical reactions, the amount of energy released is very small and hence the law of conservation of mass holds good. In nuclear reactions, however, the change in mass is quite significant because a tremendous amount of energy is released during these reactions. So, the law of conservation of mass does not hold good.
In such cases, the total sum of mass and energy remains constant. Thus, the law of conservation of mass has been modified and the modified law is known as the law of conservation of mass energy.
The law states that mass and energy are interconvertible but the total sum of mass and energy of a system before and after any physical or chemical change remains constant.
Law Of Constant Proportions Or Definite Proportions
Postulated: French chemist, Louis Proustin 1799.
Statement A pure chemical compound always consists of the same elements (irrespective of their sources & method of preparations) combined together in the same definite proportions by mass.
Law Of Constant Proportions Explanation: Suppose compound AB is prepared by two different methods. In one method, x gram of A combines a co with y gram of B while in the other method m gram of A combines with n gram of B to form the compound AB. According to the law of constant proportions,
⇒ \(x: y=m: n, \text { or, } \frac{x}{y}=\frac{m}{n}\)
Law Of Constant Proportions Example: Pure water obtained from any natural source (For example well, river, lake, etc.) or prepared artificially (For example bypassing H2 gas over heated CuO) is always found to be made up of only two elements i.e., hydrogen and oxygen combined together in the same definite ratio of 1: 8 by mass.
The converse of the law of constant proportion is not always true: The converse of the law of constant proportions can be stated as—”When the same elements combine in a constant proportion by mass, the same compound will always be formed.”
This statement is not always correct, especially for isomeric compounds. Although the isomeric compounds have the same molecular formula, their properties are not similar. For example,
Combination of carbon, hydrogen, and oxygen in the ratio of 12: 3: 8 by mass may produce either ethyl alcohol (C2H5OH) or dimethyl ether (CH3OCH3) under different experimental conditions. But their properties are different.
A combination of carbon, hydrogen, nitrogen, and oxygen in the ratio of 12: 4: 28: 16 by mass may produce two different compounds urea (NH2CONH2) and ammonium cyanate (NH4CNO) under different experimental conditions. These two compounds have different properties.
Again, the converse of the law of constant proportion is not true for monomers and polymers. Thus, acetylene (C2H2) on polymerization gives benzene (C6H6). These two compounds contain carbon and hydrogen in the same ratio by mass (12: 1) but have different properties.
Imitations of the law of constant proportions:
If two or more isotopes of an element take part separately in the formation of a particular compound, then the same compound will contain different proportions by mass of Ihe elements depending upon its isotopic mass.
For example, In 12CO2, the ratio of the masses is C:0 = 12:32 whereas in 14C02, the ratio of the masses is C.0 = 1 4: 32.
This shows different sources of carbon dioxide may contain carbon and oxygen present in different mass ratios.
There are some compounds that have variable molecular compositions. The law of constant proportions is not applicable to such compounds.
For example, cuprous sulfide may have a molecular composition from and titanium oxide may have a composition. Such compounds are called non-stoichiometric compounds.
WBBSE Class 11 Basic Concepts of Chemistry Questions and Answers
Numerical Examples
Question 1. Analysis of 30g of compound D was found to contain 10g of element A and 20g of element IS. Again analysis of 45 g of another compound E was found to contain 15g of element B and 30g of element C. Calculate the amounts of D and E formed if 15g of A, 60g of B, and 15g of C are mixed together and allowed to react with each other. Also, calculate the total mass of the mixture after the completion of the reaction. Assume that no other reaction is possible except the reaction of B with A and separately. State which laws of the chemical combination can be utilized in the calculation.
Answer:
In compound D, mass-ratio of A to B = 10: 20 =1: 2
In compound E, the mass ratio of C = 15: 30 =1:2
Now applying the law of constant proportions we have, 15g of A combined with 2 x 15 = 30 g of B to form (15 + 30) = 45 g compound D.
Similarly, 15g of C combines with x 15 = 7.5 g of B to form (15 + 7.5) = 22.5 g compound E
∴ Amount of B remaining unreacted in the mixture
= [60 -(30+7.5)] = 22.5 g
The total mass of the mixture after completion of the reaction = Mass of D + Mass of + Mass of remaining unreacted
= 45 + 22.5 + 22.5 = 90 g.
Concepts of the law of mass action and the law of constant proportions are utilized in the calculations.
Question 2. 5g of pure MgO (obtained by reaction of metallic magnesium with oxygen) contains 3g of Mg. Again 8.5 g of pure MgO (obtained by heating MgCO3) contains 5.1g of Mg. Show that these results are in accordance with the law of constant proportions.
Answer:
Given
5g of pure MgO (obtained by reaction of metallic magnesium with oxygen) contains 3g of Mg. Again 8.5 g of pure MgO (obtained by heating MgCO3) contains 5.1g of Mg.
In the first variety of MgO, the ratio of masses of Mg to O = 3 : (5-3) = 3: 2
In the second variety of MgO, the ratio of the masses of Mg to O
= 5.1: (8.5 -5.1) =5.1 : 3.4 = 3: 2
So independent of its source, MgO always contains Mg and O in the mass ratio of 3: 2 and this is in accordance with the law of constant proportions.
Law Of Multiple Proportions
Postulated by: John Dalton in 1803.
Law Of Multiple Proportions Statement
when two elements combine with each other to form two or more compounds, then the different masses of one of the elements which combine with a fixed mass of the other, bear a simple whole number ratio (Example1: 2,1 : 2: 3,1: 3: 4 etc.).
Law Of Multiple Proportions Explanation:
Let a fixed mass of the element X combine separately with a, b, and c parts by masses of another element Y to form three different compounds A, B, and C.
So according to the law of multiple proportions, the ratio a: b: c will be a simple whole number ratio.
Law Of Multiple Proportions Example: Carbon and hydrogen combine with each other to form ethane (C2Hg), ethene (C2H4), and ethyne (C2H2).
In the formation of these three compounds, 24 parts by mass of carbon combine separately with 6 parts, 4 parts, and 2 parts by masses of hydrogen respectively.
Thus, the ratio of the masses of hydrogen which combine separately with the fixed mass of carbon (24 parts) in these compounds is =6:4:2 =3:2:1 which is a simple whole number ratio.
Exception of the law of multiple proportions: in case of simple hydrocarbons—methane (CH4), ethane (C2H6), propane (C3H8), butane (C4H10), etc., the ratio of different masses of hydrogen which combines separately with 12 parts by masses of carbon is 4: 3: 2.67: 2.5. This is not a simple whole number ratio.
Numerical Examples
Question 1. Two compounds A and B consist of tin and oxygen. Compound A contains 78.77% of tin and 21.23% of oxygen while compound B contains 88.12% of tin and 1 1.88% of oxygen. Show that these data illustrate the law of multiple proportions.
Answer:
Given
Two compounds A and B consist of tin and oxygen. Compound A contains 78.77% of tin and 21.23% of oxygen while compound B contains 88.12% of tin and 1 1.88% of oxygen.
In the formation of compound B, 11.88 parts by mass of oxygen combine with 88.12 parts by mass of tin.
21.33 parts by mass of oxygen combined with \(\frac{88.12 \times 21.23}{11.88}=157.47\) parts by mass of tin.
Thus, the ratio of the masses of tin which combine separately with fixed mass (21.33 parts) of oxygen to form the compounds A and B is given by = 78.77: 157.47 = 1:2 (approx) which is a simple whole number ratio. So, the given data illustrates the law of multiple proportions.
Question 2. Two oxides of a metal, M were heated separately in hydrogen. The water obtained in each case was carefully collected and weighed. It was observed that— O 0.725 g of the first oxide gives 0.18 g of water and 2.86 g of the second oxide gives 0.36 g of water. Show that these results are in accordance with the law of multiple proportions.
Answer:
Given
Two oxides of a metal, M were heated separately in hydrogen. The water obtained in each case was carefully collected and weighed. It was observed that— O 0.725 g of the first oxide gives 0.18 g of water and 2.86 g of the second oxide gives 0.36 g of water.
The amount of water obtained by the reduction of the first oxide =0.18 g.
Now, 18 g water contains = 16 g of oxygen
∴ 0.18g of water contains = 0.16 g of oxygen.
∴ 0.725g of the first oxide contains = 0.16 g of oxygen
So, the mass of metal in the first oxide = (0.725- 0.16)
= 0.565 g
∴ The mass of oxygen which combines with 0.565g of metal, M =0.16g.
Again, 0.36 g of water is obtained by a reduction of 2.86 g of the second oxide.
Now, 0.36 g ofwater contains \(=\frac{16 \times 0.36}{18}=0.32 \mathrm{~g}\) oxygen.
So, the amount of metal in the second oxide
=(2.86-0.32) =2.54 g
The mass of the oxygen winch combines with 2.54g of metal, M = 0.32 g.
The mass of oxygen which combines with 0.565 g of metal, \(\mathrm{M}=\frac{0.32 \times 0.565}{2.54}=0.071 \mathrm{~g}\)
Thus, the ratio of the masses of oxygen which combine separately with a fixed mass (0.565 g) of the given metal to form two different oxides is given by =0.16: 0.071 = 2:1 (approx), which is a simple whole number ratio. So, the given data are in accordance with the law of multiple proportions.
Law of reciprocal proportions
Postulated by: Richter 1792
Law of reciprocal proportions Statement
When two different elements combine separately with a fixed mass of a third element, then the ratio of their masses is either the same or some simple whole number multiple of the ratio in which they combine directly with each other.
Law of reciprocal proportions Explanation:
Suppose parts by mass of the element A and b parts by mass of the element B combine separately with a fixed mass of the element C to form the compounds X and Y respectively.
Thus, the ratio of the masses of the elements A and B which combine separately with the fixed mass of C is given by a: b. Now according to the law of reciprocal proportions, if the elements A and B combine with each other, the ratio of their masses in the resulting compound will be either a: b or xa:yb (where x andy are simple whole numbers).
Law of reciprocal proportions Example: The elements, carbon and oxygen combine separately with the third element, hydrogen to form methane (CH4) and water (H2O) respectively. Analysis shows that
1. in methene (CH4), 4 parts by mass of hydrogen combines with 12 parts by mass of carbon i.e., 1 part by mass of hydrogen combines with 3 parts by mass of carbon;
2. in water (H2O), 2 parts by mass of hydrogen combined with 16 parts by mass of oxygen i.e.,1 part by mass of hydrogen combined with 8 parts by mass of oxygen.
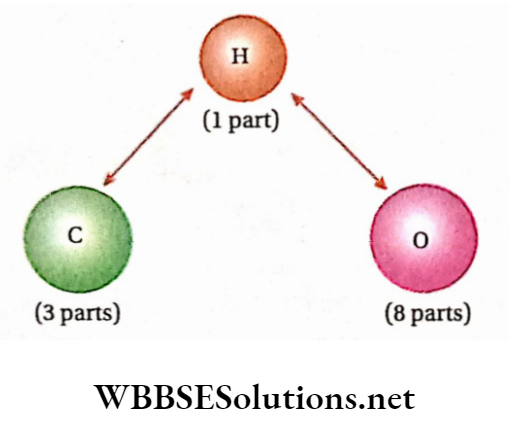
Thus, the masses of carbon and oxygen, which combine separately with a fixed mass (1 part) of hydrogen are in the ratio of 3:8.
So, according to the law of reciprocal proportions, if carbon and oxygen combine directly with each other, the ratio of their masses in the resulting compound will be 3: 8 or some simple whole number multiple of it.
Again, carbon and oxygen directly combine to form carbon dioxide (CO2). Analysis shows that in this compound, the ratio of the masses of C to 0 is = 12: 32 = 3: 8.
This ratio is found to be the same as that has been predicted earlier. Thus, the law of reciprocal proportions is illustrated.
Again, C and 0 directly combine with each other to form another compound called carbon monoxide (CO).
Analysis shows that the ratio of the masses to O in this compound is 12: 16 = 3: 4. This is a simple whole number multiple of the ratio 3: 8 as predicted earlier, 3:4 = (3×2:8xl)
Numerical Examples
Question 1. Show that the following experimental data are in agreement with the law of reciprocal proportions :
- 0.46 g of Mg on burning in air forms 0.76 g of MgO.
- 0.41 g of Mg in reaction with excess acid produces 380 cm³ of H2 at STP.
- 0.16 g of H2 reacts with excess oxygen to produce 1.45g of water.
Answer:
1. Mass of oxygen 0.76 g of MgO = (0.76- 0.46) = 030g
∴ Mass of oxygen combining within of Mg \(=\frac{0.30}{0.46}\) =0.652g
2. Mass of 22400 cm³ of H2 gas (atSTP)=2g
∴ Mass of 380 cm³ of H2 gas (at STP) =\(\frac{2 \times 380}{22400}=0.034 \mathrm{~g}\)
So, the amount of H2 gas produced by 0.41g of Mg=0.034 g
Amount of H2 gas produced by lg of Mg \(=\frac{0.034}{0.41}=0.083 \mathrm{~g}\)
From (1) and (2), it is found that the masses of hydrogen and oxygen combined with or replaced by a fixed mass are in the ratio, 0.083: 0.652 1: 8 Now, according to the law of reciprocal proportions, if the elements H and O combine, the ratio of their masses in the resulting compound will be either 1: 8 or any simple multiple of it.
0.16 g of hydrogen reacts with excess oxygen to form 1.45 g of water.
∴ Mass of oxygen combined with hydrogen
= 1.45- 0.16 = 1.29 g
So, the mass ratio of H to O in water
= 0.16: 1.29=1: 8
This is the same ratio as predicted earlier.
Thus, the given data are in agreement with the law of reciprocal proportions.
Question 2. Ammonia contains 17.65% of hydrogen, water contains 11.11% of hydrogen and nitrous oxide contains 36.36% of oxygen. Show that these data illustrate the law of reciprocal proportions.
Answer:
Given
Ammonia contains 17.65% of hydrogen, water contains 11.11% of hydrogen and nitrous oxide contains 36.36% of oxygen.
In ammonia, the amount of 11 = 17.65 %.
Amount of = (100- 17.65) = 82.35 %.
So, the mass of N that combines with 1 part by the mass of H
⇒ \(=\frac{82.35}{17.65}=4.66 \text { parts. }\)
In water, the amount of present =11.11 %,
Amount of C-present = (100- 11.11) = 88.89 %
So, the mass of O that combines with part by the mass of H
⇒ \(=\frac{88.89}{11.11}=8 \text { parts. }\)
So, according to the law of reciprocal proportions, if the elements, N and O combine together, the ratio of their masses in the die compound so formed will be either 4.66: 8 (=0.5825: 1) or any simple multiple of it.
Now in nitrous oxide, the amount of present = 36.36 %,
amount of present = (100- 36.36) = 63.64 %
So, die mass-ratio of N to O in nitrous oxide
= 63.64: 36.36 = 1.75: 1 = 3 X 0.5825: 1
It is a simple multiple of the ratio, 0.5825:1 as predicted earlier. So, the given data illustrate the law of reciprocal proportions.
Gay-Lussac’s law of gaseous volumes
Postulated by: French chemist, Gay-Lussacin 1808.
Gay-Lussac’s law of gaseous volumes Definition
When gases react with each other, they always do so in volumes that bear a simple whole number ratio to one another and to the volumes of the products, if these are also gaseous, provided all volumes are measured under similar conditions of temperature and pressure.
The volumes of gaseous reactants and products are considered to be at constant temperature and pressure because the volumes of Gay-Lussac’s gases are dependent on both temperature and pressure.
So, GayLussac’s law will not be valid if volumes are not measured under the same conditions of temperature and pressure.
Gay-Lussac’s law of gaseous volumes may be considered the law of definite proportions in terms of volume.
The law of definite proportions, discussed earlier, was with respect to mass. Gay-Lussac’s law was justified theoretically by Avogadro in 1911.
Gay-Lussac’s law of gaseous volumes Example: 1 has been experimentally found that under the same conditions of temperature and pressure, 2 volumes of hydrogen reacts with 1 volume of oxygen to form 2volume of steam. So, the volumes of the reactants (H2 and O2) and the product (steam i.e., H2O) measured under identical conditions of temperature and pressure are in the proportion of 2: 1: 2 which is a simple ratio.
It has been experimentally observed that 1 volume of hydrogen gas reacts with 1 volume of chlorine gas to form 2volume of hydrogen chloride.
So, under identical conditions of temperature and pressure, the ratio of the volumes of gaseous hydrogen, chlorine, and hydrogen chloride is 1: 1: 2, which is a simple ratio.
Characteristics of Lussac’s law of gaseous volumes:
The other laws of chemical combinations interpret the chemical combinations in terms of masses of reactants and products.
But, Gay-Lussac’s law of gaseous volumes establishes the relation between the gaseous reactant(s) and product(s) in terms of their volumes.
This law cannot be explained with the help of Dalton’s atomic theory whereas the other laws of chemical combination can be successfully explained by this theory.
Numerical Examples
Question 1. 2 volumes of 03 produce 3 volumes of O2 on complete decomposition. 40 mL of a mixture of O3 and O2 is heated at first and then brought back to the previous temperature and pressure.
The volume of the gaseous mixture is now found to be 42 mL. Find the percentage composition of Og in the gas mixture by volume.
The volume of all gases is measured under the same conditions of temperature and pressure.
Answer: Let, the volume of O3 present in the mentioned gas mixture is x mL. Volume of O2 in the gas mixture = (40- x) mL. Now, the decomposition of O3 can be represented as
⇒ \(2 \mathrm{O}_3 \longrightarrow 3 \mathrm{O}_2\)
⇒ \(\begin{array}{ll}
2 \text { volume } & 3 \text { volume } \\
2 \mathrm{~mL} & 3 \mathrm{~mL} \\
1 \mathrm{~mL} & 3 / 2 \mathrm{~mL}
\end{array}\)
Hence, x mL of 03 yields (3/2)xml of O2.
So,the total volume of O2 = \((40-x)+\frac{3}{2} x=\left(40+\frac{x}{2}\right) \mathrm{mL}\)
According to the question, 03 is completely decomposed.
So, \(40+\frac{x}{2}=42\) Or,x=4
Amount of O3 in the initial mixture =4×100/40=10%
Question 2. What is the minimum volume of oxygen that must be mixed with 100mL of carbon monoxide to convert it completely into carbon dioxide in an explosion? Find the volume of carbon dioxide formed at the same temperature and pressure. The volume of all gases is measured under the same conditions of temperature and pressure.
Answer: The formation of CO2 by the explosion of a mixture of CO and O2 follows the equation given below:
⇒ \(2 \mathrm{CO}+\mathrm{O}_2 \longrightarrow 2 \mathrm{CO}_2\)
2 volume 1 volume 2 volume
(under identical conditions of pressure and temperature)
The above equation shows that under similar conditions of pressure and temperature, 2 volumes of CO react with 1 volume of02 to produce 2 volumes of C02
According to Gay-Lussac’s law of gaseous volumes, the ratio of the volumes of CO, 02, and C02 is:
2: 1: 2 = 2 x 50: 1 X 50: 2 X 50 = 100: 50: 100
Hence, 50 mL of 02 must be muted with 100 mL of CO so that CO2 formed as a result of the reaction will be 100 mL.
Question 3. Under the same pressure and temperature, a mixture of 100 mL of water gas and 100 mL of 02 is subjected to explosion. Find the composition of the gas mixture formed by an explosion under the same conditions as AllS. pressure water gas and is temperature.
Answer: Water is gas and is a temperature. m DALTON’S ATOMIC THEORY (mixture of the same volume of CO and H2 ).
So, 100 mL of water gas contains 50 mL each of CO and H2. The reactions caused by the explosion are:
⇒ \(2 \mathrm{CO}+\mathrm{O}_2 \longrightarrow 2 \mathrm{CO}_2 \cdots[1] \quad 2 \mathrm{H}_2+\mathrm{O}_2 \longrightarrow 2 \mathrm{H}_2 \mathrm{O}\)
From equation
1. it is seen that under the same conditions of temperature and pressure, 1 Volume of 02 reacts with 2 vols. of CO to form 2 vols. of CO2.
So, under the same conditions of temperature and pressure, the volume of 02 required for 50 mL of CO = 25 in and the volume of CO2 formed = 50 mL.
According to equation [2], under the same conditions of temperature and pressure, the volume of 02 required to react with 50 mL of H2 = 25 mL and the volume of water vapor, produced by the reaction = 50 mL.
Total volume of 02 used up in the two reactions = (25 + 25) = 50 mL.
Hence after explosion, both the volumes of CO2 and H20 produced is 50 mL while the volume of unreacted 02 =(100-50) = 50 ml
Understanding the Mole Concept: Questions and Answers
Dalton’s Atomic Theory
Atoms are the building blocks of matter. John Dalton developed the concept of the atom and put forward a scientific theory regarding the constitution of matter called Dalton’s atomic theory.
The theory is based on some postulates. [A postulate means a statement accepted without proof.]
Postulates Of Dalton’s Atomic Theory
- Matter is composed of very tiny particles called atoms.
- The smallest particles of an element were termed ‘simple atoms’ and that of a chemical compound were called ‘compound atoms’.
- Atoms are indivisible and cannot be divided by any physical or chemical means.
- Atoms can neither be created nor destroyed.
- Atoms of the same element are identical in all respects i.e., in mass, size, and other properties.
- Atoms of different elements are different in all respects.
Atoms take part in chemical reactions. During chemical reactions, they combine with one another in simple whole-number ratios such as 1: 1, 1: 2, 2 : 3, etc to form compound atoms (nowadays called molecules). [When we say ‘atom,’ we imply a simple atom.]
Postulates Of Dalton’s Atomic Theory Definition
The smallest indivisible, indestructible, and discrete particle of an element that retains all the physical and chemical properties of that element and takes part in chemical reactions is called an atom.
Importance of Dalton’s atomic theory
- According to this theory, an atom is the ultimate building block of matter. It is the first scientific approach toward the constitutional aspects of matter.
- The idea that all atoms of the same element are identical in mass helped to determine the atomic masses of elements.
- It successfully explains the laws of chemical combinations involving masses.
- The idea that atoms combine with one another in a simple ratio helped to determine the formulae of chemical compounds and to express chemical reactions in the form of balanced chemical equations.
- The idea of the indivisibility of atoms has made chemical calculations easier. Still now, in all chemical calculations, the atom is considered an indivisible unit.
- This theory helped Avogadro to formulate the concept of molecules and to propose molecular theory.
Limitations Of Dalton’s Atomic Theory
- Dalton’s atomic theory did not make any distinction between the smallest particle of the elements and that of the chemical compounds having free existence.
- So, this theory created confusion regarding the nature of the ultimate particles of matter.
- Later this confusion was dispelled by Avogadro when he first introduced the concept of molecules in the substances.
- According to Dalton’s atomic theory, the atom is indivisible. However, after some fundamental scientific discoveries, it has been found that an atom is composed of sub-atomic particles like electrons, protons, neutrons, etc., i.e., the atom cannot be regarded as Indivisible.
- In the opinion of Dalton, the atom can neither be created nor be destroyed. But this proposition is not correct as an atom of an element can be transformed artificially into an atom of another element by nuclear reactions.
- According to Dalton, atoms of the same element are identical in all respects while atoms of different elements are different. This postulate was proved wrong after the discoveries of isotopes and isobars.
- Isotopes are the atoms of the same element having different atomic masses and physical properties while isobars are the atoms of different elements having the same atomic mass.
- During the formation of a chemical compound, the atoms unite together in simple whole-number ratios.
- This statement is not valid in all cases. In the case of compounds like protein, starch, cellulose, etc.,
- The atoms combine In the ratio of whole numbers but the ratios are not simple. Besides, in Berthollide compounds, the atoms do not combine in the die ratio of whole numbers.
- Gay-Lussac’s law of gaseous volumes cannot be explained due to the absence of molecular concepts in Dalton’s atomic theory.
- Compounds in which die atoms of the constituent elements are present in a simple ratio of their numbers are called Daltonide compounds. For example, CO2, H2O, FeO etc.
- There are certain compounds in which atoms of the constituent elements do not exist in a simple ratio of their numbers. These are known as Berthollide compounds such as Cu, 7S, TI07SO, etc.
Modified Form Of Dalton’s Atomic Theory
Atom is no longer considered to be indivisible. With the discovery of radioactivity, cathode rays, etc., it has been well established that atoms are composed of minute sub-atomic particles like electrons, protons, neutrons, etc.
Atoms of an element with similar chemical properties may possess different physical properties and masses ( For Example Isotopes).
Atoms of different elements with dissimilar properties may have Identical masses (For example Isobars such as 40Ca and 40Ar).
During the formation of a chemical compound, atoms of different elements may not combine in the ratio of simple whole numbers (e.g., sucrose: C2H22O).
Atom is no longer Indestructible. With the discovery of artificial radioactivity has been possible to convert atoms of one element into atoms of another element. For example
\({ }_7^{14} \mathrm{~N}+{ }_2^4 \mathrm{He} \rightarrow{ }_8^{16} \mathrm{O}+{ }_1^2 \mathrm{H}\)This is called a nuclear reaction. However, the chemical reactions fail to effect any such change.
According to Dalton’s atomic theory, atoms take part in chemical reactions—which is true even today.
But, now it has been slightly modified and it is established that the electrons in the outermost shell of an atom take part in chemical reactions.
Concept Of Molecules And Avogadro’s Hypothesis
In order to correlate Dalton’s atomic theory and Gay-Lussac’s law of gaseous volumes, Berzelius, a Swedish chemist made a generalization known as Berzelius’ hypothesis.
It states that, under the same conditions of temperature and pressure equal volumes of all gases contain the same number of atoms. Application of this hypothesis to some gaseous reactions leads to the conclusion that atoms are divisible.
This is In direct conflict with DaJton’s atomic theory, which states that atoms are the smallest particles of elements and are indivisible.
Hence, scientists discarded Berzelius’ hypothesis. While Investigating the cause of the failure of Berzelius’ hypothesis an Italian scientist Amadeo Avogadro (1811) announced that it would be possible to correlate Gay-Lussac’s law of gaseous volumes with DaJton’s atomic theory if the existence of another type of minute particle, besides atom, was conjectured.
He named this minute particle a molecule. By applying this concept of molecule, he introduced the molecular theory wherein the distinction between ‘atom’ and ‘molecule’ was mentioned explicitly. In Avogadro’s opinion—
The building blocks of matter are of two kinds—one is the atom as mentioned by Dalton while the other ultimate particle molecule as mentioned by Avogadro.
A molecule refers to the ultimate particle of a substance (element or compound) that has free existence and possesses all the characteristic properties of that substance.
An atom is the ultimate particle of an element, which takes part in a chemical reaction and may or may not exist in a free state.
Molecules may be of two types viz., elementarymolecule and compoundmolecule. Elementarymolecule is formed by atoms of the same element On the other hand, atoms of different elements form a compound molecule.
Unlike an atom, a molecule may be divided into its constituent atoms which take part in any chemical reaction.
Avogadros hypothesis
Avogadros hypothesis Statement: The same conditions of temperature and pressure, equal volumes of all gases (element or compound) contain the same number of molecules.
Avogadro’s hypothesis Explanation: If ‘n’ is the number of molecules presenting 1L of hydrogen at pressure P and temperature T, then at the temperature and pressure, 1L of carbon dioxide or 1L of any other gas will also contain the ‘ri number of molecules.
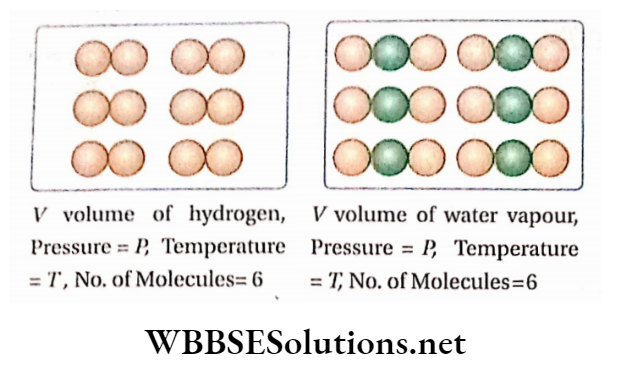
The converse statement of Avogadro’s hypothesis: All gases containing the same number of molecules will occupy the same volume under the same temperature and pressure.
Hence, if the ‘n ‘ number of hydrogen molecules occupy V volume under certain conditions of temperature and pressure, then the ‘n ‘ number of molecules of nitrogen, carbon dioxide, or ammonia will also occupy the same volume ( V), provided the temperature and pressure remain the same.
Elementary Molecule And Compound Molecule
Elementary molecule: Molecules composed of atoms of the same element are known as elementary molecules or homoatomic molecules. For example, hydrogen (H2) oxygen (O2), chlorine (Cl2), etc.
There are some solid non-metals whose molecules are composed of a single atom; example carbon (C), silicon (Si), etc., and thus monoatomic. Gases like O2, Cl2, H2, etc., are diatomic.
Again, molecules of some non-metals contain more than two atoms viz., phosphorus (P4), sulfur (S0), etc., which are polyatomic.
Compound molecule: Molecules that are composed of atoms of two or more different elements are called compound molecules or heteroatomic molecules.
For example, a water (H2O) molecule consists of 2 atoms of hydrogen and 1 atom of oxygen. Again, the sulphuric acid (H2SO4) molecule is composed of 2 atoms of hydrogen, 1 atom of sulfur, and 4 atoms of oxygen.
The number of atoms present in an elementary molecule is called the atomicity of the molecule. Thus the atomicities of argon, nitrogen, and phosphorus are 1, 2, and 4 respectively.
Correlation between Dalton’s atomic theory and Gay-Lussac’s law of gaseous volumes
Avogadro’s hypothesis helps to correlate Dalton’s atomic theory with Gay-Lussac’s law of gaseous volumes.
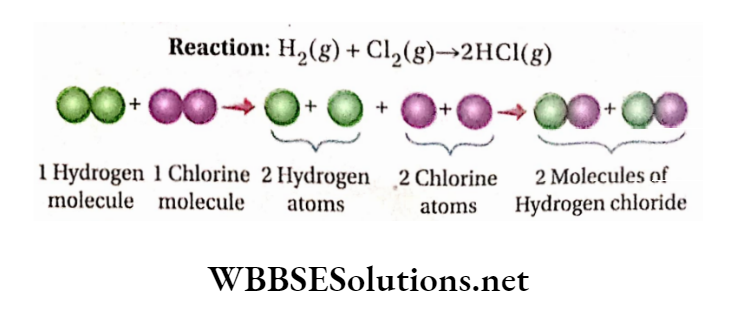
Formation of hydrogen chloride from hydrogen and chlorine gases: From actual experiments, it is known that under the same conditions of temperature and pressure, 1 volume of hydrogen (H2 ) combines with the volume of chlorine (Cl2) to produce 2 volumes of hydrogen chloride (HC1) gas; i.e., 1 volume of H2 +1 volume of Cl2 = 2 volume of HCl
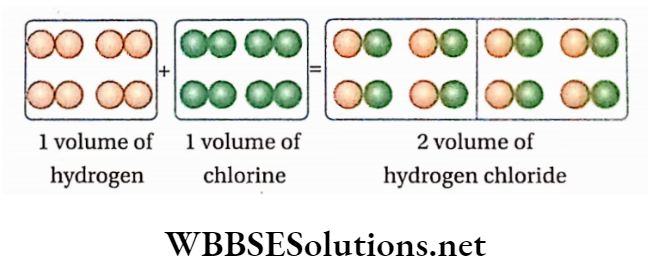
If under the experimental conditions of temperature and pressure, 1 volume of H2 contains the ‘n’ number of molecules, then according to Avogadro’s hypothesis, at the same temperature and pressure, 1 volume of Cl2 and 2 vols. of HC1 will contain ‘n ’ number of chlorine molecules and 2n number of hydrogen chloride molecules respectively
So, ‘n’ molecules of H2 +’n’ molecules of Cl2 = 2n molecules of HC1
or, 1 molecule of H2 +1 molecule of Cl2 = 2 molecules of HC1
i.e.,1/2 molecule of H2 molecule of Cl2 =1 molecule of HC1.
It has been later proved by Avogadro’s hypothesis and other experiments that elementary gases such as hydrogen, chlorine, nitrogen, oxygen, etc. are diatomic, i.e., each molecule of these gases contains two atoms only.
Therefore, 1/2 molecule of hydrogen =1 atom of hydrogen and, 1/2 molecule of chlorine = 1 atom ofchlorine Thus, the combination of one atom of hydrogen with an atom of chlorine yields one molecule of hydrogen chloride.
This deduction does not. contradict Dalton’s atomic theory because an atom is indivisible but a molecule may be divisible.
Indeed, during the chemical reaction, the molecules of hydrogen and chlorine split into their respective atoms and these atoms combine in a simple ratio to form a hydrogen chloride molecule.
Again, when Gay-Lussac’s law is applied to this gaseous reaction, the ratio of the volume of the reactants and the product becomes, H2: Cl2: HC1 =1:1: 2—it is a simple whole number ratio.
Thus in the case of the above chemical reaction, it is seen that Avogadro’s hypothesis successfully correlates Gay-Lussac’s law of gaseous volumes with Dalton’s atomic theory.
Deduction of Gay-Lussac’s law of gaseous volumes with the help of Avogadro’s hypothesis
Gay-Lussac’s law of gaseous volumes can be deduced with the help of Avogadro’s hypothesis in the following way.
Let, a molecules of a gas A, react with b molecules of another gas B, to form c molecules of another gas C at a particular temperature and pressure, where a, b, and c are small whole numbers.
Now, let’s assume that under the experimental conditions of temperature and pressure, the unit volume of gas A contains n number of molecules. So, according to Avogadro’s hypothesis, at the same temperature and pressure, the unit volume of each of the gases B and C will also contain n number of molecules.
Let n number of molecules of gas A occupy1 volume.
A number ofmolecules of gas A occupy \(\frac{a}{n}\) volume.
Similarly, b number of molecules of gas B occupy \(\frac{b}{n}\) volume.
and c number ofmolecules ofgas C occupy \(\frac{c}{n}\) volume.
Thus, the ratio of the volumes of the reacting gases, A and B to that of the gaseous product, \(C=\frac{a}{n}: \frac{b}{n}: \frac{c}{n}=a: b: c\) which is a simple ratio because a, b and c are small whole numbers.
Thus, it is observed that under the same conditions of temperature and pressure, the reacting gases combine in a simple proportion by volume and the volumes of the ga product(s) also maintain a simple ratio with the volumes of the gaseous reactants.
Therefore, the law of gaseous volumes as expressed by Gay-Lussac is established.
Modified Form of Dalto’s atomic theory based on Avogadro’s hypothesis: Molecular concept of matter.
With the introduction of Avogadro’s concept, modification of the atomic concept of matter became unavoidable.
The modified concept came to be known as the molecular concept or atomic-molecular concept of matter.
The newly embodied concept regarding the constitution of matter and its related properties is summarised below.
- The smallest particles of an element that take part in chemical reactions are known as atoms. Atoms may or may not have independent existence.
- The ultimate particles of a substance, element, or compound that can exist in the free state and possess all the physical and chemical properties of that substance are called molecules.
- Generally, molecules are composed of two or more atoms. Atoms of the same element form elementary’ molecules (For example H2 Cl2, N2, etc.) while atoms of different elements constitute a compound molecule (for example; H2O HNO3, etc.).
- Molecules are divisible.
- Molecules ofthe same substance are identical in mass and properties but the molecules of different substances differ in mass and properties.
- During chemical reactions, the participating molecules react in a simple ratio of their numbers to form molecules of new substances. However, the molecules do not react directly. At first, the reacting molecules split into their respective atoms which in turn combine mutually in a simple ratio to form molecules of a new substance.
- The formation of HCI gas by the combination of HCl2 is shown below pictorially.
Definition of molecule on the basis of Avogadro’s hypothesis
The ultimate particle of a substance (element or compound) that can exist in the free state and possesses all the properties of that substance is called the molecule of that substance.
Can Avooadro’s hypothesis be considered as a law?
Avogadro’s hypothesis originated from mere imagination. Even till now, it has not been verified by any direct experiment But the validity of this hypothesis has been well established indirectly with the help of various experiments.
The conclusions resulting from the application of this hypothesis have always been proved errorless.
No experimental results ever challenged the validity of this hypothesis. That is why, Avogadro’s hypothesis is, nowadays, called Avogadro’s law.
Atomic Mass Or Atomic Weight
The absolute mass of an atom of any element is so small that it cannot be weighed directly with the help of a chemical balance.
Moreover, it is inconvenient to express such a small mass. Thus, the mass of an atom of an element is expressed in terms of relative mass and this relative mass is called atomic mass.
Different Scales Of Atomic Mass
- In order to determine the relative mass of an atom of any element, it is necessary to take an element as a standard of reference.
- For this purpose, elements like hydrogen, and carbon are considered as the standard elements.
Hydrogen scale: At first, hydrogen was regarded as the standard element for determining the atomic masses of elements.
Hydrogen scale Definition The Atomic mass of an element may be defined as a relative number that shows how many times an atom of the element is heavier than one atom of hydrogen, taking the mass of hydrogen as unity.
⇒ \(\text { Atomic mass of an element }=\frac{\text { mass of } 1 \text { atom of the element }}{\text { mass of } 1 \text { atom of hydrogen }}\)
Example: The statement ‘atomic mass of sodium is 23 signifies that one atom of sodium is 23 times heavier than one atom of hydrogen.
Oxygen scale: Later on, instead of hydrogen, oxygen was considered the standard. In the oxygen scale, the atomic mass of an element is defined as follows.
Oxygen scale Definition The Atomic mass of an element is a relative number that denotes how many times an atom of the element is heavier than the l/16th part of the mass of an oxygen atom.
\(\begin{aligned}\text { Atomic mass of an element } & =\frac{\text { mass of } 1 \text { atom of the element }}{\frac{1}{16} \times \text { mass of } 1 \text { atom of oxygen }} \\
& =\frac{\text { mass of } 1 \text { atom of the element }}{\text { mass of } 1 \text { atom of oxygen }} \times 16
\end{aligned}\)
Example: The statement ‘the atomic mass of nitrogen is 14’ implies that an atom of nitrogen is 14 times heavier than 1/16 the part of the mass of an oxygen atom.
Reasons for taking oxygen as standard Instead of hydrogen:
- Most of the elements, metals, in particular, react with oxygen compared to hydrogen to form stable compounds.
- As hydrogen is the lightest of all elements, slight experimental errors in the determination of atomic masses in the H-scale become erroneous.
- Atomic masses of elements determined in the O-scale are mostly whole numbers compared to the fractional values as obtained from H -the scale.
Carbon (12C) scale: At present, carbon has been accepted as the standard element. This scale has been approved by the international organization 1UPAC.
On the die scale, the mass of one 12C atom is taken as 12. [On this basis the relative mass of hydrogen comes out to be 1,008 and that ofoxygen is 15.994 (or roughly 16).
Carbon (12C) scale Definition: The atom’s mass of an element is a relative number which denotes how many times an atom of that particular element is heavier than l/12th part of the mass of one 12 C atom.
That is the atomic mass of an element
⇒ \(\begin{aligned}
& =\frac{\text { mass of } 1 \text { atom of the element }}{\frac{1}{12} \times \text { mass of } 1 \text { carbon }\left({ }^{12} \mathrm{C}\right) \text { atom }} \\
& =\frac{\text { mass of } 1 \text { atom of the element }}{\text { mass of } 1 \text { carbon }\left({ }^{12} \mathrm{C}\right) \text { atom }} \times 12
\end{aligned}\)
Since the atomic mass of an element is the ratio of two Atomic masses of some elements in different scales masses, it is in fact relative atomic mass, it has no unit and is expressed as a pure number.
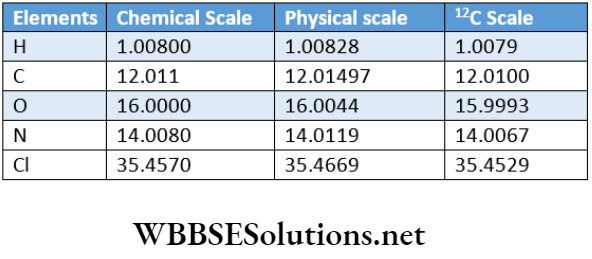
Physical and chemical scales of atomic mass: Natural oxygen consists of 3 isotopes:leO (99.759%),170 (0.037%) and 180 (0.204%). So, the true atomic mass i.e., the average atomic mass of natural oxygen
= 16×0.99759 +17×0.00037 +18 X 0.00204 = 16.00204
But the atomic mass ofthe most abundant isotope (160) of natural oxygen = 16. Chemists take the average atomic mass of natural oxygen as the standard of reference to prepare the chemical scale of atomic mass and physicists take the atomic mass of the most abundant isotope of natural oxygen as the standard of reference to prepare the physical scale of atomic mass.
Almost all elements have isotopes, therefore, in place of ‘mass of atom; ‘average mass of an atom’ is to be used.
The chemical scale of atomic mass: The scale of atomic mass, which is obtained by taking the average mass of an atom of natural oxygen as 16.0000, is called the chemical scale of atomic mass and the atomic mass, as obtained by this scale, is known as the chemical atomic mass of that element.
Physical scale of atomic mass: The scale of atomic mass, which is obtained by taking the mass of a lsO isotope in natural oxygen as 16.0000, is called the physical scale of atomic mass and the atomic mass, as obtained by this scale, is known as physical atomic mass.
According to the physical scale, the average atomic mass of natural oxygen = 16.00447. However according to the chemical scale, the average atomic mass of natural oxygen = 16.0000. So, 16.0000 units in the chemical scale = 16.00447 units in the physical scale.
1 unit in chemical scale =16.00447/16.0000=1.0002794 in physcial Scale.
Thus, the magnitude of the average atomic mass of an element on the chemical scale is slightly less than that of the mass of an atom ofthe element’s physical scale of atomic mass.
1.0002794 is the conversion factor that is used to convert the chemical atomic mass of an element to its physical atomic mass and vice-versa.
Atomic mass on a physical scale = 1.0002794 x Atomic mass on a chemical scale.
Atomic mass unit (AMU)
The atomic mass of an element is a relative number and it has no unit. So, the atomic mass of an element does not stand for the absolute or actual mass of an atom of that element order to express the actual mass of an atom, scientists introduced another unit. This unit is termed an atomic mass unit (AMU).
Atomic mass unit Definition: The unit with respect to which the actual atom of any element is expressed and whose value is equal to the mass of l/12th part of the mass of one 12C atom is called the atomic mass unit.
Atomic mass unit= \(\frac{1}{12}\) x actual mass of one C atom.
Mathematical expression: Actual mass of 6.022 x 1023 atoms of 12C isotope = 12 g.
⇒ \(\begin{aligned}\quad \text { Actual mass of one }{ }^{12} \mathrm{C} \text { atom } & =\frac{12}{6.022 \times 10^{23}} \mathrm{~g} \\
\text { So, atomic mass unit }(1 \mathrm{amu}) & =\frac{1}{12} \times \frac{12}{6.022 \times 10^{23}} \mathrm{~g} \\
& =1.6605 \times 10^{-24} \mathrm{~g}
\end{aligned}\)
1 amu = 1.6605×10-24g=1.6605×10-27kg
The actual mass of an atom: Atomic mass of an element
⇒ \(\begin{aligned}
& =\frac{\text { mass of } 1 \text { atom of the element }}{\text { mass of } 1 \text { carbon }\left({ }^{12} \mathrm{C}\right) \text { atom } \times \frac{1}{12}} \\
& =\frac{\text { mass of } 1 \text { atom of the element }}{1 \mathrm{amu}}
\end{aligned}\)
The actual mass of an atom of an element
= atomic mass ofthe element x 1 amu
= atomic mass ofthe element x 1.6605 x 10
100-24g
Thus, the atomic mass of an element, multiplied by 1 amu gives the actual mass of an atom of that element.
Examples: 1 Actual mass of atom of hydrogen
= 1.008 amu = 1.008 x 1.6605 x 10-24 g
2. Actual mass of atom of nitrogen
= 14 amu = 14 x 1.6605 x 10-24 g
The actual mass of an atom of oxygen
= 16 amu = 16 x 1.6605 x 10-24 g
In recent times, a new symbol ‘u’ (which signifies unified mass) is used in place of amu (Le., atomic mass unit)
Therefore Mass of1 H-atom = 1.008u (le., 1.008 amu)
The atomic mass of an element and the actual mass of an atom of an element are completely different: The atomic mass of an element indicates how many times an atom of that element is heavier than 1/12 part of an atom of a C isotope. It is a relative number and it has no unit.
The actual mass of an atom of an element indicates the exact mass of an atom of that element and has definite units (for example 8kg).
Example: Atomic mass of oxygen is 16 but the actual mass of an atom ofoxygen is 16 x 1.6605 x 10-24g = 2.656 X 10-23g
Average Atomic Mass
Atomic masses of most of the elements are fractional numbers because they actually represent their average atomic masses. In nature, most of the elements exist as a mixture of two or more isotopes.
The relative abundance of the isotopes of particular natural elements is more or less fixed. The atomic mass of any element is determined by taking the average of the atomic masses obtained on the basis of the abundance of various isotopes of the element in nature.
Thus, the estimated atomic mass ofthe element is a fractional value although the atomic masses of different isotopes are whole numbers.
Average atomic mass
⇒ \(=\frac{\Sigma(\text { natural abundance of isotope }(\%) \times \text { its atomic mass })}{100}\)
Let the natural abundance of the three isotopes of an element be x %, y%, and z % and their atomic masses be a, b, and c respectively.
The atomic mass (average atomic mass) of the element
⇒ \(=\frac{x \times a+y \times b+z \times c}{x+y+z}=\frac{x \times a+y \times b+z \times c}{100}\)
Example: Natural chlorine contains 2 isotopes: 35C1 and 37C1 as a mixture of 75% and 25% respectively. The atomic masses of these isotopes are 35 and 37 respectively, both of which are whole numbers. But the atomic mass ofchlorine.
⇒ \(=\frac{(35 \times 75)+(37 \times 25)}{(75+25)}=35.5, \text { which is a fraction. }\)
Gram-atomic mass and gram-atom
Gram-atomic Mass Definition The Gram-atomic mass of an element is defined as the atomic mass expressed in grams.
Atomic mass has no unit while the unit of gram-atomic mass is gram. For example, the atomic masses of nitrogen and oxygen are 14 and 16 respectively but the gram-atomic masses of these elements are 14 grams and 16 grams respectively.
The gram atomic mass is best defined as the mass in grams of an element that contains the same number of atoms as 12 presenting 12 grams of C atom.
Gram-atom: One gram-atom of an element is defined as the quantity in gram which is numerically equal to its atomic mass. For example, one gram-atom of nitrogen means 14 g of nitrogen and one gram-atom of oxygen signifies 16 g of oxygen.
One gram-atom of an element also referred to as the mass in gram of the element contains 6.022 x 1023 number (Avogadro number) of atoms.
A number of gram-atom: The given mass of an element expressed in gram, when divided by its gram-atomic mass, gives the number of gram-atoms present in that quantity ofthe element.
Therefore, the number of gram-atom of the element
⇒ \(=\frac{\text { mass of the element }(\mathrm{g})}{\text { gram-atomic mass of that element }}\)
Examples: Number of gram-atom in 42 g of N2 \(=\frac{42 \mathrm{~g}}{14 \mathrm{~g}}=3\)
Number of gram-atom in 64 g of O2 \(=\frac{64 \mathrm{~g}}{16 \mathrm{~g}}=4\)
The discussion done till now about atomic mass indicates the relative atomic mass of an element.
However, according to IUPAC, the atomic mass of any element is the mass of one atom of that element expressed in the atomic mass unit (u).
The mass of 1 atom of any element with respect to the mass of 1 atom of 12C isotope as 12u is considered as the atomic of the corresponding element.
The atomic mass of different elements can be precisely determined by using a mass spectrograph.
According to the 12C scale, atomic masses of different elements are tabulated below—
Short Answer Questions on Dalton’s Atomic Theory
Numerical Examples
Question 1. The atomic weight of ordinary hydrogen is 1.008. Ordinary hydrogen contains two isotopes JH and 11H. What is the weight percentage of 21H in ordinary hydrogen?
Answer:
Given
The atomic weight of ordinary hydrogen is 1.008. Ordinary hydrogen contains two isotopes JH and 11H.
Let in ordinary hydrogen, 11H = X%
Percentage of 21H = 100- x
Atomic mass of ordinary hydrogen \(=\frac{x \times 1+(100-x) \times 2}{100}\)
As per given data, \(\frac{x+(100-x) \times 2}{100}=1.008\)
or, 200-x = 1.008 x =99.2
∴ Ordinary hydrogen contains 99.2% of 11H and (100-99.2) = 0.8 % of 21H.
Question 2. Chlorine occurs in nature in the form of two isotopes with atomic masses 34.97 and 36.97 respectively. The relative abundance of the isotopes are 0.755 and 0.245 respectively Find the atomic mass of chlorine.
Answer:
Given
Chlorine occurs in nature in the form of two isotopes with atomic masses 34.97 and 36.97 respectively. The relative abundance of the isotopes are 0.755 and 0.245 respectively
Atomic mass of chlorine
⇒ \(=\frac{34.97 \times 0.755+36.97 \times 0.245}{0.755+0.245}=35.46\)
Question 3. Determine the mass of1 F-atom in gram (F = 19)
Answer: Gram-atomic mass of fluorine = 19 g. The mass of1 gram-atom of fluorine= 19 g Number of atoms in 1 gram-atom fluorine = 6.022 x 1023 Mass of 6.022 x 1023 atoms of fluorine = 19g
Hence, the mass of1 atom of fluorine = \(\frac{19}{6.022 \times 10^{23}} \mathrm{~g}\)
=3.1550×10-23g
Question 4. Calculate the atomic volume of sodium (atomic weight = 23 ). Density of sodium=0.972 g rnL-1.
Answer: In the case of monatomic elements (like Na), the volume of 1 gram-atom is called its atomic volume.
1 gram-atomNa =23gofNa(v atomic mass of Na = 23)
Atomic volume of sodium = \(\begin{aligned}
& =\frac{\text { gram-atomic mass }}{\text { density }} \\
& =\frac{23 \mathrm{~g}}{0.972 \mathrm{~g} \cdot \mathrm{mL}^{-1}}=23.66 \mathrm{~mL}
\end{aligned}\)
Question 5. Find out the highest and lowest masses from the following:
- 25.6g oxygen (atomic mass = 16)
- 2.86 gram-atom of sodium (atomic mass = 23)
- 0.254 gram-atom of iodine (atomic mass = 127
Answer: Mass of 2.86 gram-atom of sodium = 2.86 x 23 = 65.78g
Mass of 0.254 gram-atom ofiodine = 0.254 x 127 = 32.258g
∴ Oxygen has the lowest and sodium has the highest mass.
Question 6. A compound contains 28% of nitrogen and 72% of metal by weight. In the compound, 3 atoms of metal remain combined with 2 atoms of nitrogen. What is the atomic mass of the metal?
Answer:
Given
A compound contains 28% of nitrogen and 72% of metal by weight. In the compound, 3 atoms of metal remain combined with 2 atoms of nitrogen.
Each molecule of the compound contains 3 atoms of metal and 2 atoms of nitrogen. If the symbol ofthe metal is M, then the molecular formula ofthe compound will be M3N2.
Molecular mass ofthe compound = 3a + 2 X 14 = 3a + 28
[where a is the atomic mass ofthe metal]
∴ Quantity of nitrogen in the compound \(=\frac{28}{(3 a+28)} \times 100 \%\)
Now, according to the problem, \(\frac{28}{3 a+28} \times 100=28\)
or, 3a +28= 100 or, a= 24
Molecular Mass Or Molecular Weight
A molecule is the smallest particle ofthe substance (element or compound) which has independent existence. Molecules are formed by the combination of atoms of the same or different elements.
So, like the atomic mass, some standard should be taken to express the molecular mass. At present, the 12C isotope is taken as the standard to express both the atomic mass and molecular mass.
Molecular mass with respect to 12 C-atoms
The molecular mass of a substance (element or compound) is a relative number that denotes how many times a molecule of the substance is heavier than the l/12th part of the mass of a 12C -atom.
∴ Molecular mass
⇒ \(\begin{aligned}
& =\frac{\text { mass of } 1 \text { molecule of the element or compound }}{\frac{1}{12} \times \text { mass of } 1 \text { carbon }\left({ }^{12} \mathrm{C}\right) \text { atom }} \\
& =\frac{\text { mass of } 1 \text { molecule of the substance }}{\text { mass of } 1 \text { carbon }\left({ }^{12} \mathrm{C}\right) \text { atom }} \times 12
\end{aligned}\)
On the carbon scale, molecular masses of nitrogen and oxygen are 28.013 and 31.998 respectively. It means that one molecule of each of nitrogen and oxygen is respectively 28.0 til and 31.990 times heavier than 1/12 the part of the mass of one,2C -atom.
Determination of the molecular mass of an element and a compound: The molecular mass of a substance (element or compound) can be determined by adding the atomic of all the atoms present in a molecule of the substance (element or compound).
The molecular mass of an element: Let us consider, the molecular formula of an element to be A,(, where n = Atomicity (i.e., no. of atoms present in the molecule of the element). Therefore, in the case of an element molecular mass = atomic, mass ofthe element x its atomicity.
- In the case of monoatomic elements (n = 1), molecular mass and atomic mass will be the same. Most of the metal elements and noble gas elements belong to this group.
- In the case of diatomic elements (n =2), the molecular mass is twice its atomic mass. Most ofthe gaseous elements (H2, N2, O2, Cl2, etc.) belong to this group.
- In the case of triatomic elements (n =3), the molecular mass is thrice its atomic mass. For example, ozone (O3 ).
- In the case of tetra-atomic elements(n = 4), the molecular mass is ( four times its atom mass. For example, phosphorus (P4).
The molecular mass of the compound: Let, the molecular formula of a compound be A2 C2, where the number of atoms of A, B, and C in a molecule of the compound are x, y,z respectively.
If the atomic masses are a, b, and c respectively, then the molecular mass ofthe compound =axx+bxy+cxz.
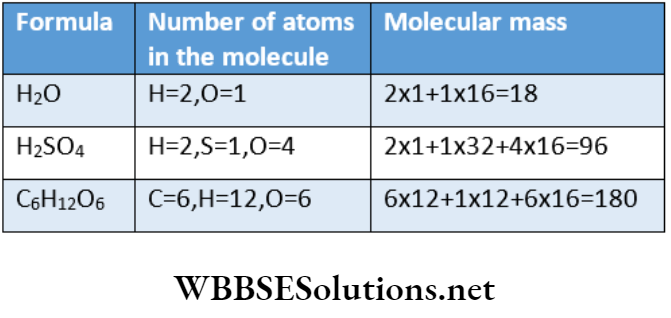
Molecular Mass Of Some Elements And Compounds In Unified Scale
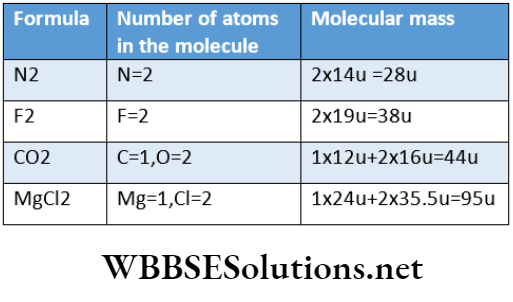
Formula Mass Or Formula Weight
Formula mass represents the sum of atomic masses of the Number of gram-molecule or gram-mole: The given present in the formula with which a substance is expressed. For example, the formula mass of sodium chloride, gram-molecular mass, gives the number of gram-mole or NaCl= (23 + 35.5) = 58.5.
Molecular mass and formula mass are not always synonymous: Molecular mass can be determined from the formula of an element or compound.
So sometimes, the formula mass is called the molecular mass. But these two terms are not always synonymous.
When a substance contains discrete molecules, then only these two terms can be used in the same sense.
Again, there are certain substances which do not exist as molecules. For example, the compound sodium chloride is represented by the formula NaCl but the existence of discrete molecules of this compound is conspicuously absent from the crystal of sodium chloride,
Na+ and Cl- ions exist in a state of aggregation where one Na+ ion is surrounded by six Cl- ions and one Cl- ion is similar and pressure, molar volumes of all gases are the same and it does surround by six Na+ ions to give rise to an octahedral not depend on the nature of the molecular mass ofthe gas.
As a result, there is no existence of Gram-molecular volume or molar Thus the statement—”the molecular mass of sodium chloride is 58.5″—has no logical basis because sodium chloride never forms a molecule.
The correct statement should be—”The formula mass of sodium chloride is 58.5″ Usually the term formula mass’ is applied to ionic compounds that do not exist as discrete molecules even in the solid state while the term ‘molecular mass’ is applied to the case of covalent compounds which remain in the molecular state even in aqueous solutions.
Gram-molecular mass
Gram-molecular mass: Molecular mass. of an element or compound expressed in gram is called gram-molecular N2 28 g 56 g —5628 = 2 2 x 22.4 = 44.8 L mass or gram-mole.
Unlike molecular mass molecular mass has a unit. For example, the gram-molecular masses of nitrogen and carbon dioxide are 28 g and 44 g respectively.
Gram-molecule or gram-mole: The quantity of a substance (element or compound), expressed in gram, which is numerically equal to its molecular mass, represents one gram-molecule or one gram-mole of that substance.
For example, 28 g of nitrogen, 32 g of oxygen, and 18 g of water represent gram-molecule or 1 gram-mole of each of the respective compounds.
Several gram-molecule or gram-mole: The given present in the formula with which a substance is expressed in gram when divided by its Therefore, number of gram-mole of substance.
⇒ \(=\frac{\text { mass of the substance }(\mathrm{g})}{\text { gram-molecular mass of that substance }}\)
Examples: 1. Number of gram-mole in 88 g of C02 \(=\frac{88 \mathrm{~g}}{44 \mathrm{~g}}=2\)
Number of gram-molein 45 g of H2O \(=\frac{45 \mathrm{~g}}{18 \mathrm{~g}}=2.5\)
Gram-molecular volume or molar volume at STP: The volume occupied by the gram-mole of all gases at STP (standard temperature and pressure) is 22.4L.
Example: Each of 2g hydrogen, 28 g nitrogen, 44 g carbon dioxide, and 18 g water vapor occupy a volume of 22.4L at STP. Conversely, it can be stated that 22.4 L of any gas at STP contains 1 gram-mole of that gas.
Volumes of different amounts of gases at STP
Numerical Examples
Question 1. Calculate the number of gram-molecules present in 14.7gH2SO4.
Answer: Number of gram-molecules of a substance
⇒ \(=\frac{\text { given mass }(\text { in } \text { g) }}{\text { gram-molecular mass }}=\frac{14.7}{98}=0.15\)
Question 2. Calculate the mass of 1.5 gram-molecule of glucose.
Answer: Gram-molecular mass of glucose(C6H12O6)
= 6X12 + 12X1 + 6X16 = 180g
Mass of 1.5 gram-molecule of glucose = 1.5 x 180 = 270g
Question 3. If the number of gram-molecules present in 4.8g of oxygen and rg of nitrogen are equal, calculate the value of x.
Answer:
Given
If the number of gram-molecules present in 4.8g of oxygen and rg of nitrogen are equal
Gram-molecular mass of oxygen (O2 ) and nitrogen (N2 ) are 32 g and 28g respectively.
Number of gram-molecule of oxygen in 4.8g = 4.8/32 and number of gram-molecules of nitrogen in xg = \(\frac{x}{28}\)
⇒ \(\text { Given, } \frac{x}{28}=\frac{4.8}{32} \text { or, } x=4.2\)
Question 4. Calculate the volume of 3.6 g of water vapor at 273 K temperature and 1 atm pressure.
Answer: Molecular mass of water vapor (H2O) =18 No. of gram-molecule in 3.6g water vapor =3.6/18 At STP (273K, late), the volume of gram-molecule of water vapour= 22.4L
At STP, the volume of 0.2 gram-molecule of water vapor = 0.2X22.4 = 4.48L
Question 5. If the density of water at 273K is i.0g-cm-3, calculate its molar volume at that temperature.
Answer: Density of water at 273K = 1.0 g-cm Gram-molecular mass of water = 18g Hence, at 273K the molar volume of water.
Question 6. How many moles of water molecules are present in 1.8 ml of water?
Answer:
At ordinary temperature, the density of water =1g mL 1.
Mass of 1.8 mL ofwater = 1.8 X 1 = 1.8 g.
Now, 1.8 g of H2O =\(\frac{1.8}{18}\)=0.1 mol H2O
Question 7. Calculate the number of moles and the volume at STP of 0.53g of acetylene.
Answer: Gram-molecular mass of acetylene is 26g.
Number of gram-mole present in 0.52g of acetylene
= 0.52/26g = 0.02
At STP, 26 g of acetylene occupies a volume of 22.4 L.
At STP, 0.52g of acetylene occupies a volume of
⇒ \(=\frac{22.4 \times 0.52}{26} \mathrm{~L}=0.448 \mathrm{~L}\)
Question 8. At STP, the volume of lg of a gaseous substance is 280 mL. Find its relative molar mass.
Answer: At STP, the mass of 22400 mL of the gas \(=\frac{1}{280} \times 22400=80 \mathrm{~g}\) Now, on the basis of Avogadro.s hypothesis, 22400mL (at STP) of any gas contains1 gram-mole ofthe substance.
The molar mass of the gas = 80
Question 9. The volume of one atom of a metal M is 1.66x 10-23 cm3. Find the atomic mass of M (Given: density of M = 2.7 g.cm-3).
Answer: Volume of 6.022 x 1023 atoms of the metal, M
= 6.022 X 1023 X 1.66 X 10-23 =9.99652 cm3
Mass of 6.022 x 1023 atoms of the metal M
= density x volume = 2.7 x 9.99652 = 26.990 g
= gram-atomic mass ofthe metal M
Hence, the atomic mass ofthe metal = 2.990.
Question 10. Haemoglobin was found to contain 0.335% iron (Atomic weight of Fe = 56). The molecular weight of haemoglobin is 1.67 X 104. Find the number of iron atoms in hemoglobin.
Answer: Molecular mass of hemoglobin = 1.67 x 104
∴ 1 gram-mole of hemoglobin = 1.67 x 104 g.
