Chemical Thermodynamics Introduction
Energy is the ability of a physical system to do work. It always remains conserved in any event, i.e., it cannot be created or destroyed. However, it can take different forms such as mechanical energy, chemical energy, electrical energy, heat energy, light energy, sound energy, etc. Energy can be converted from one form to another.
Thermodynamics is the study to find out the conditions of interconversion of different forms of energy as well as to predict the extent of their conversions.
The branch of science which deals with the interconvertibility of different forms of energy (mainly heat and work) is called thermodynamics. The Greek words ‘thermo’ means heat and ‘dynamics’ means power.
Thermodynamics is based on four fundamental rules or laws namely zeroth law, first law, second law, and third law of thermodynamics. These laws are based on natural experience gathered over centuries. As the laws of thermodynamics are derived from the direct human experience, it is sometimes called axiomatic science.
Chemical Thermodynamics
Energy which we generally use in our daily lives is mostly obtained from chemical reactions. Thus the study of energy changes in chemical reactions is considered to be an important topic in chemistry.
The branch of thermodynamics that deals with energy evolved or absorbed during physical or chemical transformation is called chemical thermodynamics or chemical energetics.
Importance Of Thermodynamics
Some important applications of thermodynamics are:
- In thermodynamics, we study the inter-relationships among various macroscopic variables like pressure, temperature, volume, etc., and the changes in these variables as a result of various processes.
- Energy changes associated with physical or chemical transformations can be explained using thermodynamics.
- Under a given set of conditions, the feasibility of a physical or chemical transformation can be predicted from thermodynamics.
- Thermodynamics can predict the extent to which a physical or chemical transformation occurs before it reaches an equilibrium state.
- The relative amount of the reactants and products at the equilibrium of a reaction as well as the value of the equilibrium constant of the reaction can be determined by thermodynamics.
- Under a given set of working conditions, the maximum efficiency of a heat engine can be determined by thermodynamics.
- The ideal condition(s) for the transformation of different types ofenergy can be determined by thermodynamics.
Limitations of thermodynamics
- Classical thermodynamics applies only to the macroscopic system (For example in the case ofa few grams of ice) and not to the microscopic system (For example in the case of a few molecules or atoms). So it cannot give us any idea regarding the structure of matter.
- Thermodynamics does not give us any information about the rate or velocity ofa process (such as chemical reaction, osmosis, etc.). Also, it cannot give us any idea about the mechanism or path ofa chemical reaction.

Terms And Concepts Related To Thermodynamics
Some terms are frequently used in thermodynamics. One should have a good knowledge of these terms before going through the subject of thermodynamics. Here are these terms and their brief explanations.
System, Surroundings, and Boundary
System: In thermodynamics, a system is defined as the part of the universe under study, which is separated from the rest of the universe by real or imaginary boundaries- System
System, Surroundings, and Boundary Explanation: If we study something 2 about the human body, then the M human body is considered to be the system. Similarly, if we perform an experiment living cell or one mole of water, then the living cell or one mole of water will be the system.

Surroundings: Everything outside the system In the universe Is called the surroundings. Once the system Is defined, the surroundings will be defined automatically.
System + Surroundings=Universe
Boundary: Heal or Imaginary surface that separates a system from its surroundings is called the boundary of the system.
Explanation: If a certain amount of water taken In a beaker is considered to be the material of our experiment, then water will be our system. The rest of the universe including the beaker will be the surroundings of the lire system.
The interface between water and glass and that between water and air are the boundaries between the system and its surroundings.
If a gas enclosed in a cylinder fitted with a piston is considered to be the object of our analysis, then the gas will be our system, and the rest of the universe including the cylinder and the piston will constitute the surroundings of the system.
1 lore, the inner surfaces of the walls of the die cylinder, and the piston are the boundaries between the system and its surroundings.

Role of boundary: The boundary of a system plays an important role when there occurs an interaction between the system and its surroundings. During the interaction, a system exchanges energy or matter or both matter and energy with Its surroundings through its boundary.
Characteristics of the boundary of a system:
- The boundary of the n system may be real or imaginary.
- It may be rigid or non-rigid, in the case of a rigid boundary, the volume of the system does not change, while In the case of a non-rigid boundary, the volume of the system can change.
- Tim boundary of a system may be permeable or Impermeable. In the case of a permeable boundary, the exchange of matter takes place between the system and its surroundings through the boundary, whereas in the case of an impermeable Imundury, no exchange of matter can take place through the boundary.
- Exchange of beat may take place between a system and US surroundings through the boundary. A boundary that permits the cycling of heat between a system and Its surroundings Is called an antennal or diathermic boundary. On the other hand, a boundary that does not permit the exchange of heat between the system and its surroundings is called an adiabatic boundary.
- In reality, a perfect adiabatic boundary is not possible. The wall of a Dewar flask nearly behaves as an adiabatic boundary. According to the definition, the surroundings mean everything outside the system.
- But in thermodynamics, the surroundings are considered as the portions of the universe around the system, upto which a change occurring in the system has its influence, For Example, the burning of a candle produces light and heat.
- When we consider the effect of light, the tire space enclosing the candle, which is illuminated by Uie light, will be the surroundings. On the other hand, if we consider the effect of heat (produced), a narrow space affected by the produced heat, will be the surroundings
Types of system
Depending on the nature of its boundary, a system may or may not exchange matter energy, or both with its surroundings. Based on the exchange of matter and energy with the surroundings, systems may be classified as open systems, closed systems, and isolated systems.
Open system: A system that can exchange both energy and matter with its surroundings is called an open system.

Examples: Every living being in nature: Every living being (system) takes food (matter) and excretes waste materials (matter) to the surroundings. They (systems) also exchange heat (energy) with the surroundings.
Some water (or any other liquid) in an open container: In an open atmosphere, water (system) continuously evaporates and water vapor (matter) escapes from the container to the air (surroundings). Also, O2 or CO2 (matter) from the air may dissolve into water (system).
So, the exchange of matter takes place between the system and its surroundings. If the temperature of water (system) is different from that of its surroundings, then there occurs an exchange of heat (energy) between water and its surroundings.
The ocean: The Ocean Is a perfect example of an open system. Water (matter) evaporates from the ocean to the atmosphere and then again is added to it in time of rain. The ocean also absorbs heat (solar energy) and releases its energy in the form of latent heat.
Closed system: A system that can exchange energy with its surroundings but not the matter is called a closed system.

Examples: Boiling of water in a closed glass/metallic container: During boiling, water vapor (matter) cannot escape to the surroundings from the container, also any matter from the surroundings cannot enter into the system.
So, the exchange of matter is not possible between the system and its surroundings. But if the temperature of the water (system) is different from the surroundings, then heat (energy) will be exchanged between the system and its surroundings through the boundary wall of the container. So, the exchange of energy is possible between the system and its surroundings.
A gas enclosed in an impermeable metallic cylinder fitted with a piston: Since the cylinder and piston are impermeable, matter will not be exchanged between the system and its surroundings.
But heat (energy) will be exchanged between the system and its surroundings through the wall and piston of the cylinder. If a pressure greater than the pressure of the system is applied on the piston, work is done on the system. As a result, energy in the form of work is transferred to the system from its surroundings.
On the other hand, if the pressure on the piston is kept lower than that of the system, the system does the work on its surroundings, and energy in the form of work is transferred from the system to its surroundings.
Isolated system: A system that can exchange neither energy nor matter with its surroundings is called an isolated system.


An isolated system does not interact with its surroundings as its boundary is impervious to all matter and does not permit energy to pass through it As a result, any change occurring in an isolated system does not influence the surroundings, and vice-versa
Examples: Hot tea kept in a sealed & thermally insulated flask (Dewar flask): Since the flask is closed, matter cannot be transferred from the system (hot tea) to its surroundings, and vice-versa. As the walls ofthe flask are thermally insulated, the system cannot exchange energy with its surroundings.
Water taken in a closed container having rigid, impermeable, and thermally insulated walls: Since the container is closed and its walls are impermeable, no matter can exchange between the system (water) and its surroundings.
Furthermore, the container is insulated. So, no heat can flow into or out of the system. As the walls are rigid, there will be no change in the volume of the system. So, no energy in the form of work can be exchanged between the system and its surroundings. A perfectly isolated system is a hypothetical concept because no wall is perfectly adiabatic.
Based on physical properties and chemical composition, a system may further be classified as—
Homogeneous system: If the physical properties and chemical compositions are uniform throughout a system, then it is called a homogeneous system. For example, pure solid, liquid or gaseous substance, gas mixture, 2 completely miscible liquids like water, alcohol, etc.
Heterogeneous system: If the physical properties and the chemical compositions are different in different parts of a system, then it is called a heterogeneous system. For example, the mixture of two immiscible liquids (water and benzene), a mixture of two solids (sugar and NaCl) etc.
Extensive and intensive properties of a system
A system consisting of a large number of atoms, ions, or molecules is called a macroscopic system. For example, a certain amount of water, a certain volume of solution, a certain mass of sodium chloride, etc., are macroscopic systems. The properties associated with a macroscopic system are called macroscopic properties. For example, temperature, pressure, concentration, mass, density, composition, etc., are the properties ofa macroscopic system.
The macroscopic properties of a system can be classified into two categories:
- Extensive properties
- Intensive properties.
Extensive property: Tile property which depends upon the mass (or size) of the system i.e., the quantity of matter present in the system is called an extensive property. Extensive properties are additive, i.e., the total value of an extensive property of a system is equal to the sum of the extensive properties of different parts ofthe system.
Examples: Mass, volume, internal energy, enthalpy, entropy, heat capacity, Gibbs free energy, etc., are the extensive properties of a system.
The volume of a system: If V is the volume of lg substance (system) at a particular temperature and pressure, then the volume of 5 g of the same substance will be 5 x V. So the volume of a system is an extensive property.
Number of moles ofa system: If the number of moles of1 g of a substance (system) is ‘ri then the number of moles for 2 g of that substance will be ‘ 2n! So, several moles of a system is an extensive property.
The internal energy of a system: When water transforms to ice, its internal energy in the form of heat is liberated. Under an identical set of conditions, the heat liberated in the transformation of 5 g of water to 5 g of ice is found to be five times as much as that liberated when 1 g of water transforms into 1 g of ice. This means that a 5 g sample of water contains five times as much internal energy as a 1 g sample of water does. Thus, the internal energy ofa system is an extensive property.
Intensive property: The property that does not depend upon the mass (or size) of the system i.e., the amount of matter present in the system, is called an intensive property. Intensive property has the same magnitude at every point in a homogeneous system under equilibrium.
Examples: Temperature, pressure, density, viscosity, molar heat capacity, refractive index, boiling point, freezing point, surface tension, Viscosity coefficient, molar volume, molar internal energy, molar enthalpy, molar entropy, molar free energy, mole fraction, color, concentration, thermal conductivity, specific rotation, standard reduction potential, etc.
The boiling point of a liquid: If we take different amounts of pure water in two different containers and determine their boiling points at a given pressure, we get the same boiling point for both although the amount of water is different. Thus, the boiling point ofa liquid does not depend upon the amount of the liquid, implying that it is an intensive property.
The density of a substance: At a given temperature, the density of 1 kg of a pure sample of copper is the same as the density of lg of the same sample. Thus, the density of a substance does not depend upon the amount of substance, indicating that it is an Intensive property.
The concentration of a homogeneous solution: If the concentration of a homogeneous solution is 1 g.L-1 at a particular temperature, then the concentration ofa drop of that solution will also be 1 g.L-1 at that temperature. Thus the concentration of a homogeneous solution does not depend upon the amount of solution. So, it is an intensive property.
Important points regarding intensive & extensive properties:
- The ratio of two extensive properties is always an intensive property.
- Explanation: The mass (m) and volume (V) of a system are extensive properties. But the ratio ofthese two, i.e., density \(\left(\frac{m}{V}=d\right)\) is an intensive property.
- Extensive property becomes intensive when It Is expressed in terms of per unit mass or unit mole.
- Explanation: if x is an extensive property for moles of a system, then the value of X per mole, \(X_m=\frac{X}{n}\), will be an intensive property because it denotes the value of X for 1 mol of the system and is independent of the amount of substance.
- The internal energy of a system is an extensive property, but internal energy per mole (molar internal energy) or internal energy per gram (specific internal energy) is an intensive property.
- The volume of a system is an extensive property, but volume per mole (molar volume) or volume per gram (specific volume) is an intensive property. Similarly, the heat capacity of a system is an extensive property, but heat capacity per mole (molar heat capacity) or heat capacity per gram (specific heat capacity) is an intensive property.
Internal Energy And Its Change
Heat is released during the solidification of a liquid (for example when 1 g of water is transformed into ice at 0°C and late pressure, it releases 80 cal heat.) When coal burns, it produces both light and heat.
Mechanical work is done during the expansion of water vapor from high pressure to low pressure. We have many other examples like these, where we see that the system does work or produces heat or other form of energy out of its own intrinsic or inherent energy, without taking aid from an external energy source.
These observations indicate that every system contains some amount of energy intrinsically associated with it This intrinsic energy associated with every system or substance, is called its internal energy. Internal energy is denoted by the symbol either U or E.
Internal Energy And Its Change Definition: Every system, for its existence, is associated with an amount of energy in exchange for which it can do work or produce heat or another form of energy without the help of external energy. This energy is termed internal energy.
Origin of internal energy: The constituent particles in a system possess kinetic and potential energies arising from different internal modes of motion such as translational motion, vibrational motion, rotational motion, and electronic motion. In addition, they also possess nuclear energy, bond energy, and energy due to intermolecular attractions or repulsions.
All these energies contribute to the internal energy ofthe system. Therefore, the sum of all forms of energies ofthe constituent particles in a system gives rise to the internal energy ofthe system.
The absolute value of the internal energy of a system cannot be measured experimentally because it is not possible to determine all the types ofenergy associated with the internal energy of a system. However, in a process, the change in the internal energy (AU) of a system can be determined experimentally.
Some important points about internal energy
Internal energy is an extensive property.
Explanation: Internal energy ofa system is an extensive property because it increases as the amount of substance present in the system increases. For example, the internal energy of 5g of water is five times that of lg of water.
Internal energy depends upon the nature ofthe system.
Internal Energy And Its Change
Internal Energy And Its Change Explanation: The constituent particles (atoms, ions, and molecules) are different for different systems. This makes magnitudes of translational energy, rotational energy, vibrational energy, electronic energy, binding energy, etc. different for different systems.
Therefore, the values of internal energy will be different for different systems, even under identical conditions. For example, under identical conditions of temperature and pressure, the internal energy of one mole of O2 gas Is different from that of one mole of N2 gas.
Internal energy Increases with an increase in temperature. Explanation: With increasing temperature, the magnitudes of the translational motion, rotational motion, vibrational motion, etc., of constituents of a system increase. This results in an increase in energies associated with these motions. Consequently, the internal energy of the system increases.
Internal energy ofa system is a state function.
Explanation: In a process, the change in internal energy (All) ofa system only depends upon the initial and final states of the system. It does not depend upon the path followed for carrying out the process. A process with a given initial and final states can be carried out in different paths but the change in internal energy will be the same in all paths.
For example, the combustion of glucose, \(\mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_6(s)+6 \mathrm{O}_2(\mathrm{~g}) \rightarrow 6 \mathrm{CO}_2(\mathrm{~g})+6 \mathrm{H}_2 \mathrm{O}(l)\), can be carried out either by its direct combustion in a reaction vessel, or by oxidizing it in presence of enzyme. However, the change in internal energy in each case will be the same as the initial and final states for both processes are identical.
Change in the internal energy of a system in a process
Change in the internal energy of a system in a process Definition: process, the change in internal energy of a system is defined as the difference in internal energies between the final and initial states of the system in the process.
Let us consider a process in which the internal energy of the system at the initial and final states are U1 and U2, respectively. Therefore, the change in internal energy of the system, Δ17 = U2-Ul. Change in internal energy of a system in a chemical reaction: In a chemical reaction, the system is considered to be made up of reactants and products involved in the reaction.
At the beginning of the reaction, the system contains only reactants, and after the completion of the reaction, the system contains only products. Therefore, the change in internal energy in a chemical reaction equals the difference between the internal energies of the products and the reactants.
Let us consider a reaction: A →B The change in internal energies of the reaction is given by, \(\Delta \boldsymbol{U}=\overline{\boldsymbol{U}}_{\boldsymbol{B}}-\overline{\boldsymbol{U}}_A ; \text { where } \bar{U}_A \text { and } \bar{U}_B\) are the molar internal energies (internal energies per mole) of the product (B) and reactant (A), respectively
if then AU = negative, indicating an exothermic reaction.
if then ΔU = positive, indicating an endothermic reaction.
Both calorimeters are used to determine the change in internal energy in a combustion reaction.
The change In internal energy in an isothermal expansion or compression of an ideal gas (system) is zero. When an ideal gas undergoes isothermal expansion or compression, the average distance between the molecules in the gas changes, but this change does not affect the internal energy of the gas because the molecules of an ideal gas do not experience intermolecular forces of attraction. Therefore, an isothermal expansion or compression of an ideal gas (system) does not cause any change in the internal energy of the gas.
The change in internal energy of an ideal gas depends only upon temperature. At a constant temperature, the internal energy of an ideal gas is independent of its volume or pressure
Heat And Work In Thermodynamics
A system can interact with its surroundings by exchanging energy either in the form of heat or work or both. The interaction brings about changes in the properties ofthe system
In thermodynamics, heat is defined as the energy flowing across the boundary of a system by the temperature difference between the system and its surroundings.
Work In thermodynamics, work is defined as the energy transferred between the system and its surroundings due to the existence of unbalanced forces between the two.
Some important features of heat and work:
- Transfer of heat, work occurs only at the boundary of a system.
- Both heat and work are the forms of energy in transit. Heat or work appears only when a system undergoes a process. There is no existence of heat or work before or after the process. This means that heat and work are the forms ofenergy that are not stored within a system. Therefore, heat and work are not the properties of a system.
- Heat flows from a region of high temperature to a low temperature. If the temperature of a system is higher or lower than that of its surroundings, then heat flows from the system to the surroundings or vice-versa.
- A system can do work on its surroundings. When it occurs, we say that work is done by the system on its surroundings. Conversely, work can be done on the system by its surroundings.
- When heat flows from the surroundings to the system, the internal energy of the system increases. On the contrary, when heat flows from the system to its surroundings, then the internal energy ofthe system decreases. If work is done by the system on its surroundings, the internal energy of the system decreases. Conversely, if work is done on the system by its surroundings, the internal energy ofthe system increases.
- Heat and work are not the state functions of a system: When a system changes its state, the amount of heat transferred between the system and its surroundings or the amount of work involved in the change depends not only on the initial and the final states ofthe system but also on the path the system follows during the change. So, heat and work are not the state functions of a system. These are called path-dependent quantities.
Explanation: In the following example it can be shown that the work and heat are not the state functions ofa system. Let us consider, that 1 mol ofice at (0°C and 1atm) is to be converted into water at the same temperature and pressure.
1 mol of ice (0°C and 1 atm) → 1 mol of water (0°C and 1 atm) This transformation can be carried out by following the two alternative ways given below.
Work is done during rubbing but no heat is transferred. But in heat is transferred but no work is done. Although the initial and final states of the system are the same in both processes, the amount of work or heat involved in these processes is different. Thus, work done or heat transferred in a process depends on the route followed to carry out the process.
In a process, the change in a property of a system is calculated by subtracting the final value from the initial value. Generally, this difference is denoted by the symbol ‘A’. Heat and work are not the properties of the system. Heat and work are the energies in transit. There is no existence of heat and work before or after a process. Thus, we can write AP or AV, but not Aq or Aw.
Sign of heat and work in thermodynamics:
Sign of heat: The amount of heat transferred is expressed by Q If heat is transferred from the surroundings to the system, the system gains energy. The gain of heat by the system is represented by the +ve sign. For example, if a system absorbs local of heat from the surroundings, then q =+10 cal.
If heat is transferred from the system to the surroundings, the system loses energy. The heat rejected by the system is represented by the -ve sign. For example, if a system rejects 10 cal of heat to the surroundings, then q = -10 cal.