Here, Fe -content haemoglobin = 0.335%
∴ Fe-content 1.67 x 104 g of haemoglobin
⇒ \(=\frac{0.335 \times 1.67 \times 10^4}{100}=55.945 \mathrm{~g} .\)
Hence, the number of gram-atom of Fe present in haemogobin= \(=\frac{55.945}{56} \approx 1\)
The atomic weight of =
∴ In 1 gram-mole of hemoglobin, the number of gram atoms of Fe =1
So, 1 mol of hemoglobin contains only one atom of Fe.
Question 11. The mass of 0.1 mol of X2Y is 4.4 g and the mass of 0.05 of XY2 is 2.3g. Find the Atomic mass of X and Y.
Answer:
Mass of 0.1 mol of X2Y = 4.4 g.
∴ Mass of 1 mol of X2Y \(=\frac{4.4}{0.1}=44 \mathrm{~g}\)
Similarly, mass of 1 mol of XY2 =2.3/0.05 =46g
So, the molecular mass of X2Y and XY2 are 44 and 46 respectively. Let, the atomic masses of X and Y be a and b respectively. So, the molecular mass of X2Y = (2a + b) and the molecular mass of XY2 =(a + 2b).
According to the question, 2a + b =44 and a + 2b = 46.
By solving the equations, a = 14 and b = 16.
∴ The atomic masses of X and Y are 14 and 16 respectively.
Question 12. A plant virus is found to consist of uniform cylindrical particles whose diameter is 150A and length is 5000A. The specific volume of the virus is 0.75cm³/g. If the virus is considered to be a single particle, then find its molecular mass.
Answer:
Given
A plant virus is found to consist of uniform cylindrical particles whose diameter is 150A and length is 5000A. The specific volume of the virus is 0.75cm³/g. If the virus is considered to be a single particle,
Volume ofthe virus =nr2 xl
⇒ \(=3.14 \times\left(\frac{150}{2} \times 10^{-8}\right)^2 \times 5000 \times 10^{-8}=0.884 \times 10^{-16} \mathrm{~cm}^3\)
⇒ \(\begin{aligned}
\text { Mass of single virus } & =\frac{\text { volume }}{\text { specific volume }} \\
& =\frac{0.884 \times 10^{-16}}{0.75}=1.178 \times 10^{-16} \mathrm{~g}
\end{aligned}\)
The molar mass of virus = Mass of single virus x NA
= 1.78 x 10-16x 6.023 x 1023 =7.095 x 10 7
Avogadro’s number
Avogadro’s number Definition: Avogadro’s number may be defined as the number of molecules present in one gram-mole of any substance, or element Similarly, mass of 1 mol of XY2 =2.3/0.05 =46g or compound (solid, liquid, or gas).
Avogadro’s number is usually denoted by the letter ‘N’ or ‘Na’- and its value is 6.022 x 1023. R. A. Millikan determined its value by the oil drop experiment’ in 1913.
Example: 28 g of N2, 32 g of O2, 18 g of H2O, or 100 g of CaCO3 —each indicates 1 gram-mole of substance containing 6.022 x 1023 number of molecules.
The value of Avogadro’s number does not depend on temperature and pressure because the mass and the number of molecules do not change with the variation in temperature and pressure.
Alternative definition of Avogadro’s number: The number of atoms present in the gram-atom of an elementary substance is called Avogardro’s number.
Example: 16 g O2 contains 6.022 x 1023 number atoms and in 12g carbon, the number of constituent atoms is 6.022 x 1023.
As 1 gram-mole of any gaseous substance (element or compound) occupies a volume of 22.4 L at STP, Avogadro’s number can further be expressed as—the number of molecules present in 22.4 I. of any gaseous substance (element or compound) at STP is called Avogadro’s number.
Modern Definition Of Avogadros Number
The Number of Od atoms Present In exactly 12 g carbon (12C) is designated as Avogadro’s number.

Airogadro’s constant: ‘Avogadro’s number/mole’ is called Avogadro’s constant i.e., ‘6.022 X 1023 ‘.
Avogadro’s number has no unit. But Avogadro’s constant has the unit ‘per mole’ or mol-1.Itis auniversal constant.
Definition of Gram-mole, Gram-atom, Molecular mass, Atomic mass in terms of Avogadro’s number.
Gram-mole: The quantity of a substance (element or compound) expressed in gram which contains 6.022 x 1023 molecules is defined as 1 gram of that substance.
Gram-atom: The quantity of an element expressed in gram which contains 6.022 x 1023 atoms is called 1 gram-atom of that element.
Molecular mass: The molecular mass of a substance (element or compound) may be defined as the number which when expressed in grams contains 6.022 x 1023 number of molecules that substance.
Atomic mass: The atomic mass of an element indicates the number which when expressed in grams contains 6.022 x 1023 atoms of that element.
Applications of avocados number
Calculation of actual mass of a molecule: if the molecular mass of a substance is known, the actual mass of one molecule of that substance can be calculated in the following way: Let, the molecular mass of a substance be M.
∴ 1 gram-mole ofthe substance =M g of that substance. Now, we know that 1 gram-mole of a substance contains Avogadro’s number of molecules.
∴ Mass of 6.022 x 1023 molecule M g.
∴ Mass of 1 molecule = \(\frac{M}{5.022 \times 10^{23}} \mathrm{~g}\)
∴ The actual mass of a molecule of any substance
⇒ \(=\frac{\mathrm{gram}-\text { molecular mass of the substance }}{\text { Avogadro’s number }}\)
Examples: Actual muss of a molecule of oxygen: The molecular mass of oxygen = 32.
∴ Actual mass of I oxygen molecule
⇒ \(\begin{aligned}
=\frac{\text { Gram-molecular mass of oxygen }}{\text { Avogadro’s number }} & =\frac{32 \mathrm{~g}}{6.022 \times 10^{23}} \\
& =5.313 \times 10^{-23} \mathrm{~g} .
\end{aligned}\)
Actual mass of 1 molecule of water:
The molecular mass of water = 18.
∴ Actual mass of I molecule of water
⇒ \(=\frac{18 \mathrm{~g}}{6.022 \times 10^{23}}=2.989 \times 10^{-23} \mathrm{~g}\)
Calculation of actual mass of an atom: From the known value of the atomic mass of an element, the actual mass of an atom of that element can be determined in the following way: Let, the atomic mass of an element = A.
1 gram-atom of that element = A g.
We know, 1 gram-atom of an element contains Avogadro’s number of atoms, i.e., 6.022 x 1023 atoms.
Mass of 6.022 x 1023 atoms =A g.
∴ Actual mass of 1 atom of an element
= \(=\frac{\text { gram-atomic mass of the element }}{\text { Avogadro’s number }}\)
Example 1. The actual mass of 1 atom of nitrogen:
Atomic mass ofnitrogen = 14.
∴ The actual mass of 1 atom of nitrogen:
⇒ \(\begin{aligned}
& =\frac{\text { gram-atomic mass of nitrogen }}{\text { Avogadro’s number }} \\
& =\frac{14 \mathrm{~g}}{6.022 \times 10^{23}}=2.324 \times 10^{-23} \mathrm{~g}
\end{aligned}\)
An alternative method for determination of the actual mm of an atom: Let the molecular mass of a substance= M 1 gram-molecule ofthe substance = Mg.
Now, 1 gram-molecule of any substance contains
Avogadro’s number of molecules.
Hence, mass of 6.022 x 1023 molecules =M g
Mass of 1 molecule \(=\frac{M}{6.022 \times 10^{23}} \mathrm{~g} .\) If the atomicity of the element is n, then one molecule ofthe element consists of n number of atoms.
Hence, the mass of n atoms of the element \(=\frac{M}{6.022 \times 10^{23}} \mathrm{~g}\) and the mass of I atom of the element \(\frac{M}{n \times 6.022 \times 10^{23}} g\)
Hence, the actual mass of an atom of the element
⇒ \(=\frac{\text { gram-molecular mass of the element }}{\text { atomicity of a molecule of the element } \times \text { Avogadro’s number }}\)
Example: Determination of the actual mass of a nitrogen atom—Gram-molecular mass of nitrogen = 28 and atomicity of a nitrogen molecule =2. Hence, the actual mass of a nitrogen atom.
⇒ \(=\frac{\text { gram-molecular mass of nitrogen }}{\text { atomicity of a nitrogen molecule } \times \text { Avogadro’s number }}\)
⇒ \(=\frac{28 \mathrm{~g}}{2 \times 6.022 \times 10^{23}}=2.324 \times 10^{-23} \mathrm{~g}\)
Number of molecules in a definite mass of a substance: Let, the mass of a certain amount of an element or a compound = W gram and its gram-molecular mass M gram. Now, the number of molecules presenting M gram of the substance = 6.022 X 1023.
W gram ofthe substance will contain \(=\frac{W}{M} \times 6.022 \times 10^{23}\) number of molecules.
Some important relations: If Avogadro’s number =NA and gram-molecular mass of an element or a compound =M, then—
Mass of 1 atom of an element \(=\frac{\text { gram-atomic mass }}{N_A}\)
Mass of 1 molecule of the substance \(=\frac{M}{N_A}\)
No. of molecules in W gram ofthe substance \(=\frac{W \times N_A}{M}\)
Number of molecules present in V L of gas at STP \(=\frac{V \times \mathrm{N}_{\mathrm{A}}}{22.4}\)
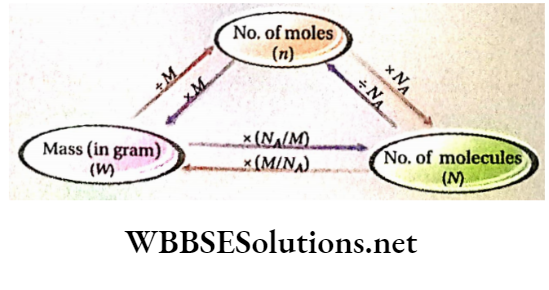
No. of molecules in n mole of any substance = n x NA
lu (or, 1 amu)=\(\frac{1}{N_A}\)– g. = 1.6605 x lO-24 g (This amount is called 1 program or 1 dalton)
1 gram-mole of any substance contains the same number of molecules:
1. Let, the actual mass of atom hydrogen =x g
∴ Actual mass of a molecule hydrogen -2xg hydrogen is diatomic]
∴ Now,1 gram-mole ofhydrogen = 2 g ofhydrogen
∴ Number of molecules present in 1 gram-mole (or 2g) ofhydrogen= 2/2x = l/x
The molecular mass of oxygen = 32
2. A molecule of oxygen is 32 times heavier than 1 atom of hydrogen.
If the actual mass of an atom of hydrogen is xg, then the actual mass of 1 molecule ofoxygen will be 32x g.
Now,1 gram-mole ofoxygen =32 g of oxygen.
∴ Number of molecules present in 1 gram-mole (or 32g) of oxygen of oxygen
⇒ \(=\frac{32}{32 x}=\frac{1}{x}\)
Let, the molecular mass of a substance be M. Therefore, a molecule of that substance will be M times heavier than an atom of hydrogen.
If the actual mass of 1 atom of hydrogen is x g, then the actual mass of 1 molecule of that substance will Now, be 1 gram-mole of that substance =M g Number of molecules constituting 1 gram-mole of the substance = \(=\frac{M}{M \times x}=\frac{1}{x}\)
Therefore, it can be concluded that 1 gram-mole of any substance contains the same number of molecules.
The ratio of the number of molecules present In equal masses of two different substances (solid, liquid, or gas) with different molecular masses: Let the molecular masses of two substances A and B be MA and MB respectively and the mass of each of them be = W g.
1 gram-mole of any substance contains = 6.022 x 1023 number of molecules.
∴ Number of molecules present in MA g of A = 6.022 x 1023 Number of molecules presentin’ W g of \(A=\frac{6.022 \times 10^{23} \times W}{M_A}\)
Similarly, the number of molecules present in Wg of B \(=\frac{6.022 \times 10^{23} \times W}{M_B}\)
The ratio of the number of molecules present in equal masses of A and B
⇒ \(=\frac{6.022 \times 10^{23} \times W}{M_A}: \frac{6.022 \times 10^{23} \times W}{M_B}=M_B: M_A\)
Thus, the ratio of the number of molecules in equal masses of two different substances Is equal to the inverse ofthe ratio of their molecular masses.
Mole Concept
Mole is a Latin word meaning quantity, heap, or collection. In the SI system, mole (symbol: mol) was introduced as the seventh base quantity for the amount of a substance.
Mole Concept Definition: A mole is defined as the number of particles (like atoms, molecules, ions, or radicals) that is exactly equal to the number of 12C atoms present in 0.012kg of carbon.
Mole Concept Explanation: 0.012kg of 12C atoms contains Avogadro’s number of carbon atoms. So, 1 mol of any substance i.e., molecule, atom, ion, or radical contains Avogadro’s number of the specified species.
Irrespective of the ultimate particles, one mole always contains the same number of constituent particles i.e., 6.022 x 1023.
Number of molecules in 1 mol of molecule 1
= Number of atoms in 1 mol of the atom
= Number of mol of ion
= Number ofradicalsin1 mol of radical
= 6.022 X 1023
Mole Concept Discussion: While using the word ‘mole’, the name and nature (i.e., molecule, atom, ion, or radical) should be mentioned.
For example, the number of molecules in 1 mol of an oxygen molecule is equal to the number of atoms present in 1 mol of oxygen atom and in both cases, this number is 6.022 x 1023.
But oxygen is diatomic and hence the number of oxygen atoms in a 1 mol oxygen molecule is double the number of oxygen atoms in a 1 mol atom of oxygen.
So, the statement “1 mol oxygen” may lead to unnecessary confusion because it indicates both 1 mol of oxygen molecule and 1 mol of oxygen atom. But quantitatively they are altogether different although the number of ultimate particles in both cases is the same.
Often, quantity of substance are expressed in decimole, centimole or millimole where, 1 decimol = 10-1 mol, 1 centimol = 10-2 mol,1 millimol = 10~3 mol.
‘ Mole’ is also used in the case of electrons e.g., 1 mol electron = 6.022 X 1023 electrons and 1 millimol electron =6.022 X 1020 electrons.
Useful relations used in the mole-related calculation
Relation between ‘1 mol’ molecule and gram-mole: Mol molecule of any substance (element or compound) denotes 6.022 x 1023 molecules of that substance.
Again, the mass of 6.022 x 1023 molecules = 1 gram molecular mass or 1 gram-mole.
Hence, 1 mol molecule of an element or compound — 1 gram-mole of that substance.
Mole Concept Examples:
l mol nitrogen molecule = 6.022 x 102J nitrogen molecules= 28 g nitrogen (=1 gram-mole of nitrogen)
1 mol carbon dioxide molecule = 6.022 x 1 023 carbon dioxide molecules= 44 g carbon dioxide (= 1 gram-mole carbon dioxide)
1 million oxygen molecule = 10~3 mol oxygen molecule = 10-3 X 6.022 x 1023 oxygen molecules = 10~3 X 32 g oxygen = 3.2 x 10-2 g of oxygen
1 mol sodium chloride = 6.022 X 1023 formula units of sodium chloride
Relation between ‘1 mol’ atom And gram-atomic mass or gram-atom: 1 mol of an element denotes = 6.022 x 1023 atoms.
Again, a mass of 6.022 x 1023 atoms =1 gram-atomic mass (or, 1 gram-atom) So,1 mol atom of an element = 1 gram-atom of that element.
Mole Concept Examples:
- l mol oxygen atom = 6.022 x 1023 oxygen atoms =16 g oxygen (=1 gram-atom ofoxygen)
- 1 mol sodium atom = 6.022 x 1023 sodium atoms =23 g sodium(=1 gram-atom of sodium)
- Relation between ‘1 mol’ ion and gram-ion: 1 mol ion indicates 6.022 x 1023 ions.
- Again, number ofionsin1 gram-ion = 6.022 x 1023. 1 million =1 gram-ion (gram-formula mass of)
Examples:
l mol Cl- ion = 6.022 x 1023 Cl- ions
= 35.5 g Cl- ions (=1 gram-ion of Cl-) (v mass of an electron is negligible)
1 mol \(\mathrm{SO}_4^{2-}\) ion = 6.022 x 1023 \(\mathrm{SO}_4^{2-}\) ions = 96 g
\(\mathrm{SO}_4^{2-}\) ions (=1 gram-ion of \(\mathrm{SO}_4^{2-}\) )
1 mol Al3+ ion = 6.022 x 1023 Al3+ ions =27 g Al3+ ions (=1 gram-ion of Al3+)
The volume occupied by the ‘1 mol’ molecule of a gas at STP: For any substance, 1 mol molecule stands for 6.022 x 1023 number of molecules. Moreover, 22.4 L of any gas at STP contains 6.022 x 1023 molecules.
Hence, the volume occupied by 1 mol molecule of any gaseous substance at STP = 22.4 L.
Examples: 1 mol oxygen molecule = 6.022 x 1023 oxygen molecules =22.4L of oxygen at STP
1 mol carbon dioxide molecule= 6.022 x 1023 carbon dioxide molecules =22.4 L ofcarbon dioxide at STP.
Various relationships regarding the mole concept

Number of electrons in terms of mol: 6.022 x 1023 number of electrons are present in 1 mol of the electron. The charge carried by 1 mol electron is 96500 coulomb or faraday.
Calculation of mole number
In the case of monatomic elements: 23 g sodium (1 gram atom sodium) = 1 mol sodium
W g sodium = mol\(=\frac{W}{23}\) sodium
Mole number monatomic element
⇒ \(=\frac{\text { mass of the element }(\mathrm{g})}{\text { gram-atomic mass of the element }}\)
In the case of ions: 35.5 g (1 gram-ion) of Cl =1 mol Cl.
Wg Cl- ions\(=\frac{W}{35.5}\) mol CP ions.
Similarly, 96 g (1 gram-ion) of \(\mathrm{SO}_4^{2-}\) ions = 1 mol \(\mathrm{SO}_4^{2-}\)
W g \(\mathrm{SO}_4^{2-}\) ions \(=\frac{W}{96}\) mole \(\mathrm{SO}_4^{2-}\) Ions
Mole number of ion = \(=\frac{\text { mass of ion (g) }}{\text { mass of } 1 \text { gram-ion }}\)
2. In case of any gas: 22.4 L of any gas at STP =1 mol of gaseous molecules.
VL of any gas at STP =\(=\frac{V}{22.4}\) mol of gaseous molecules.
In case of any gas: 22.4 L of any gas at STP =1 mol of
gaseous molecules.
VL of any gas at STP = mol of gaseous molecules.
Mole number of gaseous molecule
⇒ \(=\frac{\text { volume of the gas at STP (L) }}{22.4 \mathrm{~L}}\)
Calculation of mass of substance from mole number
Mass of monoatomic element = mole number of molecule or atom of the element X gram-atomic mass of the element.
Mole number of gaseous molecules
⇒ \(=\frac{\text { volume of the gas at STP }(\mathrm{L})}{22.4 \mathrm{~L}}\)
Calculation of mass of substance from mole number
1. Mass of monoatomic element = mole number of molecule or atom of the element X gram-atomic mass of the element.
For example, 0.5 mol of Na = ( 0.5 x 23) = 11.5 g Na
2. Mass of polyatomic substance (element or compound) = mole number of the substance X gram-molecular mass of that substance
For example, 0.5 mol H2SO4 =(0.5 x 98) = 49g H2SO4
Mass of ions = mole number of Ions x mass of 1 gram-ion or gram-ionic mass
For example, 0.5 mol SO2-4 =(0.5 x 96) =48 gSO2- ion
Determination of the volume of a gas from mole number:
The volume of any gas at STP (L) = a number of moles of gaseous molecules x 22.4 L.
Determination of number of particles (molecules, atoms, or ions): Number of particles (molecule, atom, or ion) = mole number x 6.022 x 1023.
Advantages of the mole concept
Chemical calculations can be worked out in a much simpler way by using mole numbers instead of the masses or volumes of the reactants and products. To be more specific, (2 x 2.016) g of 112 react with 32 g of O2 to form (2 x 18.016) g of water (H2O). If the reaction is expressed in terms of mole, then we will say that 2 mol of hydrogen molecules combine with 1 mol of oxygen molecules to produce 2 mol of water molecules.
⇒ \(2 \mathrm{H}_2+\mathrm{O}_2 \rightarrow 2 \mathrm{H}_2 \mathrm{O}\)
Valuable Information like the number of molecules, atoms, or ions present In a certain amount of any substance is represented by the number of moles.
The word ‘mole’ finds extensive use in expressing the quantity of molecules, ions, or other tiny particles present in the solution.
If the mole number of molecules of a gaseous substance is known, then its volume at STP can be determined.
Numerical Examples
Question 1. If we spend 10 lakh rupees per second then how much time will be required to spend an amount of money which is equal to Avogardo’s number?
Answer: 10000000 rupee will be spent in 1 sec
6.022x 1023 rupees will be spend in ,\(\frac{1 \times 6.022 \times 10^{23}}{10^6} \text { sec. }\)
⇒ \(=\frac{6.022 \times 10^{17}}{60 \times 60 \times 24 \times 365} \text { years }=1.91 \times 10^{10} \text { years }\) years = 1.91×10 10 years
Question 2. Find the weight of 12.046 X 1025 number of ammonia molecules.
Answer: 1 gram-mole ammonia = 17 g of ammonia.
∴ Number of molecules contained in 1 gram-mole of ammonia = 6.022 X 1023.
Hence, 6.022 x 1023 molecules weigh 17 g
∴ 12.046 X 1025 molecules weight
⇒ \(=\frac{17 \times 12.046 \times 10^{25}}{6.022 \times 10^{23}}=3400 \mathrm{~g}=3.4 \mathrm{~kg}\)
Question 3. What is the quantity of charge carried by 1 mol of electron?
Answer: 1 mol electron =6.022×1023 number of electrons
Charge carried by 1 electron =1.6x 10-19 coulomb
The total charge carried by 1-mole electron
=(6.022x 1023×1.6×10-19)= 96352 coulomb.
Question 4. Calculate the number of molecules left when 1021 molecules of CO2 are removed from 200 mg of CO2.
Answer: 200mg of CO2 =0.2 g of (Gram – molecular mass of CO2=44g). 44g CO2 Contains 6.022×1023 molecules.
∴ 0.2g CO2 Contains \(\begin{aligned}
& =\frac{6.022 \times 10^{23} \times 0.2}{44} \text { molecules } \\
& =2.7372 \times 10^{21} \text { molecules }
\end{aligned}\)
On removing 1021 molecules, number of C02 molecules remaining = 2.7372 X 1021 – 1 x 1021 = 1.7372 X 1021
Question 5. Find the number of atoms of hydrogen and oxygen present in one spherical drop of water with radius I mm at 4°C.
Answer:
The volume of one spherical drop of water
⇒ \(=\frac{4}{3} \times \pi \times(0.1)^3=\frac{4}{3} \times \frac{22}{7} \times 10^{-3}=4.19 \times 10^{-3} \mathrm{~cm}^3\)
Further, the density of water at 4°C = lcm-3
Mass of 4.19 x 10-3cm3 water at 4°C =4.19 x 10~3 g.
Now, the mass of 1 gram-molecule of water = 18 g.
∴ Number of molecules in 18g water =6.022 x 1023
∴ Number of molecules present in 4.19 x 10-3 g of water
⇒ \(=\frac{6.022 \times 10^{23} \times 4.19 \times 10^{-3}}{18}=1.4017 \times 10^{20}\)
1 molecule of water contains 2 hydrogen and 1 oxygen atom.
∴ The number of hydrogen atoms in 1.4017 x 1020
∴ Molecules of water = 2 x 1.4017 x 1020 = 2.803 x 1020
So, the number of oxygen atoms present in 1.4017 x 1020
molecules of water = 1.4017 x 1020
Question 6. Find the number of electrons in a drop of sulphuric acid weighing 4.9 x 10-3 mg [assume it to be cent percent pure].
Answer: 4.9 X 10-3 mg = 4.9 X 10-6 g Molecular mass of H2S04 = 98 Its gram-molecular mass = 98 g. Number of molecules in 98 g of H2S04 = 6.022 x 1023 So, number of molecules in 4.9 x 10-6 g of H2S04
⇒ \(=\frac{6.022 \times 10^{23} \times 4.9 \times 10^{-6}}{98}=3.011 \times 10^{16}\)
Atomic numbers of H, S, and O are 1, 16, and 8 respectively. So, the number of electrons in them is 1, 16, and 8.
Total number of electrons present in 1 molecule of H2SO4 =(2×1=1×16=4×8)=50
3.011 x 1016 number of H2S04 molecules contain = 3.011 X 1016 x 50 = 1.5055 X 1018 electrons.
Thus, 4.9 x 10-3 mg of H2S04 has a 1.5055 X 1018 number of electrons.
Question 7. Find the number of 0 atoms and 0 molecules present in 1 g of oxygen.
Answer: 1 g oxygen= gram-mole ofoxygen [v = 32 ]
Number of moleculesin1 g ofoxygen = 6.022 x 1023 x — = 1.882 x 1022
As oxygen molecule is diatomic, the number of atoms present
in 1g of oxygen = 2 X 1.882 X 1022 = 3.764 X 1022
Question 8. Calculate the number of O-atoms present in 112 L of COz gas at STP.
Answer: At STP, the number ofmoleculespresentin 22.4 L of C02 gas
= 6.022 x l023
Number of molecules present in 112L of C02 gas =112. x 6.022 X 1023 = 5 X 6.022 X 1023 22.4
Now, each molecule of C02 contains 2 atoms of oxygen.
Number ofO-atoms presenting 112L of C02 gas (at STP)
= 2 X 5 X 6.022 X 1023 = 6.022 X 1024
Question 9. Find the number of neutrons present in 5 x 104 mol of 14 C Isotope.
Answer: Atomic number of carbon = 6
Number of neutrons in one C atom =14-6 = 8.
Now, in 1 mol of 14C, the number of atoms = 6.022 x 1023
In 5 x 10 4 mol of C, the number of atoms
= (6.022 X 1023 x 5 X 10-4) = 3.011 X 1020 14
Again, one C atom contains 8 neutrons.
3.011 x 1020 number of atoms of 14C contain
= (8 x 3.011 x 1020) =2.4088 x 1021 neutrons.
Number of neutrons in 5 x 10-4 mol of 14C =2.4088 x 1021.
Question 10. Find the number of hydrogen and oxygen atoms present in 0.09 g of water.
Answer: -09 g water =\(=\frac{0.09}{18}\) =5×10 3 gram-mole ofwater [ Molecular mass ofwater = 18
Number of molecules in 1 gram-mole water = 6.022 x 1023
Number of molecules in 5 x 10-3 gram-mole water
⇒ \(=6.022 \times 10^{23} \times 5 \times 10^{-3}=3.011 \times 10^{21}\)
The number of hydrogen and oxygen atoms in 1 molecule of water (H2O) are 2 and 1 respectively.
The number of hydrogen atoms in 3.011 x 1021 number
of water molecules = 2 x 3.011 x 1021 =6.022 x 1021
The number ofoxygen atoms =1 x 3.011 x 1021
= 3.011 X 1021
Question 11. What is the mass of 1 millimol of ammonia? Also, find the number of ammonia molecules presenting it.
Answer: 1 millimol = 10-3 mol.
Mass of1 mol of ammonia = 17 g
Mass of 10-3 mol of ammonia = 17 x 10-3g
Again, the number of ammonia molecules present in 1 mol of ammonia = 6.022 x 1023
The number of ammonia molecules in 10-3 mol of ammonia = 6.022 x TO23 x 10-3 = 6.022 x 1020
Hence, the number of ammonia molecules present in millimolar of ammonia =6.022 X 1020
Question 12. What will be the number of
- Moles of ethylene,
- Molecules of ethylene,
- Atoms of carbon and
Atoms of hydrogen in 0.28 g of ethylene contained in a cylinder?
Answer: 0.28 g of ethylene \(=\frac{0.28}{28}\) = 011 gram-mole ethylene [since molecular mass of ethlene= 28]
Quantity of ethylene in the cylinder= 0.01 mol.
1 gram-mole contains 6.022 x 1023 ethylene molecules.
1 gram-mole contains 6.022 x 1023 ethylene molecules.
1 gram-mole contains 6.022 x 1023 ethylene molecules.
1 gram-mole contains 6.022 x 1023 ethylene molecules.
2 x 6 022 X 1°21 carbon atoms
So, the number of carbon atoms present in the cylinder
⇒ \(=2 \times 6.022 \times 10^{21}=1.2044 \times 10^{22}\)
1 molecule of ethylene contains 4 atoms of hydrogen.
Number of hydrogen atoms in 6.022 x 1021 ethylene molecules = 4 x 6.022 x 1021 =2.4088 X 1022 Thus, the number of hydrogen atoms present in the cylinder = 2.4088 x 1022.
Question 13. Find the number of moles and quantifying gram, contained in 100 m of ammonia at STP.
Answer: At STP, the volume of gram-mole of ammonia = 22.4L
∴ Number of the in 100 of ammonia \(=\frac{1 \times 100}{22.4}=4.464\)
Now 1 gram-mole of ammonia = 17 g of ammonia [since the molecular mass of ammonia = 17 ]
∴ 4.464 gram moles of ammonia = (17 x 4.464) =75.888g of ammonia
Thus, 100 l of ammonia at STP contained = 75.888g
Question 14. Suppose the human population of the world is 3 X 1010. If 100 molecules of sugar are distributed per head, what is the total quantity of sugar required for distribution?
Answer: Gram-molecular mass of sugar
=(12×12+22×1+16×11)=(144+22+176)= 342g
Number of molecules in 342 g of sugar =6.022×1023
∴ Mass of 100 molecules of sugar \(\begin{aligned}
& =\frac{342 \times 100}{6.022 \times 10^{23}} \\
& =5.679 \times 10^{-20} \mathrm{~g}
\end{aligned}\)
So, 5.679 x 10-20 g of sugar will be required per head.
For 3 x 1010 number of people, the quantity of sugar
required =5.679 x 10-20 X 3 x 1010 = 1.7037 X 10-9 g.
Question 15. Find the number of atoms of nitrogen in 1 grantmole of NO and 0.5 gram-mole of N02. Which one will be heavier—1 gram-mole of NO or 0.5 gram-mole of N02?
Answer: Number of molecules present 1 gram-mole of NO = 6.022 X 1023.
Each molecule of NO contains a nitrogen atom.
therefore 6.022×1023 No molecules will contain 6.022×1023 atoms of nitrogen.
Again, the number of N02 molecules present in 0.5 gram-mole of N02 =\(=\frac{1}{2} \times 6.022 \times 10^{23}\) x 6.022 x 1023 =3.011 X 1023
Again, 1 gram-mole NO2= 30g of NO [∴ MNO = 30]
∴ 0.5 gram-mole of NO2 = 0.5×46 =23g NO2 [since M NO2=46]
So, 1 gram- mole NO2 is heavier than 0.5 Gram mole NO2.
Question 16. A mixture contains 02 and N2 in the proportion of 1: 4 by weight. What will be the ratio of the number of molecules of 02 and N2 in the mixture?
Answer:
Given
A mixture contains 02 and N2 in the proportion of 1: 4 by weight.
Let, the mass ofthe mixture be W g
In the mixture, mass of 02 =\(\frac{W}{5} \mathrm{~g}\) and mass of N2 \(=\frac{4 W}{5} \mathrm{~g}\)
Number of gram-mole \(\mathrm{N}_2=\frac{4 W / 5}{28}=\frac{4 W}{140}\)
So, number of gram-mole of \(=\frac{4 W / 5}{28}=\frac{4 W}{140}\)
So, number of 002 molecules = \(=\frac{W}{160} \times 6.022 \times 10^{23}\)
number of N2 molecules = x 6.022 x 1023
The ratio of the number of molecules of 02 to the number of molecules of N2 in the mixture
⇒ \(=\frac{W \times 6.022 \times 10^{23}}{160}: \frac{4 W \times 6.022 \times 10^{23}}{140}\)
⇒ \(=\frac{1}{160}: \frac{4}{140}=14: 64=7: 32 \text {. }\)
Common Questions on Laws of Chemical Combination
Question 17. A young man has given ills bride a tin engagement ring containing a 0.50-carat diamond. How many atoms of carbon are present in that ring? |1 carat = 200 mg).
Answer:
Given
A young man has given ills bride a tin engagement ring containing a 0.50-carat diamond.
1 carat = 200 mg = 0.2 g. So,0.5 carat = 0.5 x 0.2 = 0.1 g Diamond is composed of only carbon atoms
i.e., 0.1 g diamond = 0.1 g carbon
Number of carbon atoms in 12 g of carbon = 3.022 x 1023
[ since Atomic mass of carbon = 12 ]
Number of carbon atoms in 0.1 g of carbon
⇒ \(=\frac{6.022 \times 10^{23}}{12} \times 0.1=5.018 \times 10^{21}\)
Therefore, the young man gave his bride-to-be 5.0018×1021 atoms of carbon
Question 18. What is the number of O-atoms present in 44.8 L of Ozone gas at STP?
Answer:
At STP, the number of molecules present in 22.4 L of ozone gas = 6.022 X 1023
Number of molecules present in 44.8 L of ozone gas at
⇒ \(\mathrm{STP}=\frac{6.022 \times 10^{23} \times 44.8}{22.4}=2 \times 6.022 \times 10^{23}\)
∴ Number of oxygen atoms present in the given volume of ozone gas = 3 X 2 X 6.022 X 1023 = 3.6132 X 1024
[Since each molecule of ozone contains 3 O-atoms]
Applications Of Avogadro’s Hypothesis
The following important corollaries which are of great significance in chemistry have been established by the application of Avogadro’s hypothesis:
Molecules complementary gases (e.g., hydrogen, oxygen, etc.) except inert gases are diatomic.
The molecular mass of a gaseous substance (element or compound) is twice its vapor density.
The gram-molecular volume of any gaseous substance (element or compound) at STP is 22.4L.
The molecular formula of any gaseous compound can be determined from its volumetric composition.
The atomic masses of elements can be determined from the value of their vapor density.
All elementary gases are diatomic except inert gases
1. Hydrogen and chlorine molecules are diatomic: From experiments, it is observed that under the same conditions of temperature and pressure, 1 volume of hydrogen combines chemically with 1 volume of chlorine to form 2 volume of hydrogen chloride gas.
∴ 1 volume of hydrogen + 1 volume ofchlorine = 2 volumes of hydrogen chloride Let, ‘n’ be the number of hydrogen molecules in 1 volume of hydrogen gas under the experimental condition.
According to Avogadro’s hypothesis, at the same temperature and pressure, 1 volume of chlorine and 2 volumes of hydrogen chloride will contain ‘n’ molecules of chlorine and ‘2n’ molecules of hydrogen chloride respectively.
Therefore, ‘n ‘ molecules of hydrogen +’n’ molecules ofchlorine = 2n molecules of hydrogen chloride. or, 1 molecule of hydrogen +1 molecule ofchlorine = 2 molecules of hydrogen chloride.
molecule ofhydrogen +i molecule of chlorine =1 molecule ofhydrogen chloride.
Hence, 1/2 molecule of hydrogen and 1/2 molecule of chlorine are present molecules of hydrogen chloride.
We know, a molecule of hydrogen chloride is composed of hydrogen and chlorine atoms only.
Therefore, 1 molecule of hydrogen chloride must contain at least 1 atom of hydrogen and 1 atom of chlorine because the atom is indivisible.
Thus 1 atom of hydrogen and 1 atom of chlorine must have come from 1/2 molecule of hydrogen and 1/2 molecule of chlorine respectively i.e., 1/2 molecule of hydrogen and 1/2 molecule of chlorine contain one hydrogen atom and one chlorine atom respectively.
So, two atoms of hydrogen and two atoms of chlorine are present in hydrogen and chlorine molecules respectively i.e., hydrogen and chlorine molecules are diatomic.
2. Nitrogen molecule is diatomic: Actual experiments show that under the same conditions of temperature and pressure, 3 volumes of hydrogen and 1 volume of nitrogen combine chemically to produce 2 volumes of ammonia.
1 vol. of nitrogen + 3 vol. hydrogen = 2 vol. of ammonia If under the experimental conditions of temperature and pressure 1 volume of nitrogen contains ‘n’ nitrogen molecules, then according to Avogadro’s hypothesis, at the same temperature and pressure 3 volumes of hydrogen and 2 volume of ammonia will also contain ‘3n’ and ‘2n’ molecules of hydrogen and ammonia respectively.
‘n’ molecules ofnitrogen +’ 3n’ molecules ofhydrogen = ‘2n’ molecules ofammonia
i.e.,1 molecule ofnitrogen + 3 molecules ofhydrogen = 2 molecules of ammonia
or, 1/2 molecule ofnitrogen + 3/2 molecules ofhydrogen =1 molecule ofammonia.
Now, at least 1 atom of nitrogen must be present in 1 molecule of ammonia and this atom of nitrogen must come from 1/2 molecule of nitrogen.
So, in a molecule of nitrogen, the number of nitrogen atoms must be at least 2 i.e., the nitrogen molecule is diatomic.
3. Oxygen molecule is diatomic: From actual experiments, it has been found that under identical conditions of temperature and pressure, 2 volumes of hydrogen react with 1 volume ofoxygen to give 2 volumes of steam.
2 vol. of hydrogen +1 vol. ofoxygen = 2 vol. of steam If at the experimental conditions of temperature and pressure, 1 volume of oxygen gas contain ‘n ‘ molecules, then according to Avogadro’s hypothesis, at that temperature and pressure 2 volume of each of hydrogen and steam must contain’ 2n’ molecules each. Therefore, ‘ 2n’ molecules of hydrogen +’n’ molecules of oxygen
= ‘2n’ molecules of steam or, 2 molecules of hydrogen +1 molecule of oxygen
= 2 molecules of steam.
According to Dalton’s atomic theory, the atom is indivisible. Therefore, at least one atom of oxygen must be present in one molecule of steam and this atom of oxygen must come from 1/2 molecule of oxygen,
So, the number of oxygen atoms present in one molecule of oxygen is two. Hence, oxygen molecules are diatomic.
The molecular Mass Of A Gas Is Twice Its Vapour Density
Absolute density of a gas: The mass (in grams) of L of a gas at a certain temperature and pressure is called its absolute density at that temperature and pressure. Absolute density changes with temperature and pressure.
The normal density of a gas: The normal density of a gas may be defined as the mass (in grams) of 1 L of the gas at STP; e.g., the normal density of hydrogen is 0.089 g- L-1.
Relative density or vapor density of a gas: The vapor density of a gas is a relative value that shows how many times it is heavier than the equal volume ofthe lightest gas i.e., hydrogen under similar conditions of temperature and pressure. Its value is independent of temperature and pressure.
Relative density or vapor density of a gas Definition: Vapour density or relative density of a gaseous substance is defined as the ratio of the mass of a certain volume of the gas to the mass of the same volume of hydrogen, measured under the same conditions of temperature and pressure.
Relation between normal density & relative density of a gas: For any gas, relative density of a gas: for any gas, relative density
⇒ \(\begin{aligned}
& =\frac{\text { mass of } V \text { volume of the gas at STP }}{\text { mass of } V \text { volume of } \mathrm{H}_2 \text { gas at STP }} \\
& =\frac{\text { mass of } 1 \mathrm{~L} \text { of the gas at STP }}{\text { mass of } 1 \mathrm{~L} \text { of } \mathrm{H}_2 \text { gas at STP }} \\
& =\frac{\text { normal density of the gas }}{\text { normal density of } \mathrm{H}_2 \text { gas }}=\frac{\text { normal density of the gas }}{0.089} \\
& \quad\left[\text { Normal density of } \mathrm{H}_2 \text { gas }=0.089 \mathrm{~g} \cdot \mathrm{L}^{-1}\right]
\end{aligned}\)
Thus for any gas, normal density = 0.089 x relative density
Relation between molecular mass and vapor density (D):
Vapour density of a gas \(=\frac{\text { mass of } V \text { volume of a gas }}{\text { mass of } V \text { of } \mathrm{H}_2 \text { gas }}\)
[At same temperature and pressure]
According to Avogadro’s hypothesis, under the conditions of temperature and pressure, equal volumes of all gases contain the same number of molecules.
So, If the V volume of a gas under certain conditions of temperature and pressure contains ‘n ‘ molecules, then at the same temperature and pressure, the V volume of hydrogen gas will also contain ‘n’ number of molecules.
Vapor density of a gas(D)
⇒ \(\begin{aligned}
& =\frac{\text { mass of ‘ } n \text { ‘ molecules of a gas }}{\text { mass of ‘ } n \text { ‘ molecules of hydrogen }} \\
& =\frac{\text { mass of } 1 \text { molecule of a gas }}{\text { mass of } 1 \text { molecule of hydrogen }}
\end{aligned}\)
⇒ \(\begin{aligned}
& =\frac{\text { mass of } 1 \text { molecule of a gas }}{\text { mass of } 2 \text { atoms of hydrogen }}[\text { hydrogen is diatomic }] \\
& =\frac{1}{2} \times \frac{\text { mass of } 1 \text { molecule of a gas }}{\text { mass of } 1 \text { atom of hydrogen }}
\end{aligned}\)
Now by definition, the molecular mass (M) of a substance
⇒ \(=\frac{\text { mass of } 1 \text { molecule of the substance }}{\text { mass of } 1 \text { atom of hydrogen }}\)
Therefore, vapour density of a gas (D) = \(=\frac{1}{2} x\)
its molecular mass (M) i.e., D= M/2 Or, M=2D
So, the molecular mass of any gas is twice its vapor density. [If the atomic mass of hydrogen is taken as 1.008, molecular mass (M)’= 2.016 x vapor density (D) ]
Effect of temperature and pressure on the vapor density of a gas: The absolute density of a gas depends on both temperature and pressure.
This is because even though the mass of gas is kept fixed, the volume changes with variations in temperature and pressure.
But the vapor density of a gas is independent of temperature and pressure because it is a ratio ofmass of a certain volume of gas to the mass of the same volume of hydrogen at a fixed temperature and pressure.
Thus, vapor density is a mere number. Temperature or pressure has no effect on it.
Vapor density of a gas with respect to another gas: Let MA and MB be the molecular masses of the gases A and B respectively. So, the vapor density of gas A with respect to gas B, t(DA)B] is given by
⇒ \(\left(D_A\right)_B=\frac{M_A}{M_B}\)
Examples: Vapour density of 02 with respect to N2
⇒ \(=\frac{\text { molecular mass of } \mathrm{O}_2}{\text { molecular mass of } \mathrm{N}_2}=\frac{32}{28}=1.14\)
Vapor density of NH3 with respect to air
⇒ \(=\frac{\text { molecular mass of } \mathrm{NH}_3}{\text { average molecular mass of air }}=\frac{17}{29}=0.59\)
The gram-molecular volume of any (JASOOIIS substance (element or compound) at STP Is 22.4 L
With the help of Avogadro’s hypothesis, It can be deduced that the molar volume of any gaseous substance at STP Is 22.4 L
Vapour density (D) of any gas \(=\frac{\text { mass of } V \text { volume of the gas }}{\text { mass of } V \text { volume of } \mathrm{H}_2 \text { gas }}\)
If the gas is kept at STP, then the vapor density (D) of the gas
⇒ \(\begin{aligned}
& =\frac{\text { mass of } V \text { volume of the gas at STP }}{\text { mass of } V \text { volume of } \mathrm{H}_2 \text { gas at STP }} \\
& =\frac{\text { mass of } 1 \mathrm{~L} \text { gas at STP }}{\text { mass of } 1 \mathrm{~L} \mathrm{H}_2 \text { gas at STP }}=\frac{\text { mass of } 1 \mathrm{~L} \text { gas at STP }}{0.089 \mathrm{~g}} \\
& \qquad\text { Mass of } 1 \mathrm{~L} \text { of } \mathrm{H}_2 \text { gas at STP }=0.089 \mathrm{gl}
\end{aligned}\)
Mass of L of the gas at STP =D x 0.009 g
According to Avogadro’s hypothesis, D= M/2
[where, M = molecular mass of the gas]
Mass of1 L ofthe gas at STP = \(=\frac{M \times 0.089}{2} \mathrm{~g}\)
Or, Volume occupied by \(\frac{M \times 0.089}{2}\)g of the gas at STP =1l
Volume occupied by M gram gas at STP = \(=\frac{2}{0.089}\)= 22.4 L
The exact atomic mass of hydrogen on a 12 C -scale is 1.008.
Thus, the true molecular mass of hydrogen is 2.016.
For an accurate result, the correct value of the normal density of hydrogen i.e., 0.09 g-L-1 is to be considered.
Therefore, the volume occupied by M gram of the gas at
g of the gas at STP \(=\frac{2.016}{0.09} \mathrm{~L}=22.4 \mathrm{~L}.\)
So,1 gram-mole ofthe gas occupies 22.4L at STP.
Thus, it is proved that the molar volume of any gaseous substance at stp =22.4 l.
The molar volume of all gases Is same under the identical Conditions of temperature and pressure: On the basis of
Avogadro’s hypothesis, this important corollary can established in the following way:
Let, the actual mass of 1 atom of hydrogen = x g.
Mass of1 hydrogen molecule = 2x g [hydrogen is diatomic] Now, 1 gram-mole hydrogen =2 g of hydrogen
Number of hydrogen molecules in 1 gram-mole (or 2g) of hydrogen \(=\frac{2}{2 x}=\frac{1}{x}\)
The molecular mass of oxygen = 32
So, the actual mass of ] molecule of oxygen = 32 x actual
mass of1 atom ofhydrogen = (32 X x)g
Now, I gram-mole oxygen 32 % oxygen
Number of oxygen molecule* in J gram-mole for 32 g oxygen = \(=\frac{32}{32 x}=\frac{1}{x}\)
let, the molecular: m of any gaseous substance = M
Therefore, the I molecule of the above gas is A/ times heavier than the I atom of hydrogen.
So, the actual mass of the molecule of the gas
= M x actual mass of hydrogen atom =(M x x)g
Now, I gram-mole of the gas =M g
Number of molecules in 1 gram- mole of the gaseous substance = \(=\frac{M}{M x}=\frac{1}{x}\)
Thus, it shows that 1 gram-mole of any gaseous substance contains the same number of molecules.
Again according to Avogadro’s law, at the same temperature and pressure, the volume occupied by this same number of molecules Is the same.
Hence, under identical conditions of temperature and pressure, the volume occupied by 1 gram-mole of all gaseous substances. i.e., the molar volume of all gases is the same.
Volume ovmple At STP,l gram-mole of any gas occupies a volume of 22.4L Again, 1 gram-mole of any gas contains 6.022 x 1023 molecules.
1 molecule occupies the voulume of \(\frac{22.4}{5.022 \times 10^{23}} \mathrm{~L}\)
1 molecule occupies the volume of \(\frac{22.4}{6.022 \times 10^{23}} \mathrm{~L}\) = 3.719 x 10-23 l at STP.
Determination of the molecular formula of any gaseous compound from its volumetric composition
If the volumetric compositions ofthe constituent elements and the vaPour density of the gaseous compound are known, the the molecular formula of the compound can easily be determined by the application of Avogadro’s hypothesis.
Determination of molecular formula of hydrogen chloride: From experiments, it is found that at a certain temperature and pressure, 1 volume of hydrogen combines with 1 volume of chlorine to form 2 volumes of hydrogen chloride.
If under the experimental conditions of temperature and pressure, ‘n ‘ molecules of hydrogen are present in its I volume, then according to Avogadro’s law, under the same conditions of temperature and pressure, ‘n ‘ molecules ofchlorine and ‘2n’ molecules of hydrogen chloride will be present in 1 volume of chlorine and 2 volumes of hydrogen chloride respectively.
‘ n ‘ molecules ofhydrogen+’n’ molecules ofchlorine
= ‘2n’ molecules of hydrogen chloride
or, 1 molecule of hydrogen + 1 molecule ofchlorine
= 2 molecules of hydrogen chloride
or, I molecule of hydrogen + 1/2 molecule ofchlorine
= 1 molecule of hydrogen chloride
According to Avogadro’s law, both hydrogen and chlorine are diatomic.
atom of hydrogen +1 atom of chlorine
=1 molecule of hydrogen chloride
The molecular formula of hydrogen chloride =HC1.
The molecular mass, determined from the deduced formula =1 + 35.5 = 36.5
[ V Atomic masses of hydrogen and chlorine are 1 and 35.5 respectively]
Again, the vapor density of hydrogen chloride = 18.25
Thus, the molecular mass of hydrogen chloride = 2X18.25 =36.5
So, the molecular mass, determined from the deduced formula of hydrogen chloride is exactly the same as calculated from the measured vapor density of hydrogen chloride.
Therefore, it is proved that the molecular formula of hydrogen chloride is HCl.
2. Determination of molecular formula of carbon dioxide: Carbon dioxide is made up of carbon and oxygen.
From experiments, it is found that under similar conditions of temperature and pressure, a certain volume of carbon dioxide dissociates to give an equal volume of oxygen.
So, the volume of carbon dioxide contains the volume of oxygen.
If at a certain temperature and pressure, 1 volume of carbon dioxide contains an ‘n’ number of molecules, then according to Avogadro’s law, at that temperature and pressure, 1 volume of oxygen also contains an ‘n’ number of molecules.
So, 1 molecule of carbon dioxide contains 1 molecule of oxygen or 2 oxygen atoms (as oxygen is diatomic).
Let, the molecular formula of carbon dioxide be Cx02 where x stands for the number of carbon atom(s) present in 1 molecule of carbon dioxide.
Now, Molecular mass of CxO2 =12X x+16X2 =(12x+32)
[since atomic masses of and O are 12 and 16 respectively]
Further, the vapor density of earphone dioxide is 22, and hence its molecular mass = 2 x 22 = 44 Therefore, 12x + 32 = 44 or, x =
The molecular formula of carbon dioxide is CO2
3. Determination of molecular formula of ammonia:
Experimental observations show that under similar conditions of temperature and pressure, 1 volume of nitrogen and 3 volumes of hydrogen combine chemically to produce 2 volumes of measure, the number of molecules present in 3 volummonia.
Let, under experimental conditions of temperature and pressure, ‘n’ be the number of molecules present In I volume of nitrogen.
Then according to Avogadro’s law, under the same conditions of temperature and pressure of hydrogen, 2 volumes of ammonia will be 3n and 2n respectively.
‘n’ molecules of nitrogen + ‘3n ‘ molecules of hydrogen.
= ‘2n’ molecules of ammonia
i.e.,1/2 molecule of nitrogen +3/2 molecules of hydrogen
=1 molecule of ammonia
or, 1 atom of nitrogen + 3 atoms of hydrogen = 1 molecule of ammonia [v hydrogen and nitrogen are diatomic So, 1 molecule of ammonia is composed of 1 atom of nitrogen and 3 atoms of hydrogen.
The molecular formula of ammonia =NH3
The molecular mass calculated from the deduced formula = 14 +(1×3) = 17
[ since Atomic masses of and H are 14 and 1 respectively]
Again, the vapor density of ammonia = 8.5
therefore Molecular mass of ammonia = 8.5 x 2 = 17
So, the molecular mass determined from the deduced formula of ammonia is exactly the same as the molecular mass calculated from the value of the vapor density of ammonia.
Hence, it is proved that the molecular formula of ammonia is NH3
Determination of the atomic mass of a gaseous element from its vapor density
If the vapor density of a gaseous element is known then its atomic mass can be estimated with the help of Avogadro’s law.
The molecular mass of a gaseous element = atomic mass of the element x atomicity of its molecule.
The atomic mass of the gaseous element.
⇒ \(\begin{aligned}
& =\frac{\text { molecular mass of gaseous element }(M)}{\text { atomicity }} \\
& =2 \times \frac{\text { vapour density of gaseous element }(D)}{\text { atomicity }}[\quad M=2 D]
\end{aligned}\)
Example: Vapour density and atomicity of oxygen are 16 and 2 respectively.
Therefore Atomic mass of oxygen \(=2 \times \frac{16}{2}=16\)
Importance of Avogadro’s hypothesis
The contributions of Avogadro’s hypothesis towards the development of chemistry are mentioned below:
This hypothesis made a clear distinction between the ultimate particle of matter (atom) and the smallest particle having independent existence (molecule).
This hypothesis modified the drawbacks of Dalton’s atomic theory and proposed the molecular theory.
This theory successfully explained Gay-Lussac’s law of gaseous volumes.
The different corollaries derived from this hypothesis served as important tools for the purpose of chemical calculations.
Determination of the atomic mass of an element, the molecular formula of a gaseous compound, and the expression of a chemical reaction by an equation was made possible with the help of this hypothesis.
Numerical Examples
Question 1. The vapor density of a gaseous element is 5 times that of oxygen. If the element is triatomic, find its atomic mass.
Answer: Vapour density of oxygen \((D)=\frac{M}{2}=\frac{32}{2}=16\)
Therefore Vapour density of the gaseous element = 5×16 =80
Therefore Molecular mass of the gaseous element = 80 x 2 =16
So, atomic mass of the element \(\frac{\text { molecular mass }}{\text { atomicity }}\)
=160/3=53.33
Question 2. 100 mL of ay weighs 0.144 g at STP. What is the vapor density of the gas?
Answer: Mass of 1 00mL of the gas at STP = 0.144 g
therefore 22400mL ofthe gas at STP weighs \(=\frac{0.144 \times 22400}{100}\)
= 32.256 g
Hence, the molecular mass of the gas = 32.256 g
Vapour density of the gas = \(\frac{\text { molecular mass }}{2}\)
⇒ \(=\frac{32.256}{2}=16.128\)
Question 3. The vapor density of sulfur relative to nitrogen gas at STP is 9.143. Determine the molecular formula of sulfur vapor
Answer: If the molecular masses of two gases are M1 and M2 then
the vapor density of the first gas, relative to the second gas [(D1)2]=M1/M2
So, vapor density of sulfur vapor relative to nitrogen gas \(=\frac{\text { molecular mass of sulfur vapor}}{\text { molecular mass of nitrogen gas }}\)
∴ Molecular mass of sulphur vapour = 9.143×28 =256.004
Now, the atomic mass of sulfur = 32
∴ Atomicity of sulphur vapour= \(=\frac{256.004}{32} \approx 8\)
Hence, the molecular formula of sulfur vapor =S8
Question 4. At STP, 250 cm3 of a gas weighs 0.7317 g. If the density of H2 gas at STP is 0.08987 g L”1 then what will be the vapor density of the gas? Determine the molecular mass of the gas.
Answer: Mass of250 cm3 of H2 gas at STP
⇒ \(=\frac{0.08987 \times 250}{1000} \mathrm{~g}=0.0224 \mathrm{~g}\)
[since At STP, the density of H2 gas = 0.08987 g.L-1 1
∴ The vapor density of the gas
⇒ \(=\frac{\text { mass of } 250 \mathrm{~cm}^3 \text { gas at STP }}{\text { mass of } 250 \mathrm{~cm}^3 \mathrm{H}_2 \text { gas at STP }}=\frac{0.7317}{0.0224}=32.66\)
Thus, its molecular mass = 2×32.66 =65.32.
Question 5. Volumes of N2 and 02 in any gas mixture are 80% and 20% respectively. Determine the average vapor density of the gas mixture.
Answer: Vapour density of N2 \(=\frac{M_{\mathrm{N}_2}}{2}=\frac{28}{2}=14 \text { and }\)
Vapour density of 02 \(=\frac{M_{\mathrm{O}_2}}{2}=\frac{32}{2}=16\)
Since the Average vapor density of the gas mixture
⇒ \(\begin{aligned}
& =\frac{80 \times \text { vapour density of } \mathrm{N}_2+20 \times \text { vapour density of } \mathrm{O}_2}{100} \\
& =\frac{80 \times 14+20 \times 16}{100}=14.4
\end{aligned}\)
Question 6. At 26.7°C, the vapor density of a gaseous mixture containing N2O4 and N2O4 is 38.31. Calculate the number of moles of N2O4 in 100g of that mixture.
Answer: Let the amount of N02 in 100 g of the mixture x g
Therefore Amount of N204 = (100 -x) g
Therefore Number of moles ofN02 in mixture \(=\frac{x}{46}\)
And number of moles of N90i( in mixture \(=\frac{(100-x)}{92}\)
since MNO2 = 46 MN2O4=92]
Total number ofmoles ofN02 and N204 in the mixture \(=\frac{x}{46}+\frac{100-x}{92}=\frac{100+x}{92} \cdots(1)\)
Now,molecular mass ofthe mixture = (2 x 38.3) = 76.6
Total number of moles of the mixture \(=\frac{100}{76.6}\)
Now from 1 and 2 \(\frac{100+x}{92}=\frac{100}{76.6} \quad \text { or, } x=20.1\)
So, number of moles of N02 in mixture \(=\frac{20.1}{46}=0.4369\)
Question 7. The vapor density of a gas, relative to air is 1.528. What is the mass of 2L of the gas at 27°C temperature and 750 mm Hg pressure? [Vapour density of air, relative to hydrogen = 14.4.]
Answer: Vapour density of a gas \(=\frac{\text { mass of certain volume of a gas }}{\text { mass of same volume of } \mathrm{H}_2 \text { gas }}\)
Therefore Vapour density of the gas
⇒ \(=\frac{\text { mass of } V \mathrm{~cm}^3 \text { of gas }}{\text { mass of } V \mathrm{~cm}^3 \text { of air }} \times \frac{\text { mass of } V \mathrm{~cm}^3 \text { of air }}{\text { mass of } V \mathrm{~cm}^3 \text { of } \mathrm{H}_2 \text { gas }}\)
[at same temperature and pressure]
=1.528×14.4 =22
Therefore Molecular mass ofthe gas = 2 x vapour density = 44
Again, volume of1 gram-mole of any gas at STP = 22.4 L
So, mass of22.4L of given gas at STP = 44 g
Now, let the volume ofthe gas be V L at STP. i.e.,
P1 = 750 mm; P2 = 760 mm; Vx = 2L; V2 = VL;
Tx =27 °C =(27 + 273) =300 K; T2 = 273 K
⇒ \(\quad \frac{750 \times 2}{300}=\frac{760 \times V}{273} \quad \text { or, } V=\frac{750 \times 2 \times 273}{300 \times 760}=1.796 \mathrm{~L}\)
Now,mass of 22.4 L ofthe gas at STP = 44 g
Therefore Mass of 1.796l of the gas at stp \(=\frac{44 \times 1.796}{22.4}=3.528 \mathrm{~g}\)
Hence, the mass of the gas= 3.258g.
Question 8. Under the same conditions of temperature and pressure, complete combustion of the volume of a gaseous hydrocarbon produces 3 volumes of carbon dioxide and 4 volumes of steam. What is the die formula of die hydrocarbon?
Answer: Volume ofhydrocarbon+ 02 =3 volumes of C02 +4 volumes of H.,0 (steam) [under same conditions of temperature and pressure]
If 1 volume of hydrocarbon contains ‘n’ molecules, then under identical conditions of temperature and pressure 3 volumes of C02 will contain ‘3n’ molecules and 4 volumes of steam will contain ‘4n’ molecules.
Therefore, ‘n’ molecules hydrocarbon = ‘3n’ molecules C02 + ‘4n’ molecules H2O Now, 3 molecules of C02 = 3 atoms of C
[ ∴ 1 atom of C is present in 1 molecule of C02 and 4 molecules of H2O = 8 atoms of H [v 2 atoms of I-I are presentin1 molecule of H2O
So, 1 molecule of hydrocarbon contains 3 atoms of C and 8 atoms of H.
Molecular formula of the hydrogen = C3H8
Question 9. Under the same conditions of temperature and pressure, a gaseous hydrocarbon contains hydrogen which is twice its volume. If the vapor density of that hydrocarbon is 14, then what will be its molecular formula?
Answer: According to a problem, at the same conditions of temperature and pressure, 1 volume of gaseous hydrocarbon contains 2 volumes of hydrogen.
Since n molecules of gaseous hydrocarbon contain 2n molecules of hydrogen or, 1 molecule of gaseous hydrocarbon contains 2 molecules of hydrogen =4 atoms of hydrogen.
(According to Avogadro’s hypothesis)
Therefore Molecular formula hydrocarbon is CXH4
[Where x stands for the number of c-atoms]
The molecular formula of hydrocarbon is CXH4 = 12xx+4
[since the atomic mass of C=12, H=1]
Again, the vapor density of the hydrocarbon = 14
So, molecular formula of the hydrocarbon = 2 x 14 = 28
CxH4 = 14 x 2
therfore, 12x+4=28 or, x=2
The molecular formula hydrocarbon is C2H4.
Question 10. The weight of 350mL of a diatomic gas at 0°C temperature and 2 atm pressure is lg. Calculate the weight of its own atom.
Answer: According to the given condition, the volume of diatomic gas at 0°C and 2 atm pressure = 350 mL.
Let, the volume ofthe gas at 0°C and atm pressure be V mL
Now, according to Boyle’s law, P1 V1 =P2 V2
or, 2 X 350 =1 X V V = 700 mL
i.e, mass of 700mL of the gas at 0°C and 1 atm pressure =1 g
∴ Mass of 22400mL ofthe gas at 0°C and 1 atm pressure
⇒ \(=\frac{22400}{700} \times 1=32 \mathrm{~g}\)
Molar volume of all gases at STP = 22400 mL.
Hence, the gram-molecular mass ofthe given gas = 32 g.
So, the mass of 6.022 x 1023 molecules ofthe given gas= 32 g.
Therefore, the mass of 2 x 6.022 x 1023 atoms ofthe gas= 32 g
[since the gas is diatomic]
Thus, the mass of 1 atom of the given gas
⇒ \(=\frac{32}{2 \times 6.022 \times 10^{23}}=2.656 \times 10^{-23} \mathrm{~g} .\)
Question 11. For complete combustion, 24g of a solid element requires 44.8L of 02 at STP. The gaseous oxide produced in combustion occupies a volume of 4.8 L at STP. What is the molecular mass of the produced gaseous oxide?
Answer: Molar volume of any gas at STP = 22.4L.
∴ 22.4L of 02 at STP =1 gram-molecule of 02 = 32 g 02
∴ Mass of 44.8L of 02 at STP \(=\frac{32}{22.4}\) 44.8 = 64 g 02
Now, 24 g of solid element reacts completely with 64 g of
02 to produce a gaseous oxide.
In this case, the total mass ofthe reactants = (24 + 64) =88 g.