Sign of work: Work done on the system or by the system is denoted by w. According to the IUPAC convention, the energy ofthe system decreases when work is done by it. So, w is negative. On the other hand, the energy ofthe system increases if work is done on it. So it is positive, Example if 10 kj of work is performed by a system, then w = -10 kl. On the other hand, if 10 kj of work is performed on a system, w = +10 kj


Units of heat and work:
Units of heat: The CGS unit of heat is calorie. The SI unit of heat is joule. [1 cal = 4.184 joule]
Units of work: The traditional unit of work is erg. lerg =1 dyn. cm. SI unit of work is joule. 1 joule =1 N. m = 107 erg.
Pressure-Volume Work
A chemical reaction, is generally, associated with an absorption or evolution of heat However, many reactions also involve mechanical work along with the absorption or evolution of heat.
This work is done either by the system on its surroundings or by the surroundings of the system. The mechanical work associated with a chemical reaction is due to the change in the volume ofthe reaction system.
If the volume of a system increases against an external pressure or decreases by an external pressure, then some amount of mechanical work is performed because of the change in the volume of the system. This work is called pressure-volume work (or P-Vwork).
In a reaction, if one or more gaseous products are formed [example Zn(s) + 2HCl(aq)→ZnCl2(aq) + H2(g) ] or the number of moles of gaseous substances increases [example PCl5(g)→PCl3(g) + Cl2(g) ], then the volume of the system increases against external pressure (usually against atmospheric pressure). The work is thus performed by the system on the surroundings.
On the other hand, in a reaction, if the gaseous reactants are consumed [example 2H2(g) + O2(g) 2H2O(1) ] or the number of moles of gaseous substances decreases [example N2(g) + 3H2(g) 2NH3(g) ], then work is performed on the system! by the surroundings under the influence of external pressure.
Calculation of pressure-volume (P-V) work: According to mechanics, if dx is the displacement of the point of application of force F acting on a particular body, then [ work done = Fcost? dx [where 6 is the angle between the direction of applied F and the direction of displacement (dx) of the point, FcosB is the component of force along the direction ofdisplacement] If the applied force (F) and f . the displacement (dx) be in the same direction, then. 9 = 0 and cos# = 1 . Hence, in this case, we can write,

Again, when (lie applied force (P) and displacement are In the opposite direction, then and COHO m In this case, work done = Pdx.
Force And displacement are both vector quantities, but their product (work) Is a scalar quantity. The equation for pressure-volume work: Let us consider, that a gas is kept In a cylinder fitted with a weightless and frictionless piston. The piston Is held up by the stops (s,s).

Suppose, an external force is applied to the piston. If the cross-sectional area of the piston is A then external pressure, Pex \(=\frac{f_{e x}}{A}\) Suppose, Pex is less than the pressure of the gas.
Now, if stops (s, s) are removed, the gas will expand against external pressure till the piston is again held up by the stops (s1, S1). Ifthe piston moves a distance of dx, then work is done by the system on the surroundings
\(\delta w=-f_{e x} d x=-P_{e x} A d x\) [since \(f_{e x}=P_{e x} A\)
[According to the convention, work done bythe system is -ve ] Now, A dx – dV = increase in volume ofthe gas (system) due to the displacement of the piston. So, \(\delta w=-P_{e x} d V\) Let us consider the gas is expanded from its initial volume K1 to final volume V2. If this expansion is carried out in several steps, then the total work done by the gas.
⇒ \(w=-\int_{V_1}^{V_2} P_{e x} d V\)
[since Since work is not a state function, the value of \(\int_1^2 \delta w\) cannot be expressed as (u/2- u/1, instead we represent it as w .] Equation [1] is the general expression of P-Vwork. Work done by the gas and work done on the gas both can be calculated by using equation [1].
Expansion work of a gat: if a ga expands from iu initial volume V1 to final volume V2 against a constant eternal pressure of V2, then work done
⇒ \(w=-\int_{V_1}^{V_2} p_{e x} d V=p_{e x}\left(V_2-V_1\right)\left\{p_{s x}=\text { constant }\right]\)
⇒ \(=-P_{e x} \Delta V\)
⇒ \(w=-P_{e x} \Delta V=-v e\)
Since \(V_2>V_1, \Delta V=V_2-V_1=+v e j\)
Conventionally, Work done bythe system is negative.
Compression work of a gas: If a gas is compressed from its initial volume V1 to final volume V2 where a constant external pressure is Pex, then work done, \(w=-\int_{V_1}^{V_2} P_{e x} d V=-P_{e x}\left(V_2-V_1\right)\)
since Pex constant
⇒ \(=-P_{e x} \Delta V\)
∴ \(w=-P_{e x} \Delta V=+v e\)
Since \(V_2<V_1 \text { and } \Delta V=V_2-V_1=-v e\)
Conventionally, work done on the die system is positive.
Pressure-volume work in a reversible process
A reversible process is completed by an infinite number of small steps. In each step of this process, the driving force is infinitesimally greater than the opposing force.
Let V1 be the volume of a certain amount of a gas confined in a cylinder fitted with a weightless & frictionless piston. The gas is expanded reversibly from volume V1 to V2.
If the external pressure (Pex) on the gas is equal to the pressure ofthe gas (P), then there will be no displacement ofthe piston. Consequently, the volume ofthe gas does not change and the system remains in equilibrium.

Now, external pressure is diminished by an infinitesimal amount of DP. As a result, the gas will start to expand against pressure (Pex- dP) until the pressure of the gas equals the pressure (Pgx- dP). Suppose the volume is increased by an amount of dV. At the end of this expansion, the system regains its equilibrium state.
Again, external pressure is further decreased by an amount of DP. Consequently, the volume ofthe gas also increases by an infinitesimal amount of dV, and at the end of the expansion, the equilibrium of the system is restored. In this way, the gas is made to expand in an infinite number of small steps until the volume ofthe gas reaches V2.
Expression of pressure-volume work in a reversible process: In a reversible expansion of a gas, in each step, external pressure (Pex) is infinitesimally smaller than the pressure of the gas (P). Thus, in each step, Pgx is considered to be almost equal to P. So, in each step work done, 8wrgv = -PgxdV =-PdV [P = pressure ofthe gas at respective step, dV= infinitesimal increase in volume due to infinitesimal decrease in pressure & ‘rev’ = reversible]. If in a reversible expansion, the volume ofa gas is increased from V1 to V2, then-
⇒ \(w_{r e v}=-\int_{V_1}^{V_2} P d V\)
Equation [1] can be used to calculate the work done by a gas in its reversible expansion. To integrate equation [1], it is necessary to know the variation of pressure ofthe gas with volume. Work done on the gas in a reversible compression can also be calculated using equation [1], in this case, V2 < V1.
Work done by an ideal gas in its isothermal reversible expansion
Let us consider that n mol of an ideal gas is enclosed in a cylinder fitted with a weightless and frictionless piston. The initial pressure, temperature, and volume of the gas are P1, T, and V1 respectively. Now the gas is expanded reversibly from volume V1 to V2 under isothermal conditions. Suppose, due to this expansion the pressure of the gas decreases from P1 to P2.
Calculation of work done: Work done due to isothermal reversible expansion,
⇒ \(w_{r e v}=-\int_{V_1}^{V_2} P d V=-\int_{V_1}^{V_2} \frac{n R T}{V} d V \quad\left[P=\frac{n R T}{V}\right]\)
As the process is isothermal, so Tremains constant
∴ \(w_{r e v}=-n R T \int_{V_1}^{V_2} \frac{d V}{V}\)
∴ \(w_{r e v}=-n R T \ln \frac{V_2}{V_1}=-2.303 n R T \log \frac{V_2}{V_1}\)
At constant temperature for an ideal gas \(\frac{V_2}{V_1}=\frac{P_1}{P_2}\)
In equations [1] and [2], wrong is negative because during expansion V2 > & P1 > P2. This conforms with the convention because the sign of work done by the system is negative. Therefore, applying equations [1] and [2], it is possible to calculate the amount of work done by an ideal gas (system) in an isothermal reversible expansion.
Work done on an ideal gas in its isothermal reversible compression
Let us consider that n mol of an ideal gas is enclosed in a cylinder fitted with a weightless and frictionless piston. The initial pressure, temperature, and volume (before compression) are Px, T, and V respectively. Now the gas is compressed reversibly from volume Vj to V2 under isothermal conditions.

Suppose, the pressure of the gas increases from P1 to P2 because of this compression.
Calculation of work done: Work done in the isothermal reversible compression,
⇒ \(\begin{gathered}w_{r e v}=-n R T \ln \frac{V_2}{V_1} \\text { or, }_{r e v}=-n R T \ln \frac{P_1}{P_2}=-2.303 n R T \log \frac{P_1}{P_2}\end{gathered}\)
In equations [1] and [2], the sign is positive because V2 < V1 and P2 > P1. This conforms with the convention because the sign of work done on the system is positive. Therefore, applying equations [1] and [2], it is possible to calculate the amount of work done on an ideal gas (system) in its isothermal reversible compression.
Work done by a gas in its irreversible expansion
Let us assume that a certain amount of gas is kept in a cylinder fitted with a weightless and frictionless piston. Suppose, the initial volume and pressure of the gas are V and P1 respectively

If the external pressure is suddenly reduced to P2 (where P2 is much less than P1 ), the gas will go on expanding till the internal pressure becomes equal to P2. Let the volume of the gas increase from V1 to V2 due to the lowering of pressure from P1 to P2.
Calculation of Work done: Work done by the gas
\(w_{i r r}=-\int_{V_1}^{V_2} P_{e x} d V=-\int_{V_1}^{V_2} P_2 d V\left[\text { [External pressure, } P_{e x}=P_2 ; \text { irr }=\text { irreversible. }\right]\)
∴ \(w_{i r r}=-P_2\left(V_2-V_1\right)\)
As \(v_2>v_1, w_{i r r}\) = negative. This agrees with the convention as the sign of work done by the system is negative. The amount of work done by the gas in its irreversible expansion against a constant external pressure can be calculated by using the above equation [1],
Work done by n mol of an Ideal gas in Its isothermal irreversible expansion: From equation [1] we obtain \(w_{i r r}=-P_2\left(V_2-V_1\right) \text {; where, } V_2>V_1 \text {. For } n \mathrm{~mol} \text { of an }\) \(\text { ideal gas, } V=\frac{n R T}{P} \text {. At the initial state, } V_1=\frac{n R T}{P_1} \text { and at }\) \(\text { final state, } V_2=\frac{n R T}{P_2} \text {. Therefore, } w_{i r r}=-P_2\left(\frac{n R T}{P_2}-\frac{n R T}{P_1}\right)\)
∴ \(w_{i r r}=-n R T\left(1-\frac{P_2}{P_1}\right)\left[P_2<P_1\right]\)
The amount of work done by n mol of an ideal gas is due to its isothermal irreversible expansion can be calculated by using equation [1] or [2],
Work done on a gas in its irreversible compression
Let us consider a gas kept in a cylinder fitted with a frictionless and weightless piston is in thermodynamic equilibrium. Suppose, the initial pressure and volume of the gas are P1 and V1 respectively.
As the gas is in the state of thermodynamic equilibrium, the internal and external pressures are equal. If the external pressure is suddenly increased from (P1 to P2 (where P2 is much greater than P1 ), the volume of the gas will keep on decreasing until the internal pressure becomes equal to the external pressure P2. Suppose the volume of the gas decreases from V1{ to V2 due to an increase in pressure from P1 to P2.