Now According to the law of constant proportion (in the case) an oxide is only formed) or the law of conservation ofmass, the total mass ofthe product will be 88 g.
∴ Mass of 44.8 L ofthe gaseous oxide formed at STP = 88 g.
∴ Mass of22.4L ofthe gaseous oxide at STP = y = 44 g.
Hence, the gram-molecular mass of the gaseous oxide formed = 44 g
∴ The molecular mass ofthe gaseous oxide formed = 44.
Question 12. A sample of hard water contains 20 mg of Ca2+ ions per liter. How many millimoles of Na2C03 would be required to soften the L of the sample? Also, calculate the mass of.Na2C03
Answer: Reaction \(\mathrm{Ca}^{2+}+\mathrm{Na}_2 \mathrm{CO}_3 \rightarrow \mathrm{CaCO}_3+2 \mathrm{Na}^{+}\)
Number of moles ofNa2C03 molecules
= Number of moles of Ca2+ ion \(=\frac{20 \times 10^{-3}}{40}=5 \times 10^{-4}\)
∴ Number of millimoles of Na2C03 required to soften 1L of the sample = 5 x 10-4 x 103 = 0.5
∴ Mass of Na2C03 =5xl0-4xl06 =0.05g
Valency
Valency Definition: The capacity with which an atom of an element combines chemically with the atom(s) of another element is called the combining capacity or valency of the element.
It is determined by the number of hydrogen atoms that combine with an atom of the element or are displaced from a hydrogenated compound by one atom of that element.
There are some elements that do not combine directly with hydrogen or can’t displace hydrogen from any hydrogenated compound.
In such cases, the valency of an element can be determined by the number of atoms ofthe element that combine with an element of known valency.
Elements with different valencies are mentioned in the following table
Valency of radicals: Like an element, the valency of a radical can also be explained in the same way.
Valency of radicals Definition: Valency of a radical refers to the number of hydrogen atoms with which a radical can combine.
Thus, the valency of nitrate radical (NOJ) is because combines with 1 atom of hydrogen to produce an HN03 molecule. Similarly, sulfate (SO1-) and phosphate (PO1-) radicals exhibit di and tri valencies respectively.
This is because they combine with 2 and 3 atoms of hydrogen to form H2S04 and H3P04 molecules respectively.
Except NH+, other radicals behave like nonmetals. NH+ behaves like a monovalent metal. Like elements, radicals may also be monovalent, divalent, trivalent, etc.
Some Radicals And Their Valencies

In the case of a binary compound, the ratio of the number of atoms of the constituent elements is the inverse of the ratio of their valencies.
Let, x atoms of element A combine with y atoms of element B to form a compound and the valencies of A and B are a and b respectively.
So, the total valency of A in the compound =xa [For x atoms] and that often that compound =yb [For y -atoms
Now, in the case of a compound composed of two elements, the total valency of one element is equal to that ofthe other.
⇒ \(x a=y b \quad \text { or, } \frac{x}{y}=\frac{b}{a}\)
Thus, the tint formula of the compound formed by the elements A and I will be A;(Hrt.
So, It In evident that In a molecule of a binary compound, the ratio of (lie number of atoms of the constituent elements is Inverse of the ratio of their valencies.
Equivalent Weight Or Equivalent Mass Or Chemical Equivalent Of An Element
Equivalent Weight Definition: The equivalent weight or mass of an element Is the number of parts by mass of the element which combines with 1.008 parts by mass of hydrogen or 8 parts by mass of oxygen or 35.5 ports by mass of chlorine or can displace the same amount of hydrogen, oxygen or chlorine respectively from their compounds.
Thus, the equivalent mass of an element
⇒ \(\begin{aligned}
& =\frac{\text { mass of the element }}{\text { mass of hydrogen combined or displaced }} \times 1.008 \\
& =\frac{\text { mass of the element }}{\text { mass of oxygen combined or displaced }} \times 8 \\
& =\frac{\text { mass of the element }}{\text { mass of chlorine combined or displaced }} \times 35.5
\end{aligned}\)
The equivalent mass is a ratio of two masses. So it is a Pure number and has no unit.
Alternative Definition: The equivalent weight of an element may be defined as the number of parts by weight of the element which combines with 11.2L of hydrogen or 5.6L of oxygen or 11.2L of chlorine (at STP) or displaces the above-mentioned volume of hydrogen or oxygen or chlorine (at STP) from any compound.
Hence, the Equivalent mass of an element
⇒ \(\begin{aligned}
& =\frac{\text { mass of the element }(\text { in gram })}{\text { volume of hydrogen combined }} \times 11.2 \\
& \text { or displaced (in L) at STP } \\
& =\frac{\text { mass of the element }(\text { in gram })}{\text { volume of oxygen combined }} \times 5.6 \\
& \text { or displaced (in L) at STP } \\
& =\frac{\text { mass of the element }(\text { in gram })}{\text { volume of chlorine combined }} \times 11.2 \\
& \text { or displaced ( In L) at STP } \\
&
\end{aligned}\)
Gram-equivalent mass (or weight) and gram-equivalent: The equivalent mass of a substance (element, radical, or compound) expressed in gram is called gram-equivalent mass. This particular amount represents 1 gram equivalent of the corresponding substance (element, radical, or compound)
Gram-equivalent mass (or weight) and gram-equivalent Example: The equivalent masses of Na and Mg are 23 and 12 respectively, so their gram-equivalent masses are 23 g and 12 g respectively. Again, 1 gram-equivalent of Na= 23g and 1 gram-equivalent of Mg= 24g.
The number of grams- the equivalent of an element
⇒ \(=\frac{\text { mass of the element (in gram) }}{\text { gram-equivalent mass of the element }}\)
Law of equivalent proportions: Elements combine with one another or displace the other from their compounds in the ratio of their respective equivalent mass or in simple multiples of their equivalent masses.
The law of equivalent proportions Is a direct corollary ofthe type of reciprocal proportions: Both calcium and chlorine combine separately with oxygen to form calcium oxide (CaO) and chlorine monoxide (C120) respectively.
In calcium oxide (CaO), 16 parts by mass of O combine with 40 parts by mass of Ca.
∴ 8 parts by mass of O combine with 20 parts by mass of a. So, equivalent mass of Ca= 20 In chlorine monoxide (C120), 16 parts by mass of O combines with (2 X 35.5) parts by mass of Cl.
∴ 8 parts by mass of nO combines with 35.5 parts by mass of Cl.
So, the equivalent mass ofchlorine = 35.5 From the above data, it is clear that 8 parts by mass of oxygen combine separately with 20 parts by mass of Ca and 35.5 parts by mass of Cl.
So according to the law of reciprocal proportion, if the elements Ca and Cl combine together, the ratio of their masses in the resulting compound will be either 20: 35.5 or any simple multiple of it.
Again according to the law of equivalent proportion, Ca and Cl will combine with each other in the ratio of their equivalent masses i.e., in the ratio of 20: 35.5 (because their equivalent masses are 20 and 35.5 respectively) In practice, it is also found that Ca and Cl combine with each other in the mass-ratio of 20: 35.5 to form calcium chloride (CaCl2).
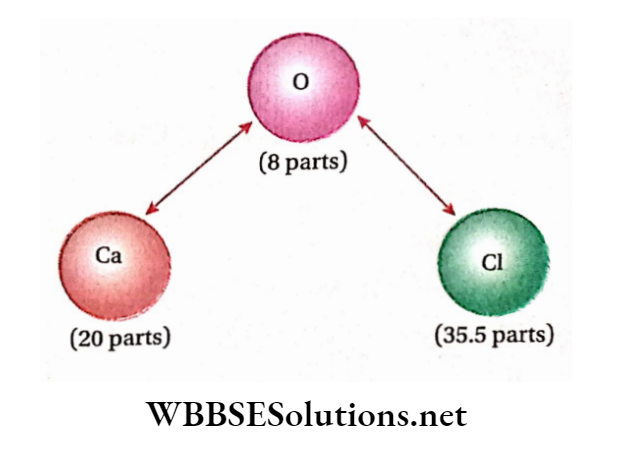
It is thus seen that by using the law of reciprocal proportions and the law of equivalent proportions arrive at the same conclusion regarding the chemical combination of two or more elements. So, these two laws are different versions ofthe same proposition.
Relation Between Equivalent Mass And Atomic Mass Of An Element
Let, the atomic mass of an element =A, its equivalent mass
=E and its valency = V.
We know that the valency of an element indicates the number of hydrogen atoms that combine with one atom of that particular element.
∴ V atoms of hydrogen combine with the atom ofthe element. So, V X 1.008 parts by mass of hydrogen combine with A parts by mass of that element.
∴ 1.008 parts by mass of hydrogen combined with
⇒ \(\frac{A \times 1.008}{V \times 1.008}=\frac{A}{V}\) parts by mass of that element.
Thus, according to the definition of equivalent mass, A/v stands for the equivalent mass ofthe element.
⇒ \(E=\frac{A}{V} \quad \text { or, } \quad A=E \times V\)
The atomic mass of an element = equivalent mass of that element x its valency
If the valency of an element (V) =1, then A = E.
∴ For monovalent elements, atomic mass and equivalent mass are equal.
Example: Atomic mass of Na = 23. As Na is monovalent, the value of its equivalent mass will also be 23.
The equivalent mass of an element can never be more than that of its atomic mass: From equation number (1), A = E=a/v From this equation, it is clear that if the value of E exceeds that of A then the value of V will be less than 1. But valency is always a whole number and its value can never be less than 1.
∴ The equivalent mass of an element can never be more than its atomic mass.
The equivalent mass of an element can never be zero:
- From equation no. (1), E = A/v
- Now, E will be zero only when A = 0.
- But the atomic mass of an element can never become zero.
- Hence, the equivalent mass of an element can never be zero.
The equivalent mass of an element is inversely proportional to its valency:
⇒ \(=\frac{\text { atomic mass of the element }(A)}{\text { its valency }(V)}\)
But, for a given element, A = constant.
⇒ \(E=\frac{\text { constant }}{V} \text { or, } E \propto \frac{1}{V}\) Therefore Equivalent mass of an element varies inversely with its valency.
Equivalent Mass Of An Element May Vary
Equivalent mass of an element= \(=\frac{\text { atomic mass of the element }}{\text { valency of that element }}\)
The atomic mass of an element has a fixed value. However, the element may have variable valencies. In such cases, the equivalent mass ofthe elements may vary.
Thus, while mentioning the equivalent mass of an element having variable valencies, the compound that contains the element or the reaction in which the element participates must be mentioned.
Examples: Copper exhibits more than one valency; For Example in cuprous compounds (e.g., Cu20), the valency of Cu is 1 while in cupric compounds (For example CuO), the valency of Cu is 2.
Therefore, the equivalent mass of copper in cuprous compounds
⇒ \(=\frac{\text { atomic mass of } \mathrm{Cu}}{\text { valency }}=\frac{63.5}{1}=63.5\)
And the equivalent mass of copper in cupric compounds
⇒ \(=\frac{\text { atomic mass of } \mathrm{Cu}}{\text { valency }}=\frac{63.5}{2}=31.75\)
Similarly, the valency of metallic iron in ferrous compounds such as FeO, FeCl2, etc., is 2 while in ferric compounds such as Fe203, FeCl3, etc., is 3.
∴ Equivalent mass of Fein ferrous compounds \(=\frac{\text { atomic mass of } \mathrm{Fe}}{\text { valency }}=\frac{55.85}{2}=27.925\)
And the equivalent mass of Fein ferric compounds \(=\frac{\text { atomic mass of Fe }}{\text { valency }}=\frac{55.85}{3}=18.616\)
The equivalent mass of the elements that exhibit variable equivalent masses can be determined if the reactions in which they participate are known.
For example, in the reaction of Fe with HC1, ferrous chloride and H2 gas are produced.
⇒ \(\begin{aligned}
& \mathrm{Fe}+2 \mathrm{HCl} \longrightarrow \mathrm{FeCl}_2+\underset{2 \times 1.008 \text { parts by mass }}{+\mathrm{H}_2} \\
& 55.85 \text { parts by mass } \\
&\text { Equivalent mass of } \mathrm{Fe}=\frac{55.85 \times 1.008}{2 \times 1.008}=27.925
\end{aligned}\)
Again, red-hot Fe reacts directly with Cl2 to form FeCl3.
⇒ \(\begin{aligned}
& 2 \mathrm{Fe}+3 \mathrm{Cl}_2 \quad \rightarrow \quad 2 \mathrm{FeCl}_3 \\
& 2 \times 55.85 \text { parts by mass } \quad 3 \times 71 \text { parts by mass } \\
& \text { Here, equivalent mass of } \mathrm{Fe}=\frac{2 \times 55.85 \times 35.5}{3 \times 71}=18.616 \\
&
\end{aligned}\)
Examples of some other elements having variable equivalent mass are Sn, Pb, Hg, Cr, Mn, As, etc. Elements like Na, K, Mg, Ca, etc., are not characterized by variable valencies, and hence their equivalent masses are always fixed.
Thus while mentioning the equivalent masses of such elements, it is not necessary to mention the compound containing the element or the reactionin which the element participates.
Equivalent mass of radicals, acids, bases, salts, oxidants, and reductants
Equivalent mass of radicals: Equivalent mass of a radical denotes the number of parts by mass of a radical which combine with 1.008 parts by mass ofhydrogen, 8 parts by mass of oxygen, or 35.5 parts by mass of chlorine, or one equivalent of any other element or a radical.
Examples:
In HN03, 1.008 parts by mass of hydrogen combine with 62 parts by mass of nitrate (N03) radical, and hence the equivalent mass of nitrate (N03 ) radical = 62. 0 In H2S04, 2 x 1.008 parts by mass of hydrogen remain associated with 96 parts by mass of sulfate (SO2-4) radical.
Hence the equivalent mass of sulphate (SO2-4) radical= =\(\frac{96 \times 1.008}{2 \times 1.008}=48\)
⇒ \(\text { Equivalent mass of a radical }=\frac{\text { formula mass of the radical }}{\text { valency of the radical }}\)
Equivalent mass of an acid: The equivalent mass of an acid is defined as the number of parts by mass of the acid which contains 1.008 parts by mass irreplaceable hydrogen.
⇒ \(\begin{aligned}
\text { Equivalent mass of an acid } & =\frac{\text { molecular mass of the acid }}{\begin{array}{l}
\text { no. of replaceable } \mathrm{H} \text {-atoms } \\
\text { present per molecule of the acid }
\end{array}} \\
& =\frac{\text { molecular mass of the acid }}{\text { basicity of the acid }}
\end{aligned}\)
Examples:
Equivalent mass of HN03 = 63/1 = 63
Equivalent mass of H2S04 =98/2 = 49
Basicity of HN03 and H2S04 are1 and 2 respectively]
Equivalent mass of a base (or alkali): The equivalent mass of a base (or alkali) is defined as the number of parts by mass of the base (or alkali) which requires one equivalent mass of an acid for complete neutralization.
Equivalent mass of a base (or alkali)
⇒ \(=\frac{\text { molecular mass or formula mass of the base }}{\text { acidity of the base }}\)
Examples: Equivalent mass of NaOH =40/1= 40
Equivalent mass of CaO =56/2=28
[∴ Acidity of NaOH and CaO are1 and 2 respectively]
Equivalent mass of salt: The equivalent mass of a normal salt denotes the number of parts by mass of that salt which contains one equivalent mass of active cation or anion.
The equivalent mass of a slat
⇒ \(\begin{aligned}
& =\frac{\text { molecular mass or formula mass of the salt }}{\text { (number of cations or anions present per molecule }} \\
& \text { of the salt } \times \text { valency of that cation or anion) } \\
& =\frac{\text { molecular mass or formula mass of the salt }}{\text { total valency of the cation or anion in one molecule of the salt }} \\
& =\frac{\text { molecular mass or formula mass of the salt }}{\text { total charge of the cation or anion in one molecule of the salt }}
\end{aligned}\)
Example: In A12(S04)3, the valency of Al3+ =3
∴ Equivalent mass of A12(S04)3 \(=\frac{342}{2 \times 3}=57\)
Similarly, in A12(S04)3, valency of SO2-4 = 2
∴ Equivalent mass of A12(S04)3 \(=\frac{342}{3 \times 2}=57\)
The equivalent mass of salt is also defined as the stun of the equivalent masses ofthe radicals present in that salt
∴ Equivalent mass of ofsalt= equivalent mass of cation + equivalent mass opinion
Examples: 1. Equivalent mass of Na2S04 = Equivalent mass of Na+ + Equivalent mass of SO2-4 =23 + 48 =71
Equivalent mass of A12(S04)3 = equivalent mass of Al3+ +Equivalent mass of SO2-4 =9 + 48 =57
Equivalent mass of Ca3(P04)2
= Equivalent mass of Ca2+ + Equivalent mass ofPO3-4
= 20 + 31.67 =51.67
Equivalent maw of oxidant and reductant: Equivalent muss of oxidant turd reductant tilth In determined by the following two methods. These are:
Electronic method:
⇒ \(\begin{aligned}
& \text { Equivalent mass of an oxidant } \\
& =\frac{\text { molecular mass or formula mass of the oxldant }}{\text { mumber of electron( } \mathrm{s}) \text { gained per molecule }}
\end{aligned}\)
Examples: Equivalent mass of KMnO4:0 Acidic medium, KMn04 Is reduced by suitable reductant to manganous (Mn2+) salt.
Reactions \(\mathrm{MnO}_4^{-}+8 \mathrm{H}^{+}+5 e \rightarrow \mathrm{Mn}^{2+}+4 \mathrm{H}_2 \mathrm{O}\)
∴ In an acidic medium, the equivalent mass of KMn04
⇒ \(=\frac{\text { molecular mass of } \mathrm{KMnO}_4}{\text { number of electrons gained }}=\frac{158.1}{5}=31.6\)
In a neutral medium, KMn04 Is reduced by a suitable reductant to Mn02.
Reaction: \(\mathrm{MnO}_4^{-}+2 \mathrm{H}_2 \mathrm{O}+3 e \rightarrow \mathrm{MnO}_2+4 \mathrm{OH}^{-}\)
∴ In a neutral medium, the equivalent mass of KMn04 Is reduced by a suitable reductant to Mn02.
In a strongly alkaline medium, KMn04 is reduced to K2Mn04.
Reaction: \(\mathrm{MnO}_4^{-}+2 \mathrm{H}_2 \mathrm{O}+3 e \rightarrow \mathrm{MnO}_2+4 \mathrm{OH}^{-}\)
Therefore In a neutral medium, the equivalent mass of KMn04
⇒ \(=\frac{158.1}{3}=52.7\)
In a strongly alkaline medium, KMn04 is reduced to K2Mn04.
Reaction: \(\mathrm{MnO}_4^{-}+e \rightarrow \mathrm{MnO}_4^{2-}\)
In strongly alkaline medium,
Equivalent mass of KMn04 \(=\frac{158}{1}=158\)
Equivalent mass of K2Cr207: K2Cr207 on reduction by a suitable reductant produces chromic salt (Cr3+).
Reaction: \(\mathrm{Cr}_2 \mathrm{O}_7^{2-}+14 \mathrm{H}^{+}+6 e \rightarrow 2 \mathrm{Cr}^{3+}+7 \mathrm{H}_2 \mathrm{O}\)
Equivalent mass of K2Cr207 \(=\frac{294.18}{6}=49.03\)
Equivalent Mass Of A Reductant
⇒ \(=\frac{\text { molecular or formula mass of the reductant }}{\text { number of electron(s) lost per molecule }}\)
Examples: In the presence of a suitable oxidizing agent, FeS04 gets oxidized to Fe2(S04)3.
Reaction: \(\mathrm{Fe}^{2+} \rightarrow \mathrm{Fe}^{3+}+e\)
Equivalent mass of FeS04 \(=\frac{151.85}{1}=151.85\)
In the presence of a suitable oxidizing agent, oxalic acid (C2H204 2H20) is oxidized to C02.
Reaction: \(\mathrm{C}_2 \mathrm{O}_4^{2-} \rightarrow 2 \mathrm{CO}_2+2 e\)
Here, the molecular mass of C2H204.2H20 = 126 and number of electrons lost = 2
∴ Equivalent mass of oxalic acid \(=\frac{126}{2}=63\)
Oxidation Mass of oxidant \(=\frac{\text { molecular or formula mass of the oxidant }}{\text { change in oxidation number }}\)
Examples: In the presence of d .H2S04, KMnCO4 Is reduced to manganous sulplutto (MnS04).
∴ \(\stackrel{+7}{\mathrm{KMnO}_4 \rightarrow} \stackrel{+2}{\mathrm{MnSO}_4}\)
Here, change In oxidation number of Mn in KMn04 = 7-2 = 5 units and molecular mass or formula mass of KMn04 =150.
Equivalent mass of KMn04 \(=\frac{158}{5}=31.6\)
In presence of dil. H2S04, K2Cr207 is reduced by a suitable reductant to Cr2(S04)3.
\(\mathrm{K}_2 \stackrel{+6}{\mathrm{Cr}_2 \mathrm{O}_7} \stackrel{+3}{\mathrm{Cr}_2}\left(\mathrm{SO}_4\right)_3\)Here, change in oxidation number of 2 Cr -atoms in K2Cr2O7 =2x(+6)-2x(+3)=6 units.
Since the Equivalent mass of k2crO7 = 294/6=49
Equivalent mass of a reductant \(=\frac{\text { molecular or formula mass of the reductant }}{\text { change in oxidation number }}\)
Examples: 1 FeS04 is oxidized by an oxidant to Fe2(S04)3.
⇒ \(\stackrel{+2}{\mathrm{FeSO}_4} \stackrel{+3}{\rightarrow} \mathrm{Fe}_2\left(\mathrm{SO}_4\right)_3\)
Here, the change in oxidation number of Fe in FeS04 = 3-2 = 1 unit
∴ Equivalent mass of FeS04 \(=\frac{151.85}{1}=151.85\)
Sodium thiosulphate (Na2S203-5H20) is oxidized by an oxidant to sodium tetrathionate (Na2S406)
⇒ \(2 \mathrm{Na}_2 \stackrel{+2}{\mathrm{~S}_2} \mathrm{O}_3 \rightarrow \mathrm{Na}_2 \mathrm{~S}_4 \mathrm{O}_6+2 \mathrm{Na}^{+}\)
Here, changein oxidation number of4 S-atoms in Na2S203 = 4 X (+2.5)- 4 x (+2) = 18- 8 = 2 units.
∴ Changes in oxidation number for two S-atoms =1 unit Now, molecular mass of Na2S203.5H20 = 248
Equivalent mass of Na2S203-5H20 \(=\frac{248}{1}=248\)
In any chemical reaction, the number of equivalent or gram-equi-valent of reactants and products are always equal: In the chemical reaction, the reacting substances unite together in accordance with their equivalent masses. By analyzing the chemical reaction, it has been observed that the number of equivalents or gram-equivalents of constant(s) and produces) are always equal.
Example 1 \(\begin{aligned}
& \text { (1) } \underset{56 \mathrm{~g}}{\mathrm{Fe}}+\underset{2 \times 36.5 \mathrm{~g}}{2 \mathrm{HCl}} \longrightarrow \underset{129 \mathrm{~g}}{\mathrm{FeCl}_2}+\underset{2 \mathrm{~g}}{\mathrm{H}_2} \\
& 2 \text { gram-eqv. } 2 \text { gram-eqv. } 2 \text { gram-eqv. } 2 \text { gram-eqv. } \\
&
\end{aligned}\)
Generally, if in the case of the reaction, A + B-+C +D, the gram-equivalent of A reacts with .v gram-equivalent of B then, the gram-equivalent of C and .v gram-equivalent of D will be produced.
Methods for the determination of the atomic mass of an element:
Determination of atomic mass by Dulong and Petit’s law: At ordinary temperature, the atomic heat of all the solid elements (except C, B, Si, and Be) is always the same and numerically equal to 6.4 (approximately). The product of atomic mass and specific heat is known as atomic heat.
Hence, atomicmass x specific heat = 6.4 (approx) Approximate atomic mass \(=\frac{6.4}{\text { specific heat }}\)
The approximate atomic mass so obtained is divided by the equivalent mass ofthe element to get its valency. Since the atomic mass is not accurate, the value of valency may be fractional.
So, the nearest whole number should be considered as its valency which on multiplication with equivalent mass gives accurate atomic mass.
Determination of atomic mass of an element by applying Mitscherlich’s law of isomorphism: Let, a salt of an element E be isomorphous with K2S04, and the salt contains a % of E.
The atomic mass of E is to be determined. Let, the atomic mass of E be r. Since the salt of E is isomorphous with K2S04, the formula of the salt, according to Mitscherlich’s law, would be K2E04. Now, the molecular mass of K2E04
= 2×39+x + 4x 16 = 78+X + 64 = 142 + x
∴ Percentage of E in the salt \(=\frac{x}{142+x} \times 100\)
By the above assumption \(\frac{100 x}{142+x}=a\)
As ‘a’ is known, x can easily be calculated.
An alternative method (hy Isomorphous replacement): Experimental results show that different key dementia (l.a, the elements which are different) forming an Isomorphous compound can replace each other, atom for atom, without changing the crystal structure.
Let A and li be the key elements (having atomic masses a and respectively) In a pair of isomorphous compounds.
If W g of 1 is replaced hy W2 g of, then according to the law oflsomorphlsm.
⇒ \(W_1 / a=W_2 / b \text { or, } W_1 / W_2=a / b\)
⇒ \(\text { i.e., } \frac{\text { mass of the substituted element, } A}{\text { mass of the substituting element, } B}=\frac{\text { atomic mass of } A}{\text { atomic mass of } B}\)
This relation can be used to determine the atomic mass of A if that of B is known or vice versa.
Two compounds having the following characteristics are said to be isomorphous crystals:
- Both have similar external crystalline structures.
- They together can form mixed crystals.
- One can form overgrowth on the other if a small crystal of the latter is placed in the saturated solution of the former.
The property by virtue of which isomorphous crystals are formed is called isomorphism (iso: similar, morph: form).
Examples of isomorphous crystals:
White vitriol (FeS04-7H20), Epsom salt (MgS04- 7H20), green vitriol (FeS04-7H20)
Potassium permanganate (KMn04), potassium perchlorate (KC104)
Potash alum [K2S04-A12(S04)3-24H20]; chrome alum [K2S04-Cr2(S04)3-24H20]
It is observed from the formulae of isomorphous crystals that the total number of atoms in their molecules is the same.
Mitscherlich’s law of isomorphism: The same number of atoms combine in the same way to produce isomorphous crystals.
The crystalline form of these crystals depends only on the number of atoms in their molecules and the way by which they combine but is independent of their chemical properties.
Question 1. The atomic mass & equivalent weight of an element are 27 & 9 respectively. Find the formula of its chloride
Answer: Valency of element \(=\frac{\text { atomic mass }}{\text { equivalent weight }}=\frac{27}{9}=3\)
∴ Formula of its chloride = MC13 [since valency of Cl =1]
Question 2. x gram of an element forms y gram of its chloride. Calculate the equivalent weight of the element.
Answer: Mass ofthe chloride =y g and mass of element = x g
Mass of chlorine =(y- x) g
So, (y- x) g of chlorine combines with x g of the element.
∴ 35.5 g ofchlorine combines with \(\frac{35.5 \times x}{y-x} g\)
∴ Equivalent weight ofthe element \(=\frac{35.5 x}{y-x}\)
Example 3. Calculate the relative equivalent weight (£) of copper in cuprous oxide.
Answer: Formula of cuprous oxide = Cu20
∴ 16 g of oxygen combined with 2 x 63.5 g of Cu
So, 8 g ofoxygen combines with 63.5 g of Cu
∴ Equivalent weight (E) = 63.5
Example 4. A metallic oxide contains 60% metal. Calculate the equivalent weight of the metal.
Answer: 100 g of metallic oxide contains 60 g of metal. 100 g of metallic oxide contains (100 – 60) = 40 g of oxygen So, 40 g of oxygen combines with 60 g of metal.
8 g of oxygen combines with \(\frac{60 \times 8}{40}\)
Therefore, the equivalent weight of the metal = 12
Stoichiometry Practice Questions for Class 11
Question 5. 0.56 g of metallic oxide contains 0.16 g of oxygen. Determine the equivalent weight of that metal.
Answer: Mass of metallic oxide = 0.56 g and mass of oxygen = 0.16 g
So, mass ofmetalin the metallic oxide = (0.56- 0.16) g = 0.40 g
Equivalent weight of the metal
⇒ \(=\frac{\text { mass of the metal }}{\text { mass of combined oxygen }} \times 8=\frac{0.40 \times 8}{0.16}=20\)
Question 6. Determine the equivalent weight of O carbonate radical Ferrous Sulphate [Fe = 56]
Answer: Formula mass of carbonate radical =12 + 3×16 = 60
Therefore Equivalent weight of carbonate radical
⇒ \(=\frac{\text { formula mass }}{\text { valency }}=\frac{60}{2}=30\)
Formula mass of ferrous sulphate =56 + 32 + 64 = 152
Therefore Equivalent weight formula of ferrous mass sulphate
⇒ \(\begin{aligned}
& =\frac{\text { formula mass }}{\text { total valency of cation or anion per molecule }} \\
& =\frac{152}{2}=76
\end{aligned}\)
Question 7. When 0.3 g of a metal is dissolved in dilute IIC1 the volume of H2 gas liberated is 1 10mL at 17°C and 755 35.5 g ofchlorine combined with g ofthe element Hg of pressure. [Aqueous tension at 17°C = 14.4 Hg] Determine the equivalent mass of the metal.
Answer: Pressure of dry hydrogen gas= 755.14.4
If the volume of H2 gas produced at STP is VmL, then
⇒ \(\frac{110 \times 740.6}{(273+17)}=\frac{V \times 760}{273}\)
So, \(V=\frac{110 \times 740.6 \times 273}{290 \times 760} \mathrm{~mL}=100.91 \mathrm{~mL}\)
Now,mass of22400 mL of H2 at STP = 2x 1.008g
Mass of100.91 mL of H,2 at STP \(=\frac{2 \times 1.008 \times 100.91}{22400} \mathrm{~g}\)
=0.00908g
So, 0.00908 g of H2 is displaced by 0.3 g of metal.
1.008 g of H2 is displaced by \(\frac{0.3 \times 1.008}{0.00908}\) =33.3 g of mental
Therefore Equivalent mass of the mental=33.3
Question 8. The equivalent mass of a metal is 11.6. When 0.177g of that metal is allowed to react completely with dilute HC1, what will be the volume of H2 gas liberated at 12°C and 766mm Hg pressure?
Answer: The equivalent mass ofthe metal is 11.6.
∴ 11.6 g metal displaces 11200 mL of H2 at STP
0.177 g metal displaces \(\frac{11200 \times 0.177}{11.6}\)=170.89 ml of H2
Let volume by V at 12°C and 760 mm Hg pressure
∴ \(\frac{V \times 766}{(12+273)}=\frac{170.89 \times 760}{273}\)
Or, \(V=\frac{170.89 \times 760}{273} \times \frac{285}{766}=177.0 \mathrm{~mL}\)
Question 9. 20 g of a metal reacts with dilute H2S04 to liberate 0.504 g of H2 gas. Calculate the amount of metal oxide formed from 2.0 g of the metal.
Answer: 0.504 g of H2 is liberated by 20 g of the metal
∴ 1.008 g H2 is liberated by \(\frac{20 \times 1.008}{0.504}\)
So, the equivalent mass ofthe metal = 40
∴ 40 g of metal combined with 8 g of oxygen
So, 2.0 g metal combines with \(=\frac{8 \times 2}{40} g=\) 0.4 of oxygen
∴ Amount of metal oxide = mass of metal + mass of oxygen
= (2.0 4-0.4) = 2.4 g
Question 10. 3.26 g of zinc reacts with acid to liberate 1.12L of hydrogen gas (H2) at STP. Calculate the relative equivalent weight of zinc.
Answer: 1-12L of H2 (at STP) is liberated by 3.26 g of zinc, therefore, 11.2L Of H2 is Liberated By \(\frac{3.26 \times 11.2}{1.12}\) =32.6g of Zinc.
So, the equivalent weight of zinc = 32.6
Question 11. 15 g of an element reacts completely with 30 g of We know, A =Ex V A = 9.02 x V another element. Calculate the specific equivalent weight of A if that of B is 60.
Answer: We know that two elements A and B will react with each other in the ratio of their equivalent weight (EA and EB).
So, in the reaction between A and B Or, 15/30 = Ea/Eb
∴ Ea=1/2 X Eb =1/2 X 60 =30
So, the equivalent weight of A = 30
Question 12. 0.362g of metal is added to an aqueous solution of AgNOs. Consequently, 3.225 g of silver is precipitated. What is the equivalent mass of the metal? [Atomic mass of Ag = 108, valency = 1]
Answer: Equivalent mass of Ag \(=\frac{\text { mass of the metal }}{\text { mass of } \mathrm{Ag}}\) =108/1 =108
Now, the displacement of Ag by the metal from a compound of the metal occurs in proportion to their equivalent masses.
So, \(\frac{\text { equivalent mass of the metal }}{\text { equivalent mass of } \mathrm{Ag}}=\frac{\text { mass of the metal }}{\text { mass of } \mathrm{Ag}}\)
Let the equivalent mass of the metal be E
According to the problem \(\frac{E}{108}=\frac{0.362}{3.225}\)
Or, E=12.12
∴ Equivalent mass ofthe metal =12.12
Question 13. A metallic oxide contains 53% neutral. The vapor density of the chloride of the metal is 66. Find the atomic weight of the metal.
Answer: The metallic oxide contains 53% metal.
∴ The metallic oxide contains (100-53) = 47 % oxygen.
Hence, the equivalent weight (£) of the metal.
⇒\(=\frac{\text { mass of the metal }}{\text { mass of oxygen combined }} \times 8=\frac{53 \times 8}{47}=9.02\)
As given, vapour density of the metallic chloride = 66 Molecular weight ofthe chloride = 2 x 66 =132
since M=2xD
Let, the molecular formula of the metallic chloride be MCIv [where, V = valency of the metal]
∴ Molecular weight of MClv = A 4- 35.5 V [where, A = atomic weight of the metal]
We know, A =Ex V A = 9.02 x V
Hence, 9.02 X V4- 35.5 x V = 132 or, V \(=\frac{132}{44.52} \approx 3\)
So, atomic weight ofthe metal =ExV = 9.02 X 3 = 27.06
Question 14. A metallic chloride contains 20.2% by mass of metal (M). If the atomic mass of the metal is 27. What is the molecular formula of the metallic chloride?
Answer: Metal content in metallic chloride = 20.2 %
∴ Amount ofchlorine in the metallic chloride=(100- 20.2) = 79.8%
So, 79.8 parts by mass of chlorine combined with 20.2 parts by mass of the metal (M)
∴ 35.5 parts by mass of chlorine combined with \(\frac{20.2 \times 35.5}{79.8}\) = 8.986 Parts by mass of the mental.
Hence, the Equivalent mass of mental (M)=8.986
\(\quad V=\frac{A}{E}=\frac{27}{8.986}=3 \text { (approx.) }\)So, the valency of the metal in the metallic chloride = 3
∴ The molecular formula of the metallic chloride = MC13
[Therefore Valency of chlorine =1]
Question 15. 8.08g of metallic oxide on being reduced by H2, produces 1.8g of water. Find the quantity of Oz In the above oxide and the equivalent mass of the metal.
Answer: Amount of oxygen present in 18 g of water = 16 g
Amount of oxygen present 1.8 g of water = 1.6 g
Oxygen content in 8.08 g ofthe metallic oxide = 1.6 g
[since all of this oxygen comes from the metallic oxide]
Hence, the amount ofthe metal present in that oxide =(8.08-1.6)=6.48g
∴ 1.6g of O2 COmbine with 6.48g of mental
Or, 8g Of O2 Combine with \(\left(\frac{6.48}{1.6} \times 8\right)\) g of the mental
Therefore, the equivalent mass of the metal = \(=\frac{6.48}{1.6} \times 8=32.4\)
Question 16. In the following reaction, determine the equivalent
weight of H3P04.
\(\begin{array}{r} \mathrm{Ca}(\mathrm{OH})_2+\mathrm{H}_3 \mathrm{PO}_4 \rightarrow \mathrm{CaHPO}_4+2 \mathrm{H}_2 \mathrm{O} \\{[\mathrm{Ca}=40 ; \mathrm{P}=31] \text { [II 82] }} \end{array}\)
Answer: In this acid-base neutralization reaction, 2 hydrogen atoms of H3P04 are replaced by atoms of calcium.
Equivalent weight of H3P04
⇒ \(\begin{aligned}
& =\frac{\text { molecular mass of } \mathrm{H}_3 \mathrm{PO}_4}{\text { number of displaced hydrogen atoms }} \\
& =\frac{3 \times 1.008+31+4 \times 16}{2}=49.012
\end{aligned}\)
Question 17. A metallic bromide weighing 1.878 g is heated in a current of hydrogen chloride. Consequently, 1.0 g of metallic chloride is obtained. What is the equivalent mass of the metal?
Answer:
Let the equivalent mass ofthe metal be the Equivalent mass ofthe metallic bromide =E+ 80
[∴ Equivalentmass ofbromine = 80 ] Equivalent mass ofmetallic chloride =E + 35.5
[∴ Equivalent mass ofchlorine = 35.5]
So, the number of gram-equivalent of metallic bromide and metallic chloride is \(\frac{1.878}{E+80} \text { and } \frac{1.0}{E+35.5}\) Respectively.
Now, in any chemical reaction, the gram-equivalents of the reactant and the product are equal.
So, in the reaction, the gram-equivalent of metallic bromide is equal to the gram-equivalent of metallic chloride.
∴ \(\frac{1.878}{E+80}=\frac{1.0}{E+35.5}\)
or, E(1.878-1) = 80-35.5X 1.78 =80-66.669
or, 0.878 x £ = 13.331 or, E= 15.18
∴ Equivalent Mass of mental = 15.18
Question 18. The hydride of element A contains 25% of hydrogen by mass. The percentages of oxygen in two oxides of that element are 57.1 and 72.7 respectively. If the atomic mass of the element is 12 determine the formula of the hydride and the two oxides.
Answer: The quantity of the element A in the hydride of element
A =(100-25)% = 75%
So, in the hydride of element A, 25 parts by mass ofhydrogen combine with 75 parts by mass element A.
∴ 1.008 parts by mass of hydrogen combined with
75/25 X 1.008 = 3.024 Parts by mass of element A
∴ Equivalent mass of ‘A’ anhydride = 3.024
∴ Quantity of A in the first oxide = (100- 57.1) % = 42.9 %
and in the second oxide = (100- 72.7) % = 27.3 %
So, in the first oxide, 57.1 parts by mass ofoxygen combine with
42.9 parts by mass of A
In the first oxide, 8 parts by mass of oxygen combine with
⇒ \(\frac{42.9}{57.1} \times 8\)=60.1 parts of mass of A.
Similarly, in the second oxide, 8 parts by mass of oxygen react with \(\frac{27.3 \times 8}{72.7}=\) = 3 parts by mass of A.
∴ The valency of A in the first hydride.
⇒ \(=\frac{\text { atomic mass of } \mathrm{A}}{\text { equivalent mass of } \mathrm{A} \text { in the hydride }}=\frac{12}{3.024}=4\)
Formula of the hydride = AH4
Valency of A in the first oxide
⇒ \(=\frac{\text { atomic mass of } \mathrm{A}}{\text { equivalent mass of } \mathrm{A} \text { in the first oxide }}\)
=12/6.01 =2
Formula of first oxide = AO [Therefore Valency of oxygen =2]
Valency of A in the second oxide
⇒ \(=\frac{\text { atomic mass of } \mathrm{A}}{\text { equivalent mass of } \mathrm{A} \text { in the second oxide }}\)
=12/3=4
therefore Formula ofthe second oxide = AO2
Question 19. The specific equivalent weight of a solid element is 17.8 and its specific heat is 0.124 cal .K-1. g-1. Find its valency and correct specific atomic mass.
Answer: As perDoulong and Petit’s law, the approximate atomic mass
ofthe solid element = \(\frac{6.4}{\text { specific heat }}=\frac{6.4}{0.124}=51.61\)
therefore Valency of element = \(=\frac{\text { atomic mass (approx) }}{\text { equivalent weight }}=\frac{51.61}{17.8} \approx 3\)
Therefore Correct atomic mass ofthe element
= equivalent weight X valency = 17.8 X 3 = 53.4
Question 20. The specific heat of magnesium is 0.262 and magnesium chloride contains 25.5% by weight of magnesium. Determine the atomic mass, valency of magnesium, and also the formula of magnesium chloride.
Answer: Percentage of magnesium chloride= 25.5
∴ Percentage of chlorine in the salt = 74.5
∴ Equivalent weight of magnesium
⇒ \(=\frac{\text { mass of magnesium }}{\text { mass of chlorine }} \times 35.5\)
⇒ \(=\frac{25.5}{74.5} \times 35.5=12.15\)
According to Dulong and Petit’s law,
Atomic mass ofthe metal x specific heat = 6.4 (approx)
So, approximate atomic mass of magnesium \(=\frac{6.4}{0.62}=24.42\)
∴ Valency of Mg \(=\frac{\text { approximate atomic mass }}{\text { equivalent weight }}\)
Therefore, the atomic mass of magnesium = equivalent weight x valency = 12.15×2 = 24.30, and the formula of magnesium chloride is MgCl2.
Question 21. An aqueous solution contains 0.22 g of metallic chloride. 0.51 g of AgNOa is required for the complete precipitation of chloride from that solution. If the specific heat of the metal is 0.057 then what will be the correct atomic mass of that metal? What is the formula of that metallic chloride?
Answer: S. Let the equivalent mass ofthe metal be E.
Equivalent mass of the metallic chloride =E + 35.5 [ v Equivalent mass ofchlorine = 35.5 ]
Equivalentmass of AgN03
= Equivalent mass of Ag+ + Equivalent mass of N03
= 108 + 62 =170.
In any chemical reaction, the reactants combine together in the proportion of their equivalent masses.
⇒ \(\text { So, } \quad \frac{E+35.5}{170}=\frac{0.22}{0.51} \quad \text { or, } E=37.83\)
Determination of atomic mass ofthe metal: According to Dulong and Petit’s law, the approximate atomic mass of metal
⇒ \(=\frac{6.4}{\text { specific heat }}=\frac{6.4}{0.057}=112.28\)
Therefore Valency of the metal \(=\frac{\text { approx. atomic mass of the metal }}{\text { equivalent mass }}\)
⇒ \(=\frac{112.28}{37.83}=2.968 \approx 3\)
Therefore, the correct atomic mass of M =37.83×3 = 113.49
So, the molecular formula of chloride salt = MC13.
Question 22. The percentage by weight of chromium in green-colored chromium oxide (chromic oxide) is 68.43 and it is isomorphous with ferric oxide. Estimate the = 74.5— X35.5 = 12.15 correct atomic weight of chromium.
Answer: Chromium oxide is isomorphous with Fe203. So, according to Mitscherlich’s law of isomorphism, the formula chromium oxide will be Cr203.
Since the valency of Fe in Fe203 is 3 the valency of Cr in Cr203 will also be 3.
Now, the quantity of chromium in chromium oxide = 68.43 % Quantity of oxygen =(100-68.43) = 31.57 % Equivalent weight of chromium
⇒ \(=\frac{\text { weight of chromium }}{\text { weight of combined oxygen }} \times 8=\frac{68.43}{31.57} \times 8=17.34\)
Hence, the correct atomic weight chromium
= equivalent weight x valency = 17.34 x 3 = 52.02
Question 23. Potassium manganate and potassium chromate (K2Cr04) Percentage of manganese by mass present in potassium manganate is 27.86. Determine the atomic mass of manganese.
Answer: Potassium chromate (K2Cr04) is isomorphous with potassium manganate. So according to Mitscherlich’s law of isomorphism, the formula of potassium manganate should be K2Mn04.
Suppose, the atomic mass of Mn = a
Molecular mass of K2Mn04 = 2×39 + a + 4xl6 = a + 142
Therefore Percentage of Mn in K2Mn04 \(=\frac{a}{a+142} \times 100\)
As given in the question \(\frac{a \times 100}{a+142}=\)
Therefore Atomic mass of manganese = 54.84
Question 24. An element (X) reacts with KOH to form a salt. The salt is isomorphous with potassium permanganate (KMn04). The oxide of the element X contains 61.2% of oxygen. Determine the formula of the oxide and atomic mass of X.
Answer: Percentage of oxygen in the oxide of X = 61.2
Therefore Percentage of X in the oxide of X = (100- 61.2) = 38.8
Therefore \(\text { Equivalent mass of } \mathrm{X}=\frac{\text { mass of } \mathrm{X}}{\text { mass of combined oxygen }} \times 8\)
⇒ \(=\frac{38.8}{61.2} \times 8=5.07\)
In reaction with KOH, the element X produces a salt that is isomorphous with KMn04. Since the valency of Mn in KMn04 is 7, according to Mitscherlich’s law, a formula of the salt produced will be KX04 where the valency of X is 7.
Therefore Atomic mass of X = equivalent mass x valency
∴ = 5.07X7 = 35.49.
∴ The formula of the oxide would be X207
Question 25. Oxides of two metals A and B are isomorphous. The atomic weight of B is 43.5 and the vapor density of its chloride is 75. The amount of oxygen present in 1.02g ofthe oxide of A is 0.48g. Determine the atomic weight of A.
Answer: Determination of equivalent weight of A: Quantity of oxygen present 1.02g of the oxide of A = 0.48 g
So, the quantity of A in that oxide = (1.02- 0.48) = 0.54 g
Equivalent weight of the metal A
\(=\frac{\text { mass of } \mathrm{A} \times 8}{\text { mass of combined oxygen }}=\frac{0.54 \times 8}{0.48}=9\)Determination of valency of B: Vapour density ofthe chloride metal B = 75
Molecular mass ofthe chloride of metal B = 2 x 75 = 150 If the valency of B is x, then the formula of the chloride
of B = BC1X
Molecular mass ofthe compound BC1X = 43.5 + 35.5 X x
Therefore 43.5 + 35.5 X x = 150 or, x = 3
Therefore Valency of B = 3 and the formula of its oxide is B203
Determination of valency of A: Oxides of A and B are isomorphous. So according to the law of isomorphism, the formula ofthe oxide of A is A203 and the valency of A is 3. Hence, the atomic mass of A.
= equivalent weight x valency = 9×3 = 27
Quantity of water associated with 1 11 g or 1 mol of the anhydrous salt \(=\frac{1.48 \times 111}{1.52}=108.07 \mathrm{~g}\)
Therefore, the quantity of water associated with 1 mol of anhydrous CaCl2 is 108.07g.
Percentage Composition Empirical And Molecular Formula
Percentage Composition
The percentage composition of a compound means the parts by mass of each of the constituent elements in every 100 parts by mass of that compound.
Chemical analysis of water reveals that in every 100 parts by mass of water, 11.1 parts by mass of hydrogen, and 88.9 parts M1 Gram-formula mass of Ca3(P04)2 by mass of oxygen is present the percentage composition of water is: hydrogen (H) = 11.1 % and oxygen (O) = 88.9%.
Hence, it is clear that the percentage composition of the = [3 X 40 + 2(31 + 64)] = 310g
Ca3(P04)2 can be regarded as constituent elements in a compound independent of the Gram-molecular mass of P205 = (2 X 31 + 5 x 16) = 142g quantity of the compound taken.
For example, irrespective of the quantity of water taken % of P2O5 in calcium phosphate = x 100 = 45.8 (say 4g or 18g or 50g, etc.), the percentage of hydrogen and
oxygen by mass will always be 11.1 and 88.9 respectively.
Percentage composition from the molecular formula:
- The molecular mass or formula mass of the compound is first calculated from its molecular formula.
- The mass of each element or radical present in 1 gram ofthe compound is then calculated separately.
- Finally, the percentage of the mass of each element present in the compound is computed separately.
% of an element or radical present in the compound
\(\begin{aligned}& \text { mass of the element or radical in } 1 \text { gram-mole } \\
& =\frac{\text { of the compound }(\mathrm{g})}{\text { gram-molecular mass or gram-formula mass }} \times 100 \\
& \text { of the compound (g) } \\
&
\end{aligned}\)
Question 1. 3 g of hydrated calcium chloride yields 1.52 g of the anhydrous salt on heating. What is the percentage of water present in the hydrated salt? Find the quantity of water associated per mole ofthe anhydrous salt.
Answer: Amount of water present in 3g of hydrated calcium chloride =(3- 1.52) =1.48g
∴ Percentage of water in hydrated salt= \(\frac{1.48}{3} \times 100\) 100 = 49.33
Gram-formula mass of any. CaCl2 =(40 + 2 x 35.5) = 392 = 111g
Quantity of water associated with 1. 52g anhydrous =1.48g
∴ Quantity of water associated with 1 11 g or 1 mol of the anhydrous salt \(=\frac{1.48 \times 111}{1.52}=108.07 \mathrm{~g}\)
∴ The quality of water associated with 1 month 1 mol of anhydrous CaCl2 is 108.07g
Question 2. What is the percentage of P205 in calcium phosphate [Ca3(PO4)2]?
Answer: Gram-formula mass of Ca3(P04)2 = [3 X 40 + 2(31 + 64)] = 310g
Ca3(P04)2 can be regarded as [3CaO P205]
Gram-molecular mass of P205 = (2 X 31 + 5 x 16) = 142g
∴ % of P2O5 in calcium phosphate \(=\frac{142}{310} \times 100=45.8\)
Question 3. A compound contains 28% of nitrogen and 72% of metal mass. In the compound, 3 atoms of the metal remain combined with two atoms of nitrogen. Find the atomic mass of the metal.
Answer: The formula of the compound is M3N2. [metal =M]
∴ Molar mass of M3N2 =3a + 28 [a = atomic mass ofM]
So, percentage of metal in the compound \(=\frac{3 a}{3 a+28} \times 100\)
∴ \(\left(\frac{3 a}{3 a+28}\right) \times 100=72 \text {, or } a=24\)
Therefore, the atomic mass of the metal = 24
Question 4. Give the percentages of ammonium and sulfate radicals in Mohr salt [(NH4)2S04 -FeS04 -6H20 ].
Answer: Molecular mass of Mohr salt = (2 X 18) + 96 + 56 + 96 + (6 x 18) =392
In each mole of Mohr salt, the amount of ammonium
⇒\(\left(\mathrm{NH}_4^{\oplus}\right)\)
radical=2×18 =36g and amount of sulphate \(\left(\mathrm{SO}_4^{2-}\right)\) radical
= 2×96 = 192g
∴ Percentage of ammonium \(\left(\mathrm{NH}_4^{\oplus}\right)\) radical in mohar salt.
\(=\frac{36 \times 100}{392}=9.18\)Percentage of sulphate \(\left(\mathrm{SO}_4^{2-}\right)\) radical in Mohr Salt \(=\frac{192 \times 100}{392}=48.98\)
Question 5. Haematite (Fe203) contains some water in addition to Fe203. 4.0 kg of this mineral contains 2.5 kg of iron. Find the purity of Fe2Os in the mineral.
Answer: Gram-molecular mass of Fe203 =2×55.85 + 3×16 =159.7g
Now, (2 x 55.85) g Fe= 159.7 g Fe2O3
∴ 2500g Fe = \(\frac{159.7 \times 2500}{2 \times 55.85} \mathrm{~g} \mathrm{Fe}_2 \mathrm{O}_3=3574 \mathrm{~g} \mathrm{Fe}_2 \mathrm{O}_3\)
So, 4000 g mineral contains 3574 g of pure Fe203.
∴ Purity of Fe203 in haematite \(=\frac{3574 \times 100}{4000}=89.35 \%\)
Empirical And Molecular Formula
Empirical formula: The empirical formula of a compound indicates the number of atoms of different elements present in a molecule of the compound, expressed in a simple whole number ratio.
The empirical formula merely indicates the ratio of the atoms of elements constituting the molecule of a compound. Thus, this formula may not represent the actual number of atoms in the molecule of a compound.
Example: The empirical formula of glucose is CH2O. This shows that in the molecule of glucose, C, H, and O-atoms are present in the simplest ratio of 1:2: 1.
Molecular formula: The molecular formula of a compound indicates the actual number of atoms of various constituent elements present in a molecule of the compound.
The molecular formula represents the actual formula of the molecule of a compound as it gives the actual number of atoms constituting the molecule.
Example: The molecular formula of glucose is C6H1206. This shows that a molecule of glucose consists of six C-atoms, twelve H-atoms, and six O-atoms.
However, in certain cases, the empirical formula and the molecular formula may be identical. For example, formaldehyde has both empirical and molecular formula CH20.
Relation between empirical formula and molecular formula: The molecular formula of a compound is a whole number multiple ofthe empirical formula of that compound.
Molecular formula of a compound = (empirical formula)n
[where n = 1, 2, 3, etc. are simple whole numbers]
Example: The empirical formula of glucose is CH20 while its molecular formula is C6H1206 or CH12O6 When n — 1, both the empirical formula and the molecular formula will be the same.
In the case of formaldehyde, the empirical formula i.e., CH20 and the molecular formula [i.e., CH20 (HCHO)] are the same.
The sum of the atomic masses of the constituent elements present in the molecular formula of a compound indicates the molecular mass of that compound.
On the other hand, the sum of the atomic masses of the constituent elements present in the empirical formula of a compound indicates the empirical formula mass of that compound.
The molecular formula of a compound is either equal to or is a simple multiple of its empirical formula mass Molecular mass of a compound =n x empirical formula mass.
Now, if n is 1, i.e., molecular mass = empirical formula mass of a compound, then the empirical formula and the molecular formula ofthe compound would be identical.
Basis of determination of the empirical formula of a compound: Let the molecular mass of a compound, composed of two elements A and B, is M and the percentage composition by mass of A and B in that compound are x and y respectively.
Therefore Amount of A in M gram i.e., 1 gram-mole compound \(=\frac{x \times M}{100}\) gram. Similarly, the amount of B in Mgram i.e., 1 gram-mole ofthe compound gram.
Let, the atomic masses of A and B be a and b respectively.
∴ Number of gram-atoms of A in 1 gram-mole of the \(=\frac{x \times M}{100 \times a} .\)atoms of B in1 gram-mole of the compound \(=\frac{x \times M}{100 \times a}: \frac{y \times M}{100 \times b}=\frac{x}{a}: \frac{y}{b}\)
In 1 molecule of the compound, the ratio of the number of atoms of A and B
Hence, in a molecule of the compound, the ratio of the number of atoms of A to the number of atoms of B \(=\frac{\text { mass of } \mathrm{A} \text { in the compound }(\%)}{\text { atomic mass of } \mathrm{A}}: \frac{\text { mass of } \mathrm{B} \text { in the compound }(\%)}{\text { atomic mass of } \mathrm{B}}\)
Similarly, the number of grams of A in the compound(%). mass of B in the compound(%) atomic mass of A atomic mass of B
Therefore, the ratio of the quotients, obtained by dividing the percentage mass of each constituent element by its corresponding atomic mass, gives the ratio of the atoms of the elements present in a molecule of that compound. From this ratio, the empirical formula can be determined.
Determination of the empirical formula of a compound: The empirical formula ofany compound can be determined the following steps
The percentage by mass of each element in the compound is accurately evaluated by suitable methods.
The percentage by mass of each element is divided by its atomic mass in order to get the relative number of different types of atoms present in the molecule.
Each of the numbers obtained is divided by the smallest of these numbers to get the simplest ratio of atoms.
In determining the ‘simplest ratio of atoms; the ‘rule of approximation’ is followed. For example, if the quotient is 2.99 or 4.01, then the nearest whole numbers, i.e., 3 or 4 respectively are accepted as the required quotient.
But when the ‘rule approximation’ cannot be applied, e.g., if any quotient is 1.5 or 1.6, each of the quotients obtained is to be multiplied with a suitable factor so as to convert all the quotients into lowest whole numbers.
The ratio, thus obtained, expresses the ratio of the number of atoms of the elements constituting a molecule and thus gives the empirical formula of the compound.
Example: In an acetic acid molecule, constituent atoms are C, H, and O. The ratio of the number of atoms is 1:2:1 as determined by the preceding steps. Thus, the empirical formula acetic acid will be CH20.
Method of determination of molecular formula:
- The empirical formula of a compound is first determined. by the method described above.
- The molecular mass of the compound is determined experimentally.
- In the case of volatile compounds, the molecular mass is evaluated using the equation, M = 2D, where D = experimentally determined value of the vapor density of the compound.
- [It is to be noted that the equation, M = 2D is applicable only in the case of those stable compounds that do not undergo dissociation or decomposition reaction in the vapor state.
- From the empirical formula of the compound, the empirical formula mass is calculated.
- The empirical formula mass of any compound is the sum of the atomic masses of atoms of different elements represented by the empirical formula.
- The molecular mass of the compound. n x empirical formula mass
- So, in order to evaluate ‘n ’, the molecular mass of the compound is to be divided by its empirical formula
- Finally, the empirical formula ofthe compound multiplied by n ’, gives the molecular formula of the compound.
Numerical Examples
Question 1. A compound of carbon, hydrogen, and oxygen contains 40% of carbon and 6.67% of hydrogen. The vapor density of the compound, when vapourised, is 2.813 times the vapor density of oxygen. Determine the empirical formula and molecular formula of the compound.
Answer:
Given
A compound of carbon, hydrogen, and oxygen contains 40% of carbon and 6.67% of hydrogen. The vapor density of the compound, when vapourised, is 2.813 times the vapor density of oxygen.
In the compound, C = 40%, H = 6.67% and thus O = [100 -(40 + 6.67)]% =53.33%
Now, the ratio of the number of atoms of C: H:O in the compound, \(\mathrm{C}: \mathrm{H}: \mathrm{O}=\frac{40}{12}: \frac{6.67}{1}: \frac{53.33}{16}\)
Therefore Atomic masses of C, H, and O are 12, 1, and 16 respectively]
= 3.33: 6.67: 3.33 as 1: 2: 1
[dividing by the lowest number 3.33 ]
Therefore Empirical formula ofthe compound = CH20.
Molecular formula =(CH20) n [where n is an integer]
∴ Molecular mass ofthe compound=(12 + 2 + 16)n =30 n.
Again, the vapor density (D) of the compound
= vapour density of oxygen x 2.813 = 16 x 2.813 = 45.008
∴ The molecular mass of the compound =2 x D
= 2×45.008 =90.016
Hence, 30n= 90.016 i.e., n=3
∴ Molecular formula =(CH20)3 = C3Hg03.
Question 2. A gaseous hydrocarbon contains 75% carbon by moss, 100 cm3 of this gas at STP weighs 0.072 g. What is the molecular formula of the hydrocarbon? [Weight of litre hydrogen at STP = 0.09 g]
Answer:
Given
A gaseous hydrocarbon contains 75% carbon by moss, 100 cm3 of this gas at STP weighs 0.072 g.
Hydrocarbon consists ofhydrogen and carbon only.
Let, in the hydrocarbon, the mass of carbon = 75 g The mass ofhydrogen = ( 100- 75) g = 25 g Ratio of the number of carbon (C) atoms and hydrogen (H) atoms in the compound, C: H \(=\frac{75}{12}: \frac{25}{1}\)
or, C: H =6.25: 25 =1:4
∴ The empirical formula of the hydrocarbon = CH4
∴ Its molecular formula = (CH4)n
Molecular mass of hydrocarbon = (12 + 4)n = 16n
Vapor density (D) of hydrocarbon
\(=\frac{\text { mass of } 100 \mathrm{~cm}^3 \text { hydrocarbon at STP }}{\text { mass of } 100 \mathrm{~cm}^3 \text { hydrogen at STP }}\) \(=\frac{0.072}{0.009}=8\)Molecular mass hydrocarbon =2×0 =2×8 =16
So, 16n = 16 or, n = 1
Molecular formula ofthe hydrocarbon = (CH4)1 = CH4
Question 3. A compound on analysis gives the following percentage composition: K=31.83, CI= 28.98, and 0=39.19. Find the molecular formula of the compound if its molecular mass is 122.5
Answer:
Given
A compound on analysis gives the following percentage composition: K=31.83, CI= 28.98, and 0=39.19.
Empirical formula of the compound = KC103, molecular formula =(KC103)n
Molecular mass = n X (39 + 35.5 + 48) = 122.5 x n 122.5 x n = 122.5 or, n = 1
So, the molecular formula ofthe compound = KC103.
Question 4. 0.93 g of a compound containing C, H, and N, on C : H: N = burning, produces 2.64 g C02 and 0.63 g HzO. In another experiment, 0.186 g of that compound yields 24.62 cm3 of nitrogen at 1 atmospheric pressure at 27°C. Molecular weight ofthe compounds 93. What is its molecular formula?
Answer:
Given
0.93 g of a compound containing C, H, and N, on C : H: N = burning, produces 2.64 g C02 and 0.63 g HzO. In another experiment, 0.186 g of that compound yields 24.62 cm3 of nitrogen at 1 atmospheric pressure at 27°C. Molecular weight ofthe compounds 93.
Molecular masses: C02 =44 and H20 = 18
Quantity of carbon in 44g of C02 = 12 g
Quantity ofcarbon in 2.64g of C02= \(=\frac{12 \times 2.64}{44}=0.72 \mathrm{~g}\)
Again, quantity of H2 in 18g of H20 =2g
Quantity of H2 in 0.63g of H2O \(=\frac{2}{18} \times 0.63=0.07 \mathrm{~g}\)
Therefore, 0.93g ofthe compound contains 0.72g carbon and 0.07g hydrogen.
In another experiment, it was found that the volume of N2 produced at 1 atm pressure and 27°C temperature from 0.186g of that compound is 24.62 cm3.
Let, the volume ofthe gas at STP be V2 cm3.
As given in the question,
P1 = 1 atm, Kj = 24.62cm3, P2 = 1 atm
T1 = (273 + 27) = 300 K, T2 = 273 K, V2 = ?
we know \(\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\)
∴ \(V_2=\frac{P_1 V_1 T_2}{P_2 T_1}=\frac{1 \times 24.62 \times 273}{1 \times 300}\) = 22.4 cm3
1 mol or 22400 cm3 of nitrogen at STP weighs 28 g
∴ 22.4 cm3 of nitrogen at STP weighs \(\frac{28 \times 22.4}{22400}\) = 0.08g
So, the mass of nitrogen obtained from 0.186 g of the compound = 0.028 g
∴ Mass of nitrogen obtained from 0.93 g of compound
⇒ \(=\frac{0.028 \times 0.93}{0.186}=0.14 \mathrm{~g}\)
Hence, the mass of C, H, and N in 0.93 g of the compound is 0.72, 0.07, and 0.14 g respectively.