Calculation of work done:
⇒ \(w_{i r r}=-\int_{V_1}^{V_2} P_{e x} d V=-\int_{V_1}^{V_2} P_2 d V \quad l \begin{aligned}
& {[\text { External pressure, }} \\
& \left.P_{e x}=P_2\right]
\end{aligned}\)
∴ \(w_{i r r}=-P_2\left(V_2-V_1\right)\)
∴ \(w_{i r r}=\text { positive }\left[V_2<V_1\right]\)
This agrees with the convention because the sign of work done on the system is positive.
The amount of work done on the gas (by the surroundings) in an isothermal irreversible compression can be calculated try the equation 11 {. Work done by n mol of an Ideal gnu In Its Isothermal Irreversible compression: Prom the equation |1| we obtain
⇒ \(w_{i r r}=-P_2\left(V_2-V_1\right) \quad\left|V_2<V_1\right|\)
⇒ \(\text { Initial volume, } V_1=\frac{n R T}{P_1} \text { and the final volume } V_2=\frac{n R T}{P_2}\)
Therefore \(w_{i r}=-P_2\left(\frac{n n T}{P_2}-\frac{n R T}{P_1}\right) .\)
∴ \(w_{i r r}=-n R T\left(1-\frac{P_2}{P_1}\right)\left[P_2>P_1\right]\)
The amount of work done on n mol of an ideal gas in its isothermal irreversible compression can be calculated by the equation [1] or [2].
The magnitude of work done by a gas in its reversible expansion is always greater than that in its irreversible expansion provided the initial and final states of the system arc are identical in hodi die cases.
Fit tu on Reversible expansion of gas occurs through a large number of infinitesimal steps, and in each step, the external pressure differs from the pressure of the gas by an infinitesimal amount. Therefore, in each step of a reversible expansion, the gas is expanded against the maximum possible pressure. As a result, The work obtained in a reversible expansion is maximum.
On die other hand, in an irreversible expansion of a gas, the external pressure differs from the pressure of the gas by a finite amount. In this expansion, since the gas expands against an external pressure fairly less than the pressure of the gas, the work obtained is always less than the reversible expansion.
Work done by a gas in its free expansion
In free expansion, the gas expands against zero external pressure. The work done \(\boldsymbol{w}=-\int_{V_1}^{V_2} \boldsymbol{P}_{e x} d V=-\int_{V_1}^{V_2} 0 \times d V=0\)
[Since the gas expands against zero pressure, Pex = 0] Therefore, the work done by a gas in its free expansion is zero.
Pressure-volume work in a chemical reaction
At a particular temperature and pressure, the change in volume of a reaction system is considered to be made up of reactants and products involved in the reaction depending primarily on the change in the number of moles of gaseous substances that participated in the reaction.
This is because the change in the number of moles of solid or liquid substances that participated in a reaction has a negligible effect on the volume of the reaction system.
Example: When I mol of Zn reacts completely with dilute HCl, 1 mol of H2 gas is produced. In this reaction, the change in volume of the reaction system will be approximately equal to the volume of 1 mol of H2 since the contribution of other constituents towards the volume change is negligible. Suppose, at constant temperature ( T) and pressure, the difference in volume between the gaseous products and reactants in a reaction is AV’. So. work done, w = -PAV. If the difference in the number of moles between the gaseous products and the gaseous reactants Is An and these gases behave like an ideal gas, then work done, \(w=-P \Delta V=-\Delta n R T\)
If An > 0, (Example \(\left.\mathrm{Zn}(s)+2 \mathrm{HCl}(a q) \rightarrow \mathrm{ZnCl}_2(a q)+\mathrm{H}_2(g)\right) .\). then w is negative. In this case, work is done by the system on die surroundings.
If An < 0 , [Example \(2 \mathrm{H}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l)\). then w is positive, in this case, work is done on the system by the surroundings.
Heat Capacity, Specific Heat Capacity And Molar Heat Capacity
Heat capacity of a substance
The heat capacity is the quantity of heat required to raise the temperature ofa substance by 1°C (1 K).
The heat capacity is usually denoted by If of the amount of heat required to raise the temperature of a given amount of substance, then its heat capacity, \(C=\frac{δ q}{d T} \).
characteristics of the heat capacity of a substance
The higher the heat capacity of a substance, the smaller the increase in temperature of a given amount of the substance when a certain amount of heat is added to it. For example, the heat capacity of water is higher than that of copper. So more heat will be required to raise the temperature of 1 g of water than lg of copper by 1 K (or 1°C).
The heat capacity of a substance depends on its nature.
The heat capacity ofa substance depends on its amount. Thus, it is an extensive property.
The heat capacity is a path-dependent quantity. The heat required to raise the temperature of a substance by IK depends on the process by which the substance is heated. For example, the amount of heat required to raise the temperature of1 mol of N2 gas by IK depends on whether the heating is done at constant volume or constant pressure.
Unit: \(\text { cal } \cdot{ }^{\circ} \mathrm{C}^{-1}\left[\text { or, cal } \cdot \mathrm{K}^{-1}\right] \text { or, } \mathrm{J} \cdot{ }^{\circ} \mathrm{C}^{-1}\left[\text { or, } \mathrm{J} \cdot \mathrm{K}^{-1}\right] \text {. }\)
Specific heat or specific heat capacity of a substance
The amount of heat required to raise the temperature of the unit mass ofa substance by 1°C [or IK] is called specific heat or specific heat capacity ofthe substance. It is represented by d. Specific heat capacity is an intensive property ofthe system.
⇒ \(Unit: cal \cdot \mathrm{g}^{-1} \cdot{ }^{\circ} \mathrm{C}^{-1} or, cal \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1} or, \mathrm{J} \cdot \mathrm{g}^{-1} \cdot{ }^{\circ} \mathrm{C}^{-1} or, \mathrm{J} \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1}.\).
The specific heat capacity of water (4.]8J-g-1.°C-1) is considerably higher than that of other common substances. Thus, a large amount of heat as well as time is required to warm a given amount of water. For the same reason, hot water takes a long time to cool.
Molar heat capacity
The amount of heat required to raise the temperature of I mol of a substance by 1°C (or IK) is called the molar heat capacity of the substance. The molar heat capacity is denoted by’ Cm’ (the suffix’ m ’ refers to molar), it is an Intensive property.
⇒ \(\mathrm{cal} \cdot \mathrm{mol}^{-1} \cdot{ }^{\circ} \mathrm{C}^{-1} \text { or, cal } \cdot \mathrm{mol}^{-1} \cdot \mathrm{K}^{-1}\)
⇒ \(\text { or, } \mathrm{J} \cdot \mathrm{mol}^{-1} \cdot{ }^{\circ} \mathrm{C}^{-1} \text { or, } \mathrm{J} \cdot \mathrm{mol}^{-1} \cdot \mathrm{K}^{-1} \text {. }\)
Molar heated panacea substance(Cm)
=Specify Lear capacity of me substance(c) x Molar mass (M)
Heat absorbed or released by substance (q)
= mass of the substance (m)>specificheat capacity (c) x increase or decrease in temperature <Ar> thus, q= mxcxT
[where AT =final temperature- initial temperature] Using fee equation [1], we can calculate c if we know the values of, m and AT. The heat released or absorbed (q) can be calculated by using equation [1] if we know the values of c, m, and AT. An increase or decrease in temperature (AT) can be calculated by using equation[1] if we know the values of q,c, and m.
Heat Capacity Of A Substance At Constant Volume And Pressure


Comparison of the values of molar heat capacities at constant pressure and constant volume
Comparison for gaseous substances: For gaseous substances, the molar heat capacity at constant pressure is greater than die molar heat capacity at constant volume.
Addition of heat at constant volume: As the volume of the system is constant, no external work by the system is possible. So, all die heat added to the system will be used for increasing the internal energy of the system, which in turn increases the temperature of the die system.

Suppose, the addition of 8a amount of heat to 1 mole of gas causes an increase in the temperature of the gas by dT. Therefore, by definition,
⇒ \(C_{V, m}=\left(\frac{\delta q}{d T}\right)_V\)
Addition of heat at constant pressure: When heat is added to a gas at constant pressure the added heat is used up in two ways. One part of it is expended for the external work done by the system, and the other part of it goes to increase the internal energy of the system.
As a result of an increase in internal energy, the temperature of the system also increases Suppose, when 5q amount of heat is added to l mole of a gas at constant pressure, the temperature of the gas is increased by dT’.
So, by definition \(C_{P, m}=\left(\frac{\delta q}{d T^{\prime}}\right)_P\) Therefore, if Ihe same amount of heat Is added to I mol of a given gas separately at constant volume and constant pressure, then the Increase in temperature will be small at constant pressure than at constant volume so dt’,dt and cp, m. Cv, m. As a result of this, the molar heat capacity of a gas at constant pressure(cp,m) will be greater than its molar heat capacity at constant volume (cv,m).
Comparison in the case of solids and liquids: when heat is added to a solid or liquid, there occurs no significant change in their volumes. Thus, the work involved in the process of heating a solid or liquid at constant pressure is negligible. This is why the values of Cp m and Cv m are found to be almost the same in the case of a liquid or solid.
For a gas, the ratio of the molar heat capacity at constant pressure (Cp m) to the molar heat capacity at constant volume (Cp,m) is termed as heat capacity ratio (7). Therefore, 7 =CPf m/C V, m
Some relations of Cp and Cv for an ideal gas 1) Internal energy ( U) of an ideal gas depends only on the temperature (T), neither on the pressure (P) nor the volume (V). When an ideal gas undergoes a process involving only P-V work, the heat absorbed by the gas is equal to its internal energy change. Therefore, for an ideal gas
⇒ \(C_V=\left(\frac{\delta q}{d T}\right)_V=\frac{d U}{d T} \text { or, } d \boldsymbol{d}=\boldsymbol{C}_V \boldsymbol{d} T\)
Enthalpy (H) of an ideal gas depends only on temperature (T). It does not depend either on pressure (P) or on volume (V). When an ideal gas undergoes a process involving only P-V work, the heat absorbed by the gas is equal to its enthalpy change. Therefore, for an ideal gas, \(C_P=\left(\frac{\delta q}{d T}\right)_P=\frac{d H}{d T} \text { or, } d H=C_P d T\)
For an ideal gas, the difference between the moon’s heat capacities at constant pressure and constant volume is equal to the universal gas constant (R). Thus, CP, m -CV, m = R.
For 1 mole of an ideal gas, PV = RT
Substitution RT for PV in the relation H = U+ PV gives the enthalpy for1 mol of an ideal gas, i.e., H = U+ RT Differentiating both sides, we have dH = dU + RdT.
Dividing both sides by dT gives
⇒ \(\frac{d H}{d T}=\frac{d U}{d T}+R \quad \text { or, } C_{P, m}=C_{V, m}+R\) Or, \(C_{P, m}-C_{V, m}=R\)
[For an ideal gas, dU = CydT and dH = CpdT}
The change in internal energy (A) with the change in temperature of an ideal gas: Suppose, the ‘n’ mole of an ideal gas undergoes a process in which its temperature changes from T1 to T2 and so does its internal energy from U1 to U2. Therefore, the change in internal energy ofthe gas,
⇒ \(\int_{U_1}^{U_2} d U=\int_{T_1}^{T_2} n C_{V, m} d T\)
⇒ \(\text { or, } U_2-U_1=\int_{T_1}^{T_2} n C_{V, m} d T \text { or, } \Delta U=\int_{T_1}^{T_2} n C_{V, m} d T\)
If CV,m is considered to be independent of temperature within the temperature range to T2, then | AU = nCV/Therefore, the change in internal energy (AI7) with the temperature change can be calculated by using equation [1], If T2> T1, then AU = positive, ie., with increasing temperature, the internal energy of the system increases. If T2<Ty then A U = negative i.e., with decreasing temperature, the internal energy of the system decreases.
The change in enthalpy (AH) with the change in temperature for an ideal gas: Suppose, an ideal gas undergoes a process in which its temperature changes from T1 to T2.
As a result of which its enthalpy changes from H1 to H2. Therefore, the change in enthalpy of the gas in this process is:
⇒ \(\int_{H_1}^{H_2} d H=\int_{T_1}^{T_2} n C_{P, m} d T\)
⇒ \(\text { or, } H_2-H_1=\int_{T_1}^{T_2} n C_{P, m} d T \text { or, } \Delta H=\int_{T_1}^{T_2} n C_{P, m} d T\)
If Cp m is considered to be independent of temperature within the temperature range T1 to T2, then, \(\Delta H=n C_{P, m}\left(T_2-T_1\right)\) So, the change in enthalpy (AH) with the change in temperature can be calculated by using equation [1],
Numerical Examples
Question 1. How much 90g of water from 30°C to 100°C? [Molar heat capacity of water at constant pressure = 75.3 J.mol-1 K-1]
Answer: The specific heat of water at constant pressure,
⇒ \(c_P=\frac{C_{P, m}}{M}=\frac{75.3}{18}=4.18 \mathrm{~J} \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1}\)
where, Cp m = molar heat capacity at constant pressure, M = molar mass ofthe substance Mass of water, m = 90 g and AT = (373-303)K =70K
∴ q =mx cpx AT = 90 x 4.18 x 70 =26334 J =26.33 kj
∴ 26.33 kl of heat is required to raise the temperature of 90g of water from 30 °C to 100 °C.
Question 2. How much heat will be released when the temperature of1 mol of water changes from 90 °C to 80 °C? Given: Specific heat of water 4.18 J.g-1.K-1
Answer: We know, q = mx cx AT The amount of water = lmol. Therefore, m = 18g, c = 4.18 J.g-1.K-1 and \(\Delta T=[(273+80)-(273+90)] \mathrm{K}=-10 \mathrm{~K}\)
∴ q = 18 X 4.18 X (-10) J = —752.40J
So, the amount of heat that will be liberated when the temperature of1 mol water changes from 90 °C to 80 °C is 752.40 J.
Question 3. Specific heats of an ideal gas at constant volume & constant pressure are 0.015 and 0.025 cal. g-1 .K-1 respectively. Determine the molar mass of the gas.
Answer: \(c_V=0.015 \mathrm{cal} \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1}, c_P=0.025 \mathrm{cal} \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1}\)
∴ Molar heat capacity at constant volume, Cy m=M x Cy, and that at constant pressure, Cp m = M x cp[Af= molar mass] Again we know, for an ideal gas, Cp m- Cy m = R.
∴ M(Cp-Cy) = R
⇒ \(\text { or, } \quad M(0.025-0.015) \mathrm{cal}^{-1} \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1}=1.987 \mathrm{cal}^{-1} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}\)
or, Af = 198.7g-mol-1 \(\left[ R=1.987 \mathrm{cal} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}\right]\)