In the given compound,
Percentage of C by mass =(0.72/0.93) x 100 =77.42,
percentage of by mass = (0.07/0.93) X 100 = 7.52,
percentage ofN by mass = (0.14/0.93) x 100 = 15.05 .
The ratio of the number of atoms of C, H, and N,
⇒ \(C: H: N=\frac{77.42}{12}: \frac{7.52}{1}: \frac{15.05}{14}\)
= 6.451:7.52:1.075 =6:7:1 [dividing by the lowest number 1.075 ]
∴ Empirical formula compound = C6H7N
∴ Molecular formula =(C6H2N)n [where n is an integer]
∴ Molecular mass = (6 X 12 + 7 x 1 + 1 x 14)/t = 93 n
According to the given condition, 93n = 93 n = 1
Therefore Molecular formula of compound =(C6H7N)1 = CgH7N
Question 5. A compound composed of carbon, hydrogen, and chlorine contains C = 10.04% and Cl = 89.12%. The vapor density of the compound is 59.75. Determineitsmolecular formula.
Answer:
Given
A compound composed of carbon, hydrogen, and chlorine contains C = 10.04% and Cl = 89.12%. The vapor density of the compound is 59.75.
Amount of hydrogen in the compound = 100- (10.04 + 89.12) = 0.84% Ratio of number of atoms of carbon (C), hydrogen (H) and chlorine (Cl) in the compound,
\(\begin{aligned}
\mathrm{C}: \mathrm{H}: \mathrm{Cl} & =\frac{10.04}{12}: \frac{0.84}{1}: \frac{89.12}{35.5} \\
& =0.836: 0.84: 2.510 \approx 1: 1: 3
\end{aligned}\) dividing by the lowest number 0.836 ]
∴ Empirical formula compound = CHC13
∴ Molecular formula ofthe compound = (CHC13)n
Molecular mass ofthe compound = (12 +l + 3x 35.5)n = 119.5n
Again, vapour density (D) ofthe compound =59.75
Molecular mass (Af) = 2 x D = 2 x 59.75 = 119.5
Hence, 119.5n = 119.5
∴ n = 1
Molecular formula of compound = (CHC13)1 = CHC13
Question 6. lg of phosphorus on combustion produces 1.77g oxide. What is the empirical formula of the compound? If the vapour density ofthe compound is 110, what is its molecular formula?
Answer: Quantity of phosphorus (P) in 1.77g of oxide =1 g The percentage-mass of phosphorus in its oxide
⇒ \(=\frac{1}{1.77} \times 100\) = 46.49 and that of oxygen in the oxide
= 100-56.49 =43.51
The ratio of the number of atoms of P and O in 1 molecule of the oxide
\(P: O=\frac{56.49}{31}: \frac{43.51}{16}\)
=1.822: 2.719
=1: 1.492 [dividing by the lowest number]
=2:3 [to get the lowest whole number, the
the ratio is multiplied by 2]
1.24 X 100
= 40%
∴ The empirical formula of compound = (P203)n, where n is an integer.
So, molecular mass of compound =(2 x 31 + 3 X 16)n = 110 n
Now, the vapour density (D) ofthe oxide = 110
Therefore The molecular mass (M) ofthe oxide = 2 X D
= 2 x 110 = 220
So, HOn = 220
∴ n =2
Thus, molecular formula ofthe compound = (P203)2 or P4Og
Question 7. The empirical formula of a gaseous compound is CH2C1.0.12 g ofthe compound occupies 37.20 cm3 at 105°C 12 1 16 Empirical formula of compound = CH20 temperature and 768 mmHg pressure. Find the molecular formula of the compound.
Answer:
Given
The empirical formula of a gaseous compound is CH2C1.0.12 g ofthe compound occupies 37.20 cm3 at 105°C 12 1 16 Empirical formula of compound = CH20 temperature and 768 mmHg pressure.
The volume of 0.12 g of the compound is 37.20 cm3 at 105°C and 768 mm pressure. Let, the volume of 0.12 g ofthe compound be V cm3 at STP.
⇒ \(\text { So, } \frac{768 \times 37.20}{(273+105)}=\frac{760 \times V}{273} \quad \quad V=27.15 \mathrm{~cm}^3\)
Again, mass of 27.15 cm3 of gaseous compound at STP = 0.12 g
Therefore Mass of 22400 cm3 i.e., 1 mol ofthe compound at STP
⇒ \(=\frac{0.12 \times 22400}{27.15}=99 \mathrm{~g}\)
[Since the volume of 1 gram-mole of gas at STP = 22400 cm3 ] The molecular mass ofthe compound = 99 Now, the empirical formula ofthe compound = CH2C1.
The molecular formula of the compound = (CH2Cl)n.
[where n is a whole number]
Its molecular mass = (12+1×2 + 35.5)n = 49.5n
So, 49.5n = 99 or, n = 2
Molecular formula ofthe compound =(CH2C1)2 = C2H4C12
Question 8. An organic compound contains C, H, and O as its constituents. On heating in the absence of air, 3.10 g of this compound, produces 1.24g of carbon. But if 0.5 g of the compound is burnt in the presence of air, 0.3 g of H2O is formed. 0.05 gram-mole of the compound contains 4.8 g of oxygen. What is the molecular formula ofthe compound?
Answer:
Given
An organic compound contains C, H, and O as its constituents. On heating in the absence of air, 3.10 g of this compound, produces 1.24g of carbon. But if 0.5 g of the compound is burnt in the presence of air, 0.3 g of H2O is formed. 0.05 gram-mole of the compound contains 4.8 g of oxygen.
3.10 g of the compound when heated in the absence of air, produces 1.24 g of carbon.
∴ Carbon content of the compound \(=\frac{1.24 \times 100}{3.1}=40 \%\)
Again, 0.5 g ofthe compound yields 0.3 g of H20
Now, amount of hydrogen in 18 g of H20 = 2 g
∴ 0.3 g of H2O contains,\(=\frac{2 \times 0.3}{18}\) = 0.033 g
∴ Hydrogen content ofthe compound \(=\frac{0.033 \times 100}{0.5}=6.6 \%\)
∴ Oxygen content ofthe compound = 100- (40 + 6.6) = 53.4%
The ratio of the number of atoms of C, H, and O in a molecule ofthe compound
⇒ \(\begin{aligned}
& =\frac{40}{12}: \frac{6.6}{1}: \frac{53.4}{16} \\
& =3.33: 6.6: 3.33=1: 2: 1
\end{aligned}\)
∴ The empirical formula of the compound = CH2O
Let, the molecular formula of the compound be (CH2O)n, where n is an integer.
So, molecular mass of the compound = (12 + 2 + 16)n =30n
∴ Its gram-molocular mass = 30n g
Mass of O2 present In 1 gram-mole ofthe compound = 16n g. Now, most of 02 In 0.05 gram-mole ofthe compound =4.0 g. Mass of 02 in 1 gram-mole of compound = 96 g.
Hence, 16 n – 96 i.e., n = 6.
∴ Molecular formula = (CH20)G = C6H1206.
Question 9. A hydrocarbon contains 10.5 g of carbon and lg of hydrogen. The weight of 1 liter of the hydrocarbon at 127°C and atm pressure is 2.8 g. Determine the molecular formula ofthe compound.
Answer:
Given
A hydrocarbon contains 10.5 g of carbon and lg of hydrogen. The weight of 1 liter of the hydrocarbon at 127°C and atm pressure is 2.8 g.
Let, V Lbe the volume of the gas at STP
Thus \(\frac{1 \times 1}{(273+127)}=\frac{V \times 1}{273}\)
therefore V=0.6825l
Now, the mass of 0.6825 L of hydrocarbon at STP = 2.8 g
Therefore Mass of 22.4 L of hydrocarbon at STP= \(\frac{2.8 \times 22.4}{0.6825}=91.9 \mathrm{~g}\)
Therefore Molecular mass ofthe compound = 91.9
The ratio of the number of atoms of C and H in a molecule of the compound \(=\frac{10.5}{12}: \frac{1}{1}\) = 0.875:1 =1:1.143 =7:8
[subsequently dividing by the smallest number and then multiplying by 7]
∴ The empirical formula ofthe compound = C7H8
∴ Molecular formula ofthe compound = (C7H8)n
∴ Molecular mass = (7 X 12 + 8 x l)n =92 n
Hence, 92n = 91.9 \(\text { or, } n=\frac{91.9}{92} \approx 1\)
Molecular formula of the compound =(C7H8)1 = C7H8
Question 10. Combustion of 0.2 g of a monobasic organic acid produces 0.505 g C02 and 0.0892 g HzO. 0.183 g of the above acid requires 15 cm3 of (N/10) NaOH solution for complete neutralization. Determine the molecular formula ofthe organic acid.
Answer:
Given
Combustion of 0.2 g of a monobasic organic acid produces 0.505 g C02 and 0.0892 g HzO. 0.183 g of the above acid requires 15 cm3 of (N/10) NaOH solution for complete neutralization.
15 cm3 0.1(N)NaOH= 0.183g of acid
Therefore 1000 cm3 1(N) NaOH \(\equiv \frac{0.183 \times 1000}{15 \times 0.1}\) =122 g of acid.
So, the equivalent mass of acid = 122
As the acid is monobasic, its molecular mass will be equal to its equivalent mass.
∴ The molecular mass ofthe acid =122
Again, 0.2 g of the acid on combustion produces 0.505 g of C02 and 0.0892 g of H20.
∴ 0.505 g of C02 contains = \(=\frac{12 \times 0.505}{44}\) = 0.1377g carbon
[ 44 g of C02 contains 12 g carbon] and the amount of hydrogen present in 0.0892 g of \(\mathrm{H}_2 \mathrm{O}=\frac{2 \times 0.0892}{18}\)
= 9.911 X 10-3 g [ V 18 g of H20 contains 2 g of H2].
Therefore 0.2 g of the acid contains 0.1377 g of carbon and 9.911 x 10-3 g of hydrogen.
Therefore In the acid, mass percent of C \(=\frac{0.1377 \times 100}{0.2}\) = 68.85
and mass percent of H \(=\frac{9.911 \times 10^{-3} \times 100}{0.2}=26.2\)
Therefore Percentage of O in the acid = 100- (68.25 + 4.95) = 26.2
∴ The Ratio of the number of atoms of C, H, and O in 1 molecule ofthe acid, C : H: O
\(=\frac{68.85}{12}: \frac{4.95}{1}: \frac{26.2}{16}\)
= 5.7375 : 4.95: 1.6375
= 3.5: 3.02: 1 a: 7: 6: 2
[Multiplication by 2 gives whole numberration]
Therefore Empirical formula ofthe acid = C7Hg02
Let the molecular formula of the acid be (C7H602)
Molecular mass ofthe acid =(7xl2 +6xl + 2x 16)n
= 122 n
Therefore 122n = 122/122 or, n = = 1
∴ Molecular formula of the acid = (C7H602)1 = C7H602
Chemical Calculations Based On Chemical Equations: Stoichiometry Calculations based on the quantitative relationship between the reactants [substance(s) participated in a chemical reaction] and products [substance(s) produced in the chemical reaction] in terms of their mole numbers, masses, and volumes in any chemical transformation, are known as stoichiometry.
Now, the quantity of reactants or products is expressed in terms of mass or volume (if gaseous). Thus in any chemical reaction, there exists three types of relationship between the reactants and the products, viz.
- Mass-mass
- Mass-volume and
- Volume volume.
Naturally, there can be three possible modes of calculations on the basis of chemical equations. These are
Calculations involving mass-mass relationship: In these calculations, the mass of product(s) formed from a given; mass of reactant(s) or the mass of reactant(s) required to produce a certain mass of product(s) can be determined. For example, carbon burns produce carbon dioxide gas.
⇒ \(\mathrm{C}(12 \mathrm{~g})+\mathrm{O}_2(32 \mathrm{~g}) \rightarrow \mathrm{CO}_2(44 \mathrm{~g})\)
Evidently, 44 parts by mass of C02 are produced from 12 parts by mass of C, or in other words, 12 parts by mass of C is to be burnt to obtain 44 parts by mass of C02.
Calculations involving mass-volume relationship: In these calculations, any unknown volume of product that is produced from a given mass of reactant or any unknown mass of reactant required to obtain a known volume of product can be determined. For example, when KC103 is heated, oxygen gas (O2) is liberated.
⇒ \(\underset{245 \mathrm{~g}}{2 \mathrm{KClO}_3} \longrightarrow 2 \mathrm{KCl}+\underset{3 \times 22.4 \mathrm{~L} \text { (at STP) }}{30_2}\)
So, 67.2 L of 02 at STP is produced from the decomposition of 245 g of KC103, or in other words, 245 g of KCIO3 is required to yield 67.2 L of Oz at STP.
Calculations involving volume-volume relationship: If both the reactants and products are in the gaseous state, such calculations are done.
For example, carbon monoxide on burning produces carbon dioxide.
⇒ \(\underset{2 \text { volume }}{2 \mathrm{CO}}+\underset{\text { 1 volume }}{\mathrm{O}_2} \longrightarrow \underset{2 \text { volume }}{2 \mathrm{CO}_2}\)
The above equation shows that under the same conditions of temperature and pressure, 2 volumes of CO combine with 1 volume of O2 to produce 2 volumes of CO2.
Some Significant Information Regarding Chemical Calculations
During chemical reactions, reactant molecules interact with each other in a simple ratio of whole numbers.
The product molecules also bear a simple whole-number ratio with the reactant molecules.
The whole numbers representing the moles of reactants and products involved in a chemical equation are called stoichiometric coefficients.
The simplest whole number ratio of moles reactants and products involved in the reaction is called stoichiometric ratio.
Density (d)= \(\frac{\text { mass of the substance }(\mathrm{m})}{\text { volume of the substance }(\mathrm{V})}\)
Relative Density \(=\frac{\text { mass of the substance }}{\text { mass of equal volume of water at } 4^{\circ} \mathrm{C}}\)
In the CGS unit, relative density or specific gravity of the substance = density of the substance So in the CGS unit, the mass of a substance
=relative density of the substance x the volume of the substance
Mass of 1 L hydrogen gas at STP = 0.089 g
Molecular mass (M) of any gas =2 X vapor density (D)
Mass of 1 L of any gas at STP
= vapour density ofthe gas x mass of1 L ofhydrogen gas
at STP = vapour density of the gas x 0.089 g
Percentage: The percentage of a constituent in a mixture refers to the amount of that constituent by parts present in 100 parts of the mixture.
For a solid mixture, percentage denotes percentage by mass. For example, an alloy of copper contains 70% copper. It means that 70 parts by mass of copper are present in 100 parts by mass ofthe alloy.
For a liquid mixture or solution, the percentage is expressed in terms of mass or volume. In other words, the percentage indicates parts by mass of the substance dissolved in 100 parts by mass or 100 cm3 of the liquid or solution. For example, 10% H2S04 by mass means.
10g of H2S04 is dissolved in 100 g of H2S04 solution. Again, 10% H2S04 by volume means 10g of H2S04 is dissolved in 100 cm3 of H2S04 solution.
For a gas mixture, percentage means the percentage by volume.
In all chemical calculations, similar types of units are used.
The general method of calculation on the basis of chemical equations involves the following steps:
A balanced chemical equation is to be written first.
The relative number of moles of the relative masses (gram-atomic or gram-molecular masses) of gaseous reactants and products are written below their formulae.
Relative volumes (in multiples of 22.4 L at STP) of gaseous reactants and products are written below their formulae.
Unitary methods applied for calculations.
Mass-mass calculations
At the time of calculations involving masses, the points mentioned below should be kept in mind.
A properly balanced equation of the corresponding chemical reaction should be written.
The respective relative masses (sometimes gram-mole or mole-number) of each of the reactants and products that appear in the equation should be written below their corresponding formulae.
The molecular mass of the reactant or product when multiplied by gram-mole or mole number, gives their relative mass.
For monoatomic substances, atomic mass when multiplied by the number of gram-atoms gives the relative mass.
From the relative masses or gram-moles of the reactants and the products in the equation, a quantitative relationship between the reactants and the products is obtained. From this relationship and the given data, the desired mass can be calculated.
Limiting reagent: Sometimes in chemical reactions, one ofthe participating reactants is taken in excess of the stoichiometric requirement according to the balanced chemical equation to ensure the completion of the reaction. In such cases, the amount ofthe product formed is divided by the reactant which is present in the least amount (i.e., the reactant which is consumed completely during the reaction).
This reagent is called the limiting reagent. It is so-called since it makes the participation of the other reactants and also the amount of products formed in the reaction limited. The reactants that are taken in excess are partially left behind at the end of the reaction.
Limiting reagent Definition: The Reagent that is present in the least proportionate amount (in the reaction mixture) and hence gets completely consumed in the chemical reaction under consideration, is called the limiting reagent.
On the basis of the amount of the limiting reagent, the product and the reactant left unreacted can be quantitatively estimated.
Example: N2 and H2 gases react to form NH3 gas.
The corresponding reactions: N2 + 3H2→NH3 According to the above equation, 1 mol of N2 reacts with 3 mol of H2 to produce 2 mol of NH3.
Now, let us consider that the reaction has been initiated by taking 2 mol of N2 and 4 mol of H2. According to the above equation, for 2 mol of N2, 6 mol of H2 is required.
Here, only 4 mol of H2 has been used. So, the limiting reagent, in this case, is H2.
Here, the amount ofthe product is to be calculated on the basis ofthe amount of H2. According to the equation, 4 mol of H2 given \(\left(\frac{2}{3} \times 4\right)=\frac{8}{3}\) mol of NH3.
Again, 4/3 mol of H2 combine with| mol of N2.
∴ At the end of the reaction, amount of N2 left unreacted \(=\left(2-\frac{4}{3}\right)=\frac{2}{3} \mathrm{~mol} \text {. }\)
Numerical Examples
Question 1. How many grams of carbon is to be burnt to produce 33g of CO2?
Answer: Carbon burns in oxygen to produce C02 gas.
Reaction: C (12g) + O2 (32g)→CO2 (44g)
The reaction shows that for the production of44g of CO2,
Amount of carbon required = 12 g
∴ For the production of 33 g of CO2, the amount of carbon required
⇒ \(=\frac{12 \times 33}{44}=9 \mathrm{~g}\)
Question 2. Find the mass of CaO obtained by heating 100 g of a reactant which sample of CaC03 is 95%pure.
Answer: The thermal decomposition of CaCO3 into CaO and CO2 can be represented by the equation—
∴ \(\begin{array}{cc}
\mathrm{CaCO}_3(s) & \mathrm{CaO}(s) \\
(40+12+48) \mathrm{g} & (40+16) \mathrm{g} \\
=100 \mathrm{~g} & =56 \mathrm{~g}
\end{array}\)
According to the given condition, CaCO3 is 95% pure. So, 100 g of the sample contains 95 g of pure CaCO3.
The above reaction shows that the amount of CaO that can be obtained from 100 g of pure CaCO3 = 56 g
Amount of CaO that can be obtained from 95 g pure
⇒ \(\mathrm{CaCO}_3=\frac{56}{100} \times 95=53.2 \mathrm{~g}\)
∴ A sample of 100 g of CaC03 which is 95% pure, on heating, produces 53.2 g of CaO.
Question 3. Calculate the volume of C02 (at STP) that can be obtained from 2kg CaC03.
Answer:
CaC03 decomposes as follows:
⇒ \(\begin{array}{cc}
\mathrm{CaCO}_3(s) \longrightarrow & \mathrm{CaO}(s)+\mathrm{CO}_2(\mathrm{~g}) \\
100 \mathrm{~g} & 22.4 \mathrm{~L}(\text { at STP) }
\end{array}\)
So at STP volume of C02(at STP) obtained from 2 kg or 2000 g of CaC03
\(=\frac{22.4}{100} \times 2000=448 \mathrm{~L}\)
Question 4. How much KC103 must be heated to produce as much oxygen as that would be obtained from 200g of HgO? [Hg= 200.5,K= 39]
Answer: HgO when heated produces O2
⇒ \(\underset{(2 \times 216.5) \mathrm{g}}{2 \mathrm{HgO} \longrightarrow 2 \mathrm{Hg}}+\underset{\mathrm{O}_2}{32 \mathrm{~g}}\)
Therefore Amount of 02 obtained from 200g of HgO \(=\frac{32 \times 100}{2 \times 216.5} \mathrm{~g}\)
Preparation of O2 from KC103 \(\begin{aligned}
& 2 \mathrm{KClO}_3 \\
& (2 \times 122.5) \mathrm{g}
\end{aligned} \longrightarrow 2 \mathrm{KCl}+{ }_{96 \mathrm{~g}}^{3 \mathrm{O}_2}\)
96 g 02 is obtained by heating 2 x 122.5 g of KC103
therefore \(\frac{32 \times 200}{2 \times 216.5} \text { g O }_2\) O2 Is obtained by heating
⇒ \(\frac{2 \times 122.5}{96} \times \frac{32 \times 200}{2 \times 216.5} \mathrm{~g}=\) 37.72g of KCLO3
Question 5. A solution of nitric acid contains 60% nitric acid. The specific gravity of the solution is 1.46. How many grams of HN03 solution will be required to dissolve 5 g of copper oxide?
Answer:
The reaction between CuO and HN03 takes place as per the following equation—
⇒ \(\begin{aligned}
& \mathrm{CuO}+2 \mathrm{HNO}_3 \longrightarrow \mathrm{Cu}\left(\mathrm{NO}_3\right)_2+\mathrm{H}_2 \mathrm{O} \\
& (63.5+16)=79.5 \mathrm{~g} \quad 2(1+14+48)=126 \mathrm{~g} \\
&
\end{aligned}\)
∴ Amount of HN03 required to dissolve 79.5 g CuO = 126g
∴ Amount of HNO3 required to dissolve 5 g of CuO
⇒ \(=\frac{126}{79.5} \times 5=7.924 \mathrm{~g}\)
As given in the problem, 100 cm3 HNO is a solution containing 60 g of pure HN03.
The specific gravity of IIN03 = 1.46
Mass of100 cm3 HN03 solution = 100 x 1.46 = 146 g
Thus, 60 g of HN03 is present in 146 g IINO., solution.
∴ 7.924 g HN03 is present in \(\frac{146 \times 7.924}{60} \mathrm{~g}\) g of HNO3 solution /.e., 19.282 g ofHN03 solution.
19.282 g HN03 solution will be required for the dissolution of 5 g of CuO
Question 6. How much water will be produced when the electric spark is passed through a mixture of 20 g ofhydrogen and 200g of oxygen? What amount of oxygen will remain unreacted?
Answer: The corresponding reaction is:
⇒ \(2 \mathrm{H}_2+\mathrm{O}_2 \longrightarrow 2 \mathrm{H}_2 \mathrm{O}\)
In this reaction, 4 g of H2 produces 36 g of water.
⇒ 20 g of H2 will produce \(\frac{36 \times 20}{4}\)= 180 g of water
Again, 4 g of H2 reacts with 32 g of 02
20 g of H2 reacts with \(\frac{32 \times 20}{4}\) = 160 g of 02
⇒ Amount of O2 left unreacted = (200- 160)g = 40 g
Question 7. An astronaut needs 34 g of sucrose per hour to maintain his physical strength. What quantity of oxygen should he carry if he has to stay 1 day in the spacecraft?
Answer:
Given
An astronaut needs 34 g of sucrose per hour to maintain his physical strength.
The energy obtained due to the oxidation of sucrose serves as the source of energy for the astronaut. The oxidation reaction of sucrose is-
\(\begin{gathered}\mathrm{CaCO}_3+2 \mathrm{HCl} \longrightarrow \mathrm{CaCl}_2+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O} \\
2 \mathrm{~mol} \\
2(1+35.5)=73 \mathrm{~g}
\end{gathered}\)
Amount of sucrose required per day =34×24 = 816g
[since Requirement of sucrose per hour= 34 g]
According to the reaction, the amount of 02 required for the oxidation of 342 g of sucrose = 384 g Amount of 02 required for the oxidation of 816 g of sucrose
\(=\frac{384 \times 816}{342}=916.2 \mathrm{~g}\)
Therefore, the astronaut will have to carry 916.2 g of 02 if he has to stay in the spacecraft for 1 day.
Question 8. 25.4g of I2 and 14.2g of Cl2 react together to form a mixture of IC1 and IC13. What is the ratio of the number of moles of IC1 and IC13 in the product mixture?
Answer:
Given
25.4g of I2 and 14.2g of Cl2 react together to form a mixture of IC1 and IC13.
25.4 g of I2 \(=\frac{25.4}{254}=\) 0.1 mol and 14.2 g of Cl2 \(=\frac{14.2}{71}\)
= 0.2 mol [ v = 254, = 71 ]
The reaction between, and Cl2 is—
⇒ \(\underset{1 \mathrm{~mol}}{\mathrm{I}_2}+\underset{2 \mathrm{~mol}}{2 \mathrm{Cl}_2} \longrightarrow \underset{1 \mathrm{~mol}}{\mathrm{ICl}}+\underset{1 \mathrm{~mol}}{\mathrm{ICl}_3}\)
The reaction indicates that 1 mol of I2 and 2 mol of Cl2 react together to form 1 mol of each IC1 and ICI3.
So, 0.1 mol of, and 0.2 mol of Cl2, will react together to form 0.1 mol of IC1 and 0.1 mol of IC13.
The ratio of number of moles of IC1 and IC13 in the resultantmixture= 0.1: 0.1 =1:1
Question 9. 1L of a sample of hard water contains 1 mg of each of CaCÿ and MgCl2. Express the hardness of the sample in ppm, in terms of CaC03.
Answer:
⇒ \(\begin{aligned}
1 \mathrm{mg} \mathrm{CaCl}_2 & \equiv \frac{1 \times 10^{-3}}{111} \mathrm{~mol} \mathrm{CaCO}_3 \\
& =\frac{10^{-3} \times 100}{111} \mathrm{~g}=0.9009 \times 10^{-3} \mathrm{~g} \mathrm{CaCO}_3 \\
1 \mathrm{mg} \mathrm{MgCl}_2 & \equiv \frac{1 \times 10^{-3}}{95} \mathrm{~mol} \mathrm{CaCO}_3=\frac{10^{-3} \times 100}{95} \mathrm{~g}^2
\end{aligned}\)
∴ Amount of CaC03 equivalent to (1 mg of CaCl2 + 1 mg ofMgCl2) = (0.9009 + 1.052) x 10-3 g = 1.9529 X 10-3 g
Hardness of water \(=\frac{1.9529 \times 10^{-3}}{1000} \times 10^6=1.9529 \mathrm{ppm}\)
(1 Lwater = 1000 g water; density of water =lg-mL-1)
Question 10. What amount of calcium oxide will react with 852g of P4 O10?
Answer: The reaction of P4O10 with CaO is
⇒ \(\begin{aligned}
& 6 \mathrm{CaO}+\mathrm{P}_4 \mathrm{O}_{10} \longrightarrow 2 \mathrm{Ca}_3\left(\mathrm{PO}_4\right)_2 \\
& 6 \times 56 \mathrm{~g} \quad 284 \mathrm{~g}
\end{aligned}\)
284 g P4O10 reacts with 6 x 56 g of CaO
So, 852 g P4 O10 reacts with \(\frac{6 \times 56 \times 852}{284}\) =1008 g of CaO.
Question 11. What percent by mass of lead nitrate [Pb(N03)2] is reduced when heated strongly?
Answer:
⇒ \(\begin{array}{cl}
2 \mathrm{~Pb}\left(\mathrm{NO}_3\right)_2 \longrightarrow & 2 \mathrm{PbO}+4 \mathrm{NO}_2 \uparrow+\mathrm{O}_2 \uparrow \\
2(207+2 \times 14+6 \times 16) & 2(207+16) \\
=662 \mathrm{~g} & =446 \mathrm{~g}
\end{array}\)
On strong heating, Pb(N03)2 loses its mass as N02 and 02 escape out as gases, and only PbO is left behind as solid residue (yellow colored).
The above equation shows that 446 g of PbO is left behind as residue when 662 g of Pb(N03)2 is strongly heated.
∴ Loss in mass = (662- 446) g = 216 g.
∴ Percentage of loss by mass \(=\frac{216}{662} \times 100=32.62\)
Question 12. When a mixture of KI and KC1 is heated repeatedly with H2S04, iodine escapes completely and K2S04 is produced quantitatively. In the case of such a mixture, it is observed that the mass of K2S04 is equal to the mass of the mixture of K3 and KC1 taken. What is the ratio ofthe masses of KI and KC1 in this mixture?
Answer: Let, the masses of KI and KC1 in the mixture be x g and y g respectively. Now, the reactions ofKI and KC1 with H2S04 are as follows
⇒ \(\begin{aligned}
& 2 \mathrm{KI}+2 \mathrm{H}_2 \mathrm{SO}_4 \longrightarrow \mathrm{K}_2 \mathrm{SO}_4+\mathrm{I}_2+\mathrm{SO}_2+2 \mathrm{H}_2 \mathrm{O} \\
& 2 \times(39+127) \quad 174 \mathrm{~g} \\
& =332 \mathrm{~g} \\
&
\end{aligned}\)
Therefore \(x \mathrm{~g} \text { KI produces }\left(\frac{174}{332} \times x\right) \text { g of } \mathrm{K}_2 \mathrm{SO}_4\)
⇒ \(\begin{aligned}
& \underset{2 \times(39+35.5)}{2 \mathrm{KCl}}+\mathrm{H}_2 \mathrm{SO}_4 \longrightarrow \underset{2}{\mathrm{~K}_2 \mathrm{SO}_4+2 \mathrm{HCl} \uparrow} \\
& =149 \mathrm{~g}
\end{aligned}\)
∴ y g KC1 produces \(\left(\frac{174}{149} \times y\right)\)g of K2SO4
∴ Total amount of K2S04 produced from x g of KI and y g of KCl
\(=\frac{174 \times x}{332}+\frac{174 \times y}{149}\)As given in the question \(\frac{174 x}{332}+\frac{174 y}{149}=x+y\)
\(\text { or, } \quad \frac{158}{332} x=\frac{25}{149} y \text { or, } \frac{x}{y}=\frac{25 \times 332}{158 \times 149} \text { or, } x: y=1: 2.836\)∴ The ratio ofthe masses of KI and KCl in the mixture is 1: 2.836
Question 13. How much of 5% impure NaN03 and 98% H2S04 will be required to produce 7.5kg nitric acid by the chemical reaction between them?
Answer:
The reaction between NaN03 and H2S04 is—
⇒ \(\begin{aligned}
& 2 \mathrm{NaNO}_3+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{Na}_2 \mathrm{SO}_4+2 \mathrm{HNO}_3 \\
& 2(23+14+48)=170 \mathrm{~g} \quad(2+32+64)=98 \mathrm{~g} \quad(2 \times 63)=126 \mathrm{~g} \\
&
\end{aligned}\)
7.5 kg or 7500 g HN03 is produced from
⇒ \(\frac{170}{126}\) X 7500 = 10119 g ofpure NaN03
Therefore Amount of impure NaN03 required to produce
7.5kg HN03 = 10119 =10651 g = 10.651 kg
7.5 kg or 7500 g HN03 is produced from
7500 =5833.33 g ofpure H2S04.
∴ Amount of 98% H2S04 required to produce 7.5 kg of HN03 =\(\frac{100}{98} \times\) 5833.33 g = 5952 g = 5.952 kg
Question 14. 3 g of HCl is present per liter of gastric juice produced in the human body. If a person produces 2.5 L gastric juice per day, then how many antacid tablets are required to neutralize HCI produced per day? [Assume that each tablet contains 400mg of Al(OH)3]
Answer:
Given
3 g of HCl is present per liter of gastric juice produced in the human body. If a person produces 2.5 L gastric juice per day,
1 L gastric juice contains 3 g of HCI Molecular mass of P4Og = 4 x 31 + 6 X 16
Therefore 2.5 L gastric juice contains (3 X 2.5)g = 7.5HCL
The reaction between Al(OH)3 and HCI is as follows—
⇒ \(\begin{array}{cc}
\mathrm{Al}(\mathrm{OH})_3 & +3 \mathrm{HCl} \\
\begin{array}{c}
1 \mathrm{~mol} \\
(27+3 \times 17)=78 \mathrm{~g}
\end{array} & \begin{array}{c}
3 \mathrm{~mol} \\
(3 \times 35.5)
\end{array}=106.5 \mathrm{~g}
\end{array}\)
Evidently, 106.5gofHCI is neutralised by78gof Al(OH)3
Therefore 7.5 g HCI is neutralised by \(\frac{78 \times 7.5}{106.5}\) = 5.493 g
= 5493 mg AL(OH)3
Now, 400 mg of Al(OH)3 is present in 1 tablet.
Therefore 5493 mg Al(OH)3 will be present in \(=\frac{1 \times 5493}{400}\)
=13.73 14 tables.
∴ 14 tablets are needed to neutralize HCI produced per day.
Question 15. (C2P4)n is a polymeric substance where n is a large number. It is prepared by polymerization in the presence of a sulfur catalyst. The final product is found to contain 0.012% of S. Find the value of n if the polymeric molecule contains three S-atoms.
Answer:
Given
(C2P4)n is a polymeric substance where n is a large number. It is prepared by polymerization in the presence of a sulfur catalyst. The final product is found to contain 0.012% of S.
Since 1 molecule of the polymer contains 3 atoms of S, 1 gram-mole of the polymer should contain 3 gram-atoms or 3 x 32 g of sulfur.
Now, 0.01 2 g of sulfur is present in 100 g of polymer.
therefore 3 x 32 g of sulphur is present in \(\frac{100 \times 3 \times 32}{0.012}\)
= 8 x 10s g of polymer
Therefore Molecular mass of the polymer = 8 x 105
Again, the molecular formula of the polymer = (C2F4)n
∴ Molecular mass =n(2 x 12 + 4 x 19) = 100n
or, 100n =8 x 105
∴ n = 8000
Question 16. Calculate the number of moles of NaOH required to neutralize the solution produced by dissolving l.lg P406 in water. Use the following reactions:
Answer:
⇒ \(\begin{aligned}
& \mathrm{P}_4 \mathrm{O}_6+6 \mathrm{H}_2 \mathrm{O} \rightarrow 4 \mathrm{H}_3 \mathrm{PO}_3 \\
& 2 \mathrm{NaOH}+\mathrm{H}_3 \mathrm{PO}_3 \rightarrow \mathrm{Na}_2 \mathrm{HPO}_3+2 \mathrm{H}_2 \mathrm{O}
\end{aligned}\)
Molecular mass of P4Og = 4 x 31 + 6 X 16 = 124 + 96 = 220 g-mol-1
Number of moles of P4Og \(=\frac{1.1 \mathrm{~g}}{220 \mathrm{~g} \cdot \mathrm{mol}^{-1}}=\frac{1}{200}\)
4 mol H3PO3 is produced by 1/200 mol P4O6
1 mol H3PO3 is produced by \(=4 \times \frac{1}{200}=\frac{1}{50} \mathrm{~mol} \mathrm{P}_4 \mathrm{O}_6\)
1/200 mol P4O6 Produce 4x 1/200 mol H3PO3
Also, 1 mol of H3PO3 requires 3 mol of NaOH.
1/50 mol H3PO3 requires = 2x 1/50 =1/25 = 0.04 mol NaOH
Mass-volume calculations
In a chemical reaction, if any substance (reactant or product or both) exists in the gaseous state, then the following procedure of calculation is considered.
Important points, relevant to this type of calculation, are given below—
A properly balanced equation representing the chemical reaction should be written first.
The relative mass (or mole number) ofthe solid reactant or product is to be written under each formula.
Molecule mass multiplied by mole number gives the relative mass. In the case of monoatomic substances, relative mass is the product of atomic mass and the number of gram-atoms.
The amount of a gaseous substance is generally expressed by its volume. If the volume at STP is not given, then the volume at STP can be calculated as follows-
⇒ \(\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\)
The volume of 1 gram-mole or molar volume of any gas at STP = 22.4L.
If the conditions of the gaseous reaction (i.e., temperature and pressure) are not mentioned, then the reaction is assumed to have occurred at STP.
Mass of 1 L of gas at STP = 0.089 g.
Mass of 1L of a gas at STP = vapor density x 0.089 g.
At any given temperature and pressure, the mass of a certain volume of gas, or the volume ofthe gas from its mass, can be calculated with the help ofthe equation:
Where w and M are the mass and molecular mass ofthe gas respectively. The relation between the volume or mass of the reactant and the volume or mass ofthe products can be determined from a chemical equation. The value of an unknown quantity can also be determined from this obtained data.
Numerical Examples
Question 1. A balloon of 1000 L capacity is to be filled up with hydrogen gas at 30°C and 750mm pressure. What amount of iron will be required to generate the required volume of hydrogen?
Answer:
Given
A balloon of 1000 L capacity is to be filled up with hydrogen gas at 30°C and 750mm pressure.
Let 1000 L H2 gas which is required to fill up the balloon of 1000L capacity occupy V L at STP (at given condition).
∴ \(\frac{750 \times 1000}{(273+30)}=\frac{760 \times V}{273}\)
∴ \(V=\frac{750 \times 1000 \times 273}{760 \times 303}=889.13 \mathrm{~L}\)
Generally, H2 gas is produced by reacting iron with steam (H20). (Consumption of Fe will be more acid is used).
⇒ \(\begin{gathered}
3 \mathrm{Fe}+4 \mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{Fe}_3 \mathrm{O}_4+4 \mathrm{H}_2 \\
(3 \times 55.85)=167.55 \mathrm{~g}
\end{gathered}\)
The reaction shows 4×22.4L of H2 gas at STP is produced from 167.55 g of Fe.
889.13L of H2 gas at STPis produced from
⇒ \(\frac{167.5}{4 \times 22.4} \times 889.13=1662.15 \mathrm{~g} \text { of } \mathrm{Fe}\)
1662.15 g Fe is required to produce the desired volume of H2.
Question 2. The volume of oxygen liberated at 26°C and 714mm pressure due to the thermal decomposition of xg of KC103 and collected over water is 760mL. What is the value of x? [Given that aqueous tension at 26°C = 26mm; K = 39;Cl =35.5; O =16]
Answer:
Given
The volume of oxygen liberated at 26°C and 714mm pressure due to the thermal decomposition of xg of KC103 and collected over water is 760mL.
Actual pressure of 02 =(714- 26) = 688mm.
Let the volume ofthe given oxygen gas at STP be V L.
⇒ \(\quad \frac{688 \times 760}{(273+26)}=\frac{760 \times V}{273}\)
Or, \(V=\frac{688 \times 760 \times 273}{299 \times 760}\)
= 628.17mL = 0.62817L
The reaction of thermal decomposition of KC103 is—
⇒ \(\begin{array}{cc}
2 \mathrm{KClO}_3 \longrightarrow 2 \mathrm{KCl} & +3 \mathrm{O}_2 \\
2 \mathrm{~mol} & 3 \mathrm{~mol} \\
2(39+35.5+48)=245 \mathrm{~g} & 3 \times 22.4 \mathrm{~L}(\mathrm{STP})
\end{array}\)
Now, the mass of KC103 required to produce 3 x 22.4 L
of 02 at STP =245 g
⇒ At STP, KCIO3 is required to produce 0.62817 L of 02
⇒ \(=\frac{245 \times 0.62817}{3 \times 22.4}=2.29 \mathrm{~g}\)
⇒ x= 2.29
Question 3. What volume of gas will be formed at 523K and 1 atm pressure by the explosive decomposition of 5g of ammonium nitrate, according to the given equation? 2NH4N03(s) = 2N2(g) + O2(g) + 4H2Q(g)
Answer:
⇒ \(\begin{array}{cccc}
2 \mathrm{NH}_4 \mathrm{NO}_3(s) & 2 \mathrm{~N}_2(g) & \mathrm{O}_2(g) & +4 \mathrm{H}_2 \mathrm{O}(\mathrm{g}) \\
2 \mathrm{~mol} & 2 \mathrm{~mol} & 1 \mathrm{~mol} & 4 \mathrm{~mol} \\
2(14+4+14+48) & 2 \times 22.4 \mathrm{~L} & 22.4 \mathrm{~L} & 4 \times 22.4 \mathrm{~L} \\
=160 \mathrm{~g} & \text { (at STP) } & \text { (at STP) } & \text { (at STP) }
\end{array}\)
⇒ Total volume ofthe gases produced by the decomposition Of 160g of NH4NO3 = 7 X 22.4 L at STP.
Let, 7 x 22.4 L ofthe gas at STP occupy a volume of V L at 523K and 1 atm pressure.
⇒ \(\frac{1 \times(7 \times 22.4)}{273}=\frac{1 \times V}{523}\)
⇒ \(V=\frac{1 \times 7 \times 22.4 \times 523}{1 \times 273}=300.39 \mathrm{~L}\)
Thus, at 523K and 1 atm pressure, 160 g of NH4N03 produces 300.39L of gas.
At 523 K and1 atm pressure, 5 g ofNH4N03 produces
⇒ \(=\frac{300.39 \times 5}{160}=9.387 \mathrm{~L} \text { of gas. }\)
Question 4. Ignition of a wooden match stick involves the combustion of P4S3 in the oxygen of the air to produce a white smoke of P4O10 and gaseous sulfur dioxide (S09). Calculate the volume of SO., formed at 27°C and 770mm Hg pressure from the combustion of 0.0546 g of P4S3. [P= 31, S = 32, 0 = 16]
Answer:
Given
Ignition of a wooden match stick involves the combustion of P4S3 in the oxygen of the air to produce a white smoke of P4O10 and gaseous sulfur dioxide (S09).
The equation for combustion reaction:
⇒ \(\mathrm{P}_4 \mathrm{~S}_3+8 \mathrm{O}_2 \rightarrow \mathrm{P}_4 \mathrm{O}_{10}+3 \mathrm{SO}_2\)
The molecular mass of P4S3 = 4×314-3×32 = 220.
Now, 220 g of P4S3 produces 3 x 22.4 L of S09 at STP.
At STP, 0.0546g of P4S3 produces \(\frac{3 \times 22.4}{220} \times 0.0546\)
= 0.0166L of S02
Let. 0.0166 L of S09 at STP occupy a volume of PL at 27°C and 770 mmHg pressure.
⇒ \(\frac{770 \times V}{(273+27)}=\frac{760 \times 0.0166}{273} \text { or, } V=\frac{760 \times 0.0166 \times 300}{770 \times 273}\)
V = 0.018 L
Therefore, the volume of S09 formed from the combustion of 0.0546 g of P4S3 at 27°C and 770 mm Hg is 0.018 L
Question 5.5 g of a pure sample of FeS reacts with dil. H9S04. H9S gas produced is then completely burnt in the air. Find the volume of SO, thus obtained, measured at 25°C and 750mm pressure of Hg.
Answer: FeS reacts with H9S04 to liberate HH2S gas—
⇒ \(\begin{array}{cc}
\mathrm{FeS}+\mathrm{H}_2 \mathrm{SO}_4 \longrightarrow \mathrm{FeSO}_4+\mathrm{H}_2 \mathrm{~S} \\
1 \mathrm{~mol} \\
\begin{array}{c}
1 \mathrm{~mol} \\
(55.85+32)=87.85 \mathrm{~g}
\end{array} & 22.4 \mathrm{~L}(\text { at STP })
\end{array}\)
Now, 87.85 g of FeS produces 22.4 L of H2S at STP.
⇒ 5.5 g of FeS produces \(\frac{22.4 \times 5.5}{87.85}\) = 1.402 L of H2S at STP.
Reaction takes place when H and S burn in the air.
So at STP, 2 x 22.4 L of H2S produces 2 x 22.4 L of S02.
At STP, 1.402 L of H9S produces 1.402 L of S02.
Let, 1.402 L of S02 at STP occupies a volume of VL at 25°C and 750 mm pressure of Hg.
Thus, \(\frac{V \times 750}{(273+25)}=\frac{1.402 \times 760}{273}\)
Hence, at 25°C and 750 mm pressure, the volume of S02 produced = 1.550 L.
Question 6. 1.78 L of chlorine gas at STP is prepared by using 40% HC1 by weight according to the following reaction— Mn02 + 4HCl→MnCl2 + 2H20 + Cl2. Find the volume of hydrochloric acid and mass of Mn02 required to produce this amount of chlorine gas. [Specific gravity of the HC1 solution= 1.12]
Answer: The corresponding ingreaction is:
⇒ \(\begin{array}{ccr}
\mathrm{MnO}_2+4 \mathrm{HCl} \longrightarrow \mathrm{MnCl}_2+2 \mathrm{H}_2 \mathrm{O}+\mathrm{Cl}_2 \\
1 \mathrm{~mol} & 4 \mathrm{~mol} & 1 \mathrm{~mol} \\
(54.94+32) & 4 \times 36.5 & 22.4 \mathrm{~L} \\
=86.94 \mathrm{~g} & =146 \mathrm{~g} & \text { (at STP) }
\end{array}\)
According to the reaction, the amount of Mn09 required for the production of 22.4 L of Cl9 at STP = 86.94 g.
Since Amount of Mn02 required for production of 1.78 L of Cl2 at STP \(=\frac{86.94 \times 1.78}{22.4}\)
Similarly, the amount of HC1 required for the preparation of 22.4L of Cl9 gas at STP = 146 g.
⇒ Amount of HC1 required for the preparation of 1.78 L of Cl9 gas at STP \(=\frac{146 \times 1.78}{22.4}=11.6 \mathrm{~g}\)
Now, 100 g HC1 solution contains 40 g of HC1.
∴ 11.6 g HC1 solution contains \(\frac{100 \times 11.6}{40}=29 \mathrm{~g} \text { of } \mathrm{HCl}\)
The specific gravity of the HC1 solution =1.12
Required volume ofthe HC1 solution
⇒ \(=\frac{\text { mass of acid solution }}{\text { specific gravity }}=\frac{29}{1.12}=25.89 \mathrm{~mL}\)
Question 7. If a particular HCl solution contains 22% of the acid by weight, then how much quantity of this acid solution will be required to produce 1L C02 gas at 27°C & 760mmpressure from pure CaC03?
Answer: Reaction between CaC03 and HCI is given by:
⇒ \(\begin{gathered}
\mathrm{CaCO}_3+2 \mathrm{HCl} \longrightarrow \mathrm{CaCl}_2+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O} \\
2 \mathrm{~mol} \\
2(1+35.5)=73 \mathrm{~g}
\end{gathered}\)
If the volume of 1L of C02 at27°C and 760 mm pressure is VLatSTP, the
⇒ \(\frac{760 \times 1}{(273+27)}=\frac{760 \times V}{273}; V=\frac{760 \times 273}{760 \times 300}=0.91 \mathrm{~L}\)
Now at STP, 22.4L of C02 is produced by 73 g of
At STP, 0.91L C02 is produced from \(\frac{73 \times 0.91}{22.4}=2.965 \mathrm{~g}\) of HCI solution.
Now, 22gHCI is present in lOOg of the given HCI solution
2.965 g ofHCI will be present in \(\frac{100 \times 2.965}{22}\) =13.48 g of HCL. solution.
So, 13.48 g ofthe given acid solution will be required.
Question 8. 1.5g of a mixture of CaC03 and MgCOs, ignition produces 360 mL of C02 at STP. Calculate the percentage composition of the mixture.
Answer: Let the mixture contains xg of CaC03 and (1.5- x) g of MgC03
The decomposition reactions involved are—
⇒ \(\underset{100 \mathrm{~g}}{\mathrm{CaCO}_3(s)} \underset{22400 \mathrm{~mL}(\mathrm{STP})}{\longrightarrow} \mathrm{CaO}(s)+\mathrm{CO}_2(g)\)
⇒ \(\begin{gathered}
\mathrm{MgCO}_3(s) \longrightarrow \mathrm{MgO}(s)+\mathrm{CO}_2(g) \\
84 \mathrm{~g} \\
22400 \mathrm{~mL} \text { (at STP) }
\end{gathered}\)
Volume of CO2 (at STP) Produced From xg OF CaCO3 \(=\frac{22400}{100} \times x=224 x \mathrm{~mL}\)
The volume of COz (at STP) produced from (1.5- x) g of
⇒ \(\mathrm{MgCO}_3=\frac{22400}{84}(1.5-x)=266.66(1.5-x)\)
Now according to the given data,
224x + 266.66(1.5- x) = 360 or, x = 0.9376
So, the given mixture contains: CaC03 = 0.9376 g
and MgC03 = 1.5- 0.9376 = 0.5624 g
In the mixture,
Percentage of CaC03 \(=\frac{0.9376}{1.5} \times 100=62.5 \text { and }\)
Percentage of MgCO3 \(=\frac{0.5624}{1.5} \times 100=37.5\)
Question 9. Air contains 21% of oxygen by volume. What volume of that air at 27°C and 750mm pressure of Hg will be required for the complete combustion of 60 g of a candle? The candle contains 80% of carbon and 20% of hydrogen by mass.
Answer: Amount of Cin 60 g of a candle \(=\frac{80}{100} \times 60=48 \mathrm{~g}\)
Therefore Amount of H \(=\frac{20}{100} \times 60=12 \mathrm{~g}\)
During the combustion of the candle, C and present are oxidized to give C02 and H20. The relevant reactions are
⇒ \(\begin{aligned}
& \mathrm{C}+\mathrm{O}_2 \longrightarrow \mathrm{CO}_2 ; \mathrm{H}_2+\frac{1}{2} \mathrm{O}_2 \longrightarrow \mathrm{H}_2 \mathrm{O} \\
& 12 \mathrm{~g} \begin{array}{c}
22.4 \mathrm{~L} \\
\text { (at STP) }
\end{array} \quad 2 \mathrm{~g} \quad \frac{1}{2} \times 22.4 \mathrm{~L} \text { (at STP) } \\
&
\end{aligned}\)
Therefore At STP, the volume of 02 required for combustion of 48 g of c \(=\frac{22.4 \times 48}{12}=4 \times 22.4 \mathrm{~L}\)
and volume of oxygen required for combustion of 12 g of hydrogen = \(\frac{22.4 \times 12}{2 \times 2}=3 \times 22.4 \mathrm{~L}\)
The volume of 02 at STP required for combustion of 60 g of candle = (4 x 22.4 + 3 x 22.4) = 156.8L.
Let, 156.8 L of 02 at STP occupy V volume at 27°C and 750mm pressure.
⇒ \(\quad \frac{V \times 750}{300}=\frac{156.8 \times 760}{273} \text { or, } \quad V=174.6 \mathrm{~L}\)
Volumetrically air contains 21% of 02.
Therefore Volume of air containing 174.60 of O2 =\(=\frac{100}{21} \times 174.60\)
Hence, for complete combustion of 60 g of a candle, 831.42L of air will be required at 27°C and 750mm pressure.
Volume-volume calculations
This type of calculation finds application in gaseous reactions. The reaction in which both the reactants and the products are in the gaseous state is called a gaseous reaction.
Eudiometry: A calculation involving the volumes of gases participating in a reaction is called eudiometry.
Eudiometry furnishes the following information:
- Volumetric relationship between the reactants and the products in any gaseous reaction.
- Volumetric composition of different constituents of the gas mixture produced in a gaseous reaction.
- The molecular formula of gaseous substances, particularly that of hydrocarbons.
Principle Of eudiometry: The principle of eudiometric calculations is based on Gay-Lussac’s law of gaseous volumes and Avogadro’s hypothesis. Now let us see how Gay-Lussac’s law of gaseous volumes and Avogadro’s hypothesis can be utilized in eudiometric calculations.
Example: In the reaction between hydrogen and chlorine gas, hydrogen chloride (HC1) gas is produced.
The reaction is: H2 + Cl2→2HC1
According to the equation, 1 molecule of H2 and 1 molecule of Cl2 react to form 2 molecules of HC1 gas, or 1 gram-mole of H2 and 1 gram-mole of Cl2 combine together to produce 2 gram-moles of HC1 gas.
If the volumes are measured at STP, 2 x 22.4 L HC1 gas is produced by the combination of 22.4 L of H2 and 22.4 L of Cl2 [ v molar volume of any gas at STP = 22.4 L].
The reaction can be expressed in terms of molecule gram-mole or volume in the following way:
⇒ \(\begin{array}{ccc}
\mathrm{H}_2 & \mathrm{Cl}_2 & 2 \mathrm{HCl} \\
1 \text { molecule } & 1 \text { molecule } & 2 \text { molecules } \\
1 \text { gram-mole } & 1 \text { gram-mole } & 2 \text { gram-mole } \\
1 \text { volume }(22.4 \mathrm{~L}) & 1 \text { volume }(22.4 \mathrm{~L}) & 2 \text { volume }[2 \times 22.4 \mathrm{~L}(\mathrm{STP})]
\end{array}\)
The ratio ofthe number ofmolecules or gram-mole of H2, Cl2 and HC1 is given by: H2: Cl2: HC1 =1:1:2 At STP, the ratio of the volumes of H2, Cl2 and HC1 = 22.4:22.4:2×22.4 =1:1:2
Hence, in the gaseous reaction, the ratio of the number of gram-moles (or molecules) of the reactants and products is the same as the ratio of their volumes, measured under the same conditions of temperature and pressure.
In order to compare the volumes of different appearing in the equation of gaseous reaction, the volume of 1 gram-mole of gas is often taken volume.
The actual value of volume is 22.4L at STP.
Eudiometer:
An eudiometer is an apparatus that can be used to carry out gaseous reactions and to measure the volumes of gaseous reactants and their products.
This apparatus consists of a U-shaped glass tube one end of which is closed and the other end is kept open.
Two platinum wires are fused inside the closed end of the tube and these are used for electric sparking which brings about the required reaction between the gaseous reactants.
The reaction usually occurs with a little explosion. For measuring the volumes of gases, the arm with closed end is graduated.
Before starting the experiment, the tube is filled with mercury. Then the reacting gas is introduced into the closed end through the open end of the tube by the displacement of mercury.
There is a stop-cock towards the lower part of the limb with an open end.
By removing mercury with the help of this stop-cock, the levels of mercury in both the limbs are made equal and the volume of the gas collected in the graduated closed limb is measured.
Similarly volume of each of the reacting gases, introduced one after another in the closed limb, is recorded.
Then these gases are made to react by passing an electric spark through the platinum wires. After the completion of the reaction, the tube is cooled down to room temperature.
If there is any contraction in volume due to cooling, the observed volume is recorded from the graduations in the closed limb. This contraction is known as the first contraction.
If there is a suitable absorbent for any gas in the gas mixture (For example KOH for C02, alkaline pyrogallate for 02, etc.), then it is introduced into the gas mixture.
As a result, the gas involved gets absorbed and the mixture again suffers contraction in volume. This is called a second contraction.
Tills contraction is recorded from the graduation of the From these experimental data regarding volume, audiometric calculation is done.
In this context, it is to be noted that all volumetric measurements are always made at the same temperature and pressure.
Some important points regarding calculations of audiometry:
A properly balanced equation of the gaseous reaction Is to be used.
Under similar conditions of temperature and pressure, the ratio of the volumes and number of gram-moles of gaseous reactant(s) and product(s) must be the same.
At STP, the volume of mole of any gas is equal to 22.4 L. At the time of comparing the volumes of gases, the volume of 1 gram-mole of any gas is considered to be 1 volume. At STP, ) volume = 22.4 L
The mass of 22.41. of a gas at STP expressed in grains is equal to the gram-molecular mass of the compound.
The volume of any solid or liquid reactant or product Involved in the gaseous reaction is considered to be zero.
In the absence of drastic conditions, nitrogen present in air generally does not take part in the gaseous reaction.
During eudiometric calculations, air Is considered to be a mixture of ()2 and N2 gases. (02 = 21% ; N2 = 79% )
In most cases, contraction in volume occurs on cooling the gas mixture produced In the eudiometer tube, to mom temperature. Expansion in volume may also occur ill some cases. Further, in some reactions, no perceptible.
limb, if there is any residual gas in the eudiometer rube after the first and second contraction, the volume of the gas left Is also recorded Change in volume is observed.
In the following table, some relevant observations have been recorded—

If the addition of an absorbent to u gas mixture produced in the gaseous reliction causes contraction In volume, then the gas can be Identified from the nature of the absorbent used and the volume of the gas may be determined from the magnitude of contact.
For this reason, prior to the addition of absorbent, the volume of the mixture is measured, if there is any contraction In volume after the addition of absorbent, then the volume of the gas mixture is again measured.
Ibe difference between the two readings gives the volume of the gas absorbed.
In this way, different gaseous constituents are removed one after another from the gas mixture by the successive addition of suitable absorbents and as a result, the contraction in volume in each case gives the measure of the volume of the absorbed gas concerned.
Numerical Examples
Question 1. 60 mL of a mixture of CO and H2, mixed with 40 ml, of 02, are subjected to explosion in a eudiometer tube. On cooling the gas mixture after the end of the reaction, the volume is reduced to 30 mL.
Determine the composition of the gas mixture Initially taken. [All volumes arc measured at the same temperature and pressure.
Answer: Let the volume of CO in the initial mixture = x mL
Volume of H2 = (60- x) mL
Reactions taking place in the eudiometer tube—
⇒ \(\underset{\substack{2 \text { volume } \\ x \mathrm{~mL}}}{2 \mathrm{CO}}+\underset{\substack{1 \\ \text { volume } \\ \frac{x}{3} \mathrm{~mL}}}{\mathrm{O}_2} \longrightarrow \underset{x \mathrm{~mL}}{2 \text { volume }} \underset{x \mathrm{CO}_2}{\longrightarrow}\)
Therefore Contraction in volume \(=\left(x+\frac{x}{2}-x\right)=\frac{x}{2} \mathrm{~mL}\)
⇒ \(\begin{array}{ccc}
2 \mathrm{H}_2 & +\mathrm{O}_2 & \underset{2}{2} \mathrm{H}_2 \mathrm{O} \\
2 \text { volume } & 1 \text { volume } & 2 \text { volume } \\
(60-x) \mathrm{mL} & \frac{1}{2}(60-x) \mathrm{mL} & 0
\end{array}\)
[as water vapor on cooling gets condensed, its volume is taken as zero]
Contraction in volume \(\begin{aligned}
& =\left[(60-x)+\frac{1}{2}(60-x)-0\right] \\
& =\frac{3}{2}(60-x) \mathrm{mL}
\end{aligned}\)
Before the reaction, the volume of the mixture = volume of the mixture of CO and Il2 + volume of 02 = (60 + 40) = 100 mL Volume of the mixture after the reaction = 30 mL.
Therefore Contraction in volume in the reaction =100- 30 = 70 mL
From reactions 1 and 2, the total contraction in volume.
⇒ \(\left[\frac{x}{2}+\frac{3}{2}(60-x)\right]=(90-x) \mathrm{mL} \quad \text { or, } 90-x=70 x=20 \mathrm{~mL}\)
In the initial mixture, volume of CO = 20 mL and volume of H2 =(60-20) = 40mL.
Question 2. 30 mL of a mixture of CO and C02, mixed with 10 mL of oxygen, was exploded by an electric spark. The gas mixture produced was mixed with KOH solution and thoroughly shaken.
5mL of oxygen was left behind. What was the composition of the original mixture? [Volume was measured at STP.
Answer: Let. the volume of CO in 30 ml, in the mixture = xmL.
∴ Volume of carbon dioxide (C02) = (30 – x) ml.
⇒ \(\begin{aligned}
& \text { Reaction involved- } \mathrm{CO}+\frac{1}{2} \mathrm{O}_2 \longrightarrow \mathrm{CO}_2 \\
& 1 \text { volume } \frac{1}{2} \text { volume } 1 \text { volume } \\
& x \mathrm{~mL} . \quad \frac{x}{2} \mathrm{~mL} . \quad x \mathrm{~mL} . \\
&
\end{aligned}\)
The reaction shows that the volume of ()., used \(=\frac{x}{2}\) mL
Volume of O2 used = volume of O2 taken – volume of O2 unused =(10-5) = 5ml..
Therefore \(\frac{x}{2}=5\)
Hence, the volume of CO in the original mixture = 10 ml.
Volume of C02 in the mixture = (30- 10) = 20 ml.
Question 3. 1 L of a mixture of CO and C02, when passed through a red hot tube containing charcoal, tire volume becomes 1.6L. All volumes are measured under the same conditions of temperature and pressure. Find the Composition of the mixture.
Answer: Volume of the mixture of CO and CO., 11.
Let, the volume of CO= xl
∴ Volume of CO2= (l-x)I.
The reaction involved CO2 +C→2CO
The volume of the mixture after passing over carbon = 1.6 L
∴ Total volume of CO =[x + 2(1 – x) ) = (2- v)L
According to the given data, 2 -x = 1.6 or, x = 0.4
∴ Volume Of Co In1 L Mixture = 0.4 L = 400 Ml And Volume
Of CO2 In 1 L Mixture = 0.6 L = 600 Ml.
Question 4. A mixture contains CO, CH4, and nitrogen. 25 cm3 of the mixture on oxidation in the presence of excess oxygen, resulted in a decrease in volume by 16 cm3. A further contraction of 17 cm3 was observed when the residual gas was treated with a KOH solution. What was the composition of the original gaseous mixture of 25 cm3 volume? [All volumes are measured at the same temperature and pressure.]
Answer: In the oxidation reaction, CO2 is obtained from CO while CH4 produces CO2 and water vapor but N2 remains unaffected by the process.
Let, the volume of CO in the original mixture = x cm3 and the volume of CH4 in the original mixture =y cm3
∴ Volume of N2 = [25- (x + y)] cm3
The reactions involved in this case are
⇒ \(\begin{aligned}
& \mathrm{CO}+\frac{1}{2} \mathrm{O}_2 \longrightarrow \mathrm{CO}_2 \\
& 1 \text { volume } \frac{1}{2} \text { volume } 1 \text { volume } \\
& x \mathrm{~cm}^3 \quad \frac{x}{2} \mathrm{~cm}^3 \quad x \mathrm{~cm}^3 \\
&
\end{aligned}\)
Therefore Contraction in volume \(=\left[\left(x+\frac{x}{2}\right)-x\right]=\frac{x}{2} \mathrm{~cm}^3\)
⇒ \(\begin{aligned}
& \mathrm{CH}_4+2 \mathrm{O}_2 \longrightarrow \mathrm{CO}_2+2 \mathrm{H}_2 \mathrm{O} \\
& 1 \text { volume } 2 \text { volume } 1 \text { volume } 0 \\
& y \mathrm{~cm}^3 \quad 2 y \mathrm{~cm}^3 \quad y \mathrm{~cm}^3 \quad 0 \\
&
\end{aligned}\)
[Water vapor condenses to liquid on cooling. So, its volume is taken as zero.]
Contraction in volume this reaction = (y + 2y- y) = 2y cm3 Total contraction in volume =\(=\left(\frac{x}{2}+2 y\right) \mathrm{cm}^3\)
This contraction is equal to the first contraction i.e., \(\frac{x}{2}+2 y=16\)
The second contraction resulted from treatment with a KOH
volume of CO2 produced by the oxidation process -gift
= 17cm3. The total volume of CO2 produced by the first and second reactions = (x + y)cm3 i.e., x + y = 17 By solving [1] & [2] we have, x = 12 and y = 5.
Volume of CO in 25 cm3 of the mixture = 12cm3 and
volume of CH4 = 5cm3
∴ Volume of N2 = [25- (12 + 5)] = 8cm3
Question 3. 100 cm3 of a mixture of CO, C2H6, and N2 is exploded in the presence of excess O2. On cooling, the observed contraction in volume and the volume of C02 formed are both equal to the volume of the original mixture. Find the volumetric composition of the original mixture.
Answer: Let, the volumes of CO, C2Hg, and N2 be x, y, and z mL respectively in the mixture.
Reactions due to explosion—
⇒ \(\begin{array}{ccc}
\mathrm{CO} & +\quad \frac{1}{2} \mathrm{O}_2 & \longrightarrow \mathrm{CO}_2 \\
\text { 1volume } & 1 / 2 \text { volume } & 1 \text { volume } \\
x \mathrm{~mL} & x / 2 \mathrm{~mL} & x \mathrm{~mL}
\end{array}\)
⇒ Contration in volume \(\left(x+\frac{x}{2}-x\right)=\frac{x}{2} \mathrm{~mL}\)
Contraction in volume in volume = \(\left(y+\frac{7 y}{2}-2 y\right)=\frac{5 y}{2} \mathrm{~mL}\)
N2 remains unaffected by the explosion.
⇒ total contraction in volume \(=\left(\frac{x}{2}+\frac{5 y}{2}\right) \mathrm{mL}\)
After the explosion, the volume of C02 produced = (x + 2y) mL. According to the given condition, observed contraction in volume after explosion = volume of the original mixture.
⇒ \(\text { i.e., } \frac{x}{2}+\frac{5 y}{2}=x+y+z\)
Volume of C02 produced due to explosion = volume of the original mixture i.e.,x + 2y = (x + y + z) or, y = z
Substituting y = z in'[l] we have,\(\frac{x}{2}+\frac{5 z}{2}\) (x + z + z)
or, x + 5z = (2x + 4z) or, x = z. Hence, x = y = z
⇒ In the mixture, vol. ofCO = vol. of C2H6 = vol. of N2.
⇒ % of each constituent in the mixture = 100/3 = 33.3
Question 6. A gaseous mixture contains 50% of H2, 40% of CH4, and 10% of 02. What additional volume of 02 at STP will be required to completely burn 200 cc of this -[2] gaseous mixture at 27°C and 750mm pressure?
Answer: In 200 cm3 of gaseous mixture, volume of H2 ,\(=\frac{50}{100} \times 200=100 \mathrm{~cm}^3 \text {; }\)
Volume of CH4 \(=\frac{40}{100} \times 200=80 \mathrm{~cm}^3 \text { and }\)
Volume Of O2 \(=\frac{10}{100} \times 200=20 \mathrm{~cm}^3\)
Now, relictions due to combustion—
⇒ \(\begin{aligned}
& 2 \mathrm{H}_2+\mathrm{O}_2 \longrightarrow 2 \mathrm{H}_2 \mathrm{O} \text { and, } \\
& 2 \text { volume } \quad \begin{array}{l}
\text { I volume } \\
100 \mathrm{~cm}^3 \\
50 \mathrm{~cm}^3
\end{array}
\end{aligned}\)
⇒ \(\begin{aligned}
& \mathrm{CH}_4+2 \mathrm{O}_2 \longrightarrow \mathrm{CO}_2+2 \mathrm{H}_2 \mathrm{O} \\
& 1 \text { volume } 2 \text { volume } \\
& 80 \mathrm{~cm}^3 \quad 160 \mathrm{~cm}^3 \\
&
\end{aligned}\)
Volume of the 02 required for complete burning of II2 and (111,| =(50+160) = 210cm3 Again, the volume of the 02 present in the mixture at 27C(! and 750 mm pressure = 20cm3.
Therefore, the additional volume of 02 required for complete combustion = (210- 20) = 190cm3.
Let, the volume ofadditional 02 he V cm3(atSTP)
⇒ \(\quad \frac{750 \times 190}{(273+27)}=\frac{760 \times V}{273}\)
Or, \(V=\frac{750 \times 190 \times 273}{300 \times 760}=170.625 \mathrm{~cm}^3\)
Therefore, the additional volume of 02 required at STP will he = 170.625cm3
Question 7. 25 ml, of a mixture containing nitrogen and nitric oxide is passed over heated copper. The volume of the gaseous mixture becomes 20mL. What is the percentage composition of the original mixture. [All volumes are measured at the same temperature and pressure.]
Answer: When the mixture of N2 and NO is passed over heated copper, NO is reduced to N2 but N2 remains unaffected in the reaction.
Let, the volume of NO in the original mixture =rmL
∴ The volume of N2 in the original mixture = (25- x) mL
⇒ \(\begin{array}{cc}
\mathrm{Cu}+ & \mathrm{NO} \longrightarrow \mathrm{CuO} \\
\begin{array}{c}
1 \text { volume } \\
x \mathrm{~mL}
\end{array} & \frac{1}{2} \mathrm{~N}_2 \\
& 1 / 2 \text { volume } \\
& x / 2 \mathrm{~mL}
\end{array}\)
Volume of the gas mixture at the end ofreduction = volume of N2 produced by reduction of NO + volume of N2 already present in the mixture \(=\left(\frac{x}{2}+25-x\right)=\left(25-\frac{x}{2}\right) \mathrm{mL} .\)
According to the given data, volume of the gaseous mixture at the end ofreduction =20 mL.
∴ \(\quad 25-\frac{x}{2}=20 \quad \text { or, } x=10\)
In the original mixture, the volume of NO = 10 ml, and the volume of N2 = (25- 1 0) =15 mL.
In the original mixture, percentage of NO \(=\frac{10}{25} \times 100=40\)
Therefore In the original mixture, percentage \(\mathrm{NO}=\frac{10}{25} \times 100=40\) and percentage of N2 \(=\frac{15}{25} \times 100=60 .\)
Question 8. Combustion of 1 volume of a compound (contains of C,H and N)In air produces 3 volumes of C02 and 4.5 volumes water vapour and 0.5 volume of N2. Calculate the molecular formula of the compound. [All volumes are measured at same temperature and pressure].
Answer: Let the formula of the compound be CxHyNz. It undergoes combustion according to the following equation
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y \mathrm{~N}_z+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \longrightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O}+\frac{z}{2} \mathrm{~N}_2 \\
& 1 \text { volume }\left(x+\frac{y}{4}\right) \text { volume } x \text { volume } \quad \frac{y_2}{2} \text { volume } \frac{z}{2} \text { volume }
\end{aligned}\)
∴ x = 3,y=9,z=l
So, formula of the compound is: C3H9N
Determination of molecular formula of gaseous hydrocarbons from eudiometry
A known volume of the gaseous hydrocarbon is mixed with an excess of oxygen and exploded by sparking inside the closed limb of an eudiometer tube.
As a result, the hydrocarbon suffers oxidation to produce carbon dioxide (C02) and steam (H2O ).
As the combustion is completed, the gas mixture is cooled down to room temperature under atmospheric pressure.
As a result, steam condenses to water (liquid) but gaseous C02 and unused Oz remain unchanged.
At this stage, there usually occurs a contraction in volume of the gas mixture. This is called first contraction.
Such contraction in volume is due to two reasons—
On coolingdown the reaction mixture, steam condenses to water (having negligible volume).
Due to reaction between hydrocarbon and oxygen, the entire amount of the hydrocarbon and a majorportion of the oxygen disappear.
Then cone. KOH (or NaOH) solution is added to the reaction mixture. As a result, a second contraction in volume takes place because the whole amount of C02 is absorbed by the KOH solution.
∴ \(2 \mathrm{KOH}+\mathrm{CO}_2 \rightarrow \mathrm{K}_2 \mathrm{CO}_3+\mathrm{H}_2 \mathrm{O} .\)
The second contraction in the volume is equal to the volume of C02 produced by the combustion of the hydrocarbon.
Now, the unused oxygen is the only gas left in the eudiometer. The volume of unused oxygen is measured by absorbing it with alkaline pyrogallatc solution.
If the above experiment is carried out with a compound composed of carbon, hydrogen and nitrogen instead ofa hydrocarbon, then the gas left in the eudiometer after second contraction consists ofnitrogen (N2) and unused oxygen (O2).
Volumes of all gases are measured at the same temperature and pressure.
The empirical formula of the hydrocarbon is determined
From the knowledge of the following quantities:
- initial volume of the gas mixture
- magnitude of first contraction and
- Magnitude Of Second Contraction.
It should be remembered that the volume of 02 consumed is equal to the volume of C02 produced.
Furthermore, the volume of hydrogen required to form water is twice the volume of 02 consumed. This hydrogen originates from die hydrocarbon.
Calculation: Volume of hydrocarbon + volume of 02 taken volume of C02 + volume of unused 02 + H20 (becomes liquid on cooling)
First contraction in volume =Total volume of gas mixture before electric sparking-total volume of gas mixture (under cold condition) after electric sparking.
= (Volume of hydrocarbon + volume of 02 taken) – (volume of C02 generated by oxidation + volume of unused oxygen) [volume ofwater (liquid) is taken as zero].
= Volume of hydrocarbon + (volume of 02 takenvolume ofunused oxygen)- volume of C02 generated by the oxidation = (Volume of hydrocarbon + volume of oxygen consumed) -volume of C02 generated.
Again, second contraction in volume = volume of C02 generated by oxidation.
So, first contraction = (volume of hydrocarbon + volume of 02 consumed)- second contraction.
1st contraction + 2nd contraction = volume of hydrocarbon + volume of 02 consumed or, volume of 02 consumed = 1st contraction + 2nd contraction- volume ofhydrocarbon.
Example Let the formula of a hydrocarbon is Cxliy. The oxidation reaction ofthe hydrocarbon is represented as—
Answer:
⇒ \(\begin{array}{ccc}
\mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 & x \mathrm{CO}_2+\frac{y_{\mathrm{H}_2} \mathrm{O}}{2} \\
\text { 1 volume } \quad\left(x+\frac{y}{4}\right) \mathrm{mol} & x \mathrm{~mol} & \frac{y}{2} \mathrm{~mol} \\
\text { 1volume } \quad\left(x+\frac{y}{4}\right) \text { volume } & x \text { volume } & 0 \\
v \text { volume } v\left(x+\frac{y}{4}\right) \text { volume } & v x \text { volume } & 0
\end{array}\)
[The volume ofwaterin the liquid state = 0 ] Hence, volume of 02 required for complete combustion of v volume ofthe hydrocarbon \(=v\left(x+\frac{y}{4}\right)\)
Now, the volume of 02 used= (firstvolume contraction + second volume contraction)-volume ofthe hydrocarbon therefore \(v\left(x+\frac{y}{4}\right)\) = first volume contraction =second volume contraction)-V
Volume of the C02 produced by complete combustion of v volume of the hydrocarbon —vx= second contraction.
Therefore, if first volume contraction and second volume contraction as well as the volume of the gaseous hydrocarbon are known, x and y may be evaluated from equations 1 and 2 and hence the formula of the hydrocarbon (CxHy) can be determined easily.
Sometimes in the determination ofmolecular formula of a gaseous hydrocarbon by eudiometric method, the empirical formula ofthe compound is obtained from the experimental results.
In such a case, the molecular formulais determined by ascertaining the vapour density or molecular mass ofthat compound.
General equations for combustion reaction of different compounds.
The problems related to determination of molecular formula of gaseous hydrocarbons with the help of eudiometry are of three types. These are explained separately with examples.
Type-1: When first and second contractions and volume of mixed oxygen are known.
Question 1. 20cm3 of a hydrocarbon mixed with 66cm3 of oxygen is exploded. After cooling the gaseous mixture, the volume becomes 56cm3. The volume of this mixture when shaken with KOH solution reduces to 16cm3. Determine the formula of the hydrocarbon. [All volumes are measured at the same temperature andpressure.
Answer: Let, the formula ofthe hydrocarbonis. The reaction takes place due to explosion—
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \longrightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O} \\
& \begin{array}{llll}
1 \text { volume } & \left(x+\frac{y}{4}\right) \text { volume } & x \text { volume } & 0 \\
20 \mathrm{~cm}^3 & 20\left(x+\frac{y}{4}\right) \mathrm{cm}^3 & 20 x \mathrm{~cm}^3 & 0
\end{array} \\
&
\end{aligned}\)
since the volume ofwaterin the liquid state is zero.
∴ First contractionin this reaction \(=\left[20+20\left(x+\frac{y}{4}\right)-20 x\right]\)
As given, first contraction = (20 + 66)- 56 = 30 cm3.
(20 + 5y) = 30 or, y = 2
Now, second contraction = (56- 16) = 40cm3 = volume of C02 produced. Therfore 20x = 40 or, x = 2
∴ The formula ofthe hydrocarbon is C2H2.
Type-2: When first and second contractions are known but volume ofmixed oxygen is unknown.
Question 2. 20cm3 ofa gaseoushydrocarbon mixed with excess of oxygen is exploded. A contraction in the volume of 30cm3 takesplace. On treating theproduced gaseous mixture with KOH solution, it suffers a further contraction of 40cm3. Determine the molecular formula of the hydrocarbon. [All volumes are measured at the same temperature andpressure.]
Answer: Let the formula ofthe gaseous hydrocarbon = Cxliy. The reaction involvingits oxidation
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \rightarrow x \mathrm{CO}_2+\frac{y_2}{\mathrm{H}_2 \mathrm{O}} \\
& \begin{array}{llll}
1 \text { volume } & \left(x+\frac{y}{4}\right) \text { volume } & x \text { volume } & 0 \\
20 \mathrm{~cm}^3 & 20\left(x+\frac{y}{4}\right) \mathrm{cm}^3 & 20 x \mathrm{~cm}^3 & 0
\end{array} \\
&
\end{aligned}\)
since water occupies negligiblevolume., i.e., zero]
In this reaction, contractionin volume.
⇒ \(\begin{aligned}
& =\left[20+20\left(x+\frac{y}{4}\right)-20 x\right] \\
& =(20+5 y) \mathrm{cm}=\text { first contraction }
\end{aligned}\)
According to the given data, first contraction = 30 cm3
20 + 5y = 30 or y=2
Again, second contraction = 40cm3 = volume of CO,. Now, from the equation, it can be seen that volume of C02 evolved = 20x cm3.
therefore 20x = 40
therefore x=2
Hence, formula ofthe hydrocarbon is C2H2.
Type-3: When first contraction and vapour density of hydrocarbon are knownbut second contraction and volume ofmixed oxygen areunknown.
Question 3. 3cm3 of a gaseous hydrocarbon is exploded with excess of oxygen. On cooling the mixture, the observed contraction is found to be 6cm3. Vapour density of the hydrocarbon is 14. What is the molecular formula ofthehydrocarbon?
Answer: Let the formula ofthe hydrocarbon be Reaction due to explosion—
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \rightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O} \\
& \text { 1 volume }\left(x+\frac{y}{4}\right) \text { volume } x \text { volume } \quad 0 \\
& \begin{array}{llll}
3 \mathrm{~cm}^3 & 3\left(x+\frac{y}{4}\right) \mathrm{cm}^3 & 3 x \mathrm{~cm}^3 & 0
\end{array} \\
&
\end{aligned}\)
Therefore On cooling, water vapour is condensed to liquid (water) whose volume is assumed to be zero.]
∴ Contractionin this reaction = \(\begin{aligned}
& =\left[3+3\left(x+\frac{y}{4}\right)-3 x\right] \\
& =3+\frac{3 y}{4} \mathrm{~cm}^3=1 \text { st contraction }
\end{aligned}\)
∴ \(3+\frac{3 y}{4}=6\) or y=4
Its molecular mass = 2 x 14 =28
since V.D = 14
The molecular formula ofthe hydrocarbon = CXH4.
Its molecular mass =(12x+ 4)
Hence, 12x+ 4 = 28 or, x = 2
Therefore, formula of the hydrocarbon is C2H4.
Question 1. A gaseous hydrocarbon of volume 10mL at STP is mixed with 80mL of 02 and burnt completely. As a result, the volume of the gaseous mixture was reduced to 70mL. On treating the obtained gaseous mixture with KOH solution, the volume becomes 50mL. Determine the molecular formula of the hydrocarbon.
Answer:
Given
A gaseous hydrocarbon of volume 10mL at STP is mixed with 80mL of 02 and burnt completely. As a result, the volume of the gaseous mixture was reduced to 70mL. On treating the obtained gaseous mixture with KOH solution, the volume becomes 50mL.
At STP, 10 mL ofa gaseous hydrocarbon mixed with 80 mL
of 02 is burnt completely. The following reaction takes place—
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \rightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O} \\
& \text { 1 volume }\left(x+\frac{y}{4}\right) \text { volume } x \text { volume } \quad 0 \\
& 10 \mathrm{~mL} \quad 10\left(x+\frac{y}{4}\right) \mathrm{mL} \quad 10 x \mathrm{~mL} \quad 0 \\
&
\end{aligned}\)
since volume of water in the liquid state is assumed to be zero.
therefore volume contraction in this reaction
⇒ \(=10+10\left(x+\frac{y}{4}\right)-10 x=10+2.5 y \mathrm{~mL}=\text { first contraction }\)
In this case, first contraction =[(10 + 80)- 70] = 20 mL
∴ 10 + 2.5y = 20 or, y = 4.
Again, second contraction= volume of C02 produced.
So, second contraction = (70- 50) mL = 20 mL 10x = 20 i.e., x = 2 Hence, the formula ofthe hydrocarbonis C2H4.
Question 2. Volume of a gaseous hydrocarbon is 1.12L at STP. Whenitis completelyburntin air, 2.2 gof C02 & 1.8 g of water are formed. Find the volume of required at STP and also mass of the compound taken. Give the molecular formula of hydrocarbon.
Answer: Let the formula of the hydrocarbon be CzUy.
The concerned reaction of combustion is—
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \longrightarrow x \mathrm{CO}_2+\underset{2}{\chi_2} \mathrm{H}_2 \mathrm{O} \\
& 1 \mathrm{~mol} \quad\left(x+\frac{y}{4}\right) \mathrm{mol} \quad x \mathrm{~mol} \quad y_2 \mathrm{~mol} \\
&
\end{aligned}\)
Now, the volume of the hydrocarbon = 1.12 L.
No. ofmoles ofhydrocarbon in 1.12L at STP \(P=\frac{1 \times 1.12}{22.4}=0.05\)
From reaction it is found that 1 mol of gaseous hydrocarbon produces x mole of C02 and y/2 mole of H20.
therefore 0.05mol ofhydrocarbon produces 0.05x mol of C02 and \(\frac{0.05 y}{2}\) mol of H2O. Now 0.05x mol of C02= 0.05* x 44g= 2.2xg of C02 and \(\frac{0.05 y}{2}\) X 18g = 0.45g mol H2O
According to the given data, 2.2x = 2.2 , 0.45y =1.8 y = 4 and x = 1; the formula ofthe hydrocarbon = CH4 ixnoi Now at STP, 1.12L ofhydrocarbon =0.05 mol
Mass ofthe hydrocarbon taken = (0.05 x 16) = 0.8 g [ v Molecular mass ofhydrocarbon, CH4 = 16]
Again, the amount of 02 required for the combustion of 1 mol of hydrocarbon \(=\left(x+\frac{y}{4}\right) \mathrm{mol}=\left(1+\frac{4}{4}\right) \mathrm{mol}=2 \mathrm{~mol}\)
O2 required for combustion of 0.05 mol ofhydrocarbon = (0.05 x 2) = O.lmol
Now, Volume of1 mol of 02 at STP = 22.4 L
Therefore Volume of 0.1 mol of 02 at STP = 2.24 L. So, the volume
of 02 required for combustion ofhydrocarbon is 2.24 L.
Question 3. 5 mL of a gas composed of hydrogen and carbon is mixed with 30mL of oxygen and exploded with electric sparking. The volume of the gas mixture, obtained by explosion is found to be 25 mL. KOH is then added to the mixture and as a result, its volume is reduced to 15 mL. The residual gas is purely oxygen. All volumes have been measured at STP. Whatis the molecular formula ofthe gas?
Answer: Assuming the formula ofthe gas to be CÿHÿ, the reaction occured during explosion is represented as
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \longrightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O} \\
& \begin{array}{cccc}
1 \text { volume } & \left(x+\frac{y}{4}\right) \text { volume } & x \text { volume } & 0 \\
5 \mathrm{~mL} & 5\left(x+\frac{y}{4}\right) \mathrm{mL} & 5 x \mathrm{~mL} & 0
\end{array} \\
& {[\text { volume of water (liquid) }=0 \text { ] }} \\
&
\end{aligned}\)
since volume of water liquid)=0]
First contraction \(=\left[5+5\left(x+\frac{y}{4}\right)-5 x\right]=(5+1.25 y) \mathrm{mL}\)
As per given data, first contraction = (5 + 30)- 25 =10 mL
Therefore 5 + 1.25y = 10 or, 1.25y = 5
Second contraction = volume of the C02 formed = 5x mL According to the given condition, second contraction = (25- 15) = 10 mL.
Therefore, 5x = 10 or, x = 2
Thus, formula oftire gaseous compound = C2H
Question 4. 10 mL of a gaseous organic compound composed of carbon, hydrogen and oxygen is mixed with 100 mL of oxygen and subjected to explosion. Volume of tire mixtureproducedby explosion when cooledbecomes 90mL. On treatment with KOH, the volume is reduced by 20mL. Mass of1 L ofgaseous organic compound is 2.053 g at STP. Determine the molecular formula.
Answer: Let, the formula ofthe organic compound =
The mass of 1 L ofgaseous organic compound at STP = 2.053 g
Mass of 22.4L ofgaseous organic compound at STP = (2.053X22.4) =45.9872 g.
The reaction involved is as follows—
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y \mathrm{O}_z+\left(x+\frac{y}{4}-\frac{z}{2}\right) \mathrm{O}_2 \rightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O} \\
& 1 \text { volume. }\left(x+\frac{y}{4}-\frac{z}{2}\right) \text { volume } x \text { volume } 0 \\
& 10 \mathrm{~mL} \quad 10\left(x+\frac{y}{4}-\frac{z}{2}\right) \mathrm{mL} \quad 10 x \mathrm{~mL} \quad 0 \\
&
\end{aligned}\)
In the reaction, volume contraction= 10 + 10 \(\left(x+\frac{y}{4}-\frac{z}{2}\right)-10 x\)
= (10 + 2.5y- 5z) mL = first contraction x mL
In this case, first contraction = [(10 + 100)- 90] = 20 mL.
Therefore, 10 + 2.5y-5z = 20 or, 2.5y-5z =10 – [l]
Again, the second contraction in the mixture =20mL
= the volume of the C02 formed. From the equation, it is observed that the volume ofthe C02 produced = lOx.
Therefore, lOx = 20 or, x = 2
Now, the molecular mass of CxHy02 =(12x+ y+ 16z)
12.v+ y + 16z =45.9072 or, 12×2+y+16z =45.9872
or, y + 16z = 21.9072
Solving equations 1 & 3 we have, y = 6 and z = 1
Therefore The formula ofthe organic compound is C2H60.
Question 5. When 3 volume of a gaseous organic compound of carbon, hydrogen & sulphur mixed with excess oxygen is exploded, 3 volumes of carbon dioxide, 3 volumes of sulphur dioxide and 6 volumes of water vapour arc produced. What is the formula of the compound?
Answer: Let, the formula of the organic compound is On combustion, the following reaction takes place—
⇒ \( \mathrm{C}_x \mathrm{H}_y \mathrm{~S}_z+\left(x+\frac{y}{4}+z\right) \mathrm{O}_2 \rightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O}+z \mathrm{SO}_2\)
⇒ \(1 \text { volume }\left(x+\frac{y}{4}+z\right) \text { volume } x \text { volume } \frac{y}{2} \text { volume } z \text { volume }\)
⇒ \(3 \text { volume } 3\left(x+\frac{y}{4}+z\right) \text { volume } 3 x \text { volume } \frac{3 y}{2} \text { volume } 3 z \text { volume }\)
According to the given condition, 3x = 3 or, x = 1; \(\frac{3 y}{2}\)
= 6 or, y = 4 and 3z = 3 or, z = 1
The formula ofthe organic compound = CH4S.
Question 6. When an acetylenic hydrocarbon, in presence of excess oxygen is exploded, it shows a contraction in volume by 50mL. A further contraction of 75mL is observed when the obtained gas mixture comes in contact with KOH solution. Determine the molecular formulaofthe compound.
Answer: Let the formula be CnH2 and its volume = x mL
Reaction of the acetylenic compound with O2 —
⇒ \(\begin{aligned}
& \mathrm{C}_n \mathrm{H}_{2 n-2}+\left(\frac{3 n-1}{2}\right) \mathrm{O}_2 \longrightarrow n \mathrm{CO}_2+(n-1) \mathrm{H}_2 \mathrm{O} \\
& 1 \text { volume }\left(\frac{3 n-1}{2}\right) \text { volume } n \text { volume } 0 \\
& x \mathrm{~mL} \quad x\left(\frac{3 n-1}{2}\right) \mathrm{mL} \quad n x \mathrm{~mL} \quad 0 \\
&
\end{aligned}\)
Contraction in volume in this reaction
⇒ \(=x+x\left(\frac{3 n-1}{2}\right)-n x=\frac{x+n x}{2}=\frac{x(1+n)}{2} \mathrm{~mL}\)
This volume contraction is equal to the first contraction of the mixture. Therefore, \(\frac{x(1+n)}{2}\) = 50 or, x+nx=100
As per given data, the second contraction of the mixture = 75 mL =the volumeofthe C02 formed. So, nx= 75
From equations 1 and 2 we have, x = 25 and from equation 2 putting x = 25 we have, n = 3.
Formula ofthe acetylenic compound = C3H6-2 = C3H4
Determination Of Molecular Formula Of Other Gaseous Compounds With The Help Of Eudiometry
Type-1: When two elementary gases of known atomicity react together to form a gaseous compound, the molecular formula of that compound may be ascertainedif the volumes of the reacting gases and the product at STP are known.
Let, 1 L of A2 gas (diatomic) and 1 L of B2 gas (diatomic) react to produce 2L of a gaseous compound X at STP. So,
⇒ \(\begin{array}{ccc}
\mathrm{A}_2 & \mathrm{~B}_2 & 2 \mathrm{X} \\
1 \mathrm{~L} & 1 \mathrm{~L} & 2 \mathrm{~L} \text { (at STP) } \\
n \text { molecules } & n \text { molecules } & 2 n \text { molecules } \\
1 \text { molecule } & 1 \text { molecule } & 2 \text { molecules } \\
1 \text { atom } & 1 \text { atom } & 1 \text { molecule } \\
& \text { [according to Avogadro’s hypothesis] }
\end{array}\)
Hence, in 1 molecule of X, 1 atom of each A2 and B2 are present.
∴ The molecular formula of the compound is AB.
Suppose, 1 L of X2 gas (diatomic) and 2L of Y2 gas (diatomic) react to produce1 L of Z gas at STP. So,
⇒ \(\begin{gathered}
\mathrm{X}_2 \\
1 \mathrm{~L}
\end{gathered}+\underset{2 \mathrm{~L}}{2 \mathrm{Y}_2} \longrightarrow \mathrm{Z} \underset{\mathrm{L}(\text { at STP) }}{\mathrm{Z}}\)
⇒ \(\begin{array}{ccc}
n \text { molecules } & 2 n \text { molecules } & n \text { molecule } \\
1 \text { molecule } & 2 \text { molecules } & 1 \text { molecule } \\
2 \text { atoms } & 4 \text { atoms } & 1 \text { molecule }
\end{array}\)
Therefore in 1 molecule of Z gas, 2 atoms of X2 and 4 atoms of Y2 are present.
Molecular formula of Z gas is X2Y4.
Type 2. If the atomicity of the elementary gases produced by the decomposition of any gaseous compound and also the volumes of the reactants and the products at STP are known, the molecular formula of the gaseous compound maybe determined.
Example: Suppose at STP, 2 L of a gaseous compound decomposes to form1 L of A2 gas and 3 L of B2 gas (both A2 and B2 are diatomic).
∴ \(\begin{gathered}
2 \mathrm{X} \\
2 \mathrm{~L}
\end{gathered} \longrightarrow \underset{1 \mathrm{~L}}{\mathrm{~A}_2}+\underset{3 \mathrm{~L}}{3 \mathrm{~B}_2}\)
∴ \(\begin{array}{ccc}
\text { 2n molecules } & \text { n molecules } & 3 \text { n molecules } \\
2 \text { molecules } & 1 \text { molecule } & 3 \text { molecules } \\
1 \text { molecule } & \frac{1}{2} \text { molecules } & \frac{3}{2} \text { molecules } \\
1 \text { molecule } & 1 \text { atom } & 3 \text { atoms }
\end{array}\)
since A2 and B2 are diatomic]
So,1 molecule of X gas contains1 atom of A2 and 3 atoms of B2 gas. Hence, the molecular formula of X gas is AB3.
Numerical Examples
Question 1. At high temperature, gaseous compound, S4N4 decomposes to produce N2 and sulphur vapour. 1 vol. of S4N4 on decomposition gives 2.5 vol. of gaseous mixture at STP. Determine formula of sulphur.
Answer: Thermal decomposition: S4N4→2N2 + sulphur vapour 1 vol. of S4N4 produces 2.5 vol. of gaseous mixture.
From the equation,itis observed that 2 volumes of N2 are produced from 1 volume of S4N4 at STP. So, during thermal decomposition at STP, volume of the sulphur vapour produced = (2.5- 2) = 0.5 volumes. At STP, decomposition of 1 volume S4N4 results in evolution of2volumes N2 and 0.5 volumes sulphur vapour.
or, 1 molecule of S4N4 on decomposition gives 2 molecules of N2 and 0.5 molecules of sulphur.
1 molecule of S4N4 produces 4 N-atoms and 4 S-atoms.
∴ In 0.5 molecule ofsulphur, number of S-atoms = 4
∴ In 1 molecule of sulphur, number ofS-atoms = 8
Therefore Formula of sulphur = S8.
Question 2. When 100mL of ozonised oxygen is shaken with turpentine oil, volume decreases by 20mL. When lOOmL of the same sample is heated, the mixture occupies a volume of 110mL. Determine the molecular formula of ozone. [All volumes are measuredunder same temperature andpressure.]
Answer: Turpentine oil when added to ozonised oxygen, absorbs ozone and oxygen is leftbehindin the mixture.
∴ Volume of ozone in 100mL ofozonised oxygen = 20 mL.
∴ Volume of oxygenin 100 mL of ozonised oxygen = 80 mL
When Ozone Mixed With O2 is heated, it undergoes thermal decomposition to produce 02.
Volume of the gas obtained by heating 100mL of ozonised oxygen 110mL of this gas, (110-80) = 30 mL of 02 have been obtained by decomposing 20 mL of ozone.
Under the same conditions of temperature and pressure, 20mL ozone produces 30mL of 02.
i.e., 2 volumes of ozone produce 3 volumes of 02.
So, 2 molecules ofozone produce 3 molecules of O2 .
∴ 1 molecule of ozone produces 3/2 molecule of oxygen or [ v oxygen is diatomic]
Hence,1 molecule ofozone contains 3 oxygen atoms.
∴ Molecular formula of ozone is 03.
Calculations Involving Reactions Occurring In Solution
Methods of expressing concentration of solutions
A solution is a homogeneous mixture of two or more substances which do not react chemically with each other.
A solution consisting of only two components is called a binary solution. Some important modes of expressing concentration ofsolutions are indicated below.
Percentage strength It is defined as the amount of solute in grams presentin lOOg of the solution.
⇒ \(\%(W / W)=\frac{\text { mass of solute }(\mathrm{g})}{\text { mass of solution }(\mathrm{g})} \times 100\)
Example: Percentage strength (W/W) of a glucose solution is 10 indicates that lOg glucose is presentin lOOg ofsolution.
Strength in gram per litre: The strength of a solution is defined as the amount of the solute in grams present per litre ofthe solution.
Unit: g/L or g/dm3, i.e., g-L-1 or g-dm-3.
Example: If 4.0g of sodium carbonate is dissolved in 1L of sodium carbonate solution, the strength of the solution willbe 4.0 g-L-1 or 4.0 g-dm-3 Temperature, strengthin g-L 1 depends on temperature.
Mole fraction: The mole fraction of any componentin the solution is equal to the number of moles of that component dividedby the total number ofmoles of all the components.
Let us consider, n2 moles of a solute dissolvedin n 4 moles of a solvent.
Mole fraction of the solute and mole fraction ofUie solvent in the solution respectively.
⇒ \(x_2=\frac{n_2}{n_1+n_2} \& x_1=\frac{n_1}{n_1+n_2}\)
⇒ \(\text { Now, } x_1+x_2=\frac{n_1}{n_1+n_2}+\frac{n_2}{n_1+n_2}=\frac{n_1+n_2}{n_1+n_2}=1\)
The sum of mole fractions ofthe components is equal to 1.
Therefore mole fraction ofthe solvent, xx = l-x2
mole fraction of the solute, x2 = 1- x1
Effect of temperature: As mole fraction is a number, it is independent oftemperature.
PPm (partspermillion): The concentration ofvery dilute solution is expressed in terms of parts of the solute by mass present in million parts by mass of the solution (or ppm), i.e., ppmx \(=\frac{\text { mass of } x}{\text { Mass of solution }} \times 10^6\)
Pollution of the atmosphere is also expressed in terms of ppm but in that case we use volumes in place of masses i.e., volume (in cm3) of the harmful gases (i.e., S02 etc.) presenti n 106 cm3 ofthe air.
Molarity (M): It is a defined as the number ofgram-moles ofthe solute presentin1 L (or, 1000 mL) of the solution.
⇒ \(\begin{aligned}
& \text { Molarity }(\mathrm{M}) \text { of solution }=\frac{\text { number of moles of solute }}{\text { volume of the solution (in } \mathrm{L})} \\
& =\frac{\text { mass of the solute }(\mathrm{g})}{\text { gram-molecular mass of solute } \times \text { volume of solution (in } \mathrm{L})}
\end{aligned}\)
Example: If three different solutions are such that1 L of each of these solutions contain 1 mol, 0.5 mol and 0.1 mol of a dissolved solute then the molarity of the solutions will be 1, 0.5 and 0.1 respectively. Similarly, if 5L of a glucose solution contains 4 gram-moles of glucose then molarity ofthe solution will be,
⇒ \(\frac{\text { number of moles of glucose }}{\text { volume of the solution }(\mathrm{L})}=\frac{4}{5}=0.8(\mathrm{M})\)
Units ofmolarity: moles perlitre (mol-L-1 ) or moles per cubic decimetre (mol-dm’3 ) [1L = 1000 cm3 = 1dm3 ]
Molar solution: A molar solution is defined as a solution, 1 L ofwhich contains 1 gram-mole ofthe solute.
Example: 1L of sulphuric acid (H2S04) solution containing 98 g (i.e., 1 gram-mole) of H2S04 is called 1(M) H2S04 solution.
Similarly, 1 L of1(M) Na2C03 solution contains 106 g (i.e., 1 gram-mole) ofsodium carbonate. Molar, semi-molar, deci-molar and centi-molar solutions are denoted by1(M
⇒ \(\left(\frac{\mathrm{M}}{2}\right),\left(\frac{\mathrm{M}}{10}\right) \text { and }\left(\frac{\mathrm{M}}{100}\right)\) respectively.
Effect of temperature on the molarity of a solution: The mass of solute is independent of temperature but the volume of solution is dependent on temperature and hence the molarity of a solution depends on temperature.
Formality (F): It is defined as the number of gramformula mass of the dissolved solute present per litre of the solution.
⇒ \(\begin{aligned}
& \text { Formality }(F)=\frac{\text { number of gram-formula mass of solute }}{\text { volume of the solution (in } \mathrm{L} \text { ) }} \\
& =\frac{\text { mass of the solute }(\mathrm{g})}{\text { gram-formula mass of solute } \times \text { volume of solution (in } \mathrm{L})}
\end{aligned}\)
Example: If1 L of a magnesium chloride solution contains 190 g of MgCl2 then this solution is said to be 2.0 formal solution (t.e., 2.0 F solution) because the formula mass of MgCl2 is 95.
Formal solution: It is defined as a solution, 1 L of which contains 1 gram-formula mass of the solute.
Example: 1 L of 1(F) solution of calcium chloride (CaCI2) contains lllg {i.e., 1 gram-formula mass) of calcium chloride.
Effect of temperature on the formality of solution: The mass of a solute is independent of temperature but the volume of solution is dependent on temperature and hence the formality of a solution depends on temperature.
Molality (m): The molality of a solution is defined as the number of gram-moles of the solute dissolved in 1000 g {i.e., 1 kg) ofthe solvent.
⇒ \(\begin{aligned}
& \text { Molality }(\mathrm{m}) \text { of solution } \\
& =\frac{\text { number of moles of dissolved solu }}{\text { mass of the solvent }(\mathrm{kg})} \\
& =\frac{\text { number of moles of solute } \times 10}{\text { mass of the solvent }(\mathrm{g})} \\
& =\frac{\text { mass of solute }(\mathrm{g}) \times 1000}{\text { gram-molecular mass of solute } \times \text { mass of solvent }(\mathrm{g})}
\end{aligned}\)
Example: If 40 g of NaOH [i.e., 1 g-mole NaOH) is dissolved in lOOOg of water, then the resulting solution is said to be 1 molal solution [i.e., l(m) solution] of NaOH. Again, dissolution of 4g ofNaOH {i.e., 0.1 g-mole NaOH) in 1000 g water gives 0.1 molal [i.e., 0.1 (m)] solution of NaOH.
Unit of molality: moles perkilogram ( mol-kg-1)
Effect of temperature on molality of a solution: As the masses of substances are independent of temperature, so the molality ofa solution does not depend on temperature.
Molal solution: A molal solution is defined as a solution, which is prepared by dissolving 1 gram-mole of solute in 1000 g of solvent.
Example: If 98 g(i.e., 1 gram-mole) ofsulphuric acid is dissolved in 1000 g of water, then a molal solution of H2S04 [i.e., l(m) H2S04 solution] is o btained.
Molal, semimolal, deci-molal and centi-molal solutions denoted by l(m)
⇒ \(\left(\frac{\mathrm{m}}{2}\right),\left(\frac{\mathrm{m}}{10}\right) \text { and }\left(\frac{\mathrm{m}}{100}\right)\)
Normality (N): Normality of a solution is defined as the number ofgram-equivalents of the solute presentin1 L (or 1000 mL) of the solution.
⇒ \(\begin{aligned}
& \text { Normality }(\mathrm{N}) \text { of a solution } \\
& =\frac{\text { number of gram-equivalents of solute }}{\text { volume of solution }(\mathrm{L})} \\
& =\frac{\text { mass of the solute }(\mathrm{g})}{\text { gram-equivalent mass of solute } \times \text { volume of solution (L) }}
\end{aligned}\)
If 1 L of H2S04 solution contains 2 gram-equivalent (i.e.,98 g) of pure H2S04 the strength of the solution will be 2(N).
Similarly, if 1 L of Na,C03 solution contains 0.1 gram-equivalent (i.e., 5.3 g) of Na2C03 the strength of the solution will be 0.1(N).
Effect of temperature on normality ofa solution: Mass of solute is independent of temperature but volume of a solution depends on it. So, normality of a solution is dependent on temperature.
Normal solution: A normal solution is defined solution, 1 L (or 1000 mL) of which contains 1 equivalent of the solute.
Examples: 1 L of a normal solution of H,S04 contains 1 gramequivalent or 49 g H2SO4 . 1 L of a normal solution of NaOH contains 1 gram-equivalent or 40 g NaOH.
Normality Equation
Let Vx mL of a solution of normality A/j be diluted to V2 mL so that its normality changes to N2. Since number of gram-equivalents of solute before and after dilution is ame, we can write, V1 X N1 =V2XN2. This is called normality equation.
Let V1 mL of a solution of compound ‘ A ’ having normality Nx reacts completely with V2 mL of a solution of compound ‘B’ having normality N2. Since the ompounds react with each other in equivalent amounts we can write, V1 X N1 = V2XN2 This is another normality equation.
Molarity equation
Let V1 mL of a solution of molarity M1 be diluted to V2 mL so that its molarity changes to M2.
Since number of gram-moles of solute before and after dilution is same, we can write, V1 X M1 = V2 X M2 This is molarity equation.
Let V1 mL of a solution of molarity be mixed with V2 mL ofanother solution (complex ofsame components) having molarity M2. If M be the molarity of the mixed solution, (V1 + V2 ) M=V1 x M1 + V2 X M2
This is a second molarity equation.
Let V1 mL of a solution of compound ‘A ‘ having molarity M2 reacts completely with V2 mL of another solution of compound having molarity M2 according to the equation: xA(aq) + yB(aq)pC(aq) + qD(aq)
It can be shown that \(\frac{V_1 \times M_1}{x}=\frac{V_2 \times M_2}{y}\)
This is a thirdmolarity equation.
Relationship between molarity and normality of the solution ofan acid ora base: Normality of solution of an acid =Molarity x Basicity ofthe acid.
Normality of a base = Molarity x Acidity ofthe base. e.g., 1 (M) H2S04 Solution = 2 (N) H2S04 solution 1 (M) Ca(OH)2 solution = 2 (N) Ca(OH)2 solution.
Normality= No. ofg- eq L-1 = Number ofmilli eq mL-1 Molarity of a solution = Number ofmoles perlitre = Number of millimoles per litre.
Numerical Examples
Question 1. NaOH is dissolved in water and the solution is madeupto 500mL. Find themolarity of solution.
Answer: Number ofmoles of NaOH = \(\frac{\text { given mass }}{\text { molecular mass }}=\frac{8}{40}=0.2\)
∴ Molarity(M) = \(=\frac{\text { number of moles of } \mathrm{NaOH}}{\text { volume of solution }(\mathrm{L})}=\frac{0.2}{0.5}=0.4\)
Question 2. The density of 3(M) solution ofNaCl is 1.25 g • cm 3. Calculate themolarityofthe solution.
Answer: Amount ofNaN03 in 1L solution=3 mol=3×58.5g
Mass of 1 L solution= 1000 x 1.25 g = 1250 g
Mass of the solvent [i.e., water) in 1L ofthe solution
= (1250-175.5)= 1074.5g
∴ Molality \(=\frac{\text { no. of moles of solute }}{\text { mass of solvent }(\mathrm{kg})}=\frac{3}{1.0745}=2.79(\mathrm{M})\)
Question 3. Calculate the volume of 0.5(M) H2S04 solution required to dissolve 0.5 g ofcopper(2) carbonate.
Answer: Reaction \(\underset{1 \mathrm{~mol}}{\mathrm{CuCO}_3}+\underset{1 \mathrm{~mol}}{\mathrm{H}_2 \mathrm{SO}_4} \rightarrow \mathrm{CuSO}_4+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}\)
∴ 1 mol CuC03 = 63.5 + 12 + 48 = 123.5 g
∴ 1 mole of H2S04 is required to dissolve 123.5 g of CuC03
∴ Amount of H2S04 required to dissolve 0.5 g of CuC03
⇒ \(=\frac{1 \times 0.5}{123.5}=0.004 \mathrm{~mol}\)
Now, 0.5 mol of H2S04 is present in 1000 mL solution.
∴ 0.004 mol H2SO4 is present \(\frac{1000 \times 0.004}{0.5}\) = 8ml. given H2S04 solution. So, volume of H2S04 required= 8 mL.
Question 4. Calculate the volume of H2 gas (at STP) liberated by thereaction ofexcessZnwith 500mL 0.5(N) H2S04.
Answer: Reaction involved \(\mathrm{Zn}+\underset{98 \mathrm{~g}}{\mathrm{H}_2 \mathrm{SO}_4} \rightarrow \underset{22400 \mathrm{~mL}}{\mathrm{ZnSO}_4}+\underset{\mathrm{H}_2}{\mathrm{H}_2}\)
H2S04 presentin 1000 mL of0.5(N) H2S04 =0.5 g-eqv.
∴ Amount of H2S04 present in 500 mL of 0.5(N) H2S0 solution \(=\frac{0.5 \times 500}{1000}\) g-equiv \(=\frac{0.5 \times 500 \times 49}{1000} \mathrm{~g}=12.25 \mathrm{~g}\)
98gH-,S04 reacts withZn to liberate 22400mLH, (STP).
∴ 12.25 g H,S04 reacts with zinc to liberate \(\frac{22400 \times 12.25}{98}=2800 \mathrm{~mL} \mathrm{H}_2 \text { (at STP) }\)
Density of 3 molal NaOH solution is 1.110 g mL-1. Calculate themolarity ofthe solution.
3 molal NaOH solution means 3 moles of NaOH are dissolvedin1 kg solvent. So, the mass ofsolution = 1000g solvent + 120g NaOH =1120g [v Molar mass of NaOH =(23 + 16 + 1) =40]
Volume of solution = \(=\frac{\text { Mass of solution }}{\text { Density of solution }}\)
or, Volume of solution \(=\frac{1120}{1.110 \mathrm{~g} \cdot \mathrm{mL}^{-1}}=1009 \mathrm{~mL}\)
∴ \(=\frac{\text { Mole of solute } \times 1000}{\text { Vol. of solution }(\mathrm{mL})}=\frac{3 \times 1000}{1009}=2.973(\mathrm{M})\)
Question 6. If 4 g of NaOH dissolvesin 36g of H2O, then calculate the mole fraction of each component in the solution. Also, determine the molarity of the solution (specific gravityofsolutionis1 g.mL-1 ).
Answer: \(n_{\mathrm{NaOH}}=\frac{4}{40}=0.1 \mathrm{~mol} ; n_{\mathrm{H}_2 \mathrm{O}}=\frac{36}{18}=2 \mathrm{~mol}\)
⇒ \(x_{\mathrm{NaOH}}=\frac{0.1}{0.1+2}=0.0476 ; x_{\mathrm{H}_2 \mathrm{O}}=\frac{2}{0.1+2}=0.9524\)
Total mass ofsolution= (4 + 36) = 40 g
Volume of solution \(=\frac{40 \mathrm{~g}}{1 \mathrm{~g} \cdot \mathrm{mL}^{-1}}=40 \mathrm{~mL}\)
Molarity \(=\frac{\text { Mole of solute } \times 1000}{\text { Vol. of solution }(\mathrm{mL})}\)
⇒ \(=\frac{0.1 \times 1000}{40}=2.5(\mathrm{M})\)
Question 7. 1L ofa (N/2) HC1 solution was heated in a beaker and it was observed that when the volume of solution got reduced to 600 mL, 3.25 g of HC1 was lost. Calculate the the normality of the resulting solution.
Answer: Normality \(\frac{\text { Mass of } \mathrm{HCl}}{\text { Equivalent mass } \times \text { Volume of solution (L) }}\)
⇒ \(\text { or, } \quad 0.5 \mathrm{eqv} \cdot \mathrm{L}^{-1}=\frac{\text { Mass of } \mathrm{HCl}}{36.5 \mathrm{~g} \cdot \mathrm{eqv}^{-1} \times 1 \mathrm{~L}}\)
Mass of HCl=0.5 eqv L-1 X 36.5 g – eqv-1 x 1L= 18.25g
Mass of HC1 left after heating = 18.25- 3.25 = 15.0 g
Volume of the resulting solution = 600ml \(=\frac{600}{1000}=0.6 \mathrm{~L}\)
therefore normality \(=\frac{15.0 \mathrm{~g}}{36.5 \mathrm{~g} \cdot \mathrm{eqv}^{-1} \times 0.6 \mathrm{~L}}=0.685\)
Long Question And Answers
Question 1. Two substances A are B are of equal masses and = 29.28 their molecular masses are in the proportion of 2:3. What is the ratio of the numbers of their molecules?
Answer:
Given
Two substances A are B are of equal masses and = 29.28 their molecular masses are in the proportion of 2:3.
From the question, MA: = 2:3; where MA and MB are the molecular masses of A and B respectively. Now, let the mass of A = mass of B = W g
Number of molecules present in gram-mole of any substance
= 6.022 x1023
Number of molecules in w g of A \(A=\frac{W}{M_A}\)= x 6.022 x 1023 and that in W g of B \(=\frac{W}{M_B}\)= x 6.022 x 1023
∴ \(\frac{\text { Number of molecules in } W \mathrm{~g} \text { of } A}{\text { Number of molecules in } W \mathrm{~g} \text { of } B}\)
\(=\frac{W}{M_A} \times 6.022 \times 10^{23} \div \frac{W}{M_B} \times 6.022 \times 10^{23}=\frac{M_B}{M_A}=\frac{3}{2}\)Question 2. Show that, the ratio ofthe masses of equal volumes of two gases at the same temperature and pressure is directly proportional to the ratio of their molecular masses.
Answer:
Let the molecular masses of two gases, A and B be MA and MB respectively, and WA, and WB be their respective masses.
Now, number of moles of gas A = WA/MA
Number ofmoleculesin gas A = \(A=\frac{W_A}{M_A} \times\) x 6.022 x 1023
Again, number ofmoles ofgas B = WB/MB
Number of molecules in gas B = x 6.022 x 1023
Since the gases (having equal volumes) are at the same temperature and pressure, according to Avogadro’s hypothesis they will contain the same number of molecules.
∴ \(\frac{W_A}{M_A} \times 6.022 \times 10^{23}=\frac{W_B}{M_B} \times 6.022 \times 10^{23} \text { i.e., } \frac{W_A}{W_B}=\frac{M_A}{M_B}\)
Question 3. Which are heavier and lighter than air: O2, CO2, CH4, NH3, Cl2 ? [Volumetric composition of air N2 = 74%, O2 = 24%, CO2=2%
Answer:
Vapour density of a gas \(=\frac{\text { molecular mass of gas }}{\text { molecular mass of air }}\)
Volumetric composition of air: \(=\frac{(28 \times 74)+(32 \times 24)+(2 \times 44)}{100}=29.28\)
Therefore, concerning air, vapor densities of
⇒ \(\begin{aligned}
& \mathrm{O}_2=\frac{32}{29.28}>1 ; \mathrm{CO}_2=\frac{44}{29.28}>1 ; \mathrm{CH}_4=\frac{16}{29.28}<1 ; \\
& \mathrm{NH}_3=\frac{17}{29.28}<1 \text { and } \mathrm{Cl}_2=\frac{71}{29.28}>1
\end{aligned}\)
Therefore Gases lighter than air are CH4 and NH2 whereas gases heavier than air are O2, CO2, and CI2.
Question 4. The mass of a sulfur atom is twice that of an oxygen atom. Hence, the vapor density of sulfur will be twice that of the vapor density of oxygen—justify’.
Answer:
Given
The mass of an S-atom is twice that of an O-atom. So, the atomic mass of sulfur is twice that of oxygen.
Now, Molar mass =n x atomic mass [n = atomicity ofmolecule] and vapor density
⇒ \(=\frac{\text { molecular mass }}{2}=\frac{n \times \text { atomic mass }}{2}\)
Therefore Vapour density of sulphur \(=\frac{n \times \text { atomic mass of sulphur }}{2}\)
Vapour density of oxygen \(=\frac{2 \times \text { atomic mass of oxygen }}{2}\)
since atomicity of oxygen molecule =2
⇒ \(\quad \frac{\text { Vapour density of sulphur }}{\text { Vapour density of oxygen }}\)
⇒ \(=\frac{n \times \text { atomic mass of sulphur }}{2 \times \text { atomic mass of oxygen }}=\frac{n}{2} \times 2=n\)
since the atomic mass of sulfur is twice of that of oxygen] or, Vapour density of sulfur =n x vapor density of oxygen.
Thus, it is quite logical to conclude that depending on the atomicity (n), the vapor density of sulfur maybe 2, 4, 6, or 8 times the vapor density of oxygen.
Question 5. How many significant figures are there in each of the following numbers:
- 0.437
- 935100
- 2.158 x 104
- 0.00839
- 207.39
- 17.00
- 2.0100 x 104
- 6.0 x 1023
- 0.00070
Answer:
- 3 significant figures (4, 3, 7);
- 4 significant figures (9, 3, 5, 1);
- 4 significant figures (2, 1, 5, 8);
- 3 significant figures (8, 3, 9);
- 5 significant figures (2, 0 7, 3, 9);
- 4 significant figures (1, 7, 0, 0);
- 5 significant figures (2, 0, 1, 0, 0);
- 2 significant figures (6, 0);
- 2 significant figures (7,0)
Question 6. Express the following up to three Significant figures:
- 4.309251
- 49.793500
- 0.005728
- 7000
- 2.67876 x 103
- The decimal equivalent of 2/3,
- one-millionth of one.
Answer: The digit in the second decimal place is increased by 1 unit and the remaining digits after that are dropped. Hence the number obtained is 4.31.
The digit in the first decimal place is increased by 1 unit and the remaining digits are dropped. The number obtained is 49.8.
The digit in the fifth decimal place is increased by 1 unit and the remaining digits are dropped. The number obtained is 0.00573.
The given no. is first expressed in the exponential form i.e. 7000 = 7.000 X 103. Then the third digit after the decimal point of the tire’s first factor is dropped. Thus the number obtained is 7.00 X 103.
The given number is in the exponential form. Thus the digit at the second decimal place is increased by 1 and all other digits of its right side are dropped. Thus the number obtained is 2.68 x 103.
The given fraction is first expressed in the decimal form. The digit at the third decimal place is increased by 1 and all other digits on its right side are dropped. This gives the number as 0.667.
The given number is first expressed in the exponential form. Then all the digits after the second decimal place are dropped. This gives the number 1.00 x 10~6, which has three significant.
Question 7. On heating of 4.9 g of KC103, 1.92g of 02 is evolved and 2.97 g of KC1 is obtained as a residue. Show that these data illustrate the law of conservation of mass.
Answer: ,\(\text { Reaction: } 2 \mathrm{KClO}_3 \rightarrow \quad \begin{gathered}
12 \mathrm{KCl}+3 \mathrm{O}_2 \uparrow \\
4.9 \mathrm{~g}
\end{gathered} \quad \begin{gathered}
2.97 \mathrm{~g} \quad 1.92 \mathrm{~g}
\end{gathered}\)
Total mass ofthe products 02 + KC1 = (1.92 + 2.97)g
= 4.89g
Difference in mass between the reactant and products
= (4.9 -4.89)g =0.01g.
This small difference is due to experimental error. Otherwise, these data illustrate the law of conservation of mass.
Question 8. 10.1 g o/HCl is mixed with 6.3 g of NaHC03. Calculate the mass ofCO2 released if the residual mixture is found to weigh 12.1 g
Answer: \(\text { Reaction: } \mathrm{HCl}+\mathrm{NaHCO}_3 \rightarrow \underset{10.1}{6.3 \mathrm{~g}} \rightarrow \underset{12.1 \mathrm{~g}}{\mathrm{NaCl}+\mathrm{H}_2 \mathrm{O}}+\mathrm{CO}_2\)
According to the law of conservation of mass, the mass of
(HC1 + NaHC03) = mass of(NaCl + H20 + C02)
or, (10.1 + 6.3)g = mass of(NaCl + H20 + C02)
or, 16.4g = 12.1g + mass of C02 /. massofC02 = 4.3g
Question 9. Calculate the number of gram-atom present in 2.1 g of nitrogen and 0.23 g of sodium.
Answer: One gram-atom of nitrogen means 14g of nitrogen.
No. of g-atom present in 2.1 g of nitrogen \(=\frac{2.1}{14}=0.15\)
Again, One g-atom of sodium means 23g of sodium
No. of g-atom present in 0.23g of Na \(=\frac{0.23}{23}=0.01\)
Question 10. Give an example of a tetra-atomic element and calculate its molecular mass.
Answer:
Tetra-atomic element: P4 molecular mass of P4 = atomic mass of P x its atomicity
= 31 x4 = 124
Question 11. ‘1-mole oxygen = 2 gram-atom oxygen’—justify.
Answer:
1 g-atom of oxygen means 16g of oxygen
∴ 2 g-atom of oxygen = 32g
∴ 1 mol oxygen = 2 g-atom oxygen [proved].
Question 12. Which one is the volume of 2.8g of ethylene gas at STP?
- 2.24L
- 22.4L
- 224 L
- 0.224 L.
Answer: Gram-molecular mass of ethylene gas = 28g
∴ At STP 28g of ethylene gas occupies a volume of 22.4 L
∴ At STP, 2.8g ethylene gas occupy \(\frac{22.4 \times 2.8}{28}=2.24 \mathrm{~L}\)
∴ The volume of 2.8g of ethylene gas at STP is 2.24L
Question 13. Which one is the volume of lg of oxygen gas at STP?
- 0.7 L
- 4.8 L
- 1.4L
- 1.2 LW.
Answer: Gram-molecular mass ofoxygen gas at STP is 32g
At STP, volume 32 g ofoxygen gas = 22.4L
At STP volume of1 g of oxygen gas \(=\frac{22.4}{32} \mathrm{~L}=0.7 \mathrm{~L}\)
Question 14. Calculate the number of O-atoms in 0.5 mol of S02.
Answer: No. ofmolecules presentin1 mol S02 = 6.022 x 1023.
Number of molecules present in 0.5 mol S02
= 0.5 x 6.022 x 1023 =3.011 x 1023
2 oxygen atoms are present in each S02 molecule.
∴ The number of oxygen atoms present in 0.5mol of S02
= 3.011 X 1023 X 2 = 6.022 X 1023
Question 15. Calculate the mass o/lmol of electrons if the mass of one electron be 9.11 x 10-31 kg.
Answer: 1 mol of electrons = 6.022 x 1023 number of electrons.
Mass of one electron = 9.11 x 10-31kg
therefore The mass of 1 mol of electrons
= 6.022 x 1023 x 9.11 x 10-31kg = 5.486 x 10-7kg
Question 16. At a temperature of 273 K and 1 atm pressure, 1 L of a gas weighs 2.054 g. Calculate its molecular mass.
Answer:
Given
At a temperature of 273 K and 1 atm pressure, 1 L of a gas weighs 2.054 g.
22.4L of any gas at STP contains 1 mol of that gas.
1L of a gas weighs 2.054g
∴ 22.4L ofthe gas weighs = 2.054 X 22.4g = 46g
∴ Molecular mass ofthe gas is 46
Question 17. At the same temperature & pressure, two flasks of equal volume contain NH3 & S02 gas respectively. Identify the flask having a greater number of molecules of gaseous substance with greater mass and a greater number of atoms.
Answer:
Given
At the same temperature & pressure, two flasks of equal volume contain NH3 & S02 gas respectively.
According to Avogadro’s hypothesis, at temperature and pressure equal volume of all gases contains an equal number of molecules.
At the same temperature & pressure, two flasks having equal vol. contain equal no. of NH3 & S02 molecules.
(H) NH3 = 17 and S02 = 64.
The flask containing S02 gas has a greater mass.
NH3 is a tetra-atomic and S02 is a triatomic molecule.
So, the flask with NH3 contains a greater no of atoms.
Question 18. Choose the correct options: The vapor density of carbon dioxide is—
- 22
- 22g.cm3
- 22g.l-1
- 44
Answer: Vapour density of a gas =1 /2 x its molecular mass
∴ Vapour density of C02 =(1/2) x 44 =22
Question 19. The equivalent mass of an element can never be zero explained.
Answer: Vapour density ofoxygen (D) = 32/2 = 16.
∴ Vapour density ofthe gaseous element =5×16 = 80.
∴ Molecular mass ofthe gaseous element = 2 x 80 = 160.
Relative atomic mass ofthe element
⇒ \(=\frac{\text { molecular mass of the element }}{\text { its atomicity }}=\frac{160}{2}=80\)
Question 20. Calculate the percentage composition (by mass) of the constituent elements of sodium sulfate (Na2S04).
Answer: Gram-molecular mass of Na2SO4 = 142g
mass% of Na in Na2SO4 = \(\frac{2 \times 23}{142} \times 100=32.39\)
mass % of S in Na2SO4 =\(\frac{32}{142}\) x 100 = 22.54
∴ mass % of O in Na2SO4 = \(\frac{64}{142}\)x 100 = 45.07
Question 21. An oxide of iron is found to contain 69.9% iron and 30.1% dioxygen (02) by mass. Calculate its empirical formula.
Answer: Fe = 69.9% and 02 = 30.1%. Now, the ratio of the number of atoms of Fe and Oin the compound, \(\text { Fe }: O=\frac{69.9}{55.85}: \frac{30.1}{16}=1.25: 1.88=1: 1.5=2: 3\)
Its empirical formula is Fe203.
Question 22. Calculate the amount of sodium hydroxide present in 100 mL of 0.1(M) NaOH solution.
Answer: 1000 ml 1 (M) NaOH solution contains 40gNaOH
Therefore 100mL 0.1 (M) NaOH solution contains
⇒ \(\frac{40 \times 100 \times 0.1}{1000} \mathrm{~g} \text { of } \mathrm{NaOH}=0.4 \mathrm{~g} \text { of } \mathrm{NaOH}\)
Question 23. 100 mL of 3 (N) Na2C03 solution is diluted to 300 mL by adding water. Calculate the normality of this solution.