∴ The molar mass of the gas = 198.7 g-mol-1.
Question 3. Specific heats of an ideal gas at constant volume And constant pressure are 0.015 and 0.025 cal. g-1.K-1 respectively. Determine the molar mass of the gas.
Answer: \(c_V=0.015 \mathrm{cal} \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1}, c_P=0.025 \mathrm{cal} \cdot \mathrm{g}^{-1} \cdot \mathrm{K}^{-1}\)
∴ Molar heat capacity at constant volume, Cy m=M x Cy and that at constant pressure, Cp m = M x cp[Af= molar mass]
Again we know, for ideal gas, \(C_{P, m}-C_{V, m}=R.\)
∴ M(Cp-Cy) = R
Or, \(M=198.7 \mathrm{~g} \cdot \mathrm{mol}^{-1}\)
Since \(R=1.987 \mathrm{cal} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}\)
∴ Molar mass ofthe gas = 198.7 g-mol-1.
Heat Change In A Chemical Reaction
Like other processes, heat exchange with the surroundings also occurs in case of chemical reactions. The amount of heat absorbed or given off during a reaction depends on the conditions under which the reaction is carried out.
Generally, reactions are carried out either under the condition of constant volume or under the condition of constant pressure. However, the fact is that carrying out a reaction at constant volume is not as convenient as that at constant pressure.
In the laboratory, reactions are carried out in containers open to the atmosphere, so they occur under the condition of constant atmospheric pressure. According to the first law of thermodynamics, if a process involving only pressure-volume work occurs at constant volume, on heat change (qv) in the process equals the change in internal energy (AU) of the system.
Therefore, ΔU = qv or, = -qy. On the other hand, if a process occurs at constant pressure according to the first law of thermodynamics, the heat change in the process (qp) becomes equal to the change in enthalpy (AH) of the system. Therefore, ΔH = qp or, -ΔH= -qP
In a process, the change in internal energy is ΔU = U2-U1, and the change in enthalpy is ΔH – H2– H1 where subscripts 1 and 2 denote the initial state and the final state of the system, respectively, in the process. In a reaction system, only reactants are present at the beginning of the reaction and only products are present at the end of the chemical reaction.
Therefore, the internal energy or enthalpy at the beginning of a reaction means the total internal energy or enthalpy ofthe reactants undergoing the reaction. Similarly, the internal energy or enthalpy at the end of a reaction means the total internal energy or enthalpy of the products formed in the reaction. Therefore,
- For a chemical reaction occurring at constant volume, the heat change (qv) = AU = (total internal energy of the products – total Internal energy of the reactants) \(=U_{\text {products }}-U_{\text {reactants }}=U_P-U_{R^*}\)
- For a reaction occurring at constant pressure, the heat Change \(\left(q_P\right)=\Delta H=H_{\text {products }}-H_{\text {reactants }}=H_P-H_R \text {. }\)
As most of the reactions are carried out at constant pressure and heat change associated with constant pressure equals the change in enthalpy, the heat change of the reaction in a reaction usually signifies the change in enthalpy (AH) is the reaction unless the constant-volume condition is stated.
Exothermic and endothermic reactions
Exothermic reaction: Reactions associated with the evolution of heat are called exothermic reactions. In an exothermic reaction, heat is released from the reacting system to the surroundings.
Thus, when we touch the reaction container (which is the part of the surroundings), in which the exothermic reaction is taking place, we feel warm. Since heat is released in an exothermic reaction, the total energy of the reactants is greater than that of the products.
Let us consider an exothermic reaction: A + B→C + D. If the heat released in the reaction is’ q ‘ then, the total energy of the reactants = total energy of the products + q t.e., the total energy of A and H = the total energy of C and D + q.
If we imagine ‘q’ as the part of the die product, then the above equation can be written as A + B →C + D + q The amount of heat evolved is usually written with a positive sign on the right-hand side of the balanced equation of the reaction.
Endothermic reaction) Reactions Associated with the absorption of hoot are called endothermic reactions, In an endothermic reaction. In the absorbed by the reading system to the surroundings. Thus, when we touch the reaction container (which Is the pan of the surroundings), in which an endothermic reaction Is taking place, we feel cold.
Since the absorption of bent occurs In mi endothermic reaction, the total energy of products will be greater than the total energy of the reactants. Let us consider an endothermic reaction; A + B→C+D.
Heat absorbed In this reaction he then the total energy of the reactants s the total energy if the products total energy of A and If = total energy of C and D- q.
If imagine ‘q’ as the part ofthe reactant, then the above equation can be written as:
A+B+QC+D Or, A+B+C+D-q
The amount of heat absorbed is usually written with a negative sign on the right-hand side of the balanced equation ofthe reaction.
Enthalpy change in exothermic reactions; In an exothermic reaction, heat is released by the reacting system. Therefore, for any exothermic reaction occurring at constant pressure qp<0. Since qp=ΔH, ΔH<0 Or, \(\Sigma H_{\text {products }}-\Sigma H_{\text {reactants }}<0 \quad \text { or, } \quad \Sigma H_{\text {products }}\) Thus the total enthalpy ofthe products is less than that ofthe reactants.
In an exothermic reaction, the enthalpy of the reaction system decreases (AH < 0). Enthalpy change in endothermic reactions: In an endothermic reaction heat is absorbed by the reaction system. Therefore, for any endothermic reaction occurring at constant pressure, qo>0. Since \(q_p=\Delta H, \Delta H>0 \text { or, } \Sigma H_{\text {products }}-\Sigma H_{\text {reactants }}>0\text { or, } \Sigma H_{\text {products }}>\Sigma H_{\text {reactants }}\)
In an endothermic reaction, the enthalpy of the reaction system Increases (ΔH > 0).
Enthalpy diagram of exothermic and endothermic reactions In an exothermic reaction
\(\Sigma H_{\text {products }}<\Sigma H_{\text {reactants }}\) So, in its enthalpy diagram ,\(\Sigma H_{\text {products }}\) lies below the \(\Sigma H_{\text {reactants }}\) hand, In an endothermic reaction, \(\Sigma n_{\text {products }}>\Sigma H_{\text {reactants }}\) So, in Its enthalpy diagram, \(\Sigma H_{\text {products }}\) lies above the \(\Sigma H_{\text {reactions }}\)