Answer: Let S be the normality of the diluted solution. As given in the question, 100 x 3 = 300 x S S = 1
Question 24. Find the volume (in mL) of 0.2 (N) NaOH solution required to neutralize 25mL of 0.2(N) H2S04 solution.
Answer: Volume of H2SO4 solution V1 =25mL Normality of H2S04 solution S1 =0.2(N) Volume of NaOH solution = xmL Normality of NaOH solution S2 = 0.2(N) Now, V1S1 = V2S2 or, 25 X 0.2 = x x 0.2
∴ x =25
Question 25. Give the relation between normality & molarity of a solution.
Answer: Normality of an acid = Molarity x Basicity ofthe acid.
- Normality of a base = Molarity x Acidity of the base.
- Solution Of Warm Up Exercises
- Mixture (homogeneous);
- Mixture (homogeneous);
- Mixture (homogeneous);
- Compound;
- Mixture (heterogeneous);
- Element;
- Mixture (homogeneous);
- Mixture (heterogeneous);
- Compound;
- Mixture (homogeneous);
- Mixture (heterogenous)
Answer: Normality of an acid = Molarity x Basicity ofthe acid. Normality of a base = Molarity x Acidity of the base.
Question 26. The reactant which is entirely consumed in any reaction is known as the limiting reagent. In the reaction, 2A + 4B → 3C + 4D, if 5 moles of A react with 6 moles of B, then
- Which is the limiting reagent?
- Calculate the amount of C formed.
Answer: Reaction: 2A + 4B → 3C + 4D
- Here, 2 moles of A reacts with 4 moles of B.
- Therefore, 5 moles of A reacts with= \(\frac{4}{2} \times 5=10\) moles of B.
- But, we have 6 moles of B participating in the reaction.
- It means B is the limiting reagent.
- 4 moles of produce 3 moles of C.
- Hence, 6 moles ofB gives =(3/4) x 6 = 9/2 = 4.5 moles of C.
Question 27. A granulated sample of aircraft alloy (Al, Mg, Cu) weighing 8.72 g was first reacted with alkali and then with very dilute HC1, which left behind a residue. The residue after boiling with alkali weighed 2.10 g and the acid-insoluble residue weighed 0.69 g. What is the composition of the alloy?
Answer: Let us suppose, the amount of Al, Mg, and Cu in the sample box, y, and z g respectively.
Reactions:
⇒ \(2 \mathrm{Al}+2 \mathrm{NaOH} \stackrel{2 \mathrm{H}_2 \mathrm{O}}{\longrightarrow} 2 \mathrm{NaAlO}_2+3 \mathrm{H}_2\)
⇒ \(\mathrm{Mg}+2 \mathrm{HCl} \rightarrow \mathrm{MgCl}_2+\mathrm{H}_2, \mathrm{Cu}+\mathrm{HCl} \rightarrow \text { No reaction }\)
i.e., only Al reacts with NaOH and only Mg reacts with HC1.
Therefore + y+ z = 8.72 andy+ z = 2.10 (Residue left after alkali treatment)
z = 0.69 (Residue left after acid treatment)
therefore x = 6.62 g and y = 2.10- 0.69 = 1.41 g
∴ \(\% \text { of } \mathrm{Al}=\frac{6.62}{8.72} \times 100=75.9\)
⇒ \(\% \text { of } \mathrm{Mg}=\frac{1.41}{8.72} \times 100=16.2\)
⇒ \(\% \text { of } \mathrm{Cu}=\frac{0.69}{8.72} \times 100=7.9\)
Question 28. The equivalent mass of metal M is E and the formula of its oxide is MxOy. Show that the valency and atomic mass of metal M are 2y/x and 2yE/x respectively.
Answer:
Given
The equivalent mass of metal M is E and the formula of its oxide is MxOy.
Let the atomic mass ofthe metal M be a
So, the molecular mass ofthe compound, MxOy = ax + 16y
Therefore Equivalent mass (E) ofthe metal M in the compound
⇒ \(\mathrm{M}_x \mathrm{O}_y, \mathrm{E}=\frac{a x}{16 y} \times 8=\frac{a x}{2 y}\)
∴ Atomic mass of M (a) = 2yE/x
Hence, valency of M = \(\frac{\text { atomic mass }}{\text { equivalent mass }}\)
∴ \(=\frac{\frac{2 y E}{x}}{E}=\frac{2 y}{x}\)
The valency of an element in its oxide = 2 x the number of oxygen atoms combined with 1 the atom ofthe element.
In atoms of M combine with y no. of O-atoms.
With 1 atom of, no. of oxygen atoms combined = y/x
Valency of M = 2 x (y/x) = 2y/x
Question 29. Two gases, A and B having equal mass are kept in two separate vessels under identical conditions of temperature and pressure. If the ratio of their molecular masses is 2 : 3, find the ratio of the volumes of the vessels.
Answer:
Given
Two gases, A and B having equal mass are kept in two separate vessels under identical conditions of temperature and pressure. If the ratio of their molecular masses is 2 : 3,
Let, MA &Mfi be the molasses of A Ik B respectively.
As per given data, MA: MB =2:3
therefore \(M_A=\frac{2}{3} \times M_B\)
Here, mass of A = mass of B = Wg (say)
Therefore In Wg of each A and B, the ratio number of moles,
⇒ \(n_A: n_B=\frac{W}{M_A}: \frac{W}{M_B}=\frac{M_B}{M_A}=\frac{M_B}{\frac{2}{3} \times M_B}=\frac{3}{2}=3: 2\)
Question 30. Since under the identical conditions of temperature and pressure, the volumes of gases are directly proportional to their number of moles, [because according to Avogadro’s law, V at the same temperature and pressure.] the ratio of volumes of two gases, i.e., the ratio of volumes of two vessels, \(V_A: V_B=n_A: n_B=3: 2.\)
Answer: Here, 1 L of X combines with 2L of Y to form 1L of a gaseous compound.
Let, n be the number of molecules present per liter ofthe gas under the same temperature and pressure.
So according to Avogadro’s hypothesis, n molecules of X react with 2n molecules of Y to form n molecules of gaseous product.
i.e., 1 molecule of X + 2 molecules of Y =1 molecule of the gaseous productor, 2 atoms of X + 4 atoms of Y =1 molecule of the gaseous compound [since the reactants are diatomic]
Molecular formula ofthe compounds X2Y4.
Question 31. The vapor density of a gas at 25°C is 25. What will be its vapor density at 50°C?
Answer: Vapour density of gas \(=\frac{\text { mass of certain volume of gas }}{\text { mass of equal volume of } \mathrm{H}_2 \text { gas }}\)
[under similar conditions of temperature and pressure]
Now, an increase in temperature will result in a proportionate increase in the volume of both hydrogen and the given gas as all gases expand equally due to an equal rise in temperature irrespective of their chemical nature.
So, the vapor density ofthe gas does change with any change in temperature.
Thus, the vapor density of the gas at 50° C will be equal to the vapor density measured at 25°C. So, the vapour density ofthe gas at 50°C will also be 25.
Question 32. Do 1 mol 02 and 1 mol O signify the same quantity?
Answer: 1 mol 02 and 1 mol O do not signify the same quantity.
- 1 mol 02 =1 gram-mole 02
- = 2 gram-atom O =2×16 = 32 g oxygen.
- 1 mol O =1 gram-atom O =1 x 16 = 16 g oxygen.
Question 33. The experimental values of the vapor density of either NHÿCl or PCI- is less than that obtained from the equation D = M/2. Explain.
Answer: At the experimental temperature, both NHÿCl and PC15 undergo thermal dissociation.
⇒ \(\begin{aligned}
\mathrm{NH}_4 \mathrm{Cl}(\text { vap }) & \rightleftharpoons \mathrm{NH}_3(g)+\mathrm{HCl}(g) \\
\mathrm{PCl}_5(\text { vap }) & \rightleftharpoons \mathrm{PCl}_3(g)+\mathrm{Cl}_2(g)
\end{aligned}\)
Due to such dissociation, the number of molecules in NH4C1 vapor or PC15 vapor increases.
This causes an increase in volume because, at a certain temperature and pressure, the volume of a gas is proportional to die number of molecules. As die mass is fixed, an increase in volume causes a decrease in the value of vapor density measured experimentally.
Question 34. There are two natural isotopes of hydrogen ( 1H > 99 %; 2H < 1 ). Chlorine also has two natural isotopes (35C1 = 75%; 37C1 = 25%). How many different molecules of HC1 are possible? Arrange them in the decreasing order of their relative abundance.
Answer: The possible HC1 molecules are 1H35Cl,2H35C1, 1H37CI and 2H37CI. Since the abundance of the 1IT isotope of 35 hydrogens and ‘ Cl isotope of chlorine are maximum, the 1 35 1 abundance of IT Cl will also be Since the abundance of 2H and 37C1 isotopes is much less, 2I I37C1 will be the least abundant.
So, the order of different molecules of HC1 arranged in the decreasing order of their relative abundance will be—
\({ }^1 \mathrm{H}^{35} \mathrm{Cl}>{ }^1 \mathrm{H}^{37} \mathrm{Cl}>{ }^2 \mathrm{H}^{35} \mathrm{Cl}>{ }^2 \mathrm{H}^{37} \mathrm{Cl}\)Question 35. At the same temperature and pressure, a gaseous hydride contains twice of Its own volume of hydrogen. The vapor density of the hydride is 14. What is its molecular formula?
Answer: Let, at the same temperature and pressure, 2 volume of hydrogen is present in 1 volume of hydride.
According to Avogadro’s hypothesis, at the same temperature and pressure, if no. of molecules is present in 1 volume of hydride, the no. of hydrogen molecules in this hydride is 2n.
Therefore 4 hydrogen atoms or 2 molecules of hydrogen are present in 1 hydride molecule,| v hydrogen is diatomic.
Let, the number of carbon atoms in a hydride molecule he x. Therefore, the molecular formula of the hydride is CX.H4.
Molecular mass of the hydride = 2 x 14 = 28
Now, the molecular mass of CVH,1 = 12x + 4
Therefore, 12x + 4 = 28 or,x=2
Therefore Molecular formula of the hydride C2H4.
Question 36. According to Avogadro’s hypothesis, at the same temperature and pressure equal volume of all guscs contains an equal number of molecules. Can it be concluded from the given statement that all molecules have equal volume”?
Answer: Avogadro’s hypothesis states that dial equal volumes of all gases under similar conditions of temperature and pressure, contain equal numbers of molecules. This does not signify that the actual volumes of one molecule of different gases are the same.
Experimental studies have shown that the molecular diameters of different gases differ from each other depending on the molecular composition of the gas.
in fact, the total volume of any gas (i.e., the volume of the container in which the gas is enclosed) is equal to the sum of the volume of all gas molecules and the volume of the empty space which is available for free movement of the gas molecules.
Question 37. A mixture of formic acid and oxalic acid is heated with concentrated H2SO4. The gas evolved is collected and treated with KOH solution. The volume of the solution decreases by 1/H th of Its original volume. Find the molar ratio of the two acids in the original mixture.
Answer: Let, the mixture of a moles of oxalic acid and b moles of formic acid be heated with concentrated H2S04.
The concerned reactions are as follows :
⇒ \(\begin{aligned}
& (\mathrm{COOH})_2 \stackrel{\mathrm{H}_2 \mathrm{SO}_4 / \text { heat }}{\longrightarrow} \mathrm{CO}(\mathrm{g})+\mathrm{CO}_2(\mathrm{~g})+\mathrm{H}_2 \mathrm{O}(I) \\
& a \mathrm{~mol} \quad a \mathrm{~mol} \quad a \mathrm{~mol} \\
&
\end{aligned}\)
⇒ \(\underset{b \mathrm{~mol}}{\mathrm{HCOOH}} \stackrel{\mathrm{H}_2 \mathrm{SO}_4 / \text { heat }}{\longrightarrow} \underset{b \mathrm{~mol}}{\mathrm{CO}}(g)+\mathrm{H}_2 \mathrm{O}(l)\)
Total number of moles ofthe gaseous mixture
= number of moles of CO + number of moles of C02
=(a + b) mol + a mol = (2a + b) mol
Now, KOH absorbs only moles of C02, and the volume
ofthe solution decreases by l/6th of its initial volume.
According to Avogadro’s law
⇒ \(\frac{\text { number of moles of } \mathrm{CO}_2}{\text { number of moles of gas mixture }}=\frac{a}{(2 a+b)}=\frac{1}{6}\)
or, 6a = 2a + b or, 4a – b or, b/a = 4
Therefore Molar ratio of formic acid and oxalic acid =4:1
Question 38. Taking N2 and 02 as the main components of air (79% N2, 21% 02 by volume), find the average molecular mass of air.
Answer: For a mixture of different gases, the average molecular mass ofthe mixture is taken as—
Average molecular mass = \(\Sigma x_{\mathrm{i}} M_{\mathrm{i}}=x_{\mathrm{N}_2} M_{\mathrm{N}_2}+x_{\mathrm{O}_2} M_{\mathrm{O}_2}\)
where xN and xQ are mole fractions of N2 and 02 and MNÿ and MQ are their molecular masses.
At the same temperature and pressure, equal volumes of all gases contain an equal number of moles, and their molar ratio is the same as the ratio of their volumes.
Therefore XN2 = 0.79,xO2 = 0.21, Also MN2 = 28u, MO2= 32u
ATherefore Average molecular mass ofair = (0.79 x 28 + 0.21 x 32)u
= (22.12 + 6.72)u = 28.84U
Question 39. A gaseous hydrocarbon needs 6 times more volume of oxygen (02) than itself for complete oxidation. It produces 4 times more C02 (by volume) than it’s own. What is the formula of the hydrocarbon?
Answer: Equation of combustion:
⇒ \(\mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \rightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O}\)
1 volume ofhydrocarbon reacts with \(\left(x+\frac{y}{4}\right)\) volumes of oxygen to produce 2x volumes of C02.
Now, x + y/4=6 or, 4x+y=24
Given, x = 4. Therefore, y = 8
∴ The formula of the hydrocarbon is C4H8.
Question 40. What is the equivalent mass of KH(I03)2 oxidant in the presence of 4.0 (N) HC1 when IC1 becomes the reduced form? [where K = 39.0 andI = 127.0]
Answer: 103 is present in KH(I03)2 as I03
Let, the oxidation state of i be x.
Therefore x-6 = -l or, x = +5. \(\begin{array}{cc}
+5 & +1 \\
\mathrm{KH}\left(\mathrm{IO}_3\right)_2 & 2 \mathrm{ICl}
\end{array}\)
Therefore Decreasein oxidation state =(10-2) = 8
Equivalent mass of KH(103)2 \(=\frac{\text { molecular mass }}{8}\)
= 390/8= 48.75
Question 41. Pb(NO2)2 on strong heating loses 32.6% of its mass. Calculate the relative atomic mass of Pb.
Answer: Let, the relative atomic mass of Pb = x
⇒ \(\underset{\substack{x+2(14+3 \times 16) \mathrm{g} \\=(x+124) \mathrm{g}}}{\mathrm{Pb}\left(\mathrm{NO}_3\right)_2 \stackrel{\Delta}{\longrightarrow}} \underset{(x+16) \mathrm{g}}{\mathrm{PbO}}+2 \mathrm{NO}_2+\mathrm{O}_2\)
On dissociation of (x+124)g of Pb(N03)2, the weight decreases = (x+ 124- x- 16)g = 108g So, for lOOg Pb(N03)2, the weight decreases
⇒ \(=\left(\frac{108 \times 100}{x+124}\right) \mathrm{g} \text { As, } \frac{100 \times 108}{x+124}=32.6\)
Or, x= 207.3
Question 42. Find the molecular formula of the compound. [All volumes are measured under identical conditions of temperature and pressure] Fill in the blank: Equivalent weight of Min MC13 is E. Relative atomic mass of M is
- 0.7l
- 4.8l
- 1.4l
- 1.2l
Answer: 3E
Volume of lg of 02 at STP \(\frac{22.4}{32}\)= 0.7L
Question 43. What does 1 mol of electron signify?
Answer: 1 mol of electron signifies, 6.022 x 1023 number of electrons. The total charge of these electrons is 96500 C or F.
5. Find the number of neutrons in 5 x 10-4 mol of 14C isotope.
1.0 g of metal (A) displaces 1.134g of metal (B) from its salt. Determine the equivalent weights of(A) and (B). 2
Equivalent weight of A \(Equivalent weight of A \)
Equivalent weight of B \(=\frac{1.134 \times 28.006}{1}=31.7588\)
Question 44. 5 g of an impure sample of common salt on treatment with an excess of AgNO3 solution yields 9.812 g of AgCl. What is the percentage impurity of that sample?
Answer: NaCl(58.5g) + AgNO3 = NaNOg + AgCU(143.5g)
143.5g AgCl is obtained from 58.5gNaCl
Therefore 1 g ofAgCl is obtained from \(\frac{58.5}{143.5} \mathrm{~g} \mathrm{NaCl}\)
Therefore 9.812g of AgCl is obtained from \(\frac{58.5 \times 9.812}{143.5}=4 \mathrm{~g} \mathrm{NaCl}\)
Percentage of purity in the sample is \(=\left(\frac{4}{5} \times 100\right)=80 \%\)
Therefore So, percentage impurity = (100- 80)% = 20%
Find the number of neutrons in 5 x 10-4 mol of 14C isotope.
The chloride of a metal (A) contains 55.90% of chlorine.
10g of metal (A) displaces 1.134g of metal (B) from its salt. Determine the equivalent weights of(A) and (B). 2 6. 5 g of an impure sample of common salt on treatment with an excess of AgN03 solution yields 9.812 g of AgCl. What is the percentage impurity of that sample?
Question 45. A gaseous hydrocarbon contains 75% C by weight. 1L of this gas at STP weighs 0.72 g. What is the molecular formula of the hydrocarbon? [weight of 1L ofhydrogen at STP = 0.09 g]
Answer: D\(\frac{0.72}{0.09}\) 8; Molecular weight (M) = 2 x 8 = 16 = 3.0115 X 1021.
⇒ \(\mathrm{C}: \mathrm{H}=\frac{75}{12}: \frac{25}{1}=6.25: 25=1: 4\)
Empirical formula:CH4; molecular formula:(CH4)n.
Molecular weight = (12 + 4xl)n
As, 16n = 16
⇒ n = 1. So, the actual formulais CH4.
Question 46. 84 g of a mixture of.CaCO3 and MgCO. were heated to a constant weight. The constant weight of the residue was found to be 0.96 g. Calculate the percentage composition of the mixture. (Relative atomic masses of Ca and Mg arc 40 and 24 respectively)
Answer: Let, CaC03 = xg, MgC03 = (1.84 -x)g
∴ \(\mathrm{CaCO}_3(100 \mathrm{~g}) \stackrel{\Delta}{\longrightarrow} \mathrm{CaO}(56 \mathrm{~g})+\mathrm{CO}_2\)
∴ \(x \mathrm{~g} \mathrm{CaCO}_3 \longrightarrow \frac{56 x}{100} \mathrm{~g} \mathrm{CaO}\)
∴ \(\mathrm{MgCO}_3(84 \mathrm{~g}) \stackrel{\Delta}{\longrightarrow} \mathrm{MgO}(40 \mathrm{~g})+\mathrm{CO}_2\)
∴ \((1.84-x) \mathrm{g} \mathrm{MgCO}_3 \longrightarrow \frac{40}{84}(1.84-x) \mathrm{g} \mathrm{MgO}\)
∴ \(\text { As, } \frac{56 x}{100}+\frac{40}{84}(1.84-x)=0.96 \quad \text { or, } x=1 \text {. }\)
∴ \(\mathrm{CaCO}_3=\frac{1}{1.84} \times 100=54.35 \% ; \mathrm{MgCO}_3=45.65 \%\)
Question 47. 10 mL of a mixture of CH4, C2H4, and CO2 was exploded with excess oxygen. After the explosion, there was a contraction of 17 mL on cooling and there was a further contraction of 14 mL on treatment with KOH. Find out the composition of the mixture.
Answer: Let, CH4 = x mL, C2H4 = y mL, CO2 = z mL
∴ x+ y+ z = 10
⇒ \(\begin{aligned}
& \mathrm{CH}_4+2 \mathrm{O}_2 \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(l) \\
& \begin{array}{lccc}
1 \mathrm{~mL} & 2 \mathrm{~mL} & 1 \mathrm{~mL} & 0 \mathrm{~mL} \\
x \mathrm{~mL} & 2 x \mathrm{~mL} & x \mathrm{~mL}
\end{array} \quad \\
&
\end{aligned}\)
Contraction of volume =(x+2x-x)mL = 2xmL
Produced CO2 = xmL
⇒ \(\begin{array}{cccc}
\mathrm{C}_2 \mathrm{H}_4(\mathrm{~g}) & +3 \mathrm{O}_2(\mathrm{~g}) \longrightarrow & 2 \mathrm{CO}_2(\mathrm{~g}) & +2 \mathrm{H}_2 \mathrm{O}(l) \\
1 \mathrm{~mL} & 3 \mathrm{~mL} & 2 \mathrm{~mL} & 0 \mathrm{~mL} \\
y \mathrm{~mL} & 3 y \mathrm{~mL} & 2 y \mathrm{~mL} &
\end{array}\)
Contraction of volume = (y + 3y- 2y)mL = 2ymL Produced CO2 = 2ymL but the quantity of CO2 produced = z mL.
Contraction of total volume = (2x+ 2y+ 0)mL and total quantity of C02 = (x+ 2y+ z)mL
According to the problem, 2x+ 2y – 17 x+ 2y+ z = 14
From, [3]- [l] we get, y = 4mL
Putting y = 4 inequation [2], x = 4.5mL
Putting y = 4mL and x = 4.5mL we get z = 1.5mL from equation [3].
So, CH4 = 4.5mL, C2H4 = 4mL and CO2 = 1.5mL.
Question 48. Which of the following will have the largest no. of atoms?
- 1 gAu (g)
- 1g Na(s)
- 1g li(s)
- 1g of cl2 (g)
Answer: We know that, the number of atoms of an element
= number of moles x NA x atomicity
⇒ \(=\frac{\text { mass of the element }(\mathrm{g})}{\text { gram-atomic mass of element }} \times N_A \times \text { atomicity }\)
- No. atoms present in lg of Au = \(\frac{1}{197}\) 6.022 x 1023 (since Gram-atomic mass of Au= 197)
- No. atoms presenting lg of Na \(\frac{1}{23} \times 6.022 \times 10^{23}\)
- No. atoms present in lg of Li \(=\frac{1}{7} \times 6.022 \times 10^{23}\)
- No. of atoms present in lg of Cl2
⇒ \(=\left(\frac{1}{71} \times 6.022 \times 10^{23}\right) \times 2=\frac{1}{35.5} \times 6.022 \times 10^{23}\)
(Since 2 cl atoms are present in 1 molecule of Cl2) Therefore, in lg ofLi will have the largest no. atoms.
Question 49. Calculate the molarity of a solution of ethanol in water in which the mole fraction of ethanol is 0.040 (assume the density of water to be one)
Answer: The number of moles of ethanol present in 1 L of aqueous
solution indicates the molarity of the solution.
Let, 1L water be present in 1 L ethanol solution [since the solution is dilute.
Therefore the amount of water present in 1 L of water
⇒ \(n_{\mathrm{H}_2 \mathrm{O}}=\frac{1000 \mathrm{~g}}{18 \mathrm{~g} \cdot \mathrm{mol}^{-1}}=55.55 \mathrm{~mol}\)
For binary solutions, the mole fraction of the first component + the Mole fraction ofthe second component= 1.
Therefore, in this case, CH2o + xC2H5OH=1
Or, \(x_{\mathrm{H}_2 \mathrm{O}}=1-x_{\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}} \quad \text { or, } x_{\mathrm{H}_2 \mathrm{O}}=1-0.040=0.96\)
Again, XH20 \(=\frac{n}{n_{\mathrm{H}_2 \mathrm{O}}+n_{\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}}} \text { or, } 0.96=\frac{55.55}{55.55+n_{\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}}}\)
Or, 53.328 + 0.96nc2h5OH = 55.55
Therefore nC2H5OH \(=\frac{2.222}{0.96}\) =55.55
Therefore \(n_{\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}}=\frac{2.222}{0.96}=2.3146 \mathrm{~mol}\)
Therefore Molarity ofthe solution = 203146 mol-L-1.
Question 50. The following data are obtained when dinitrogen and dioxygen react together to form different compounds:
Which law of chemical combination is obeyed by the above experimental data? Give its statement.
Fill in the blanks in the following conversions:
- 1km =……….mm=………..pm
- 1mg=…………kg = ……….ng
- 1ml= …………l=………….dm3
Answer: The ratio of different masses of dioxygen which combine separately with a fixed mass (28g) of dinitrogen.
=32: 64:32: 80 = 2:4:2:5 which is a simple whole numberratio.
Therefore, the obtained result supports the law of multiple proportions.
⇒ \(\begin{aligned}
1 \mathrm{~km} & =1 \mathrm{~km} \times \frac{1000 \mathrm{~m}}{1 \mathrm{~km}} \times \frac{100 \mathrm{~cm}}{1 \mathrm{~m}} \times \frac{10 \mathrm{~mm}}{1 \mathrm{~cm}} \\
& =10^6 \mathrm{~mm}
\end{aligned}\)
⇒ \(1 \mathrm{~km}=1 \mathrm{~km} \times \frac{1000 \mathrm{~m}}{1 \mathrm{~km}} \times \frac{1 \mathrm{pm}}{10^{-12} \mathrm{~m}}=10^{15} \mathrm{pm}\)
Therefore 1km = 106mm = 1015pm
⇒ \(1 \mathrm{mg}=1 \mathrm{mg} \times \frac{1 \mathrm{~g}}{1000 \mathrm{mg}} \times \frac{1 \mathrm{~kg}}{1000 \mathrm{~g}}=10^{-6} \mathrm{~kg}\)
⇒ \(1 \mathrm{mg}=1 \mathrm{mg} \times \frac{1 \mathrm{~g}}{1000 \mathrm{mg}} \times \frac{1 \mathrm{ng}}{10^{-9} \mathrm{~g}}=10^6 \mathrm{ng}\)
Therefore long = 10~6kg = 106ng
⇒ \(1 \mathrm{~mL}=1 \mathrm{~mL} \times \frac{1 \mathrm{~L}}{1000 \mathrm{~mL}}=10^{-3} \mathrm{~L}\)
⇒ \(\begin{aligned}
1 \mathrm{~mL}=1 \mathrm{~cm}^3 & =1 \mathrm{~cm}^3 \times \frac{1 \mathrm{dm} \times 1 \mathrm{dm} \times 1 \mathrm{dm}}{10 \mathrm{~cm} \times 10 \mathrm{~cm} \times 10 \mathrm{~cm}} \\
& =10^{-3} \mathrm{dm}^3
\end{aligned}\)
Therefore 1ml = 10-3l =10-3dm3
Question 51 . In a reaction A + B2AB2. Identify the limiting reagent, if any, in the following reaction mixtures.
- O 300 atoms of A + 200 molecules of B 0
- 2 mol A + 3 mol B
- 100 atoms of A + 100 molecules of B
- 5 mol A + 2.5 mol B 0 2.5 mol A + 5 mol B
Answer: According to the given reaction, 1 atom of A reacts
88K with 1 molecule of B. Therefore, 200 atoms of A react with 200 molecules of B, and 100 no. of A atoms remain unused. In this case, B is the limiting reagent, as A is present in excess.
According to the reaction, 1 mol of A reacts with 1 mol of B. Thus, 2 mol of A reacts with 2 mol of B. Thus, A is the limiting reagent and B will remain in excess.
As per the reaction, 1 atom of A reacts with 1 molecule of B. So, 100 atoms of A reacts with 100 molecules of B. Thus, in this case, there is no limiting reagent.
Here, 2.5 mol of A reacts with 2.5 mol of B. Therefore, B is the limiting reagent and A remains in excess.
As per the reaction, 2.5 mol A reacts with 2.5 mol B. Thus, A is the limiting reagent, and B remains in excess.
Question 52. N2 and H2 react to produce ammonia according to the equation: N2(g) + H2(g)→2NH3(g) Calculate the mass of ammonia produced if 2.00 x 103g N2 reacts with 1.00 X 103 g of H2. Will any of the two reactants remain unreacted? If yes, which one and what would be its mass?
⇒ \(\begin{gathered}
\text { Reaction: } \mathrm{N}_2(\mathrm{~g}) \\
1 \mathrm{~mol}=28 \mathrm{~g}
\end{gathered} \quad+\quad \begin{gathered}
3 \mathrm{H}_2(\mathrm{~g}) \longrightarrow \\
2 \mathrm{~mol}=6 \mathrm{~g}
\end{gathered} \quad \begin{array}{r}
2 \mathrm{NH} \\
2 \mathrm{~mol}=34 \mathrm{~g}
\end{array}\)
28g of nitrogen reacts with 6g of hydrogen.
Therefore lg nitrogen reacts with (6/28)g of hydrogen.
Therefore 2000g of nitrogen reacts with \(\frac{6}{28} \times 2000\) =428.57g of hydrogen. therefore nitrogen is the limiting reagent and an excess amount ofhydrogen is present. 28g of nitrogen produces 34g of NH3
Therefore 2000g of N2 produces 34/28 x 2000 = 2428.57g of NH3
Yes, excess hydrogen does not take part in the reaction and remains unchanged.
Amount of hydrogen that does not take part in the reaction = (1000- 428.57) = 571.43g
Question 53. Calculate the number of atoms in each of the following
- 52 moles of Ar
- 52u of He
- 52 g of He
Answer: 1 molAr = 6.022 x 1023 no. of-atoms
∴ 52mol Ar = 52 X 6.022 x 1023 Ar-atoms
= 3.131 x 1025 no Ar-atoms
Mass of 1 He-atom = 4u
Therefore, 4u = mass of1 He-atom
Therefore 52uHe = mass of \(\frac{52}{4}\) He-atoms = mass of13 He-atoms
1 mol He-atoms = 6.022 x 1023 He-atoms = 4gHe
Hence, 4g He = 6.022 x 1023 He-atoms
∴ 52gHe \(=\frac{6.022 \times 10^{23} \times 52}{4}=7.8286 \times 10^{24} \mathrm{He} \text {-atoms }\)
Significant Figures: Questions and Answers
Question 54. A welding fuel gas contains carbon & hydrogen only. Burning a small sample of it in oxygen gives 3.38 g of C02, 0.690 g of H20, and no other products. A volume of 10.0L (at STP) of this welding gas is found to weigh 11.6g. Calculate the empirical formula, the molar mass of the gas, and the molecular formula.
Answer: Mass of carbon in 44g of C02 = 12g
∴ Mass of Cin 3.38g of C02 \(=\frac{12}{44} \times 3.38 \mathrm{~g}=0.9218 \mathrm{~g}\)
Similarly,18 g H2O = 2g hydrogen
Therefore, the mass of hydrogen in 0.690g of H20
=(2/18) x 0.690g = 0.0767g
therefore Total mass of fuel gas = (0.9218 + 0.0767) = 0.9985g
Therefore % amount ofC in the gas \(=\frac{0.9218}{0.9985} \times 100\) = 92.32
Therefore % amount of H in the gas \(=\frac{0.0767}{0.9985} \times 100=7.68\)
Determination of the empirical formula of the fuel gas
Therefore, the empirical formula ofthe fuel gas = CH
As given the question,
Mass of 10L gas at STP = 1 1.6g
∴ Mass of22.4L gas at STP = \(\frac{11.6 \times 22.4}{10}=25.984 \mathrm{~g}\)
Therefore Molecular mass = 25.984= 26
Mass of the empirical formula (CH) = 12+1 = 13.
Let, the molecular formula of the compound be (CH)n
Again, n = \(\frac{\text { molecular mass }}{\text { mass of empirical formula }}\)
∴ n = 26/13 =2
Therefore formula of fuel gas = (CH)n=(CH)2=C2H2
∴ n = 26/13=2
∴ Formula Of Fuel gas = (CH)n = (CH)2 = C2H2
Question 55. Calcium carbonate reacts with aqueous HC1 to give CaCl2 and CO2 according to the reaction. What mass of CaCO3 is required to react completely with 25 mL of 0.75(M) HC1?
Answer: Gram-molecular mass ofHC1 (solute) = (1 + 35.5) = 36.5g We know that the molarity ofthe solution (M)
⇒ \(=\frac{\text { mass of the solute (in gram) }}{\text { gram-molecular mass of solute } \times \text { vol. of solution }(\mathrm{L})}\)
Or, 0.75 \(\frac{\text { mass of } \mathrm{HCl}}{36.5 \times 25 \times 10^{-3}}\) So, mass of HCl = 0.6844g
⇒ \(\underset{100 \mathrm{~g}}{\mathrm{CaCO}_3(s)+2 \mathrm{HCL}(a q)} \underset{2 \times 36.5=73 \mathrm{~g}}{\rightarrow \mathrm{CaCl}_2(a q)+\mathrm{CO}_2(g)+\mathrm{H}_2 \mathrm{O}(l)}\)
73g ofHC1 reacts completely with 100g of CaCO3
Therefore 0.6844g of HC1 reacts completely with \(\frac{100 \times 0.6844}{73} \mathrm{~g}\)
= 0.9375g of CaCO3
Question 56. Chlorine is prepared in the laboratory by treating manganese dioxide (MnOz) with aqueous HC1 according to the reaction.
⇒ \(4 \mathrm{HCl}(a q)+\mathrm{MnO}_2(s)-\mathrm{2H}_2 \mathrm{O}(l)+\mathrm{MnCl}_2(a q)+\mathrm{Cl}_2(g)\) How many grams of HC1 react with 5.0g of MnO2?
Answer: The given reaction,
⇒ \(4 \mathrm{HCl}(a q)+\mathrm{MnO}_2(s) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l)+\mathrm{MnCl}_2(a q)+\mathrm{Cl}_2(g)\)
(4 x 36.5)g (55 + 32) = 87 g
According to the equation,
87g of Mn02 reacts with (4 x 36.5)g of HCl
therefore 5g of mnO2 reacts with ,\(\frac{4 \times 36.5 \times 5}{87}=8.39 \mathrm{~g} \text { of } \mathrm{HCl}\)
Question 57. Classify the following as pure substance, homogeneous mixture, heterogeneous mixture, element, and compound:
- Milk
- Air
- Petrol
- Distilled water
- Common salt
- Graphite
- Tap-water
- Smoke
- Dry ice
- Cold drinks
- Gun powder.
Answer:
- Mixture (homogeneous);
- Mixture (homogeneous);
- Mixture (homogeneous);
- Compound;
- Mixture (heterogeneous);
- Element;
- Mixture (homogeneous);
- Mixture (heterogeneous);
- Compound; Mixture (homogeneous);
- Mixture (heterogenous
Short Question And Answers
Question 1. Is the statement correct or incorrect— ‘35.5g of chlorine contains 6.022 x 1023 molecules’?
Answer: The statement is incorrect. 35.5g (lg-atom) of chlorine contains 6.022 x 1023 no. of atoms.
Question 2. Calculate the number of molecules in 0.52g of acetylene.
Answer: Gram-molecular mass of acetylene = 26g.
Number of molecules present in 0.52g of acetylene
∴ \(=\frac{6.022 \times 10^{23} \times 0.52}{26}=1.2044 \times 10^{22}\)
Question 3. Find the number of molecules in l milli mole of C02.
Answer: milli mol of C02 molecule = 10-3 mol of C02 Hence, number of molecules presentin1 millimole of
Question 4. The equivalent mass of iron in ferrous oxide is 28. Calculate its equivalent mass in ferric oxide.
Answer: Considering ferrous oxide (where valency of iron = 2),
Atomic mass of iron, A = £xF=28×2 = 56
Now, considering ferric oxide (where valency of iron = 3 ),
equivalent mass ofiron, E = A/ V = 56/3 = 18.67.
Question 5. Differentiate between accuracy and precision.
Answer:
Difference between accuracy and precision
Cu Precision refers to the closeness of the set of values obtained from identical measurements of a quantity.
- Whereas accuracy means how measurements obtained experimentally agree with the exact value.
- A measurement may have good accuracy but poor precision as different measurements may yield a correct average.
- Similarly, good precision does not mean good accuracy as the same mistake may occur repeatedly.
- The accuracy of a measurement depends on the— skill ofthe examiner& precision of the instrument.
Question 6. “Two different elements may combine together in a definite proportion by mass to form two different compounds. Again two different elements may combine together in different proportions by mass to produce two different compounds” Comment on this.
Answer: Two different elements may combine in a definite proportion by mass to form two isomeric compounds having different properties.
Thus, C and H may combine in the ratio of 12: 1 by mass to form a pair of isomeric compounds, acetylene (C2H2) and benzene (C6H6).
Again, C and H may combine in the mass ratio of 3: 1 to form methane (CH4) and in the mass ratio of 4: 1 to form ethane (C2H6). So, the given statements are quite justified.
Question 7. Why does Dalton’s atomic theory fail to explain GayI.ussac’s law of gaseous volumes?
Answer:
AflB.Dalton’s atomic theory could not explain Gay-Lussac’s law of chemical combination of gaseous substances in terms of their volumes.
This is because Dalton did not make any distinction between the smallest particle of an element (atom) and that of a compound (molecule).
Question 8. Show that the equivalent mass of a certain element varies inversely with its valency.
Answer: Equivalent mass \(=\frac{\text { atomic mass of the element }}{\text { valency }}\)
For a particular element, atomic mass = constants(say)
∴ Equivalent mass of the element \(=\frac{K}{\text { valency }}\)
∴ Equivalent mass of a certain element \(\propto \frac{1}{\text { valency }}\)
Question 9. The mass of one 12C atom is 1.99236 X 10-23 g. Express the value of 1 amu in grams.
Answer: 1 amu is 1 /12 th ofthe mass of one 12C atom.
⇒ \(1 \mathrm{amu}=\frac{1.99236 \times 10^{-23}}{12} \mathrm{~g}=1.6605 \times 10^{-24} \mathrm{~g}\)
Question 10. An element forms two compounds X and Y in which the element exhibits the valency of 2 and 3 respectively. What is the ratio of the equivalent masses of the element in the two compounds?
Answer: \(=\frac{\text { atomic mass of the element }}{\text { valency of the element in the compound } X}=\frac{a}{2}\)
Similarly, the equivalent mass of that element in the compound, Y = a/3. So, the ratio of the equivalent masses of the element the compounds X and Y = a/2: a/3 =3:2
Question 11. An element produces X, Y&Z compounds. Equivalent masses of that element in the compounds X, Y & Z are in the ratio of 1:2:3. In which of the three compounds, does the element exhibit its maximum valency?
Answer: We know, equivalent mass \(\propto \frac{1}{\text { valency }}\)
Hence, the compound in which the equivalent mass of that element is minimum exhibits the maximum valency ofthe element.
Since the equivalent mass of that element is minimum in compound X, the valency ofthe element in X is maximum.
Question 12. Out of Cl2 and O2, which one will have greater mass when taken in equal volumes at the same temperature and pressure?
Answer: Let the volume of Cl2 and O2 gas be VmL. At the same temperature and pressure, each gas contains n number of molecules.
Mass of n molecules of Cl2 = 71 xn g Mass of n molecules of O2 = 32 x n g Therefore, under the same conditions of temperature and pressure, the mass of Cl2 will be more than that of 02 when taken in equal volumes.
Question 13. At the same temperature and pressure, 1 volume of gas A reacts with 1 volume of gas B to produce 2 volumes of gas C. If the atomicity of the gases A, B, and C are x, y, and z respectively, then show that both x and y will be either odd or even numbers.
Answer: According to the given problem, 1 volume of A + 1 volume of B = 2 volumes of C Let the number of molecules present in 1 volume of A at a certain temperature and pressure be n.
So according to Avogadro’s hypothesis, n molecule of A + n molecule of B = 2n molecule of C or,1 molecule of A +1 molecule of B = 2 molecules of C or, x atoms of A + y atoms of B = 2z atoms of C
[since, x, y and z are the number ofatoms present in1 molecule of A, B and C gases respectively]
According to Dalton’s atomic theory, the no. of atoms before and after a reaction must be the same. So, x+ y = 2z. Since 2z is an even number, (x + y) will also be even.
Now the sum of x and y will be an even number if x and y are separately even numbers or x and y are separately odd numbers.
If between x and y, one is an even number and the other is an odd number, then the value of(x + y) will be odd. But it is not possible because 2z always represents an even number. Hence, both x & y will be either even or odd numbers.
Question 14. Calculate the number of hydrogen atoms present in 90 amu of ethane.
Answer: Molecular mass of ethane (C2Hg) = 2 x 12 + 6 = 30
Therefore Mass of1 molecule of ethane = 30 amu
Therefore Number of molecules in 90 amu of ethane = 90/30 = 3
Thus, no.of H-atoms present in 90 amu of ethane 3X6 =18
Question 15. “What is the equivalent mass of an element?” Is the question correct? If yes, then explain your answer.
Answer: The question isn’t correct because the equivalent masses of all the elements are not constant.
The equivalent mass of an element depends on particular reactions. If the element utilizes its different valencies in different reactions then its equivalent mass will also be different for different reactions.
For example, the equivalent mass of Cu in Cu2O is 63.5 whereas in CuO, it is 31.75. Because, in Cu2O, the valency of Cu is 1 and in CuO, the valency of Cu is 2.
According to the relation, equivalent mass = (atomic mass -e- valency’), equivalent mass changes with valency.
So, the compound in which the element under consideration is present should be mentioned while calculating the equivalent mass ofthe element.
Question 16. 21 molecules of C02 were expelled from 220 mg of C02. How many molecules of C02 were left?
Answer: No. of molecules in 220 mg of CO2
As, 1021 molecules of CO2 were expelled, so no. of
molecules left = (3.0115 x 1021- 1021) = 2.0115 x 1021.
Question 17. Calculate the percentage composition of the compound having the molecular formula of C6H12(LI = 1 C = 12, O = 16).
Answer: Molecular mass of C6H1206 =180
Percentage of = (100- 40- 6.67) = 53.33%.
Question 18. Between 100mL C02 and lOOmL NH3, which one has greater mass at constant temperature and pressure?
Answer: Molecular mass of CO2 = 12+16 x 2 = 44g
At, STP, mass of 22400 mol of CO2 is 44g
Mass of 100 mL of CO2 is \(\frac{44 \times 100}{22400} \mathrm{~g}=0.196 \mathrm{~g}\)
At STP, Molecular mass of NH3 = 17g [N = 14, H = 1]
Mass of 100 mL of NH3 is \(\frac{17 \times 100}{22400} \mathrm{~g}=0.0759 \mathrm{~g}\)
Question 19. Write the no. of molecules present in a millimole of S02.1
Answer: 1 million = 10 3 mol
10-3 mol of SO2=6.022 X 1023 x 10-3 molecules of SO2
Therefore 1 million of SO2 = 6.022 x 1020 molecules of SO2
Question 20. How many electrons are present in 1 millimole of methane?
Answer: Number of electrons present in each molecule of methane (CH2) = 6 + 4 = 10
∴ Total no. of electrons in 1 millimole or 10-3 mole of methane
\(=\left(6.022 \times 10^{23}\right) \times 10^{-3} \times 10=6.022 \times 10^{21}\)
Question 21. 2.7 grams of metal after reaction with excess acid produces 3.6 liters of H2 at NTP. What is the equivalent weight ofthe metal?
Answer: 3.36L H2 (NTP) is displaced from acid by 2.7g of metal
∴ 11.2Lof H2 (at NTP) is displaced from acid by
⇒ \(=\frac{2.7 \times 11.2}{3.36} \mathrm{~g}=9.0 \mathrm{~g} \text { of metal }\)
∴ Equivalent weight ofthe metal =9
Question 22. In two compounds of hydrogen and oxygen, hydrogen is present in 42.9% and 27.3% respectively. Show that the data supports the law of multiple proportions.
Answer: Let the given compounds be denoted by A and B respectively.
In A, 42.9ghydrogen combines with (100- 42.9) = 57.1g oxygen
In B, 27.3g of hydrogen combines with (100- 27.9)
= 72.7g oxygen
∴ B, 42.9 ofhydrogen combines with \(\frac{72.7 \times 42.9}{27.3}\)
=114.2g oxygen
So the masses of oxygen which combine with 42.9 g of hydrogen in the given compounds (A&B) are respectively 57.1g and 114.2g. These masses are in the ratio.
57.1: 114.2 =1:2
Since this is a simple ratio, the given data supports the law of multiple proportions.
Question 23. Calculate the molar mass: H2O, CO2, CH4
Answer: The sum of the atomic masses of all the atoms present in a molecule of a compound is known as the molecular mass.
- Molecular mass of HaO = 2 X 1.008u + 16.00u = 18.016u
- Molecular mass of CO2 = 12.01u + 2 x 16.00u = 44.01u
- Molecular mass of CH4 = 12.01u + 4 x 1.008u=16.042u
Question 24. Calculate the mass percent of different elements present in sodium sulfate (Na2S04) Mass % of an element
⇒ \(=\frac{\text { mass of the element present in the compound }}{\text { molecular mass of the compound }} \times 100\) The molecular mass of the compound
- Molecular mass of Na2S04 =(2 x23) + 32+ (4×16) = 142
- Mass percent of Na =(46/142) x 100 = 32.39
- Mass percent of S =(32/142) x 100 = 22.54
- Mass percent of O =(64/142) x 100 = 45.07
Answer: 3. Mass percent of S =(32/142) x 100 = 22.54
The weight of oxalic acid in 10CC mL CN (N/20) oxalic acid solution =1/20 gram – equivalent = 63g/20
Question 25. Determine the empirical formula of an oxide of iron which has 69.6% iron and 30.1% dioxygen by mass.
Answer: ∴ The empirical formula of the given iron oxide =Fe2O3
Therefore Equivalent weight of K2Cr2O7
⇒ \(=\frac{\text { molecular mass of } \mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7}{\mathrm{no} \text {. of electrons gained by one ion }}=\frac{\mathrm{M}}{6}\)
Question 26. Calculate the amount of carbon dioxide that could be produced when O 1 mole of carbon is burnt in the air.
1 mole of carbon is burnt in 16g of dioxygen.
2 moles of carbon are burnt in 16 g of dioxygen.
Answer:
⇒ \(\underset{1 \mathrm{~mol}}{\mathrm{C}(s)}+\underset{1 \mathrm{~mol}(32 \mathrm{~g})}{\mathrm{O}_2(\mathrm{~g})} \rightarrow \underset{1 \mathrm{~mol}(44 \mathrm{~g})}{\mathrm{CO}_2(g)}\)
According to the equation, 44g of CO2 is produced on complete combustion of 1 mol of carbon in air. According to the equation, 32 g of dioxygen is required for the combustion of mol of carbon.
However, only 16g of dioxygen is present in the reaction mixture which reacts with 0.5 mol of carbon. Therefore, dioxygen is the limiting reagent in this reaction. So, 22g of CO2 is produced in this reaction.
Again, in this case, dioxygen is the limiting reagent. Therefore, 16g of dioxygen completely reacts with 0.5 mol of carbon to produce 22g of CO2.
Word Problems Related to Percentage Composition
Question 27. Calculate the mass of sodium acetate (CH3COONa) required to make 500 mL of 0.375 molar solution. The molar mass of sodium acetate is 82.0245 g mol-1.
Answer:
⇒ \(\text { Molarity }=\frac{\text { mass of the solute (in g) }}{\mathrm{g} \text {-molecular mass } \times \text { vol. of the solution }(\mathrm{L})}\)
As given in the question, 0.375 = \(\frac{\text { mass of the solute }}{82.0245 \times 500 \times 10^{-3}}\)
∴ 500 ml =500 x 10-3l
Or, Mass of the solute \(\frac{0.375 \times 82.0245 \times 500}{1000} \approx 15.38 \mathrm{~g}\)
Therefore, 15.38g of sodium acetate (CH3COONa) is required to make 500 mL of 0.375 molar aqueous solutions.
Question 28. Calculate the concentration of nitric acid in moles per liter in a sample that has a density of 1.4 gml-1 and the mass percent of nitric acid in it being 69%.
Answer: Molecular mass of nitric acid = 1.008 + 14 + (3 x 16) ~ 63
Therefore Number of moles present in 69g of HNO3 \(=\frac{69}{65}=1.095\)
Given, a 69g mass of HNO3 is present in 100g of HNO3.
Vol. of 100g HNO3 solution \(=\frac{100 \mathrm{~g}}{1.41 \mathrm{~g} \cdot \mathrm{mL}^{-1}}=70.92 \mathrm{~mL}\)
= 70.92×10-3l
Concentration of nitric acid \(=\frac{1.095 \mathrm{~mol}}{70.92 \times 10^{-3} \mathrm{~L}}\)
=15.44 mol.L-1
Question 29. How much copper can be obtained from 100g of copper sulfate (CuO4)?
Answer: Molecular mass of CuSO4 \(\begin{aligned}
& =63.5+32+(4 \times 16) \\
& =159.5 \mathrm{~g} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
Amount of Cu present in 159.5g of CuSO4 = 63.5g
Therefore, Cu presenting 100g of CuSO4 \(=\frac{63.5 \times 100}{159.5}=39.81 \mathrm{~g}\)
Question 30. Determine the molecular formula of an oxide of iron in which the mass percent of iron and oxygen are 69.9 and 30.1 respectively.
Answer: To determine the empirical formula of the oxide of iron
Empirical formula ofthe compound = Fe2O3
Let, molecular formula ofthe compound be (Fe2O3)w Molecular mass ofthe compound
=n X [2 x 55.85 + 3 x 16.00] = 159.7n
As given in the question, n x 159.7 = 159.8 or, n = 1.
The molecular formula ofthe compound is Fe2O3.
Question 31. Calculate the atomic mass (average) of chlorine using the following data:
Answer: Fractional abundance of Cl isotope = 0.7577
Atomic mass = 34.9689
Fractional abundance of 37 Cl isotope = 0.2423
Atomic mass = 36.9659
∴ The average atomic mass of chlorine
⇒ \(=\frac{34.9689 \times 0.7577+36.9659 \times 0.2423}{0.7577+0.2423} \approx 35.4528\)
Question 32. In three moles of ethane (C2H6) calculate the following: Number of moles of carbon atoms.
- Number of moles of hydrogen atoms.
- Number of molecules of ethane.
Answer: Number of moles of C-atoms present in 1 mol of ethane (C2H6) = 2
∴ Number of moles of C-atoms present in 3 mol of ethane (C2Hg) = (2 x 3) = 6
(5) Number of moles of H-atoms present in 1 mol of ethane (C2Hg) = 6
∴ Number of moles of H-atoms present in 3 mol of ethane (C2Hg) = (3 x 6) = 18
(3) 1 mol ethane = 6.022 x 1023 no. of ethane molecules
∴ 3 mol ethane = 3x 6.022 x I023 = 1.8066 x 1024 no. of ethane molecules.
Question 33. What is the concentration of sugar (C12H22Ou) in mol.L-1 if its 20g is dissolved in enough water to make a final volume of up to 2L?
Answer: Molecular mass of sugar (C12H22On)
= (12 X 12) + (22 X 1.008) + (11 X 16.00) = 342
⇒ \(\text { Molarity }=\frac{\text { mass of the solute (in g) }}{\text { gram-molecular mass of the solute }} \times\)
or, Molarity ofthe solution, (M) = \(\frac{20}{342 \times 2}=0.0293\) = 0.0293
Concentration = 0.0293 mol L-1 or 0.293 (M).
Question 34. The density of methanol is 0.793 kg-L-1. What is the volume needed for making 2.5L of its 0.25(M) solution?
Answer: Molecular mass ofmethanol (CH3OH) = 32
No. of moles ofmethanolpresentin 1L 0.25 (M) solution = 0.25
Number of moles of methanol present in 2.5L 0.25(M)
Solution = 0.25 X 2.5 = 0.625
Mass of 0.625 mol CH3OH = 0.625×32 = 20g
Density of CH3OH = 0.793kg L-1
or, 0.793 X 103g-L-1
Hence, the required volume of methanol
⇒ \(=\frac{\text { mass of methanol }}{\text { density of methanol }}=\frac{20 \mathrm{~g}}{0.793 \times 10^3 \mathrm{~g} \cdot \mathrm{L}^{-1}}\)
= 0.02522L = 25.22mL
25.22mL of methanolic required.
Question 35. Pressure is determined as force per unit area of the surface. The SI unit of pressure, Pascal is as shown below: IPa = IN m-2. If the mass of air at sea level is 1034 g cm-2, calculate the pressure in Pascal.
Answer: Weight= mg
Pressure = Weight per unit area \(=\frac{1034 \times 9.8 \mathrm{~m} \cdot \mathrm{s}^{-2}}{\mathrm{~cm}^2}\)
\(=\frac{1034 \mathrm{~g} \times 9.8 \mathrm{~m} \cdot \mathrm{s}^{-2}}{\mathrm{~cm}^2} \times \frac{1 \mathrm{~kg}}{1000 \mathrm{~g}} \times \frac{100 \mathrm{~cm} \times 100 \mathrm{~cm}}{1 \mathrm{~m} \times 1 \mathrm{~m}}\) \(\times \frac{1 \mathrm{~N}}{\mathrm{~kg} \cdot \mathrm{m} \cdot \mathrm{s}^{-2}} \times \frac{1 \mathrm{~Pa}}{1 \mathrm{~N} \cdot \mathrm{m}^{-2}}\)= 1.01332 x 105Pa
Question 36. A sample of drinking water was found to be severely contaminated with chloroform. Is supposed to be carcinogenic in nature. His level of contamination was IB ppm (by mass). Kxpress this In percent by mass. Determine the molality of chloroform In the water sample.
Answer: 15ppm Indicates 15 parts in 106 parts.
∴ Mass percent \(=\frac{15 \times 100}{10^6}=1.5 \times 10^{-3}\)
Molar massof CHGl2 = 12+ 1 .008 + (3 x 35,5) =1 19,5
Amount of mass equal to 1,5 x 10-3% means 1.5 x 10-3g present in the 100g sample.
Amount of ChCl3 present in 1 kg sample =1.5 x 10-2g
Hence, no, of moles of CHC3 presenting 1kg sample
\(=\frac{1.5 \times 10^{-2}}{119.5}=1.255 \times 10^{-4}\)Therefore Molality = 12.55 x 10-4
Question 37. A metallic oxide contains 60% of metal. Calculate the equivalent weight of the metal
Answer:
60 g of the metal combined with (100-60) =40g of oxygen
Therefore 8g oxygen combines with \(\frac{60 \times 8}{40}\)= 12g of the mental
So, the equivalent weight of the metal = 12
Question 19. Calculate the equivalent weight of phosphate radical.
Answer: Equivalent weight phosphate radical (PO34-)
⇒ \(=\frac{\text { formula weight of phosphate radical }}{\text { valency }}\)
⇒ \(\frac{31 \times 4 \times 16}{3}=31.67\)
Question 38. A polymer contains 0.16% of sulfur by weight. What is the minimum molecular weight of the polymer?
Answer: Each molecule of the given polymer must contain at least one S-atom.
In other words, each gram-mole of the polymer must contain at least 32g of sulfur.
But, 0.16g sulfur is present in 100g of the polymer.
Therefore 32g sulfur is present in \(\frac{100 \times 32}{0.16}\) =20000g of the polymer.
Thus, the minimum molecular weight of the polymer is 20,000.
Question 39. The empirical formula of an organic compound is CH2O and its molecular weight is 180. What is the molecular formula of the compound? (H =1, C =12, 0 = 16)
Answer: Let the molecular formula be (CH2O)n
So its molecular weight =nx(12 + 2 + 16) = 30n 30n = 180 i.e., n = 6
Thus, the molecular formula of the compound is C6H12O6.
Question 40. How many neutrons are present in 5 X 10-4 moles of \({ }_6^{14} \mathrm{C}\)?
Answer: Each atom of \({ }_6^{14} \mathrm{C}\) contains (14-6) =8 neutrons
∴ No. of neutrons presenting 5 x 10~4 moles of \({ }_6^{14} \mathrm{C}\)
⇒ \(=\left(5 \times 10^{-4}\right) \times\left(6.022 \times 10^{23}\right) \times 8=2.4088 \times 10^{21}\)
Question 41. Mass percent of hydrogen in water (H2O) \(=\frac{2 \times 100}{18}=11.11\)
Mass percent of oxygen in water (H2O)
⇒ \(=\frac{16 \times 100}{18}=88.89\)
Question 42. What is the SI unit of mass? How is it defined?
Answer:
SI unit of mass
The SI unit of mass is kilogram (kg). It is equal to the mass of the international prototype of the kilogram. It is defined as the mass of a platinum-iridium (Pt-Ir) cylinder stored in an air-tight jar at the International Bureau of Weights and Measures in France.
Question 43. Match the following prefixes with their multiples
Answer: micro = 10-6
- Deca = 10
- mega = 106
- giga = 109
- femto =10-15
Question 44. Express the following In the scientific notation:
- 0.0048
- 234, 000
- 8008
- 500.0
- 6.001
Answer:
- 4.0 X 10-3
- 2.34 X 10s
- 8.008 X 1.03
- 5.000 X 102
- 6.0012 X 10°
Question 45. How many significant figures are present:
- 0.0025
- 208
- 5005
- 120,0000 500.0
- 2.0034
Answer:
- 2
- 3
- 4
- 3
- 4
- 5
Question 46. Round up the following upto three significant figures:
- 34.210
- 10.41070
- 0.04507
- 2808
Answer:
- 34.2
- 10.4
- 0.0460
- 2810
Question 47. If the speed of light is 3.0 x 108m s-I, calculate the distance covered by light in 2.00 ns
Answer: 2.00ns = 2.00 x 109s
Therefore, distance traversed = velocity x time
3.0 x 108m. s-1 X 2.0 X 10-9s =0.6m
Question 48. How are 0.5mol Na2CO3 and 0.5(M) Na2CO3 different?
Answer: Molar mass of Na2CO3 = (2 x 23) + 12 + (3 x 16) = 106
0.50 mol Na2CO3 = 0.50 x 106 = 53gNa2C03
0.50 (M) Na2CO3 means 53g of Na2CO3 is presentin 1L sodium carbonate solution.
Question 49. If ten volumes of dihydrogen gas react with five volumes of dioxygen gas, how many volumes of water vapor would be produced?
Answer: \(\begin{aligned}
& 2 \mathrm{H}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(g) \\
& 2 \text { volume } 1 \text { volume } 2 \text { volume }
\end{aligned}\)
According to Gay Lussac’s law of gaseous volumes, 2 volumes ofhydrogen reacts with 1 volume of oxygen to produce 2 volumes of water vapor under the same conditions of temperature and pressure.
Therefore, the 10 volumes of hydrogen react with 5 volumes of oxygen to produce 10 volumes of water vapor at the same temperature and pressure.
Question 50. Convert the following into basic units: O 28.7 pm 0 15.15 pm 0 25365 mg
Answer: \(28.7 \mathrm{pm} \times \frac{10^{-12} \mathrm{~m}}{1 \mathrm{pm}}=2.87 \times 10^{-11} \mathrm{~m}\)
Question 51. How many significant figures should be present in the answer to the following calculations?
⇒ \(\frac{0.02856 \times 298.15 \times 0.112}{0.5785}\)
- 5 x 5.364
- 0.0125 + 0.7864 + 0.0215
Answer: In the given problem, the least precise(0.112) has 3 significant. Therefore, the answer should have three significant.
Leaving the whole no. (5), the second has 4 significant figures. Hence, the answer should have 4 significant figures.
Here, the least number of decimal places in the term is Hence, the answer should have 4 significant.
Question 52. What is kg-mol? Calculate the total number of electrons present in 1 kg-mol of N2.
Answer: One kg-mol (or one kilomole) is the molecular mass ofthe substance expressed in kilogram. In the CGS system, 1 gram-mol of a substance contains Avogadro’s number of particles (6.022 x 1023).
In SI systems lkg-mol of a substance contains Avogadro’s number of particles but its value is 6.022 x 1023.
Thus, 1 kg-mol of N2 will contain 6.022 x 1026 molecules and hence, 14 x 6.022 x 1026 =8.4308 x 1027 electrons.
Multiple Choice Questions
Question 1. 2 g of metal carbonate is neutralized completely by 100 mL of 0.1(N)HC1. The equivalent weight of metal carbonate is—
- 50
- 100
- 150
- 200
Answer: 4. 200
No.of gram -equivalent of HCl in 100 ml 0.1(N) solution \(=\frac{100 \times 0.1}{100}=0.01\)
Question 2. The weight of oxalic acid that will be required to prepare a 1000 mL (N/20) solution is—
- \(\frac{126}{100} \mathrm{~g}\)
- \(\frac{63}{40} \mathrm{~g}\)
- \(\frac{63}{20} \mathrm{~g}\)
- \(\frac{126}{20} \mathrm{~g}\)
Answer: \(\frac{63}{20} \mathrm{~g}\)
The weight of oxalic acid in 1000 mL (N/20 oxalic acid solution= gram-equivalent=\(\frac{1}{20}\) gram-equivalent \(=\frac{63}{20} g\)
Question 3. The equivalent weight of K2Cr207 in an acidic medium is expressed in terms of its molecular weight (M) as—
- \(\frac{\mathrm{M}}{3}\)
- \(\frac{\mathrm{M}}{4}\)
- \(\frac{\mathrm{M}}{6}\)
- \(\frac{\mathrm{M}}{7}\)
Answer: 3. \(\frac{\mathrm{M}}{6}\)
Question 4. The number of hydrogen ions present in the 10 millionth part of 1.33 cm3 of pure water at 25°C is—
- 6.023 million
- 60 million
- 8.01 million
- 80.23 million
Answer: 3. 8.01 million
Question 5. The volume of ethyl alcohol (density 1.15g/cc) that has to be added to prepare 100cc of 0.5M ethyl alcohol solution in water is—
- 1.15 cc
- 2cc
- 2.15 cc
- 2.30 cc
Answer: 2. 2cc
Question 6. The system that contains the maximum no. of atoms is—
- 4.25g of NH3
- 8g of 02
- 2g of H2
- 4g of He
Answer: 3. 2g of H2
Question 7. In a flask, the weight ratio of CH4(g) and SO2(g) at 298IC and 1 bar is 1:2. The ratio of the number of molecules of SO2(g) and CH4(g) is—
- 1:4
- 4:1
- 1:2
- 2:1
Answer: 3. 1:2
Question 8. What will be the normality of the salt solution obtained by neutralizing xmL y(N) HC1 with ymL x(N) NaOH, and finally adding (x + y)mL distilled water—
- \(\frac{2(x+y)}{x y} \mathrm{~N}\)
- \(\frac{x y}{2(x+y)} \mathrm{N}\)
- \(\left(\frac{2 x y}{x+y}\right) \mathrm{N}\)
- \(\left(\frac{x+y}{x y}\right) \mathrm{N}\)
Answer: 2. \(\frac{x y}{2(x+y)} \mathrm{N}\)
Question 9. 0.126g of acid is needed to completely neutralize 20mL
- 53
- 40
- 45
- 63
Answer: 4. 63
Question 10. You are supplied with 500mL each of 2(N) HC1 and 5(N) HC1. What is the maximum volume of 3(M) HC1 that you can prepare using only these two solutions—
- 250mL
- 500mL
- 750mL
- 1000mL
Answer: 3. 750mL
Question 11. A metal M (specific heat =0.16) forms a metal chloride with 65% chlorine present in it. The formula of the metal chloride will be—
- MCI
- MC12
- MC13
- mci4
Answer: 2. MC12
Question 12. How many moles of electrons will weigh one kilogram
- \(6.022 \times 10^{23}\)
- \(\frac{1}{9.108} \times 10^{31}\)
- \(\frac{6.023}{9.108} \times 10^{54}\)
- \(\frac{1}{9.108 \times 6.023} \times 10^8\)
Answer: 4. \(\frac{1}{9.108 \times 6.023} \times 10^8\)
Question 13. A 5.2 molal aqueous solution of methyl alcohol is supplied. What is the mole fraction of methyl alcohol in the solution—
- 0.190
- 0.086
- 0.050
- 0.100
Answer: 2. 0.086
Question 14. The density of a solution prepared by dissolving 120 g of urea (mol. mass = 60u) in 1000 g of water is 1.15 g-mL-1. The molarity of this solution is—
- 1.78 M
- 1.02 M
- 2.05 M
- 0.50M
Answer: 3. 2.05 M
Question 15. A gaseous hydrocarbon gives upon combustion 0.72 g water and 3.08 g C02. The empirical formula of the hydrocarbon—
- C2H4
- C2H4
- C6H5
- C7H8
Answer: 4. C7H8
Question 16. A compound with a molecular mass of 180 is acylated with CH3COCl to get a compound with a molecular mass of 390. The number of amino groups present per molecule of the former compound is—
- 4
- 5
- 2
- 6
Answer: 2. 5
Question 17. The molarity of a solution obtained by mixing 750 mL of 0.5(M) HCI with 250 mL of 2(M) HCI will be—
- 0.875 M
- 1.00M
- 1.75M
- 0.975 M
Answer: 1. 0.875 M
Question 18. The ratio of masses of oxygen and nitrate in a particular gaseous mixture is 1:4. The ratio of the number of their molecules is—
- 3:16
- 1: 4
- 7:32
- 1: 8
Answer: 3. 7:32
Question 19. 3g of activated charcoal was added to 50 mL of acetic acid solution (0.06N) In a flask. After an hour was filtered and the strength of the filtrate was found to be 0.042 (N). The amount of acetic acid adsorbed (per gram of charcoal) is—
- 54 mg
- 36 mg
- 42 mg
- 18 mg
Answer: 3. 42 mg
Question 20. The molecular formula of a commercial resin used for exchanging Ions in water softening is C8H7SO3 Na (mol. wt. 206). What would be the maximum uptake of Ca2+ ions by the resin when expressed in mole per gram resin
- \(\frac{2}{309}\)
- \(\frac{1}{412}\)
- \(\frac{1}{103} 150\)
- \(\frac{1}{209}\)
Answer: 2. \(\frac{1}{412}\)
Question 21. At 300K and 1 atm, 15mL of gaseous hydrocarbon requires 375mL air containing 20% 02 by volume for complete combustion. After combustion, the gases occupy 330mL. Assuming that the water formed is in liquid form and the volumes were measured at the same temperature and pressure, the formula ofthe hydrocarbon is—
- C3H6
- C3H8
- C4H8
- C4H10
Answer: 2. C3H8
Question 22. The most abundant elements by mass in the body of a healthy human adult are oxygen (61.4%), carbon (22.9%), hydrogen (10.0%), and nitrogen (2.6%). The weight that a 75kg person would gain if all 1H -atoms were replaced by 2H atoms is
- 7.5kg
- 10kg
- 15kg
- 37.5kg
Answer: 1. 7.5kg
Question 23. 1 gram of a carbonate (M2C03) on treatment with excess HC1 produces 0.01186 mol of CO2. The molar mass of M2CO3 in g-mol-1 is—
- 118.6
- 11.86
- 1186
- 84.3
Answer: 4. 84.3
Question 24. The mole fraction of the solute in 1.00 molal aqueous solution is—
- 0.1770
- 0.0177
- 0.0344
- 1.7700
Answer: 2. 0.0177
Question 25. Which has the maximum number of molecules among the following—
- 44g CO2
- 48g O3
- 8g H2
- 64 g SO2
Answer: 3. 8g H2
Question 26. How many grams of concentrated nitric acid solution should be used to prepare 250 mL of 2.0 (M) HNO3? The concentrated acid is 70% HNO3
- 45.0 g cone. HN03
- 90.0 g cone. HN03
- 70.0 g cone. HNOa
- 54.0 g cone. HN03
Answer: 1. 45.0 g cone. HNO3
Question 27. 1.0 g of magnesium is burnt with 0.56 g 02 in a dosed vessel. Which reactant is left in excess and how much (At wt. Mg = 24, 0 = 16)—
- Mg, 0.16g
- O2,0.16g
- Mg, 0.44g
- O2,0.28g
Answer: 1. Mg, 0.16g
Question 28. When 22.4 litres of H2(g) is mixed with 11.2 litres of Cl2(g), each at STP, the moles of HCl(g) formed is equal to
- 1 mol of HCl(g)
- 2 mol of HCl(g)
- 0.5 mol ofHCl(g)
- 1.5 mol of HCl(g)
Answer: 1. 1 mol of HCl(g)
Question 29. Equal masses of H2, 02, and methane were taken in a container of volume V at a temperature of 27°C in identical conditions. The ratio of the volumes of gases H2: O2: methane would be
- 8:16:1
- 16:8:1
- 16:1:2
- 8:1:2
Answer: 3. 16:1:2
Question 30. If Avogadro’s number is changed from 6.022 x 1023 mol 1 to 6.022 X 1020 mol-1, this would change—
- The definition of mass in units in grams
- The mass of one mole of carbon
- The ratio of chemical species, to each other in a balanced equation
- The ratio of elements to each other in a compound
Answer: 2. The mass of one mole of carbon
Question 31. The number of water molecules is maximum—
- 18 molecules of water
- 1.8 g of water
- 18g of water
- 18 moles of water
Answer: 4. 18 moles of water
Question 32. 20.0g of a magnesium carbonate sample decomposes on heating to give carbon dioxide and 8.0g of magnesium oxide. What will be the percentage purity of magnesium carbonate in the sample (Mg- 24)
- 75
- 96
- 60
- 84
Answer: 4. 84
Question 33. What is the mass of precipitate formed when 50 mL of 16.9% solution of AgN03is mixed with 50 mL of 5.8% NaCl solution (Ag = 107.8, N = 14, O = 16, Na = 23 cl = 35.5)
- 28g
- 3.59
- 7g
- 14g
Answer: 3. 7g
Question 34. What is the mole fraction ofthe solute in a 1.00 m aqueous solution—
- 0. 177
- 1.770
- 0.0354
- 0.0177
Answer: 4. 0.0177
Question 35. Suppose the elements X and Y combine to form two compounds XY2 and X3Y2. When 0.1 mol of XY2 weights lOg and 0.05mol of X3Y2 weights 9g, the atomic weights of x and y are-
- 40,30
- 60, 40
- 20,30
- 30,20
Answer: 1. 40,30
Question 36. Which of the following is dependent on temperature
- Molarity
- Mole Fraction
- Weight Percentage
- Molality
Answer: 1. Let, the tire weight of X and Y be a and b. Mole no of X \(=\frac{\text { weight }}{\text { molecular mass }}\)
Therefore \(0.1=\frac{10}{a+2 b} \text { or, } a+2 b=100\)
mole no. of X3Y2 = \(\frac{\text { weight }}{\text { molecular mass }}\)
⇒ \(0.05=\frac{9}{3 a+2 b} \text { or, } 3 a+2 b=180\)
Question 37. In which case is the number of molecules of water maximum
- 10-3 mol of water
- 18mL of water
- 0.00224L of water vapours at1atm and 273K
- 0.18g of water
Answer: 1. 10-3 mol of water
Molality depends on the mass of the solvent whereas molarity is related to the volume of the solvent which is dependent on the temperature. Thus molarity depends on temperature.
Question 38. A mixture of 23 g formic acid and 43g oxalic acid is treated with a cone. H2SO4. The evolved gaseous mixture is passed. through KOH pellets. The weight (in g) of the remaining product at STP will be
- 4.4
- 1.4
- 2.8
- 3.0
Answer: 3.
⇒ \(\begin{array}{cl}
\mathrm{HCOOH} \stackrel{\text { conc. } \mathrm{H}_2 \mathrm{SO}_4}{\longrightarrow} & \mathrm{CO}(g)+\mathrm{H}_2 \mathrm{O}(l) \\
46 \mathrm{~g}=1 \mathrm{~mol} & 1 \mathrm{~mol} \\
2.3 \mathrm{~g}=\frac{1}{20} \mathrm{~mol} & \frac{1}{20} \mathrm{~mol}
\end{array}\)
⇒ \(\begin{array}{ccc}
\mathrm{COOH} \text { conc. } \mathrm{H}_2 \mathrm{SO}_4 & \\
\mathrm{I} & \mathrm{CO}(\mathrm{g}) & \mathrm{CO}_2(g)+\mathrm{H}_2 \mathrm{O}(l) \\
90 \mathrm{~g}=1 \mathrm{~mol} & 1 \mathrm{~mol} & 1 \mathrm{~mol} \\
4.5 \mathrm{~g}=\frac{1}{20} \mathrm{~mol} & \frac{1}{20} \mathrm{~mol} & \frac{1}{20} \mathrm{~mol}
\end{array}\)
When the gas mixture is passed KOH pellets, only C02 gets absorbed. Hence, the amount of residue
⇒ \(\mathrm{CO}=\left(\frac{1}{20}+\frac{1}{20}\right) \mathrm{mol}=\frac{1}{10} \mathrm{~mol}=\frac{1}{10} \times 28=2.8 \mathrm{~g}\)
Question 39. In which case is the number of molecules of water maximum
- 10-3 mol of water
- 18mL of water
- 0.00224L of water vapours at1atm and 273K
- 0.18g of water
Answer: 2. Molecules of water
= number of moles x NA – 10-3NA
[ZA = Avogadro number]
Mass of water = volume x density = 18 x lg = 18g
Therefore molecules of water
= number of moles x NA \(=\frac{18}{18} \times N_A=N_A\)
Number of moles of water \(=\frac{0.00224}{22.4}=10^{-4}\)
Molecules of water
⇒ \(=\text { number of moles } \times N_A=10^{-4} N_A\)
Molecules of water
⇒ \(=\text { number of moles } \times N_A=\frac{0.18}{18} N_A=10^{-2} N_A\)
Question 40. How much amount of CuSO4-5H20 is required for the liberation of 2.54g of I2 when titrated with KI—
- 2.5g
- 4.99g
- 2.4g
- 1.2g
Answer: 2. 4.99g
⇒ \(\begin{array}{r}
2 \mathrm{CuSO}_4 \cdot 5 \mathrm{H}_2 \mathrm{O}+4 \mathrm{KI} \longrightarrow \\
\mathrm{Cu}_2 \mathrm{I}_2+2 \mathrm{~K}_2 \mathrm{SO}_4+\mathrm{I}_2+10 \mathrm{H}_2 \mathrm{O}
\end{array}\)
Question 41. The compound that does not exist as a hydrate form
- Ferrous Sulphate
- Copper Sulphate
- Magnesium sulphate
- Sodium chloride
Answer: 4. Sodium chloride
Ferrous sulphate →FeS04-7H20
Copper sulphate→CuS04-5H20
Magnesium sulphate →MgSo4-7H20
Sodium chloride→NaC
Question 42. The number of atoms in 52g of He is—
- 78.299 x 1024 atoms
- 7.820 X 10-24 atoms
- 7.829 x 1024 atoms
- 78.234x 12025 atoms
Answer: 3. 7.829 x 1024 atoms
⇒ \(\text { Number of atoms }=\frac{N_A \times \text { mass }}{\text { at. mass }}\)
Question 43. How many significant figures representing 0.0000135
- 7
- 8
- 4
- 3
Answer: 4. Zeros to the left of the first non-zero digit in a number are not significant.
Question 44. The amount of BaS04 precipitated on mixing BaCl2 (0.5M) with H2S04 (1M) will correspond to—
- 0.5mol
- 1.0mol
- 1.5mol
- 2.0mol
Answer: 1. 0.5mol
⇒ \(\begin{gathered}
{\left[\mathrm{Ba}^{2+}\right]=0.5 \mathrm{~mol} \cdot \mathrm{L}^{-1} ;\left[\mathrm{SO}_4^{2-}\right]=1 \mathrm{~mol} \cdot \mathrm{L}^{-1}} \\
\mathrm{Ba}^{2+}(a q)+\mathrm{SO}_4^{2-}(a q) \rightleftharpoons \mathrm{BaSO}_4(s)
\end{gathered}\)
0.5mol of Ba2+ would react with 0.5mol of SO2-4 ions to form 0.5 mol of BaS04.
Question 45. 10mL of liquid carbon disulfide (specific gravity 2.63) is burnt in oxygen. Find the volume of the resulting gases measured at STP—
- 23.25L
- 22.26L
- 23.60L
- 202.08L
Answer: 1. 23.25L
1 mL of CS2 weights 2.63 g
10mL of CS2 will weigh 26.3
⇒ \(\mathrm{CS}_2+3 \mathrm{O}_2 \rightarrow \mathrm{CO}_2+2 \mathrm{SO}_2
\begin{gathered}
12+(2 \times 32) \\
76 \mathrm{~g}
\end{gathered} \quad 22.4 \mathrm{~L} \underbrace{44.8 \mathrm{~L}}_{67.2 \mathrm{~L}}\)
Question 46. A mixture of two bivalent metals A and B having a mass of 2g when dissolved in HC1 at STP, 2.24L H2 is evolved. What is the mass of A present in the mixture (Atomic mass of A = 15u, B = 30u)
- 1g
- 1.5g
- 0.5g
- 0.75g
Answer: 1. 1g
⇒ \(\begin{aligned}
& \mathrm{A}+2 \mathrm{HCl} \rightarrow \mathrm{ACl}_2+\mathrm{H}_2 \\
& \text { Mole: } \frac{x}{15}>\frac{x}{15} \\
& \mathrm{~B}+2 \mathrm{HCl} \rightarrow \mathrm{BCl}_2+\mathrm{H}_2 \\
& \text { Mole: } \frac{2-x}{30} \quad \frac{2-x}{30} \\
&
\end{aligned}\)
Question 47. The normality of 10% H2S04 solution having density 1.1 g/cc is
- 2.05N
- 1.25N
- 3.45N
- 2.24N
Answer: 4. 2.24N
Weight of H2SO4= l0g
Weight of solution l00
⇒ \(\text { Volume of solution }=\frac{\text { mass of solution }}{\text { density of soution }}\)
⇒ \(\text { Normality }=\frac{n_{\mathrm{H}_2 \mathrm{SO}_4} \times 1000}{\text { volume of solution }(\text { in } \mathrm{mL})}\)
⇒ \(=\frac{w_{\mathrm{H}_2 \mathrm{SO}_4} \times 1000}{\text { gram-equivalent mass of } \times \text { Volume of solution (in mL) }}\)
⇒ \(=\frac{10 \times 1000}{\left(\frac{98}{2}\right) \times 90.91}=2.24 \mathrm{~N}\)
Question 48. A hydrate of Na2S03 completely loses 22.2% of water by mass on strong heating. The hydrate is—
- Na2SO3- 4H2O
- Na2SO3- 4H2O
- Na2SO3- H2O
- Na2SO3- 2H2O
Answer: 4. Na2SO3- 2H2O
Question 49. 20g of an acid furnishes 0.5 moles of H30+ ions on complete ionization in its aqueous solution. The value of 1 g eq of that acid will be—
- 40g
- 20g
- 10g
- 100g
Answer: 1. 40g
Question 50. Fe2S3 →FeSO2 + SO2; in this reaction the equivalent weight of Fe2S3 (assuming Sin -2 oxidation state) is—
- \(\frac{M}{4}\)
- \(\frac{M}{16}\)
- \(\frac{M}{22}\)
- \(\frac{M}{20}\)
Answer: 4. \(\frac{M}{20}\)
Question 51. The equivalent mass of iron in the reaction is
⇒ \(2 \mathrm{Fe}+3 \mathrm{Cl}_2 \rightarrow 2 \mathrm{FeCl}_3\)
- Half of its molecular mass
- One third its molecular mass
- Same as its molecular mass
- One fourth its molecular mass
Answer: 2. One-third of its molecular mass
Question 52. A sample of Na2C03 -H20 weighing 0.62g is added to 100ml of. JN H2S04.The resulting solution will be—
- Acidic
- Basic
- Neutral
- Amphoteric
Answer: 3. Neutral
Question 53. The normality of 10% (weight/volume) acetic acid is—
- IN
- 10N
- 1.66N
- 0.83N
Answer: 3. 0.83N
Question 54. If the equivalent weight of an element is 32, then the % of oxygen in its oxide is—
- 16
- 40
- 32
- 20
Answer: 4. 20
Question 55. I- reduces \(\mathrm{SO}_4^{2-}\) to H2S in acidic medium as per the reaction, \(8 \mathrm{KI}+5 \mathrm{H}_2 \mathrm{SO}_4 \rightarrow 4 \mathrm{~K}_2 \mathrm{SO}_4+4 \mathrm{I}_2+\mathrm{H}_2 \mathrm{~S}+4 \mathrm{H}_2 \mathrm{O}\) To produce 34.0g H2S, volume of 0.20(M) H2S04 required is—
- 25.0L
- 12.5L
- 10. OL
- 5.0L
Answer: 1. 25.0L
Question 56. A mixture containing 1 mol of ethane and 4 moles of oxygen is ignited in a sealed container at 100. The reaction occurring is shown in the equation
⇒ \(\mathrm{C}_2 \mathrm{H}_4(g)+3 \mathrm{O}_2(g) \rightarrow 2 \mathrm{CO}_2(g)+2 \mathrm{H}_2 \mathrm{O}(g)\)
Calculate the total no. of mole of gas at the end of the reaction—
- 2
- 3
- 4
- 5
Answer: 4. 5
Question 57. The mass ratio of Na2SO3 and H2O in Na2SO3 x H20 is 1: 1 thus the ratio of their mole number is—
- 1:1
- 1:3
- 1:7
- 7:1
Answer: 3. 1:7
Question 58. When 0.273g Mg is heated strongly in a nitrogen (N2) atmosphere, 0.378 of the compound is formed. Hence the compound formed is—
- Mg3N2
- Mg3N
- mg2N3
- MgN
Answer: 1. Mg3N2
Question 59. NH3 is formed in the following steps:
- \(\mathrm{Ca}+2 \mathrm{C} \rightarrow \mathrm{CaC}_2 50 \% \text { yield }\)
- \(\mathrm{CaC}_2+\mathrm{N}_2 \rightarrow \mathrm{CaCN}_2+\mathrm{C} 100 \% \text { yield }\)
- \(\mathrm{CaCN}_2+3 \mathrm{H}_2 \mathrm{O} \rightarrow 2 \mathrm{NH}_3+\mathrm{CaCO}_3 50 \% \text { yield }\)
To obtain 2 moles of NH3, calcium required is—
- 1 mol
- 2 mol
- 3 mol
- 4 mol
Answer: 4. 4 mol
Question 60. Silver ions react with chloride ions
⇒ \(\mathrm{Ag}^{+}(a q)+\mathrm{Cl}^{-}(a q) \rightarrow \mathrm{AgCl}(s)\)
5cm3 of a 0.1mol/cm3 solution of the chloride of metal X needs 10cm3 of 0.1mol/cm3 silver nitrate for complete reaction. What is the formula ofthe chloride—
- XCl4
- XCl2
- XCl
- X2Cl
Answer: 2. XCl2
Question 61. An equal volume of H2S & SO2 reacts at NTP to form H2O and 2S + SO2→2H2O + 3S. In this reaction,
- H2s is the limit reactant
- S02 is the limit ingreactant
- Sulfur formed is three times of s02 reacted
- Sulfur formed is 1.5 times of H2s reacted
Select the correct statements(s).
- All except 1
- All except 2
- All except 3
- All except 2,4
Answer: 2. All except 2
Question 62. \(\mathrm{NaI}+\mathrm{AgNO}_3 \rightarrow \mathrm{AgI}+\mathrm{NaNO}_2\)
⇒ \(\begin{gathered}
\mathrm{AgI}+\mathrm{Fe} \rightarrow \mathrm{FeI}_2+\mathrm{Ag} \\
\mathrm{FeI}_2+\mathrm{Cl}_2 \rightarrow \mathrm{FeCl}_2+\mathrm{I}_2
\end{gathered}\)
(Atomic mass of Ag = 108,I = 127, Fe = 56, N = 14, Cl = 35.5 ). The above reaction is carried out by taking 75kg of Nal and 255kg of AgNO3. Therefore, the number of moles of iodine formed is
- 0.5
- 500
- 250
- 0.25
Answer: 3. 250
Question 63. The mole fraction and molarity of 46% (by weight) aqueous solution of ethanol is
- 0.25,18.52
- 0.75,1.0
- 0.46,18.52
- 0.54,1.0
Answer: 1. 0.25,18.52
Question 64. The mole fraction and molarity of 46% (by weight) aqueous solution of ethanol is
- 4/5 mol
- 3/5 mol
- 1m
- 2/5mol
Answer: 4. 2/5mol
Question 65. A sphere of radius 7 cm contains 56% iron. If the density of the sphere is 1.4g/cm3, then the approximate amount of iron present is—
- 20
- 10
- 15
- 25
Answer: 1. 20
Question 66. When 800g of a 40% solution by weight was cooled, 100g of solute was precipitated. The percentage composition of the remaining solution is
- 31.4%
- 20.0%
- 50%
- 25%
Answer: 1. 31.4%
Question 67. 0.70g mixture of (NH2)2SO4 was boiled with 100ml of 0.2N NaOH solution till all the NH3(g) evolved and get dissolved to 250mL and lOmL of this solution was neutralized by using 10mL of a 0.1N H2SO4 solution. The percentage purity ofthe (NH4)2S04 sample is—
- 94.3
- 50.8
- 47.4
- 79.8
Answer: 1. 94.3
Question 68. A blood sample is to be analyzed for calcium content If J 20mL of 0.001 (M) KMn04 solution is required to react with 10 mL of blood sample (containing calcium oxalate) then the concentration of calcium ion (in ppm)in the blood is—
- 50 ppm
- 100 ppm
- 150 ppm
- 200 ppm
Answer: 4. 200 ppm
Question 69. If in a solution NaCl is present as 5.85g per 500 of the solution, then the molarity of the solution will be
- 4 mol-.L-1
- 20 mol.L-1
- 2 mol.L-1
- 0.2 mol.L-1
Answer: 4. 0.2 mol.L-1
Question 70. KC104 can be prepared by the following reactions:
⇒ \(\begin{gathered}
\mathrm{Cl}_2+2 \mathrm{KOH} \rightarrow \mathrm{KCl}+\mathrm{KClO}+\mathrm{H}_2 \mathrm{O} \\
3 \mathrm{KClO} \rightarrow 2 \mathrm{KCl}+\mathrm{KClO}_3 ; 4 \mathrm{KClO}_3 \rightarrow 3 \mathrm{KClO}_4+\mathrm{KCl}
\end{gathered}\)
To prepare 200g KC1O4, the required amount of Cl2 is equivalent to—
- 8.95 equivalent H2SO4
- 129.02L O2 at STP
- 11.52 mol oxygen
- 410.1g Chlorine
Answer: 4. 410.1g Chlorine
Question 71. 103- , oxidises SOf- to SO2- in acidic medium 100mL solution containing 2.14g of KI03 reacts with 60mL of 0.5(N) Na2S03 solution. The final oxidation state of iodine in reduced species is—
- +5
- +3
- -1
- +1
Answer: 3. +1
Question 72. The concentration of both Na2CO3 and NaHCO2 is 5.2 x 10-3 mol in their mixture. The amount of 0.1(M) HC1 required to neutralise this mixture completely—
- 1.56L
- 1.57L
- 15.7L
- 156.0mL
Answer: 4. 156.0mL
Question 73. 0g of pyrolusite on reaction with cone. HC1 liberated 0.1 equivalent of Cl2. Percentage purity of pyrolusite sample—
- 87.0%
- 43.5%
- 21.75%
- 100%
Answer: 2. 43.5%
Question 74. 100mL of30%[mlv) NaOH solution is mixed with lOOmL of 90% (m/v) NaOH solution then the molarity of the final solution will be
- 1.3
- 13
- 1/5
- 15
Answer: 4. 15
Question 75. 5.3g of M2CO3 is dissolved in 150mL of 1(N) HC1. Unused acid required 100mL of 0.5(N) NaOH. Hence the equivalent weight of M is—
- 23
- 12
- 24
- 13
Answer: 1. 23
Question 76. Methane was burnt in an incorrectly adjusted burner. The methane was converted into a mixture of carbon dioxide and carbon monoxide in the ratio of 99 : 1, together with water vapor, what will -be the volume of oxygen consumed when y dm3 of methane is burnt—
⇒ \(\left(2 y-\frac{0.02}{2} y\right) \mathrm{dm}^3\)
(2y-0.01y)dm³
⇒ \(\left(y-\frac{0.01}{2} y\right) \mathrm{dm}^3\)
(y-0.0ly)dm3
Answer: 1. \(\left(2 y-\frac{0.02}{2} y\right) \mathrm{dm}^3\)
Question 77. The equivalent weight of MnS04 is half of its molecular mass when it is converted to—
- Mn2O3
- MnO2
- MnO-4
- MnO2-4
Answer: 2. MnO2
Question 78. The mass percent of the carbon in carbon dioxide—
- 0.034
- 27.27
- 3.4
- 28.7
Answer: 2. 27.27
Question 79. If the concentration of glucose in the blood is 0.9g-L_1, then its molarity will be—
- A
- 50
- 0.005
- 0.5
Answer: 3. 0.005
Question 80. In an experiment, 4g M2Ox oxide was reduced to 2.8g of the metal. If the atomic mass of the metal is 56g – mol-1, the number of-atoms in the oxide is
- 1
- 2
- 3
- 4
Answer: 3. 3
Question 81. The specific volume of a cylindrical virus particle is 6.02 x 10_2cc-g_1 whose radius and length are 7A & 10A respectively. Molecular mass (kg.mol-1 ) of the virus—
- 15.4
- 1.54 X 104
- 3.08 X 104
- 3.08 x 103
Answer: 1. 15.4
Question 82. 10mL of CS2 (Specific gravity 2.63) is burnt in oxygen. The volume ofthe resulting gases at STP will be
- 23.25L
- 22.26L
- 23.50L
- 20.08L
Answer: 1. 23.25L
Question 83. An element X has the following isotopic composition— X: 90%, X: 8.9%,202X: 1.1% . The weighted average atomic mass ofthe naturally occurring element X is
- 201
- 202
- 199
- 200
Answer: 4. 200
Question 84. At room temperature, the no of molecules present in a drop of water (volume 0.0018mL and density lg/cc) is—
- 6.02 X 1023
- 1.084 X 1018
- 4.84 X 10 17
- 6.02X 1019
Answer: 4. 6.02X 1019
Question 85. If a signature contains mg of C-atom then the number of C-atom in the signature is—
- 6.02 X 10 20
- 0.502 X 1020
- 5.02 X 10 23
- 5.02 X1020
Answer: 2. 0.502 X 1020
Question 86. No. of valence electron present in 4.2g nitride ion is —
- 2.4NA
- 4.2NA
- 1.6NA
- 3.2NA
Answer: 1. 2.4NA
Question 87. An element forms four compounds A, B, C & D. The Ratio of the equivalent weight of the compounds is 1: 2: 3: 4 . The compound in which the element has the highest valency is—
- A
- B
- C
- D
Answer: 1. A
Question 88. In which of the following compounds M has the highest equivalent weight—
- MO
- MO2
- M2O3
- M20
Answer: 1. MO
Question 89. The atomic mass of two compounds A and B is 30 and 60 respectively. If Xg of A contains Y atoms then no of atoms present in 2Xg of is
- 2Y
- Y
- 4Y
- Y/2
Answer: 2. Y
Question 90. Density (in g-mlr1) of a 3.60 (M) H2S04 solution that is 29% H2S04 (Molar mass = 98g mol-1 ) by mass will be—
- 1.64
- 1.45
- 1.22
- 1.88
Answer: 3. 1.22
Question 91. Polyethylene can be produced from CaC2 according to the following sequence of reactions.
⇒ \(\begin{aligned}
& \mathrm{CaC}_2+\mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{Ca}(\mathrm{OH})_2+\mathrm{C}_2 \mathrm{H}_2 \\
& n \mathrm{C}_2 \mathrm{H}_2+n \mathrm{H}_2 \rightarrow+\mathrm{CH}_2-\mathrm{CH}_2-{ }_n
\end{aligned}\)
The mass of polyethylene, which can be produced from 20kg of CaC2 is—
- 6.75Kg
- 7.75kg
- 9.75kg
- 8.75kg
Answer: 4. 8.75kg
Question 92. The weight of one molecule of a compound C6OH12 is—
- 1.3 X 10-20 g
- 1.4 x 10-21 g
- 1.9924 X 10-24 g
- 5.01 X 10-21 g
Answer: 2. 1.4 x 10-21 g
Question 93. A 2.0g mixture of Na2C03 and NaHCOa suffered a loss of 0.12g on heating. Percentage of Na2CO3 in the mixture
- 83.8
- 16.2
- 38.8
- 61.2
Answer: 1. 83.8
Question 94. Which of the following does not have the same percentage of carbon as ethene(C2H4 )—
- C4H8
- C6H12
- C6H10
- C5H10
Answer: 3. C6H10
Question 95. xL of nitrogen at STP contains 3 x 1022 molecules. The number of molecules in \(\frac{x}{2} \mathrm{~L}\) of ozone at STP will be—
- 3 x 1022
- 1.5 x 10 22
- 1.5 X 10 21
- 1.5 X 10 11
Answer: 2. 1.5 x 10 22
Question 96. A sample of A1F3 contains 3 X 1025P” Ions. The number of Al3+ in the sample would be
- 3X1025
- 1X1025
- 1.5 X 1025
- 2X1025
Answer: 2. 1X1025
Question 97. X and Y are the two elements that form X2Y3 and X3Y4. 0.2 mol of X2Y3 weighs 32g and 0.4mol of X3Y4 weighs 92.8g. The atomic masses of and Y respectively are—
- 16u and 56u
- 8u and 28u
- 56u and 16u
- 28u and 8u
Answer: 3. 56u and 16u
Question 98. The percentage of Sc (atomic weight = 78.4) in peroxidase anhydrase enzyme is 0.5% by weight the minimum molecular weight of peroxidase anhydrase enzyme is—
- 1.568 x 104
- 1.568 X 103
- 15.618 X108
- 2.136 X104
Answer: 1. 1.568 x 104
Question 99. How many moles of magnesium phosphate will contain 0.25 mol of oxygen atoms
- 0.02
- 3.125 x 10-2
- 1.25 X 10-2
- 2.5 X 10-2
Answer: 2. 3.125 x 10-2
Question 100. The amount of calcium oxide required, when it reacts with 852g of P4O10 is
- 100g
- 1008g
- 108g
- 1050g
Answer: 2. 1008g
Question 101. 25.3g of Na2CO3 is dissolved in enough water to make 250 ml of solution. If sodium carbonate dissociates completely, the molar concentration of sodium ion, Na+ and carbonate ions, \(\mathrm{CO}_3^{2-}\) are respectively—
- 0.955 (M) and 1.910 (M)
- 1.910 (M) and 0.955 (M)
- 1.90 (M) and 1.910 (M)
- 0.477 (M) and 0.477 (M)
Answer: 1. 0.955 (M) and 1.910 (M)
Question 102. How many moles of lead (II) chloride will be formed from a reaction between 6.5g of PbO and 3.2g HC1
- 0.011
- 0.029
- 0.044
- 0.333
Answer: 2. 0.029
Question 103. Which ofthe following exist together—
- NaOH + NaCl
- NaOH + NaHCO3
- SnCl2 + PbCl4
- Na2CO3 + NaOH
Answer: 1. NaOH + NaCl
Question 104. In 46% (by weight) aqueous solution of ethanol—
- The mole fraction of ethanol is 0.25
- The mole fraction of water is 0.75
- The mole fraction solvent (as water solvent) is 18.52
- Molarityis l0.00 mol-l-1
Answer: 1. The Mole fraction of ethanol is 0.25
Question 105. A 110% sample of oleum contains—
- 44.4% of SO2
- 55.6% of sulphuric acid
- 55.6% of SO3
- 44.4% of sulphuric acid
Answer: 1. 44.4% of SO3
Question 106. The volume of CO2 formed when a mixture of 2mol NaHCO3 and 1 mol Na2CO3 at STP, is—
- 2 equivalent H2 gas
- 6 equivalent O3 gas
- 4 equivalent O2 gas
- 2 equivalent Cl2 gas
Answer: 1. 2 equivalent H2 gas
Question 107. A and B are two elements which form AB2 and A2B3. If 0.18 mol of AB2 weighs 10.6g and 0.18 mol of A2B3 weighs 17.8g then—
- The atomic mass of a is 20.05
- The atomic mass of b is 20.05
- The atomic mass of a is 18.8
- The atomic mass of b is 18.8
Answer: 1. The Atomic mass of a is 20.05
Question 108. 100mL of the mixture of CO & CO2 is mixed with 30mL of oxygen and sparked in an eudiometer. The volume of residual gas after treatment with aqueous KOH was 10rnL, which remains unchanged when treated with alkaline pyrogallate. Which ofthe following is correct—
- The volume of CO2 absorbedbykoh is 90ml
- The volume of co initially was 70ml
- The volume initially present was 50
- The volume of CO2 absorbed by koh is 80
Answer: 1. The volume of CO2 absorbedbykoh is 90ml
Question 109. Which ofthe following pairs have the same no. of atoms—
- 16g of O2(g) and 4g of H2(g)
- 16g of O2(g) and 44g of CO2(g)
- 28g of N2(g) and 32g of O2(g)
- 12g of C(s) and 23g of Na(s)
Answer: 3. 28g of N2(g) and 32g of 02(g)
Question 110. 0.6 moles of K2Cr2O7 can oxidize—
- 3.6 mol of FeSO2 to Fe2(SO4)3
- 0.1mol of FeSO4 to Fe2(SO4)3
- 0.05 mol of Sn+2 to Sn+4
- 1.8 mol of Sn+2 to Sn+4
Answer: 1. 3.6 mol of FeS04 to Fe2(S04)3
Question 111. The sulfate of metal A contains 20% of M. This sulfate is isomorphic with ZnS04 7H20. Which ofthe following are true about metal M—
- The atomic mass of metal is 24
- Metal is bivalent
- Eq. Wt. Of metal is 12
- The salt of metal is mgs04 7H2O
Answer: 1. Atomic mass of metal is 24
Question 112. lol BaF2 + 2 mol H2SO4— mixture will be completely neutralized by—
- 1mol of KOH
- 2 mol of Ca(OH)2
- 4 mol of KOH
- 2 mol of KOH
Answer: 2. 2 mol of Ca(OH)2
Question 113. The reaction between H2SO4 and NaOH is given below
⇒ \(\mathrm{H}_2 \mathrm{SO}_4+2 \mathrm{NaOH} \rightarrow \mathrm{Na}_2 \mathrm{SO}_4+2 \mathrm{H}_2 \mathrm{O}\)
The amount and molarity of the Na2S04 produced when 0.1(M) 1L H2SO4 reacts with 0.1(M) 1L NaOH will be
- 7.10g
- 3.55g
- 0.025mol.L-1
- 0.1mol.L-1
Answer: 1. 7.10g
Question 114. If the density of air is 0.001293g/cm3 at STP then—
- The vapor density of air is 14.48
- The molecular mass of air is 28.96
- The vapor density of air is 0.001293g/cm3
- Vap. Density &mol. The mass of air cannot be determined
Answer: 1. Vapour density of air is 14.48
Question 115. You are provided with 1 (M) solution of NaNO3 whose density is 1.25g/mL. So, in the solution—
- The percentage by mass of nan O3 = 6.8
- The percentage of H2O = 93.2
- The molality of the solution is 10.72
- The solution has 0.2mol of nanO3
Answer: 1. The percentage by mass of nanO3 = 6.8
Question 116. 4.4 g CO2 signifies—
- 0.1 mol CO2
- 6.02 x 1022 molecules of CO2
- 8.8g of oxygen atom
- 1120mL CO2 at STP
Answer: 1. 0.1 mol CO2
Question 117. Which ofthe following have equal concentration—
- 200mL solution of 20gNaOH
- 100mL solution of 40gNaOH
- 200 mL solution of 0.5 mol KC1
- 200 mL solution of 20g KOH
Answer: 1. 200mL solution of 20gNaOH
Question 118. 8g of O2 has the same number of molecules as—
- 7gCO
- 14gN2
- llgCO2
- 16gSO2
Answer: 1. 7gCO
Question 119. At the same temperature and pressure, 10cc of an organic compound in the gaseous state was sparked with an excess of O2. 20cc of CO2 and 5cc of N2 were obtained among the products. Which of the following molecular formulas would fit these data—
- C2H7N
- C2H3N
- O2H6N2
- CH5N
Answer: 1. C2H7N
Question 120. Which of the following concentration-related terms are not affected by the change In temperature
- Molality
- molality
- Normality
- Mole function
Answer: 2. Molality
Question 121. The equivalent weights of sulfur in its oxides are—
- 32
- 8
- 24
- 5.33
Answer: 2. 8
Question 122. Two oxides of metal contain 50% and 40% metal (M) respectively. The formula of the oxides are
- M2O
- MO2
- MO3
- M2O2
Answer: 2. MO2
Question 123. Pairs of species having the same percentage of carbon are
- CH3COOH & C6H12O
- CH3COOH & C2H5OH
- HCOOH6 & C4H8O4
- C6H12O6 and C12H2O2
Answer: 1. CH3COOH and C6H12O
Question 124. Which of the following sets of compounds correctly follows the law of reciprocal proportions—
- N2O5,NH3,H2O
- N2O,NH3,H2O
- P2O3,PH3,H2O
- CH4, H2O, CO2
Answer: 3. N2O5,NH3,H2O
Question 125. Which ofthe following weigh 32g—
- 1 mol oxygen molecules
- 1 mol oxygen atoms
- 1 mol CO molecules
- 22.4L oxygen molecules (STP)
Answer: 1. 1 mol oxygen molecules
Very Short Question And Answers
Question 1. Give the relation between units of pressure in CGS & SI systems.
Answer: 1 dyne cm-2 = O.lPa
Question 2. Which quantity is measured by the ‘Angstrom’ unit?
Answer: Length
Question 3. Which quantity is measured in the ‘Pascal’ unit?
Answer: Louis Pressure
Question 4. Who proposed the law of definite proportion?
Answer: Proust
Question 5. Give an example where the law of infinite proportion fails.
Answer: Cu1.7 S
Question 6. Which law of chemical combination was proposed by Richter?
Answer: Law of reciprocal proportion
Question 7. Which measurement is more precise, 4.0 g or 4.00 g?
Answer: 4.00g
Question 8. Who is known as the father of atomic theory?
Answer: Dalton
Question 9. Give an example of a berthollide compound.
Answer: Cu1.7S
Question 10. Which scale is approved by IUPAC for measuring atomic mass?
Answer: 12 C scale
Question 11. Express1 amuin gram.
Answer: 1.66×10-24g
Question 12. What is the actual mass of a C -atom?
Answer: 12 amu
Question 13. What is the volume of 1 mol of any gas at STP?
Answer: 22.4L
Question 14. For which type of compounds, “formula mass” is used?
Answer: Ionic Compounds
Question 15. What is the mass of 1L ofhydrogen gas at STP?
Answer: 0.089g
Question 16. Who determined the value of Avogadro’s number?
Answer: Millikan
Question 17. What is the number of ions present in “1 mol ion”?
Answer: 6.022×1023
Question 18. How is the relative density of a gas related to its normal density?
Answer: Normal density = vapour deputy x 0.089
Question 19. Which type of elements have fixed equivalent masses?
Answer: Elements having a single valency.
Question 20. Divide the unit of volume (cm3) by the unit of area (m2) and then indicate the resulting unit.
Answer: The resulting unit is cm. [Here, m2 is first converted to cm2 and then the division is carried out.
Question 21. Give an example of one antibiotic drug and a tranquilizer.
Answer: Tetracycline (antibiotic) and barbituric acid (tranquilizer)
Question 22. Use the data given in the following table to calculate the molar mass of naturally occurring argon isotopes.
Answer: Average molar mass of argon = (0.00337 x 35.96755) +(0.00063 X 37.96272) + (0.99600 X 39.9624) = 39.948.
Question 23. Is moist air heavier than dry air? Explain.
Answer: Dry air is heavier than moist air because dry air contains mainly N2 and O2 whose vapor densities are higher than the vapor density of H2O present in moist air. [Vapour densities of N2, O2 and H2O are 14, 16 and 9 respectively]
Question 28. What will be the mass of one l2C atom in g?
Answer: Mass of one 12C atom = \(=\frac{12 \mathrm{~g}}{6.022 \times 10^{23}}=1.9927 \times 10^{-23} \mathrm{~g}\)
Question 24. Which is the limiting reagent in the combustion of methane and why is called so?
Answer: Methane is the limiting reagent in this case because the other reagent is atmospheric 02 which is always present in excess amount.
Thus, the amount of C02 and H20 formed will depend upon the amount of CH4 undergoing combustion.
Question 25. A box contains some identical red balls, labeled as A, each weighing 2 grams. Another box contains identical blue balls, labeled as B, each weighing 5 grams. Considering the combinations as AB, Alt., A2B, and A2B3, show that the law of multiple proportions is applicable to this case.
Answer: table-
Masses of B which combine separately with the fixed mass of A (say 4g) will be 10g, 20g, 5g, and 15g, i.e., they are in the ratio 2: 4: 1 : 3 which is a simple whole number ratio. Hence, the law of multiple proportions is applicable to this case.
Question 26. Convert 7.62 cm in inches using unit conversion factor.
Answer: 1 inch= 2.54 cm
⇒ \(\frac{\text { linch }}{2.54 \mathrm{~cm}}\)= 1 (unit conversion factor)
Therefore, 7.62 cm = 7.62 cm \(\times \frac{1 \text { inch }}{2.54 \mathrm{~cm}}\) = 3.
Question 27. The mass of a body determined by an analytical balance ( accuracy O.OOOlg) was reported to be 10.0008 g. Find the no. of significant figures in reported mass.
Answer: It has 6 significant figures (the first 5 digits are certain and the last digit is uncertain. Zeros between non-zero digits are significant figures).
Question 28. The length of a table measured by a meter scale (having an accuracy of 0.1cm) was reported to be 76.8 cm. What is the actual length of the table?
Answer: Actual length ofthe table = (76.8 ±0.1) cm
Question 29. Express the fraction 3/8 in a decimal system having two significant figures
Answer: 3/8 = 0.375 = 0.38 (rounded off to two significant figures); [Here, the rightmost digit to be deleted is 5 and the preceding digits odd. So, the preceding digit is increased by one unit.
Question 30. Is the volume of 1 gram-mole of COz greater than or equal to or less than 22.4L at 300K and 1 atm?
Answer: The volume is greater than 22.4L because the volume of 1 gram-mole of C02 at 273K and 1 atm is exactly 22.4L and this volume increases with temperature.
Question 31. Give the names ofone analgesic and antipyretic drug
Answer: Aspirin (analgesic) and paracetamol (antipyretic).
Question 32. Express 0.00340 cm in exponential form.
Answer: 0.00340 cm = 3.40 X 10-3 cm.
Question 33. Express the mass of one molecule of water in the unit of unified mass.
Answer: Mass ofonemolecule ofwater= (2×1.008+ 16)u= 18.016u.
Question 34. Calculate the no. of electrons in 2 mol of electrons.
Answer: Number ofelectrons = 2 x 6.022 x 1023 = 1.2044 x 1024.
Question 35. Calculate the mass of a diamond that will contain the same number of C-atoms as that in 0.1 g of graphite.
Answer: Both graphite and diamond are the allotropes of carbon So, 0.1 g of diamond will contain the same number of carbon atoms as that in O.lg of graphite.
Question 36. what 110 do you understand by the statement—Molecular mass of ammonia is 17 amu”?
Answer: This statement indicates that the actual mass of one molecule of ammonia is 17 x 1.6605 x 10-24g
Question 37. What do you mean by semimolar Na OH solution?
Answer: The solution in which 40/100 or 0.4g of NaOH remains dissolved in 1L solution is called semimolar NaOH solution.
Question 38. Are the species, N3 and N3 different? Justify’
Answer: N3 denotes an azide ion having one unit of negative charge while N3- denotes a nitride ion having three units of negative charge.
Question 39. 02 and N2 were present in a mixture in a ratio of 1: 7 by weight. Calculate the ratio of the number of molecules
Answer:
⇒ \(\mathrm{O}_2: \mathrm{N}_2=\frac{1}{32}: \frac{7}{28}=1: 8\)
Question 40. What is A heated compound, will strongly be the change in analysis oxygen of shows(Mgwhen=C 24)
Answer: \(2 \mathrm{Mg}(48 \mathrm{~g})+\mathrm{O}_2 \rightarrow 2 \mathrm{MgO}(80 \mathrm{~g})\)
Question 41. A compound, on analysis, shows C=40%, H=6.67% and O=53.33%. Determine the empirical formula of the compound. if the molar mass of the compound is 30g mol-1, what is its molecular formula?
Answer: \(\mathrm{C}: \mathrm{H}: \mathrm{O}=\frac{40}{12}: \frac{6.67}{1}: \frac{53.33}{16}=3.33: 6.67: 3.33=1: 2: 1\)
Question 42. Mention a compound where the constituent atoms are not present in a simple ratio.
Answer: C12H22011.In this compound, C, H, and O atoms are not
combined in a simple whole-number ratio.
Question 43. Mention the actual mass of 12C -atom.
Answer: 12 amu.
Question 44. What do you mean by ‘the mass of an H-atom is 1.008 u’? Calculate this mass in grams.
Answer: 1 amu orlu = 1.6605 x 10-24g.
Question 45. The element, boron has two isotopes ( 10B and 11B ). Calculate the natural abundance ofthe isotopes of boron if the atomic mass ofthe element is 10.8.
Answer: Let us consider the natural abundance of 10B is x%
Question 46. What is the magnitude of 1 amu?
Answer: 1 amu = 1.6605 x 10-24g
Question 47. How will you separate the constituents of a sample of gunpowder?
Answer: Gunpowder is a mixture of charcoal (carbon), sulfur, and niter (KN03). The constituents are separated on the basis of their solubility in different solvents.
Nitre (KN03) is soluble in water. Sulfur is insoluble in water but soluble in carbon disulfide (CS2). Charcoal is insoluble in both water and CS2.
Question 48. How will you separate the components from a mixture of common salt and niter?
Answer: The mixture is dissolved in water and the resulting solution is concentrated. Then the components of the mixture are separated by the fractional distillation process.
Question 49. Explain why is air sometimes considered a heterogeneous mixture.
Answer: When dust or smoke is mixed with air, it is considered a
heterogeneous mixture.
Question 50. State whether a mixture or a compound is formed when cone. H2S04 is added slowly to water.
Answer: Slow addition of cone. H2S04 to water results in the formation of a mixture.
Question 51. Name two drugs that cause intoxication when consumed.
Answer: LSD (Lysergic acid diethylamide) and heroin
Fill In The Blanks
Question 1. The number of significant figures in 2.500 is ___________________.
Answer: 4
Question 2. The law of multiple proportion was postulated by dm3 ___________________.
Answer: Dalton
Question 3. 1L= ___________________dm3
Answer: 1
Question 4. Dalton’s atomic theory can not explain the law of ___________________.
Answer: Gaseous Volumes
Question 5. The sum of2.3 and 6.54 should be reported as ___________________.
Answer: 8.8
Question 6. Quinine is an___________________ drug.
Answer: Antimalalerial
Question 7. Cu1.7 is a___________________
Answer: Bertholide
Question 8. The atomicity ofozone molecule is ___________________
Answer: 3
Question 9. Molecular mass of heliumon the___________________ drug.
Answer: 2
Question 10. ___________________ mass of sodium chloride is 58.5.
Answer: Formula
Question 11. 1 millimole = ___________________mole
Answer: 10-3
Question 12. Mass of1 gram-atom of oxygen is ___________________g.
Answer: 16
Question 13. The physical scale of atomic mass was proposed by ___________________.
Answer: Aston
Question 14. Mass of ___________________atoms(s) of nitrogen is 14 amu.
Answer: One
Question 15. Molecular mass of heavy water is___________________
Answer: 20.
Question 16. The equivalent mass of copper in CuO is___________________
Answer: 31.75
Question 17. Equivalent mass of an element varies___________________ valency.
Answer: inversely
Question 18. Law of isomorphism was postulated by___________________
Answer: Mitscherlich
Question 19. The empirical formula of glucose is ___________________
Answer: CH2O
Question 20. The S.I. unit ofmolar concentration is___________________.
Answer: mol.m-3
Question 21. The converse of the law of___________________ true for isomeric compounds.
Answer: Definite
Question 22. The equivalent mass of oxidants and reductants depends on the ___________________ of the reaction medium.
Answer: pH
Question 23. ___________________concentration of a solution doesnot depend on temperature.
Answer: Molal
Question 24. Atomic mass = X valency.
Answer: Equivalentmass
Numerical Problems
Question 1. Chlorine and oxygen form different compounds. One of them contains 81.6%’ chlorine and the other contains 59.7% chlorine. Which law of chemical combination supports these observations?
Answer:
Given
Chlorine and oxygen form different compounds. One of them contains 81.6%’ chlorine and the other contains 59.7% chlorine.
In the 1st and 2nd compound percentages of oxygen are 18.4 and 40.31 respectively. The ratio of the masses of oxygen that combine separately with a fixed mass of carbon (81.6 parts) in these compounds is
⇒ \(=18.4: \frac{40.3 \times 81.6}{59.7}=18.4: 55.08 \approx 1: 3\)
So, the observation supports the law of multiple proportions.
Question 2. Two oxides of a metal (M) contain 22.53% and 30.38% of oxygen. If the second oxide is M2O3, find the formula of the first oxide.
Answer:
Given
Two oxides of a metal (M) contain 22.53% and 30.38% of oxygen. If the second oxide is M2O3,
Let us consider, the atomic mass of M is a
Amount of oxygen in M2O3 \(=\frac{48}{2 a+48} \times 100=30.38 \%\)
Or, a=55
Let us consider, the formula of the first oxide Is M2O3
Amount of oxygen in M2 Ox \(=\frac{16 x}{2 a+16 x} \times 100=22.53 \%\)
⇒ \(\text { or, } \frac{16 x}{110+16 x}=0.2253 \%\)
∴ \(x \approx 2\)
∴ The formula of the first oxide is M2O3 or MO
Question 3. Three elements X, Y, and Z form three different compounds XY, YZ, and XZ. If XY contains 75% X, XZ contains 72.76% Z, and YZ contains 11.11% Y, then show that these results illustrate the law of reciprocal proportion.
Answer:
Given
Three elements X, Y, and Z form three different compounds XY, YZ, and XZ. If XY contains 75% X, XZ contains 72.76% Z, and YZ contains 11.11% Y,
In XY compound, the mass of X: mass of Y = 75: 25 = 3: 1
In the XZ compound, the mass of X: mass of Z = 27.24: 72.76 = 3:8
Therefore If the elements Y and Z combine, the ratio of their masses in the compound YZ will be 1: 8 or any simple multiple of. But in the compound YZ, the mass of Y: mass of Zs 11.11: 88.89 =1:8
Therefore These results illustrate the law of reciprocal proportion.
Question 4. Calculate the volume of oxygen that will react with hydrogen produced by the decomposition of 50 cm3 of ammonia. Both the reactions occur at 18°C & 76 cm of Hg.
Answer: Reaction: \(\left.2 \mathrm{NH}_3(2 \text { vol. }) \rightarrow \mathrm{N}_2 \text { (1 vol. }\right)+3 \mathrm{H}_2(3 \mathrm{vol} .)\)
In 18°Cand 76 cm Hg pressure, the volume of hydrogen produced by the decomposition of 50cm3 ammonia = 50 X (3/2) =75cm³
Reaction: \(\mathrm{H}_2(1 \mathrm{vol} .)+\frac{1}{2} \mathrm{O}_2\left(\frac{1}{2} \text { vol. }\right) \rightarrow \mathrm{H}_2 \mathrm{O}(1 \mathrm{vol} .)\)
At 18°C and 76 cm Hg pressure, the volume of oxygen that reacts with 75 cm³ of hydrogen = 75/2 = 37.5 cm3.
Question 5. Calculate the number of (a) CH4 molecules (b) C -atoms and (c) H-atoms in 25 g of CH4 gas.
Answer: 25g CH4 \(=\frac{25}{16} \mathrm{~mol} \mathrm{CH}_4\)
since the molecular mass of Ch4 is 16 g
Therefore Number of molecules in \(\frac{25}{16} \mathrm{~mol} \mathrm{CH}_4\)
⇒ \(=6.022 \times 10^{23} \times \frac{25}{16} \mathrm{CH}_4=9.41 \times 10^{23}\)
Question 6. Calculate: 1. mass, 2. volume at STP, 3. number of molecules present in 0.5 mol of CO2.
Answer: No. of C-atoms in one CH4 molecule = 1 No. ofC-atoms in 9.41 X 1023CH4 molecules = 9.41 X 1023 c) No. of H-atoms in one CH4 molecule = 4 No. of H-atoms in 9.41 X 1023 CH4 molecule.
⇒ \(=4 \times 9.41 \times 10^{23}=3.764 \times 10^{24}\)
1 mol CO2 = 44g of CO2
0.5 mol CO2 = 44 x 0.5 = 22g of CO2
At STP, volume of1 mol CO2 gas =22.4l
At STP, volume of0.5mol CO2 gas = 22.4 x 05L = 11.2L
(Number of molecules in 1 mol CO2 = 6.022 x 1023
Number of a molecules in 0.5mol CO2 = 3.011 x 1023
Question 7. Find the mass of carbon that contains the same number of atoms as contained by 560 g of iron (atomic mass = 56).
Answer: No. Fe-atoms presentin1 mol or 56g of
Fe = 6.022 x 1023
Therefore Number of ofFe-atoms present 560 g of Fe
⇒ \(=6.022 \times 10^{23} \times \frac{560}{56}=6.022 \times 10^{24}\)
Mass of 6.022 x 1023 number ofC-atoms = 12g
Mass of 6.022 x 1024 number of-atoms= 120g
Question 8. The atomic mass of a metallic element is 54.94. If its density is 7.42 g cm-3, find its atomic volume.
Answer: Atomic Volume \(\frac{\text { g-atomic mass }}{\text { density }}\)
⇒ \(=\frac{54.94 \mathrm{~g}}{7.42 \mathrm{~g} \cdot \mathrm{cm}^{-3}}=7.404 \mathrm{~cm}^3\)
Question 9. Calculate the number of O -atoms in 88 g of C02. Also, calculate the mass of CO which will contain the number of O -atoms.
Answer: 88g CO2 \(=\frac{88}{44}=2 \mathrm{~mol} \mathrm{CO}_2\)
1 mol of CO2 contains 2 x 6.022 x 1023 no. of oxygen atoms.
2 mol of C02 contains 4 X 6.022 X 1023
= 2.4088 x 1024 number of oxygen atoms.
Number of oxygen atoms in1 mol CO = 6.022 x 1023
Therefore 2.4088 x 1024 number of oxygen atoms present in
⇒ \(=\frac{2.4088 \times 10^{24}}{6.022 \times 10^{23}}\) mol of CO = 2mol of CO
= 4 x 28g of CO = 112g of CO
Question 10. At STP, how many moles of CO2 are present in 5.6L of CO2?
Answer: Number of moles present in 22.4L CO2 = 1
Number of moles present in 5.6L CO2 \(=\frac{5.6}{22.4}=\frac{1}{4}\)
Question 11. 1L of air contains 21% of oxygen by volume at STP. What is the number of moles of oxygen in 1L of air?
Answer: At STP, the volume of 02 present L air is 0.21L.
At STP, the number of moles present is 22.4L02 = 1
At STP, no. of mole present in 0.21L 02 \(=\frac{0.21}{22.4}\)
= 9.375 xlO-3
At STP, no.of moles of 02 present in 1L air = 9.3375 X 10-3
Question 12. The mass of 1 mL of H2 gas at STP is the same as the mass of 9.68 x 1017 atoms of iron. Find the atomic mass of iron.
Answer: At STP, the mass of 22400 mL H2 gas = 2g
Therefore At STP, mass of1 mL H2 gas \(=\frac{2}{22400}=\frac{1}{11200} \mathrm{~g}\)
At STP, the mass of 1ml. H2 mass of 9.68 x 1017 na of Fe atoms.
mass of 9.68 x 1017 number of-atoms =1/11200g or, mass of 6.022 x 1023 number of Fe-atoms
⇒ \(=\frac{6.022 \times 10^{23}}{9.68 \times 10^{17} \times 11200} \mathrm{~g}=55.55 \mathrm{~g}\)
Therefore Atomic mass of iron is 55.55
Question 13. Find the ratio of the masses of equal volumes of CH4, C2H4, and C2H2 gases under the same temperature and pressure.
Answer: Under the same conditions of temperature and pressure.
the ratio of the number of moles present in equal volumes of
CH4, C2H4 and C2H2 is \(\mathrm{CH}_4: \mathrm{C}_2 \mathrm{H}_4: \mathrm{C}_2 \mathrm{H}_2=1: 1:\) Ratio of their masses—
⇒ \(m_{\mathrm{CH}_4}:{ }^m \mathrm{C}_2 \mathrm{H}_4:{ }^m \mathrm{C}_2 \mathrm{H}_2=1 \times 16: 1 \times 28: 26\)
=8: 14: 13
Question 14. The atomic mass of an element is 24. Find the actual mass of one atom of that element. If the atomic number of the element is 11, then find out the number of neutrons present in 0.1 gram-atom.
Answer: \(M_{\text {atom }}=\frac{\text { g-atomic mass }}{N_A}=\frac{24}{6.022 \times 10^{23}}=3.985 \times 10^{-23}\)
Number of neutrons present in an atom whose atomic mass is 24 and atomic number is 11 = 24-11 = 13
Number of atoms present in 1 gram-atom – 6.022 x 1023
Therefore Number of atoms present in 0.1 gram-atom = 6.022 x 1022
Therefore Number of neutrons present in 6.022 x 1022 number of atoms or 0.1 gram-atom
⇒ \(=6.022 \times 10^{22} \times 13=7.829 \times 10^{23}\)
Question 15. Chlorophyll contains 2.68% of magnesium metal by mass. Calculate the number of Mg atoms present in 2.0 g of chlorophyll.
Answer: Amount of Mg present in 2g of chlorophyll
⇒ \(=\frac{2.68}{100} \times 2=0.0536 \mathrm{~g}\)
Gram-atomic mass ofMg = 24 g
Therefore Number of atoms in 24 g = 6.022 x 1023
Therefore Number of atoms present in 0.0536g of Mg
⇒ \(=\frac{6.022 \times 10^{23}}{24} \times 0.0536=1.345 \times 10^{21}\)
Question 16. At 27°C and 780 mm pressure, 1 L of a gas weighs 1.125 g. Find its vapor density-
Answer: Let, at 27°C temperature and 780 mm Hg pressure the the volume of 1L of a gas at STP be VL, then
⇒ \(\frac{760 \times V}{273}=\frac{780 \times 1}{273+27}\left[\frac{P_1 V}{T_1}=\frac{P_2 V_2}{T_2}\right] \quad V=0.934 \mathrm{~L}\)
Therefore At STP, mass of0.934L ofthe gas = 1.125g
Therefore At STP, mass of22.4L ofthe gas \(=\frac{1.125}{0.934} \times 22.4=26.98 \mathrm{~g}\)
Therefore Molecular mass = 26.98 and density \(=\frac{26.98}{2}=13.49\)
Question 17. The vapor density of mercury with respect to air is 6.92. Calculate the number of atoms present in each molecule of mercury vapor. (Given: Hg = 200 and relative density of air = 14.5)
Answer: Vapour density of mercury = 6.92 X 14.5 = 100.34
The molecular mass of mercury vapor
= 2 x 100.34 = 200.60 = atomic mass of mercury number of atoms in each molecule mercury vapor = 1
Question 18. At STP, volumes occupied by 1.0 g ofhydrogen and 9.6786 g of air are 11.2 L and 0.525L respectively. Calculate the vapor density of air concerning hydrogen.
Answer: Vapour density of air concerning hydrogen
⇒ \(=\frac{\text { average molecular mass of air }}{\text { molecular mass of } \mathrm{H}_2}\)
⇒ \(=\frac{\text { mass of } 22.4 \mathrm{~L} \text { of air at STP }}{\text { mass of } 22.4 \mathrm{~L} \text { of } \mathrm{H}_2 \text { at STP }}=\frac{\frac{0.6786}{0.525} \times 22.4}{\frac{1}{11.2} \times 22.4}=14.47\)
Question 19. At a certain temperature, the vapor density of sulfur concerning nitrogen is 9.15. Find the number of atoms present in each molecule of sulfur vapor.
Answer: V.D. sulfur vapour= vapor density of sulfur with
respect to nitrogen x vapor density of nitrogen = 9.15 X (28/2) = 120.1
The molecular mass of sulfur vapour = 2xvapour density = 256.2/32 =8
Question 20. 10 L of a gas at 0°C and 760 mm pressure weighs 13.39g. Under identical conditions of temperature and pressure, of10 L ofhydrogen is 0.8928 g. What is the molecular mass ofthe gas?
Answer: Vapour density
⇒ \(=\frac{\text { at } 0^{\circ} \mathrm{C} \text { and } 1 \mathrm{~atm} \text { pressure mass of } 10 \mathrm{~L} \text { of a gas }}{\text { at } 0^{\circ} \text { and } 1 \text { atm pressure mass of } 10 \mathrm{~L} \mathrm{H}_2 \text { gas }}\)
⇒ \(=\frac{13.39}{0.8928}=15\)
Question 21. 5 g of metal on ignition in air forms 9.44 g of its oxide. Calculate the equivalent mass of the metal.
Answer: Metal present in 9.44g of its oxide = 5g
Therefore Oxygen present in 9.44g oxide = (9.44- 5)g = 4.44
Therefore Equivalent mass of the metal \(=\frac{5 \times 8}{4.44}=9\)
Question 22. The equivalent mass of a metal is 20. How much of the metal will react with chlorine to give 5.9 g of metallic chloride?
Answer:
Equivalent mass of the metal = 20
20g of metal reacts with 35.5g of chlorine.
If 20g of metal reacts with 35.5g of chlorine, then the amount of metallic chloride (35.5+20)g = 55.5g
∴ 55.5g metallic chloride =20g metal
∴ 5.0g metallic chloride \(\equiv \frac{20 \times 5}{55.5} \mathrm{~g} \text { metal }\)
= 1.8018g mental
Therefore 1.8018g metal reacts with chlorine to give 5.0g metallic chloride.
Question 23. The equivalent mass of calcium in calcium oxide is 20. Find the percentage composition of calcium oxide.
Answer: Equivalent mass of calcium in calcium oxide = 20
∴ 20g calcium combines with fig oxygen in calcium oxide.
∴ Amount of calcium In calcium oxide \(=\frac{20}{(20+8)} \times 100\)
Amount of oxygen in calcium oxide
⇒ \(=\frac{8}{(20+8)} \times 100=28.57 \%\)
Question 24. An iron rod of 20 g is kept immersed for some time in an aqueous solution of CuSO4. Then the rod is taken out from the solution and weighed. The observed mass is found to be reduced to 13.84 g. On the other hand, this chemical reaction displaces 6.985 g of copper. If the equivalent mass of iron is 28, what will be the equivalent mass of copper?
Answer: DecreaseIn the mass of Fe = (20- J3.84)g =6.16g
Therefore Amountofdissolved Fe = 6.16g \(=\frac{6.16}{28} \text { gram-equivalent }\)
Amount of displaced = 6.985g.
Therefore \(\frac{6.985}{E}=\frac{6.16}{28} \text { or, } E=31.75\)
Question 25. 5.249 g of metallic carbonate on being strongly heated liberates 1309.28mL of CO2 at 27°C and 750mm pressure of Hg. Find the equivalent mass ofthe metal.
Answer: Let, at 27°C temperature and 755 mmHg pressure, a volume of 1309.28 mL C02 gas at STP be VmL, then
⇒ \(\frac{755 \times 1309.28}{(273+27)}=\frac{760 \times V}{273}\)
⇒ \(\left[\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\right]\)
∴ r, V = 1183.6 mL
At STP, mass of 22400 mL CO2 gas = 44g
At STP, 1183.6 mL CO2 gas \(=\frac{44}{22400} \times 1183.6=2.325 \mathrm{~g}\)
⇒ \(\frac{\text { mass of metallic carbonate }}{\text { mass of } \mathrm{CO}_2 \text { produced }}\)
⇒ \(=\frac{\text { equivalent mass of metallic carbonate }}{\text { equivalent mass of } \mathrm{CO}_2}\)
⇒ \(\text { or, } \frac{5.249}{2.325}=\frac{E+30}{22}[E=\text { equivalent mass of the metal }]\)
⇒ \(\text { Equivalent mass of } \mathrm{CO}_3^{2-}=\frac{60}{2}=30\)
⇒ \(\text { [Equivalent mass of } \frac{44}{2}=22 \text { ] }\)
∴ \(E=19.66 \approx 20\)
Question 26. 1.224 g of metallic oxide can be obtained from 1.872 g of the metallic hydroxide. Find the equivalent mass of metal.
Answer: \(\frac{\text { mass of the metallic hydroxide }}{\text { mass of the metallic oxide }}\)
Question 27. 1.256 g of metal contains 0.376g of oxygen. If the specific heat of the metal is 0.124 cal .°C-1 .g-1, find the formula of its oxide.
Answer: Amount of metal present = (1.256 – 0.376) g = 0.88 g
Therefore Equivalent mass of the metal \(=\frac{0.88}{0.376} \times 8=18.72\)
Approximate atomic mass ofthe mental = \(=\frac{6.4}{0.124}=51.61\)
∴ Valency of the metal \(=\frac{51.61}{18.72}=2.75 \approx 3\) [nearest wholenumber]
Therefore Formula ofthe metallic oxide is M2O3.
Question 28. 0.111 g of metallic chloride requires 0.34 g of AgNO3 for complete precipitation of chlorine. The specific heat of the metal is 0.152cal °C-1 g_1. Determine the formula of metallic chloride.
Answer: \(0.34 \mathrm{~g} \mathrm{AgNO}_3=\frac{0.34}{170}\)
[Molecular mass of AgNO3 = 170]
= 2 x 10-3 mol of AgNO3.
1 mol of AgNO3 reacts with1 mol of Cl- ions
therefore 2x 10-3mol of AgNO3 reacts with 2x 10-3mol of Cl- ions
2 X 10-3 mol of Cl- ion = 2 X 10-3 X 35.5
= 0.071g of Cl- ions
Therefore Amount of metal present= (0.111 – 0.071) g= 0.04g
Therefore Equivalent mass of the metal \(=\frac{0.04}{0.071} \times 35.5=20\)
Approximate atomic mass of the metal = \(=\frac{6.4}{0.152}=42.1\)
Valency ofthe metal in the chloride compound
∴ \(=\frac{42.1}{20}=2\)
=42.1/20=2
Question 29. Silver sulfide and cuprous sulfide are isomorphous compounds. Silver sulfide contains 12.94% of sulfur while cuprous sulfide consists of 20.14% sulfur. Determine the equivalent mass of copper.
Answer: Valency of Ag in silver sulfide (Ag2S) = 1. As cuprous sulfide and silver sulfide are isomorphous compounds, so valency of copper cuprous sulfide is 1. 20.14g sulfur combined with (100-20.14) g = 79.86g of
Equivalent mass of Cu \(=\frac{79.86}{20.14} \times 16=63.44\) [Es =16]
Question 30. A sulfate of a metal is isomorphous with ZnS04 7H20. In this salt, the percentage of the metal is 9.75. What is the atomic mass ofthe metal?
Answer: The metallic sulfate is isomorphous with ZnS04-7H20
Let, the atomic mass ofthe metal be x.
molecular mass of = x + 32 + 64 + 126 = x + 222
Metal present in MSO4-7H2O is 9.75%
∴ \(\frac{x}{x+222} \times 100=9.75\)
∴ x= 23.98
Question 31. Determine the percentage by mass of oxygen and Sb in the compound, Sb2O5.[Sb = 121.77]
Answer: Molecular mass of Sb2O5 = 2 x 121-77 + 5X 16 = 323.54
Therefore Amount of oxygen present= \(=\frac{5 \times 16}{323.54} \times 100=24.73 \%\)
Amount of Sb-present = \(=\frac{2 \times 121.77}{323.54} \times 100=75.27 \%\)
Question 32. 9.7 g dehydrated copper sulfate on heating loses 3.5 g of water. What is the percentage of water in crystallization?
Answer: Let, the formula of hydrated copper sulfate be CuSO4.xH2O
CUS04.XH2O = 63.5 + 32 + 64 + 18x = (159.5 + 18x)g
Amount of water presenting CUS04-XH2O = 18x g
Therefore Amount of water produced from (159.5 + 18x)g of hydrated copper sulphate on heating = 18x g
Therefore Amount of water produced from 9.7g of hydrated copper sulfate on heating \(=\frac{18 x \times 9.7}{159.5+18 x} \mathrm{~g}\)
As given in the question \(\frac{18 x \times 9.7}{159.5+18 x}=3.5 \text { or, } x=5\)
∴ The molecular formula of hydrated copper sulfate: CUSO2.5H2O
CUSO4.5H2O = (63.5 + 32 + 64 + 5 X 18)g =249.5
Therefore Amount of water present \(=\frac{5 \times 18}{249.5} \times 100=36.07 \%\)
Question 33. Determine the percentage by mass of chromium, sulfate radical, and water of crystallization in chrome alum [K2SO4-Cr2(SO4)3-24H2O]. (Cr = 52)
Answer: Gram-formula mass of K2SO4-Cr2(SO4)3-24H2O
=[(2 X 39 + 96) + (2 X 52 + 3 X 96) + 24 X 18]g = 998g
Therefore Amount of Cr in chrome alum \(=\frac{2 \times 52}{998} \times 100=10.42 \%\)
Amount of sulphate radicals \(=\frac{4 \times \text { gram-formula mass of } \mathrm{SO}_4^{2-}}{998} \times 100=38.48 \%\)
Therefore Amount of H2O \(\frac{24 \times 18}{998} \times 100=43.29 \%\)
Question 34. Percentage composition of a salt is: K = 8.23%, A1 = 5.70%, S04 = 40.51% and H2O = 45.5%. What is the empirical formula of the compound? The molecular mass of the compound is 948. What is its molecular formula?
Answer: In the compound ratio of, Al, \(\mathrm{SO}_4^{2-}\) radicals and H2O
⇒ \(\mathrm{K}: \mathrm{Al}: \mathrm{SO}_4: \mathrm{H}_2 \mathrm{O}=\frac{8.23}{39}: \frac{5.7}{27}: \frac{40.51}{96}: \frac{45.5}{18}\)
= 0.211 : 0.211 : 0.422: 2.527 =1: 1 : 2 : 12
∴ Empirical formula ofthe compound
[KA1(SO24)2(H2O)12) and molecular formula
[KA1(SO4)2(H2O)12]X
therefore Molecular mass = [39 + 27 + 3 x 96 + 12 x 18] x x =570 x x
As given the question, 570 x x = 948
∴ \(x\approx 2\)
∴ Molecular formula ofthe compound:
⇒ \(\mathrm{K}_2 \mathrm{SO}_4 \cdot \mathrm{Al}_2\left(\mathrm{SO}_4\right)_3 \cdot 24 \mathrm{H}_2 \mathrm{O}\)
Question 35. An organic compound consists of C, H, and N. 0.5 g of the compound, on combustion, forms 1.44 g of C02 and 0.3785 g of water. Find the molecular formula of the compound that fits the vapor density is 5.35.
Answer: Molecular mass of the compound = 2 x vapour density of the compound = 2 x 53.5 = 107
As given in the question, 0.5g of the compound, on combustion, forms 1.44g of CO2 and 0.3785g of water.
Therefore CO2 produced on combustion \(=\frac{1.44}{0.5} \times 107=308.16 \mathrm{~g}\)
Therefore H2O produced on combustion \(=\frac{0.3785}{0.5} \times 107=81 \mathrm{~g}\)
No. of moles of C-atoms in 308.16g of \(\mathrm{CO}_2=\frac{1 \times 308.16}{44}=7\)
No. of moles of H-atoms present in 81g of \(\mathrm{H}_2 \mathrm{O}=\frac{2 \times 81}{18}=9\)
Therefore Number of C and H-atoms present in each molecule of the compound are 7 and 9 respectively.
Let, the number of N-atoms in each molecule of the compound be JC, and then the molecular mass
= 7 X 12 + 9 X 1 + JC X 14 = 93 + 14x
Therefore 93 + I4x = 107 or, x =1
Therefore Molecular formula ofthe compound: is C7HgN
Question 36. The percentage composition of a mineral is CaO = 48%, P2O5 = 41.3%, and CaCl2 = 10.7%. Determine the formula of that mineral.
Answer: The ratio of the number of CaO, P2Os, and CaCl2 molecules present in a molecule ofthe mineral is \(\mathrm{CaO}: \mathrm{P}_2 \mathrm{O}_5: \mathrm{CaCl}_2\)
⇒ \(=\frac{48}{40+16}: \frac{41.3}{(2 \times 31+5 \times 16)}: \frac{10.7}{(40+2 \times 35.5)}\)
= 0.8571 : 0.2908 : 0.0964 = 8.9 : 3.0 : 1 =9:3:1
therefore Formula of the mineral: is 9Ca0-3P2O5-CaCl2
Question 37. An organic compound, consisting of carbon, hydrogen, nitrogen, and oxygen contains 40.67% of carbon and 8.47% of hydrogen. 0.5 g of that compound gives 94.91 mL of nitrogen at STP. What is the empirical formula of that compound?
Answer: Mass of 94.91 mLnitrogen at STP
Question 38. What amount of magnesium sulfide (MgS) will be produced from 1 g of Mg and 1 g of S?
Answer:
⇒ \(\begin{array}{lcc}
\mathrm{Mg} & +\mathrm{S} & \mathrm{SgS} \\
24 \mathrm{~g} & 32 \mathrm{~g} & 56 \mathrm{~g} \\
0.75 \mathrm{~g} & 1 \mathrm{~g} & 1.75 \mathrm{~g}
\end{array}\)
As given in the question, the Mass of both Mg and S taking part in the reaction is 1 g. So, in this case limiting reagent is S because lg of S reacts completely with 0.75g of Mg. So according to the above equation, Ig of S and 0.75g ofMg react to produce 1.75g ofMgS
Question 39. How much Na2SO4 will be required for complete precipitation of 7.336 g BaCl2 dissolved in water? What will be the mass of BaSO4 precipitated? [Ba = 137.36]
Answer:
⇒ \(\begin{array}{ccc}
\mathrm{BaCl}_2+ & \mathrm{Na}_2 \mathrm{SO}_4 \rightarrow \mathrm{BaSO}_4+2 \mathrm{NaCl} \\
(137.36+2 \times 35.5) & (2 \times 23+32+64) \mathrm{g} & (137.36+32+64) \\
=208.36 \mathrm{~g} & =142 \mathrm{~g} & =233.36 \mathrm{~g}
\end{array}\)
Now, 208.36g BaCI2 3 142gNa2S04 = 233.36gBaSO4
⇒ 7.336gBaCl2 = 5gNa2SO4 = 8.216g BaSO4
Therefore 5g of Na2SO4 will be required for complete precipitation of 7.336g BaCl2 and the mass of BaSO4 precipitate is 8.216g.
Question 40. What amount of CaC03 will be sufficient to produce 1 L of C02 at 27° C and 760 mmHg pressure? Determine the quantity of pure carbon required to yield the same amount of C02.
Answer: Let, at 27°C and 760 mm Hg pressure volume of1L C02 gas at STP be VL, then
⇒ \(\frac{V \times 760}{273}=\frac{1 \times 760}{(273+27)}\left[\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\right]\)
therefore \(V=\frac{273}{300}=0.91 \mathrm{~L}\)
Mass of 0.91L of CO2 gas at STP \(=\frac{44}{22.4} \times 0.91=1.7875 \mathrm{~g}\)
∴ \(\underset{100 \mathrm{~g}}{\mathrm{CaCO}_3(s) \rightarrow \mathrm{CaO}(s)}+\underset{44 \mathrm{~g}}{\mathrm{CO}_2(\mathrm{~g})}\)
⇒ \(\underset{100 \mathrm{~g}}{\mathrm{CaCO}_3(s) \rightarrow \mathrm{CaO}(s)}+\underset{44 \mathrm{~g}}{\mathrm{CO}_2}(\mathrm{~g})\)
therefore 44g C02= 100g CaCO3 or, 1.7875g CO2 = 4.0625g CaCOg
Therefore At 27°C and 760 mm Hg pressure 4.0625g CaCOg is required to produce 1L C02 gas at STP.
⇒ \(\underset{12 \mathrm{~g}}{\mathrm{C}(s)}+\mathrm{O}_2(\mathrm{~g}) \rightarrow \underset{44 \mathrm{~g}}{\mathrm{CO}_2(g}\)
∴ 44g CO2 = 12g C or, 1.7875 g CO2 = 0.4875g C
Question 41. A HN03 solution (specific gravity = 1.46) contains 75% HN03. How much of this acid will be needed to dissolve 5g of CuO?
Answer: Mass of100mL HNOg solution = 100 x 1.46 = 146g
⇒ \(\begin{aligned}
& \mathrm{CuO}(s)+2 \mathrm{HNO}_3(a q) \rightarrow \mathrm{Cu}\left(\mathrm{NO}_3\right)_2(a q)+\mathrm{H}_2 \mathrm{O}(l) \\
& (63.5+16) \mathrm{g} \quad 2 \times 63 \mathrm{~g} \\
& =79.5 \mathrm{~g} \quad=123 \mathrm{~g}
\end{aligned}\)
79.5g CuO = 126g HNO3
Therefore 5g CuO = 7.92g HNOg
75g HNOg is present in 146g solution.
Therefore 7.92g HNO3 is present in \(\frac{146 \times 7.92}{75}=15.417 \mathrm{~g}\) solution.
Therefore 15.417g acid will be required to dissolve 5g of cupric oxide.
Question 42. Calculate the amount of Cl2 produced by the reaction between 1 g of each of MnO2 and HC1.
Answer: \(\begin{aligned}
& \mathrm{MnO}_2(s)+4 \mathrm{HCl}(a q) \rightarrow \mathrm{MnCl}_2(a q)+\mathrm{Cl}_2(g)+\mathrm{H}_2 \mathrm{O}(l) \\
& (55+2 \times 16) \quad 4 \times 36.5 \quad 71 \mathrm{~g} \\
&
\end{aligned}\)
Therefore 0.4863g of Cl2 is produced from lg of each of MnO2 and HC1
Question 43. When a mixture of NaCl and NaBr is heated with H2SO4, the halogen components are liberated as their hydracids leaving Na2S04 as the residue. In an experiment, the mass of Na2SO4 left is equal to the mass of(NaCl + NaBr) taken at the beginning of the experiment. Calculate the percentages of NaCl and NaBrin in the given mixture.
Answer: Let, the masses of NaCl and NaBr in the mixture are xg and y g respectively.
⇒ \(x \mathrm{~g} \mathrm{NaBr}=\frac{x}{23+80}=\frac{x}{103} \mathrm{~mol} ;\)
⇒ \(y \mathrm{gNaCl}=\frac{y}{23+35.5}=\frac{y}{58.5} \mathrm{~mol}\)
2NaBr(aq) + H2SO4(a<7) Na2SO4(ag) + 2HBr(aq)
Therefore 2 mol NaBr= lol Na2SO4
Therefore \(\frac{x}{103} \mathrm{~mol} \mathrm{NaBr}=\frac{x}{206} \mathrm{~mol} \mathrm{Na}_2 \mathrm{SO}_4\)
Therefore 2mol NaCls lmol Na2SO4
Therefore \(\frac{y}{58.5} \mathrm{~mol} \mathrm{NaCl} \equiv \frac{y}{117.0} \mathrm{~mol} \mathrm{Na}_2 \mathrm{SO}_4\)
Therefore Amount of Na2SO4 produced
∴ \(=\left(\frac{x}{206}+\frac{y}{117}\right) \times \text { gram-formula mass of } \mathrm{Na}_2 \mathrm{SO}_4\)
∴ \(=\left(\frac{x}{206}+\frac{y}{117}\right) \times 142 \mathrm{~g}\)
As given, x+ y = \(\left(\frac{x}{206}+\frac{y}{117}\right) 142=0.6893 x+1.2136 y\)
or, 0.3107x = 0.2136y
Therefore y= 1.455x
Therefore Amount of NaBr \(\begin{aligned}
=\frac{x}{x+y} \times 100 & =\frac{x}{x+1.455 \mathrm{x}} \times 100 \\
& =40.73 \%
\end{aligned}\)
Therefore Amount of Nacl = \(=\frac{y}{x+y} \times 100\)
∴ \(=\frac{1.455 x}{x+1.455 x} \times 100=59.27 \%\)
Question 44. H2 gas liberated in the reaction of 13 g of zinc with dilute H2S04 is separately passed over 1. 10 g and 2. 20g of dry cupric oxide. What is the amount of residue remaining in each case?
Answer:
⇒ \(\begin{array}{cc}
\mathrm{Zn}(s) & +\mathrm{H}_2 \mathrm{SO}_4(a q) \rightarrow \mathrm{ZnSO}_4(a q)+\mathrm{H}_2(g) \\
65 \mathrm{~g} & 2 \mathrm{~g} \\
13 \mathrm{~g} & 0.4 \mathrm{~g}
\end{array}\)
In this case, 10 g CuO is the limiting reagent, so it reacts
completely with 0.25 g of H2 gas and produces 7.99g of Cu.
⇒ \(\begin{aligned}
20 \mathrm{gCuO} \equiv \frac{2}{79.5} \times 20=0.503 \mathrm{gH}_2 & \equiv \frac{63.5}{79.5} \times 20 \\
& =15.97 \mathrm{gCu}
\end{aligned}\)
In this case, H2 is limiting reagent, so 0.4g H2 reacts completely with CuO and produce ,\(\frac{15.97 \times 0.4}{0.503}=12.7 \mathrm{~g}\) of Cu.
therefore Amount of remaining CUO = \(20-\frac{20 \times 0.4}{0.503}=4.09 \mathrm{~g}\)
Question 45. 1 g of a mixture containing Na2CO3 and NaHCO3 in equal amounts is heated until a constant mass is obtained. Find the volume of C02 gas liberated at STP.
Answer: Amount of NaHCOg in the mixture = 0.5g
On heating NaHCOg dissociates to give Na2CO3, CO2, and H2O, and Na2CO3 remains unchanged.
⇒ \(\begin{gathered}
2 \mathrm{NaHCO}_3(s) \rightarrow \mathrm{Na}_2 \mathrm{CO}_3(s)+\mathrm{CO}_2(\mathrm{~g})+\mathrm{H}_2 \mathrm{O} \\
2 \times 84 \mathrm{~g} \\
22400 \mathrm{~mL}[\mathrm{STP}]
\end{gathered}\)
therefore \(168gNaHCOg= 22400mL C02(STP)\)
Therefore 0.5g NaHCOg = 66.67mL; So, CO2 liberated at STP = 66.67 mL
Question 46. 2 g of a mixture of CaCO3 and MgCO3 being treated with dilute HC1 gives 536.84 mL of CO2 at 27° C and 750 mm of Hg pressure. What is the composition of the mixture?
Answer: Let, at 27°C and 750 mm Hg pressure volume of 536.84 mL CO2 gas at STP be VmL.
⇒ \(\frac{750 \times 536.84}{(273+27)}=\frac{760 \times V}{273}\left[\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\right]\)
Therefore V = 482.10 mL
Let, CaCO3 in the mixture :xg; Amount of MgCO2 = (2-x)g
⇒ \(\begin{array}{cc}
\mathrm{CaCO}_3(s)+2 \mathrm{HCl}(a q) \rightarrow \mathrm{CaCl}_2(a q)+\mathrm{CO}_2(g)+\mathrm{H}_2 \mathrm{O}(l) \\
100 \mathrm{~g} & 22400 \mathrm{~mL}(\mathrm{STP}) \\
x \mathrm{~g} & 224 \times x \mathrm{~mL}(\mathrm{STP})
\end{array}\)
⇒ \(\begin{array}{cc}
\mathrm{MgCO}_3(s)+2 \mathrm{HCl}(a q) \rightarrow \mathrm{MgCl}_2(a q)+\mathrm{CO}_2(g)+\mathrm{H}_2 \mathrm{O}(l) \\
84 \mathrm{~g} & 22400 \mathrm{~mL}(\mathrm{STP}) \\
(2-x) \mathrm{g} & \frac{22400 \times(2-x)}{84} \\
& =266.67(2-x) \mathrm{mL}(\mathrm{STP})
\end{array}\)
As given the question,
224 X x + 266.67(2 —x) = 482.10
or, 42.67 x x = 51.24
therefore x = 1.2
therefore CaCO3 \(=\frac{1.2}{2} \times 100=60 \% \text {; }\)
MgCO3 \(=\frac{2-1.2}{2} \times 100=40 \%\)
Question 47. What will be the amounts of NH3 (excess) and Cl required to produce 1L N2 gas at 27 °C and 750mm pressure?
Answer: Let us suppose, at 27°C and 755mm Hg pressure volume of N, gas at STP be VmL.
⇒ \(\frac{755 \times 1}{(273+27)}=\frac{760 \times V}{273}\left[\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\right] \quad V=0.904 \mathrm{~L}\)
[Mass of 0.904L of, gas at STP \(=\frac{28}{22.4} \times 0.904=1.13 \mathrm{~g}\)]
⇒ \(\begin{array}{ccc}
8 \mathrm{NH}_3(\mathrm{~g}) & +3 \mathrm{Cl}_2(\mathrm{~g}) \rightarrow & \mathrm{N}_2(\mathrm{~g}) \\
8 \times 17 \mathrm{~g} & 3 \times 71 \mathrm{~g} & 28 \mathrm{~g} \\
\frac{8 \times 17}{28} \mathrm{~g} & \frac{3 \times 71}{28} \mathrm{~g} & 1 \mathrm{~g} \\
\frac{8 \times 17}{28} \times 1.13 \mathrm{~g} & \frac{3 \times 71 \times 1.13}{28} \mathrm{~g} & 1.13 \mathrm{~g}
\end{array}\)
Therefore Amounts of NH3 and Cl, gas are 5.488g and 8.596 g respectively.
Question 48. Calculate the volume of 02 gas required for the complete burning of 10 L of acetylene gas. Also, calculate the volume of C02 produced. All volumes are measured at the same temperature and pressure.
Answer:
The volume of O, gas required for complete burning of 10L of acetylene gas= 25
therefore The volume of CO, gas produced for the complete burning of 10 of acetylene gas = 20LThe volume of O, gas required for the complete burning of 10L of acetylene gas= 25
Therefore The volume of CO, gas produced for complete burning
of10L of acetylene gas = 20L
Question 49. A gas mixture having 1200mL volume at 27°C and 1 atm pressure consists of 80% methane & 20% CO. What amount of KC1O3 on thermal decomposition produces the same quantity of oxygen as needed for complete combustion of that mixture?
Answer: Let, the volume ofthe gas mixture at STP be V mL.
⇒ \(\frac{1 \times V}{273}=\frac{1 \times 1200}{300}\left[\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\right]\)
Therefore Volume of CH4 in the mixture \(=1092 \times \frac{80}{100}=873.6 \mathrm{~mL}\)
and volume of CH4 in the human = \(=1092 \times \frac{20}{100}=218.4 \mathrm{~mL}\)
⇒ \(\begin{array}{rl}
\mathrm{CH}_4(\mathrm{~g}) & +2 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(\mathrm{g}) \\
1 \mathrm{~mL} & 2 \mathrm{~mL} \\
873.6 \mathrm{~mL} & 2 \times 873.6 \mathrm{~mL}=1747.2 \mathrm{~mL}
\end{array}\)
⇒ \(\begin{array}{rl}
\mathrm{CO}(\mathrm{g}) & +\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) \\
1 \mathrm{~mL} & 0.5 \mathrm{~mL} \\
218.4 \mathrm{~mL} & 218.4 \times 0.5=109.2 \mathrm{~mL}
\end{array}\)
therefore At STP, a volume of 02 is required for the complete combustion of gas mixture = (1747.2 + 109.2)mL = 1856.4mL
Therefore At STP, 3 x 22400mLO, = 2mol KC1O3 = 245gKC1O3
∴ \(1856.4 \mathrm{~mL} \mathrm{O} \mathrm{O}_2 \equiv \frac{245}{3 \times 22400} \times 1856.4 \equiv 6.766 \mathrm{~g}\)
Therefore 6.768g of KC1O3 is required for complete combustion.
Question 50. The volume ofthe gas mixture obtained bypassing 400 mL C02 over red hot coke was measured to be 600 mL. Find the composition of the gas mixture.
Answer: Reaction: C(s)4- CO,(g)→2CO(g). Let us consider.
400mL CO, gas reduction produces x mL CO gas.
According to the above equation, xmL CO, = 2xmLCO Volume of residual CO, in the mixture= (400—x) mL Total volume ofthe mixture.
= (400- x+ 2x)mL= (400+ x)mL
As given the question, 400 + x = 600
∴ x= 200
200 mL of CO, and 2 x 200mL = 400mL of CO are present
Question 51. 100 mL mixture of CO, CH4, and C2H4 was ignited with 300 mL O2 gas using an electric spark. On cooling the volume of the mixture becomes 285 mL. Adding KOH to the mixture 205 mL O2 remains in the mixture. Find out the volume of CO, CH4 and H2 in the gas mixture, [CO: 50 mL, CH4: 30 mL H2: 20]
Answer: Let, the volume of CO and CH4 in the mixture be x and y respectively.
Volume ofH, in the mixture = [100 — (x — y)]mL
⇒ \(\begin{array}{c|c}
\mathrm{CO}+\frac{1}{2} \mathrm{O}_2 \rightarrow \mathrm{CO}_2 & \mathrm{CH}_4+2 \mathrm{O}_2 \rightarrow \mathrm{CO}_2 \div 2 \mathrm{H}_2 \mathrm{O} \\
x \mathrm{~mL} 1 / 2 x \mathrm{~mL} x \mathrm{~mL} & y \mathrm{~mL} 2 y \mathrm{~mL} \quad y \mathrm{~mL}
\end{array}\)
On cooling volume of the mixture is 285 mL and on adding KOH to the mixture 205mL O, remains in the mixture. The first contraction in the mixture Volume of the gas mixture- Volume ofthe gas mixture after ignition.
⇒ \(\begin{aligned}
=x+\frac{x}{2}+y+2 y+100 & -(x+y) \\
& +\frac{1}{2}[100-(x+y)] \mathrm{mL}-(x+y) \mathrm{mL}
\end{aligned}\)
⇒ \(=\left[\frac{x}{2}+2 y-\frac{x}{2}-\frac{y}{2}+150-x-y\right] \mathrm{mL}=\left(\frac{y}{2}-x+150\right) \mathrm{mL}\)
⇒ \(\text { As given, }\left(\frac{y}{2}-x+150\right) \mathrm{mL}=[(100+300)-285] \mathrm{mL}\)
⇒ \(\mathrm{O}_2 \text { used }=\frac{x}{2}+2 y+\frac{1}{2}[100-(x+y)]=\left(\frac{3}{2} y+50\right) \mathrm{mL}\)
⇒ \(\text { As given, } \frac{3}{2}+50=300-205=95 \text { or, } \frac{3}{2} y=45\)
Therefore y =30
Putting y = 30 in equation [1] 2x= 100
Therefore x=50
Volume of CO, CH4 and H2 are 50mL, 30mL and [100- (50 + 30)]mL = 20 mL respectively
Question 52. 10 mL of a gaseous hydrocarbon is completely combusted with 60 mL of oxygen by passing an electric spark. The volume of the gaseous mixture thus yielded on cooling becomes 46mL. If the vapor density of the compound is 15, what will be its molecular formula? [All volumes were measured under the same conditions of temperature and pressure.] explosion
Answer: Let, the molecular formula ofthe hydrocarbon: CxHy
- Molecular mass -2 x 15 = 30
- Therefore 12x + y =30
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y+\left(x+\frac{y}{4}\right) \mathrm{O}_2 \rightarrow x \mathrm{CO}_2+\frac{y_2}{2} \mathrm{H}_2 \mathrm{O} \\
& 10 \mathrm{~mL} \quad 10\left(x+\frac{y}{4}\right) \mathrm{mL} \quad 10 x \mathrm{~mL} \\
&
\end{aligned}\)
Ignoring the volume of water, construction in the volume of the mixture \(=\left[10+10\left(x+\frac{y}{4}\right)-10 x\right] \mathrm{mL}\) ml. As given in the question
or.\(10+10\left(x+\frac{y}{4}\right)-10 x=[(60+10)-45] \mathrm{mL}\)
therefore \(10+\frac{10}{4} y=25\)
Therefore y=6; y=6 Again, 12x=y =30 or, 12x +6 =30
Therefore x=2
∴ The formula of the hydrocarbon: is C2H6.
Question 53. 2 volume CJCHyNz + 7volume02 4 volume C02+ 6 volume water vapor + 2volume N2 Determine the values of x, y, and z. [All volumes were measured at the same temperature and pressure.]
Answer:
⇒ \(\begin{aligned}
& \mathrm{C}_x \mathrm{H}_y \mathrm{~N}_z+\mathrm{O}_2 \rightarrow \mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}+\mathrm{N}_2 \\
& \begin{array}{ccccc}
\text { 2volume } & 7 \text { volume } & 4 \text { volume } & 6 \text { volume } & 1 \text { volume } \\
2 \text { molecules } & 7 \text { molecules } & 4 \text { molecules } & 6 \text { molecules } & 7 \text { molecules }
\end{array} \\
&
\end{aligned}\)
Therefore 2 molecules of C2 = 4 molecules of CO2 = 6 molecules of H2O = 2 molecules of N2
Number of C-atoms in 2 molecules of compound = Number ofC-atoms in 4 molecules of C02
Therefore 2x =4 or, x =2
Number of H -atoms in 2 molecules of CxHyNz compound = Number ofH-atoms in 6 molecules of H2O 2y = 6x2ory = 6
In the same way, in the case of N, 2z – 2 x 2 or z= 2
Therefore 2y = 6 and z = 2
Question 54. Calculate the strength in—percentage (W/V), moles per litre or molarity, number of gram equivalents of the solute, and normality ofthe following solutions —
- 9.125 g of HC1in 200 mL of aqueous solution
- 60 g of NaOH in 3 L of its aqueous solution
- 14.7 g H2SO4 in 500mL of its aqueous solution
- 296 g Ca(OH)2 in 10 L of its aqueous solution
Answer: Strength (IV/ V) of this solution
⇒ \(=\frac{9.125}{200} \times 100=4.56 \%\)
Molar strength \(=\frac{9.125}{36.5} \times \frac{1000}{200}=1.25(\mathrm{M})\)
Gram-equivalent of the solute \(=\frac{9.125}{36.5}=0.25\)
[since Equivalent mass of HC1 = gram-molecular mass]
Normal strength \(=\frac{9.125}{36.5} \times \frac{1000}{200}=1.25(\mathrm{~N})\)
Strength(W/ V)of this solution \(=\frac{60}{3000} \times 100=2 \%\)
Molar strength \(=\frac{60}{40} \times \frac{1}{3}=0.5(\mathrm{M}) \text {; }\)
Gram-equivalent \(=\frac{60}{40}=1.5\)
[ since Equivalent mass of HCl = Gram-molecular mass]
Normal strength \(=\frac{60}{40} \times \frac{1}{3}=0.5(\mathrm{~N})\)
⇒ \(14.7 \mathrm{gH}_2 \mathrm{SO}_4=\frac{14.7}{98}=0.15 \mathrm{molH}_2 \mathrm{SO}_4=0.3 \mathrm{~g} \text {-eqv. }\)
[MH2S04 = 98 and EH2SO4 = 49]
Strength \(\left(\frac{W}{V}\right)\) of the solution \(=\frac{14.7}{500} \times 100=2.94 \%\) =2.94%
Molar strength \(=\frac{0.15}{500} \times 1000=0.3(\mathrm{M})\)
Number of gram-equivalent of the solute = 0.3
Normal strength \(=\frac{0.3}{500} \times 1000=0.6(\mathrm{~N})\)
Strength \(\left(\frac{W}{V}\right)\) of this solution \(=\frac{296}{10 \times 10^3} \times 100=2.96 \%\)
Molar strength \(\frac{296}{74} \times \frac{1}{10}=0.4(\mathrm{M})\); Gram-equivalent ,\(=\frac{296}{37}\)
Normality strength \(=\frac{8}{10}=0.8(\mathrm{~N})\)
Question 55. Calculate the molality of a solution prepared by equal volumes of a 40% H2SO4 solution (W/V) (density 1.5 g-cm-3) and 60% H2SO4 solution ( W/V) (density 1.8 gems “3).
Answer: Amount of H2SO4 in 40% H2S04 solution = 40g
Mass of 100 mL solution = 100 x 1.5 = 150g
Amount of H2SO4 in 60% \(\left(\frac{W}{V}\right)\) H2SO4 solution = 60g
Mass of 100mL solution =100×1.8=180
Total mass of the solution mixture =(150 + 180)g =330g
The total mass of H2SO4 in the solution mixture
= (40 + 60)g
= 100 g and mass ofwater = (330- 100)g = 230g
∴ Number of moles of H2SO4 in the solution mixture = \(=\frac{100}{98} \mathrm{~mol}\)
Molality ofthe solution mixture = X \(=\left(\frac{100}{98}\right) \times\left(\frac{1000}{230}\right)\)
= 4.436(m)
Question 56. An impure sample of (NH4)2S04 is supplied to a laboratory. 3.6 g of the supplied (NH4)2S04 salt on heating with NaOH forms NH3 gas. 100mL of 0.5(N)H2SO4 is required for the complete neutralisation of the libarated NH3 gas. Calculate the percentage purity of the impure sample. Let us assume, that the impurity of the sample is insoluble in NaOH.
Answer:
⇒ \(\mathrm{H}_2 \mathrm{SO}_4(a q)+2 \mathrm{NH}_3(a q) \rightarrow\left(\mathrm{NH}_4\right)_2 \mathrm{SO}_4(a q)\)
According to this equation— 1000 mL 1 (N) H2SO4= 17gNH3
100 mL 0.5 (N)H2SO4 \(\equiv \frac{17 \times 100 \times 0.5}{1000}=0.85 \mathrm{~g} \mathrm{NH}_3\)
34gNH3=132g(NH4)2S04
0.85gNH3 = X 0.85 EE 3.3g(NH4)2SO
⇒ \(0.85 \mathrm{gNH}_3 \equiv \frac{132}{34} \times 0.85 \equiv 3.3 \mathrm{~g}\left(\mathrm{NH}_4\right)_2 \mathrm{SO}_4\)
Question 57. A sample of 3.95g of 60% pure chalk (impurities in the chalk are insoluble in HC1), is dissolved in 250 mL of 0.2(M) HC1. What volume (cm3) of 0.01 (N) NaOH is required to neutralize the excess acid?
Answer:
Amount Of CaCO3 \(=\frac{60 \times 3.95}{100}=2.37 \mathrm{~g}\)
⇒ \(2.37 \mathrm{~g} \mathrm{CaCO}_3 \equiv \frac{2.37}{100} \equiv 2.37 \times 10^{-2} \mathrm{~mol} \mathrm{CaCO}_3\)
⇒ \(\mathrm{CaCO}_3+2 \mathrm{HCl} \rightarrow \mathrm{CaCl}_2+\mathrm{H}_2 \mathrm{O}+\mathrm{CO}_2\)
⇒ \(1000 \mathrm{~mL} 2(\mathrm{M}) \mathrm{HCl} \equiv 1 \mathrm{~mol} \mathrm{CaCO}_3\)
2.37 X 10_2mol CaC03 = (1000 X 2.37 X 10_2)mL 2(M)HC(1)
= 23.7mL 2(m)HCl = 237mL 0.2(M)HC1
Amount of HC1 remained = (250- 237)mL 0.2(M)HC1 =13mL0.2(M)HCl
If Vml of 0.01 (N) or 0.01 (M) NaOH solution is required to neutralize the excess acid, then
⇒ \(13 \times 0.2=V \times 0.01\left[\mathrm{~V}_1 S_1=V_2 S_2\right]\)
∴ V=200 ML
Question 58. 500 mL of a mixed solution of KOH and Na2C03 was first titrated using phenolphthalein as an indicator. 20 mL of 0.2(M) HC1 was required to reach the endpoint. Methyl orange indicator was then added and a further 10 mL of the same HC1 was required to reach the next endpoint. Find out the composition of the given mixture.
Answer:
Let, the amount of KOH and Na2CO3 in the mixture be x and y mol. Reaction occurred in the first titration,
⇒ \(\begin{aligned}
& \mathrm{KOH}+\mathrm{HCl} \rightarrow \mathrm{KCl}+\mathrm{H}_2 \mathrm{O} \\
& \mathrm{Na}_2 \mathrm{CO}_3+\mathrm{HCl} \rightarrow \mathrm{NaHCO}_3+\mathrm{NaCl}
\end{aligned}\)
HC1,in 20mL 0.2 (M) solution= \(=\frac{0.2}{1000} \times 20\) 10-3mol.
According to equation [1] 1 mol KOH Jmol HCl
∴ x mol KOH=x mol HC1
According to equation [2] 1 mol Na2CO3= lol HC1
∴ y mol Na2C03 = ymolHCl
∴ x + y = 4 X 10-3 (HC1 required in first titration)
Reaction occurred in the second titration,
\(\mathrm{NaHCO}_3+\mathrm{HCl} \rightarrow \mathrm{NaCl}+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}\)10 mL 0.2 (M) HC1 is required in this reaction. Amount of HC1in 10 mL 0.2 (M) HC1 = X 10 = 2 X 10-3mol
According to the equation [2], y mol of NaHCO3 will be produced from a symbol of Na2CO3
According to the equation [4], y mol NaHCO3 symbol HC1
∴ 2 X 10-3 mol HC1= 2 X 10-3 mol NaHCO3
∴ y = 2 x 10-3mol; Putting y in equation
x = 2 x 10-3mol
∴ Amount of KOHin the mixture = 2 x 10-3mol
= 2 X 10-3 x 56g = 0.112g and amount of
Na2C03 = 2 x 1-3mol = 2 x 10~3 x 106g = 0.212g
Question 59. 50 mLofa solution of NaHCO3 and Na2CO3 requires 10 mL 0.2(N)H2SO4 for neutralisation purpose in presence of phenolphthalein indicator. 50 mL of the same 0.2(N)H2SO4 can neutralize the supplied 50 mL NaHCO3 andNa2CO3 solution completely in the presence of methyl orange as an indicator. Find out the strength of the mixture gL-1.
Answer:
Let, the amount of NaHCO3 and Na2CO3 in 50 mL solution be x and ymol. Reactioninpresence of phenolphthalein indicator,
⇒ \(2 \mathrm{Na}_2 \mathrm{CO}_3+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow 2 \mathrm{NaHCO}_3+\mathrm{Na}_2 \mathrm{SO}_4\)
10 mL 0.2 (N) H2SO4= 10mL0.1(M)H2SO4
H2S04 in 10 mL 0.1 (M) solution
\(=\frac{0.1}{1000} \times 10=10^{-3} \mathrm{~mol}\)From equation [1]
⇒ \(10^{-3} \mathrm{~mol} \mathrm{H}_2 \mathrm{SO}_4 \equiv 2 \times 10^{-3} \mathrm{~mol} \mathrm{Na}_2 \mathrm{CO}_3\)
From equation [1],
⇒ \(10^{-3} \mathrm{~mol} \mathrm{H}_2 \mathrm{SO}_4 \equiv 2 \times 10^{-3} \mathrm{~mol} \mathrm{Na}_2 \mathrm{CO}_3\)
∴ y = 2 x 10-3mol
Reactions in the presence of methyl orange indicator,
⇒\(\begin{aligned}
& 2 \mathrm{NaHCO}_3+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{Na}_2 \mathrm{SO}_4+2 \mathrm{CO}_2+2 \mathrm{H}_2 \mathrm{O} \\
& \mathrm{Na}_2 \mathrm{CO}_3+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{Na}_2 \mathrm{SO}_4+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O} \\
& \mathrm{H}_2 \mathrm{SO}_4 \text { in } 50 \mathrm{~mL} 0.1(\mathrm{M}) \text { solution } \\
& =\frac{0.1 \times 50}{1000}=5 \times 10^{-3} \mathrm{~mol}
\end{aligned}\)
Amount of Na2CO3 In The Solution Forming By the Combination combination of NaHCO2 and Na2CO3 = 2x 10-3mol
According to equation [3], 2 x 10-3 mol of H2SO4 is required to neutralise 2 x 10-3mol of Na2CO3
Remaining (5 x 10-3- 2 x 10-3) = 3 x 1(T3 mol of H2SO4 is required to neutralise NaHCO3.
According to equation [2], 3 x 10-3molof H2SO4 is required to neutralise 2 x 3 x 10-3 = 6 x 10-3 mol NaHC03.
x = 6 x 10-3mol
5.8 X 1000
In 50 mL solution, amounts of NaHCO3 and Na2CO3 are (6 X 10~3 X 84)g = 0.504g And (2 x 10-3 x 106)g = 0.212g respectively.
Concentration of NaHC03 in g/L unit = 10.08 g/L and concentration of Na2C03 in g/Lunit = 4.24 g/L.
Question 60. 1L each of three samples of H2O2 labeled as 10 volume, 20 volume, and 30 volume are mixed and then diluted to 5L using water. Find out the relative strength of the resultant solution.
Answer:
Given:
1L each of three samples of H2O2 labeled as 10 volume, 20 volume, and 30 volume are mixed and then diluted to 5L using water.
At STP, lOmL 02 = 1 mL H2O2 solution
22400 mL O2 = 2240mL H2O2 solution
∴ \(\begin{gathered}
2 \mathrm{H}_2 \mathrm{O}_2 \rightarrow 2 \mathrm{H}_2 \mathrm{O}+\mathrm{O}_2 \\
68 \mathrm{~g} \quad 22400 \mathrm{~mL} \text { (at STP) }
\end{gathered}\)
∴ Amount of H2O2 in 2240 mL H2O2 solution = 68 g
∴ H202 in 1000 mL H2O2 solution \(=\frac{68 \times 1000}{2240}=30.36 \mathrm{~g}\)
In the same way, amount of H2O2 in 1000 mL 20 volume solution \(=\frac{68 \times 1000}{1120}=60.72 \mathrm{~g}\) and amount of H2O2 in 1000 mL 30 volume solution \(=\frac{68 \times 1000}{746.67}=91.07 \mathrm{~g}\)
Total amount of H2O2 in 5 L or 5000 mL solution mixture.
= (30.36 + 60.72 + 91.07)g = 182.15g
182.15g H2O2 = 5000mLH202 solution
⇒ \(68 \mathrm{gH}_2 \mathrm{O}_2 \equiv \frac{5000 \times 68}{182.15} \equiv 1866.6 \mathrm{mLH}_2 \mathrm{O}_2 \text { solution }\)
At STP, 22400mL of O2 is produced from 68g H20 68gof H2O2 is present in 18.66mL O2 (at STP)
∴ lmL H2O2 solution \(\equiv \frac{22400}{18.66} \equiv 12 \mathrm{~mL} \mathrm{O}_2(\text { at STP })_{\mathrm{s}}\)
∴ strength of the resultant solution = 12 volume
Question 61. The mass of an empty LPG cylinder is 14.8 kg and when it is filled with n -n-butan gas, the mass is 29.0 kg and the internal pressure is 2.5 atm. After a few days mass of the filled gas cylinder decreased to 23.2 kg. Calculate the volume of gas used in m3 (at 27 °C &1 atm).
Answer:
Given:
The mass of an empty LPG cylinder is 14.8 kg and when it is filled with n -n-butan gas, the mass is 29.0 kg and the internal pressure is 2.5 atm. After a few days mass of the filled gas cylinder decreased to 23.2 kg.
Mass of the used n -butane = (29.0- 23.2)kg = 5.8 kg
∴ Number of moles of the gas \(=\frac{5.8 \times 1000}{58}=100\)
∴ Volume ofthe gas, \(V=\frac{n R T}{P}=\frac{100 \times 0.0821 \times(27+273)}{1.0}\)
= 2463L = 2.463 m3 [since lm3=103L]
Question 62. 0.3g of a metal reacts with dilute acid and produces 110 mL of H2 which is collected above water at 17°C temperature and 755 mm Hg pressure. Find the equivalent mass of the metal. [Pressure of water vapor at 17°C = 14.4 mmHg).
Answer:
Given:
0.3g of a metal reacts with dilute acid and produces 110 mL of H2 which is collected above water at 17°C temperature and 755 mm Hg pressure.
Actual pressure of H2 gas = (755-14.4) = 740.6mmHg
Let, the volume of H2 gas at STP be VmL, then
\(\frac{110 \times 740.6}{(273+17)}=\frac{V \times 760}{273}\)At STP, mass of 100.91mL H2
\(=\frac{2 \times 1.008 \times 100.91}{22400}=0.00908 \mathrm{~g}\)∴ 1.008g H2 replaces \(\frac{0.3 \times 1.008}{0.00908}=33.3 \mathrm{~g}\) = 33.3g metal \(E_{\text {metal }}=33.3\)
Question 63. Find the volume of ammonia gas (at STP) which on passing through 30mL 1.0 (N) H2S04 solution, the acidity of the solution decreases to 0.2 (N).
Answer:
Let, Wg NH3gas will be passed through the acid solution. Amount of H2S04 in 30mL 1 (N) solution
= 30 X 1 = 30 meq
Amount of H2S04 on passing Wg of NH3 gas through
the solution = 30 x 0.2 = 6 milli gram-equivalent
∴ Amount of neutralised H2S04 = 30-6 = 24 milligramequivalent. Now, 17gNH3 = 1 gram-equivalent
\(W \mathrm{~g} \mathrm{NH}_3=\frac{W}{17} \text { g-eqv. }=\frac{W \times 1000}{17} \text { milligram-equivalent }\) \(\frac{W \times 1000}{17}=24 \text { or, } W=\frac{17 \times 24}{1000}=0.408 \mathrm{~g}\)Volume of 17g NH3 at STP = 22.4L
Therefore Volume of0.408g of NH3 at STP
\(=\frac{22.4 \times 0.408}{17}=0.538 \mathrm{~L}\)Question 64. A hole is formed on a 0.1 mm thick aluminum sheet by pouring of1 mL 12 (M) HC1. If HC1 is completely used, then find the area (cm2) of the hole. (Density of A1 is 2.7g/cm3).
Answer:
Given:
A hole is formed on a 0.1 mm thick aluminum sheet by pouring of1 mL 12 (M) HC1. If HC1 is completely used
HC1in 1ml I2 (M) HC1 solution
= 12 millimpl = 12 x 10 3 mol
2Al(2mol = 54g) + 6HCl(6mol) 2AlCl3 + 3H2
Mass of A1 dissolved by 12 x 10~3mol of HCl.
\(=\frac{54 \times 12 \times 10^{-3}}{6} \mathrm{~g}\)Let, the area of the hole on the Al-sheet be x cm2.
∴ Volume of A1 dissolved by HC1 = (0.01 x x)cm3 and mass of this amount of’Al = (0.01 x xx 2.7)
⇒ \(\begin{aligned}
& 0.01 \times x \times 2.7=\frac{54 \times 12 \times 10^{-3}}{6} \\
& x=\frac{54 \times 12 \times 10^{-3}}{6 \times 0.01 \times 2.7}=4 \mathrm{~cm}^2
\end{aligned}\)
Question 65. 52.5 millimol LiAlH4 reacts with 15.6g (210 millimol) tertbutyl alcohol. In the following reaction, 157.5 millimol H2 is produced.
Answer:
Given:
52.5 millimol LiAlH4 reacts with 15.6g (210 millimol) tertbutyl alcohol.
⇒ \(\mathrm{LiAlH}_4+3\left(\mathrm{CH}_3\right)_3 \mathrm{COH} \rightarrow 3 \mathrm{H}_2+\mathrm{Li}\left[\left(\mathrm{CH}_3\right)_3 \mathrm{O}_3 \mathrm{AlH}\right.\)
On adding extra methanol or alcohol in the above reaction, displacement of the 4th hydrogen atom LiAlH4 will be observed by the following reaction.
⇒ \(\begin{aligned}
\mathrm{Li}\left[\left(\mathrm{CH}_3\right)_3 \mathrm{O}_3 \mathrm{AlH}+\mathrm{CH}_3 \mathrm{OH} \longrightarrow\right. & \mathrm{H}_2+\mathrm{Li}\left[\left(\mathrm{CH}_3\right)_3 \mathrm{O}_3\left[\mathrm{CH}_3\right] \mathrm{Al}\right.
\end{aligned}\)
How much H2 will evolve on adding methanol?
Answer: 1 mol of H2 will be produced from 1 mol of L(CH3)aCO]3AlH.
1 mol of Llt(CM3)3O]3 AIM will be produced from 1 mol of 1,1AIM,
From second equation, 1 mol of Ll[(CH3)3CO] A1H will produce 1 mol of H2 52.5 mmol of Li[(CM3)3CO]3AlH will react with excess of CH4 to produce 52.5 million of H2
Question 66. 0.19g of an impure H202 sample is dissolved in a 20 mL solution. 0.316 g of the solution reacts completely with KMn04 in the presence of H2SO4. Find the purity of the H2O2 sample.
Answer:
Given : 0.19g of an impure H2O2 sample is dissolved in a 20 mL solution. 0.316 g of the solution reacts completely with KMn04 in the presence of H2S04.
\(\begin{aligned}& 2 \mathrm{KMnO}_4+5 \mathrm{H}_2 \mathrm{O}_2+3 \mathrm{H}_2 \mathrm{SO}_4 \longrightarrow \\
& 2 \times 15 \mathrm{Bg} \quad 5 \times 34 \mathrm{~g} \quad \mathrm{~K}_2 \mathrm{SO}_4+2 \mathrm{MnSO}_4+5 \mathrm{O}_2+8 \mathrm{H}_2
\end{aligned}\)
H2O2 is required to react completely
\(=\frac{(5 \times 34) \times 0.316}{2 \times 158} \mathrm{~g}=0.17 \mathrm{~g}\)∴ Percentage purity of H2O2 sample \(=\frac{0.17 \times 100}{0.19}=89.47\)
Question 67. 20 mL of CH3COOH reacts with 20.1 mL of C2H5OH and produces CH3COOC2H5 according to the following reaction [density of CH3COOC2H5 is 0.902g/mL].
⇒ \(\mathrm{CH}_3 \mathrm{COOH}+\mathrm{C}_2 \mathrm{H}_5 \mathrm{OH} \rightarrow \mathrm{CH}_3 \mathrm{COOC}_2 \mathrm{H}_5+\mathrm{H}_2 \mathrm{O}\) Which one is the limiting reagent in this reaction?
If 27.5 mL of pure CH3COOC2H5 is produced, then find the percentage amount of production [density of CH3COOH and C2H2OH are 1.05g/mL and 0.789g/mL respectively]
Answer: CH3COOH + C2H5OH→CH3COOC2H5 + H2
⇒ \(\begin{aligned}
& \mathrm{CH}_3 \mathrm{COOH}=\frac{20.2 \times 1.05}{60}=0.353 ; \\
& \mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}=\frac{20.1 \times 0.789}{46}=0.344
\end{aligned}\)
CH3COOH and C2H5OH react in a ratio of 1:1.
As the number of moles of C2H5OH is less than CH3COOH. So, C2H5OH is the limiting reagent.
If C2H5OH is the limiting reagent, then several moles of CH3COOC2H5 = 0.3447(theoretical) Experimentally, CH3COOC2H5 formed.
\(=\frac{27.5 \times 0.902}{88}=0.2819 \mathrm{~mol}\)Percentage amount of CH3COOC2H5
⇒ \(=\frac{0.2819}{0.3447} \times 100=81.78\)
Question 68. How much H2 will evolve on adding methanol? W. 0.19g of an impure H202 sample is dissolved in a 20 solution. 0.316 g of the solution reacts completely with KMn04 in the presence of H2SO4. Find the purity of the H2O2 sample.
Answer:
⇒ \(n_{\mathrm{CO}}=\frac{P V}{R T}=\frac{750 \times 20}{760 \times 0.0821 \times 300 \times 1000}=8.01 \times 10^{-4}\)
⇒ \(\mathrm{CO}+\frac{1}{2} \mathrm{O}_2 \rightarrow \mathrm{CO}_2, n_{\mathrm{CO}^{\prime} / n_{\mathrm{O}_2}}=2\)
∴ Moles of O2 required (nO2) \(=\frac{n_{\mathrm{CO}}}{2}=\frac{8.01 \times 10^{-4}}{2}\)
Reaction involved: \(2 \mathrm{KClO}_3 \rightarrow 2 \mathrm{KCl}+3 \mathrm{O}_2\)
3 mol O2 is produced by 2 mol KC1O3.
∴ \(\mathrm{O}_2 \text { formed }=\frac{2}{3} \times \frac{8.01 \times 10^{-4}}{2}=2.66 \times 10^{-4} \mathrm{~mol}\)
∴ Weight of KC103 = 2.66 X 10-4 x 122.5 g = 3.27 x 102-2g
∴ % of KCIO2 in the mixture \(=\frac{3.24 \times 10^{-2}}{0.5} \times 100=65.4\)
Question 69. A metal M of atomic mass 54.94 has a density of 7.42g/cm3. Calculate the volume occupied and the radius ofthe atom of this metal assuming it to be a sphere.
Answer:
Given:
A metal M of atomic mass 54.94 has a density of 7.42g/cm3.
We know, the volume of atom x its density
\(=\frac{\text { atomic weight. }}{\text { Avogadro Number }}\)Ionic radius of atom =r, its volume \(=\frac{4}{3} \pi r^3\)
\(\frac{4}{3} \pi r^3 \times 7.42=\frac{54.94}{6.023 \times 10^{23}}\)therefore 1.432 x 10-8 cm
Volume \(=\frac{4}{3} \pi r^3=\frac{4}{3} \pi \times\left(1.432 \times 10^{-8}\right)^3\)
= 1.23 x 10-23cm3




























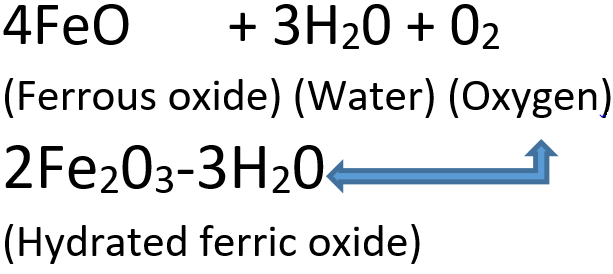

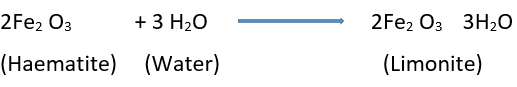
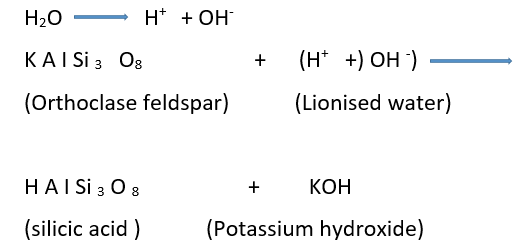
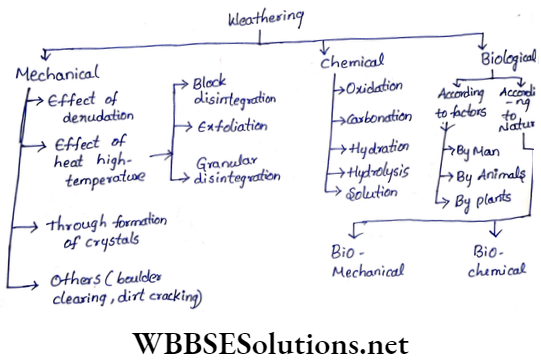
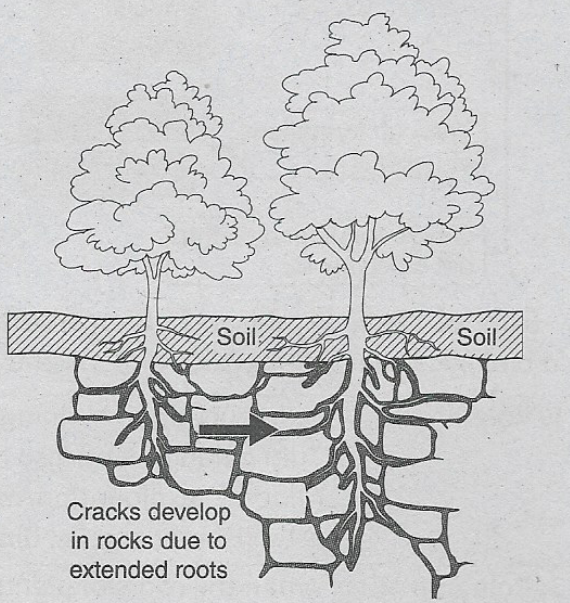

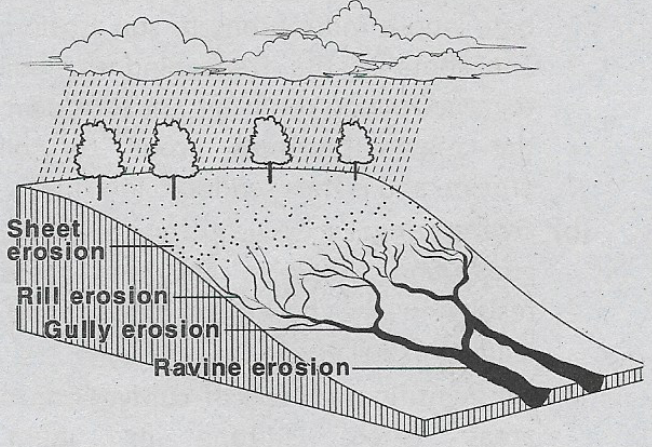






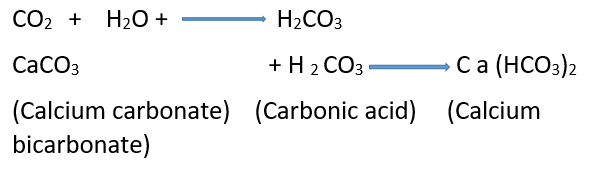 Question 21. What is hydration?
Question 21. What is hydration?