Thermochemical equations
Thermochemical equations Definitions: The thermochemical equation is a balanced chemical equation, in which the physical states of the reactant(s) and product(s) as well as the amount of heat evolved or absorbed in the reaction are mentioned.
Conventions for writing a thermochemical equation: To indicate the physical states ofthe reactant (s) and the product(s), symbols s, l, and g are used for solid, liquid, and gaseous states, respectively. These symbols are to be placed within parentheses just after the chemical formulae ofthe substances concerned.
For any reactant or product dissolved in aqueous solution, the term ‘ aq ’ (short form of the word aqueous) is to be placed within a parenthesis just after its formula.
The amount of heat evolved or absorbed (or the enthalpy change) in a reaction is to be written with a proper sign (+ or -) on the right-hand side immediately after the balanced equation.
In an exothermic reaction, if x kj of heat is evolved, then +x kj or AH = -x kj is to be written on the right-hand side immediately after the balanced chemical equation.
Example: \(\mathrm{CH}_4(\mathrm{~g})+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(l)+890.3 \mathrm{~kJ}\) or, \(\text { or, } \mathrm{CH}_4(\mathrm{~g})+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(\mathrm{l}) ; \Delta H=-890.3 \mathrm{~kJ}\)
In an endothermic reaction, if +x kj of heat is absorbed, then -x kj or AH = +xkj is to be written on; the right hand immediately after the balanced equation.
Example \(\mathrm{N}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{NO}(g)-180.5 \mathrm{~kJ}\) or, \(\mathrm{N}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{NO}(g), \Delta H=+180.5 \mathrm{~kJ}\)
The coefficients of the reactants and the products in a thermochemical equation indicate their respective number of moles This allows us to use fractional coefficients for reactants and products. Because AH is an extensive property, when the balanced equation is multiplied by a factor, the value of AH will also be multiplied by that factor.
⇒ \(2 \mathrm{H}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l), \Delta H=-571.6 \mathrm{~kJ}\) or, \(\mathrm{H}_2(\mathrm{~g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{H}_2 \mathrm{O}(l), \Delta H=-285.8 \mathrm{~kJ}\)
Unless otherwise stated the value of ΔH mentioned in a thermochemical equation is considered to be the value at standard states.
A thermochemical equation can be written in this manner. When this is done, the magnitude of AH remains the same but its sign becomes the opposite
Example: \(\mathrm{N}_2(g)+\mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{NO}(\mathrm{g}) ; \Delta H=+180.5 \mathrm{~kJ}\) or \(2 \mathrm{NO}(g) \rightarrow \mathrm{N}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}) ; \Delta H=-180.5 \mathrm{~kJ}\)
Numerical Examples
Question 1. At a fixed temperature & pressure, the heat released in the formation of 3 mol SO3(g) from SO2(g) and 2(S) is 291 kj. What will be the change in enthalpies in the formation of1 mol & 4 mol SO3(g)?
Answer: The thermochemical equation for the formation of 3 mol of SO3(g) from the reaction between SO2(g) and O2(g) is given \(3 \mathrm{SO}_2(\mathrm{~g})+\frac{3}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow 3 \mathrm{SO}_3(\mathrm{~g}), \Delta H=-291 \mathrm{~kJ}\)
The thermochemical equation for the formation of1 mol of \(\mathrm{SO}_3(g) \text { is: } \mathrm{SO}_2(\mathrm{~g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{SO}_3(\mathrm{~g}) \text {, }\) \(\Delta H=\frac{1}{3}(-291) \mathrm{kJ}=-97 \mathrm{~kJ}\)
Therefore, the change in enthalpy for the formation of lmol and 4mol S03(g) will be -97 kj and 4x(-97) =- 388 kj, respectively.
Question 2. At a particular temperature and pressure, the heat produced in the formation of 2 mol of C2H6(g) from the reaction between C2H2(g) and H2(g) is 626 kj. What amount of H2(g) will react with the required amount of C2H2(g) to produce 939 kj of heat at the same temperature and pressure?
Answer: The Thermochemical equation for the formation of 2 mol of 2H6(g) from the reaction between C2H2(g) and H2(g) is:
⇒ \(2 \mathrm{C}_2 \mathrm{H}_2(\mathrm{~g})+4 \mathrm{H}_2(\mathrm{~g}) \rightarrow 2 \mathrm{C}_2 \mathrm{H}_6(\mathrm{~g}) ; \Delta H=-626 \mathrm{~kJ}\)
∴ 626 kj of heat = 4 mol H2(g)
∴ \(939 \mathrm{~kJ} \text { of heat } \equiv \frac{4}{626} \times 939 \equiv 6 \mathrm{~mol} \mathrm{H}_2(\mathrm{~g})\)
=6×2= 12g H2(g)
Therefore, if 12 g H2(g) reacts with the required amount of C2H2(g) to produce C2H6(g), then 939 kJ of the heat is evolved.
Question 3. At a particular temperature and pressure, N2(g) and O2(g) react to form 4 mol of N2O. The heat absorbed in this reaction is 328 kj. What would the change in enthalpy be due to the formation of 2 mol of N2(g) and 1 mol of O2(g) from N2O(g) at the same temperature and pressure?
Answer: The thermochemical equation for the formation of
⇒ \(\mathrm{N}_2 \mathrm{O}(g): 4 \mathrm{~N}_2(g)+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow 4 \mathrm{~N}_2 \mathrm{O}(g) ; \Delta H=+328 \mathrm{~kJ}\)
Writing this equation in the reverse manner, we obtain,
⇒ \(4 \mathrm{~N}_2 \mathrm{O}(\mathrm{g}) \rightarrow 4 \mathrm{~N}_2(\mathrm{~g})+2 \mathrm{O}_2(\mathrm{~g}) ; \Delta H=-328 \mathrm{~kJ}\)
Therefore, the thermochemical equation for the formation of 2 mol of N2(g) and 1 mol of O2(g) from N2O(g) will be: \(2 \mathrm{~N}_2 \mathrm{O}(\mathrm{g}) \rightarrow 2 \mathrm{~N}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}); \Delta H=-164 \mathrm{~kJ}\) Thus, the change in enthalpy for the formation of 2 mol of N2(g) and 1 mol of O2(g) is -164 kJ.
Heat Of Reaction
Heat Of Reaction Definition: The Amount of heat absorbed or evolved in a reaction when the stoichiometric number of moles of reactions indicated by the balanced chemical equation, is completely converted into products at given conditions is called the heat of reaction at that conditions.
At a particular temperature, the heat of a reaction depends on the conditions under which a reaction is occurring. Generally, chemical reactions are carried out either at constant pressure or at constant volume. Accordingly, the heat of reaction is of two types, namely Heat of reaction at constant volume and heat of reaction at constant pressure.
The heat of reaction at constant volume: The amount of heat absorbed or evolved in a reaction when the stoichiometric number of moles of reactants, indicated by the balanced chemical equation of the reaction, is completely converted into products at a fixed temperature and volume is called the heat of reaction at constant volume.
Explanation: Let us consider a chemical reaction that is occurring at constant temperature and volume.
⇒ \(a A+b B \rightarrow c C+d D\)
According to die first law of thermodynamics, if a reaction occurs at constant volume then the heat change (qv) is equal to the change in internal energy (AU) ofthe system (provided only pressure-volume work is performed), qv= ΔU
Therefore, for the reaction
⇒ \(q_V=\Delta U=\Sigma U_{\text {products }}-\Sigma U_{\text {reactants }}=\Sigma U_P-\Sigma U_R\)
where ΣUp = total internal energy of products and I UR = total internal energy of reactants.
∴ \(q_V=\Sigma U_p-\Sigma U_R=\left(c \bar{U}_C+d \bar{U}_D\right)-\left(a \bar{U}_A+b \bar{U}_B\right)\)
Where \(\bar{U}_A, \bar{U}_B, \bar{U}_C \text { and } \bar{U}_D\) die molar internal energies of A. B. Cand D, respectively. Thus, the heat of reaction for a reaction at constant temperature and volume is the difference between the total internal energy of tire products and the total internal energy of the reactants.
Example: \(\mathrm{CH}_4(\mathrm{~g})+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(\mathrm{l})\)
For the above reaction, the heat of the reaction at a constant volume:
⇒ \(q_V=\left[\bar{U}\left(\mathrm{CO}_2, g\right)+2 \bar{U}\left(\mathrm{H}_2 \mathrm{O}, l\right)\right]-\left[\bar{U}\left(\mathrm{CH}_4, g\right)+2 \bar{U}\left(\mathrm{O}_2, g\right)\right]\)
Where \(\bar{U}\left(\mathrm{CO}_2, \mathrm{~g}\right), \bar{U}\left(\mathrm{H}_2 \mathrm{O}, l\right), \bar{U}\left(\mathrm{CH}_4, g\right) \text { and } \bar{U}\left(\mathrm{O}_2, g\right)\) are die molar internal energies of CO,(g), H2O(I), CH4(g) respectively.
Heat of reaction at constant pressure: The amount of heat absorbed or evolved in a reaction when the stoichiometric number of moles of reactants, indicated by a balanced chemical equation of the reaction, is completely converted into products at constant pressure and temperature is called the heat of reaction at constant pressure.
Heat of reaction at constant pressure Explanation: Let us consider a reaction that is occurring at constant temperature and pressure: aA + bB →+ cC + dD. According to the first law of thermodynamics, if a reaction occurs at constant pressure, then the heat change in the reaction is equal to the change in enthalpy of the system (provided only pressure-volume work is performed). Thus, qp = AH. Therefore, for the reaction,
⇒ \(q_P=\Delta H=\Sigma H_{\text {products }}-\Sigma H_{\text {reactants }}=\Sigma H_P-\Sigma H_R \text { ; }\)
where Hp = total enthalpy ofthe products and HR = total enthalpy of the reactants
∴ \(q_P=\Sigma H_P-\Sigma H_R=\left(c \bar{H}_C+d \bar{H}_D\right)-\left(a \bar{H}_A+b \bar{H}_B\right)\)
where, \(\bar{H}_A, \bar{H}_B, \bar{H}_C \text { and } \bar{H}_D\) are the molar enthalpies of A, B, C, and D, respectively.
Thus, for a reaction, the heat of the reaction at constant temperature and pressure is equal to the difference between the total enthalpy of products and the total enthalpy of reactants.
Example: For the Reaction, \(2 \mathrm{H}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l)\) the heat ofreaction at constant pressure is given by;
⇒ \(q_P=2 \bar{H}\left(\mathrm{H}_2 \mathrm{O}, l\right)-\left[2 \bar{H}\left(\mathrm{H}_2, \mathrm{~g}\right)+\bar{H}\left(\mathrm{O}_2, \mathrm{~g}\right)\right]\)
Where \(\bar{H}\left(\mathrm{H}_2 \mathrm{O}, l\right), \bar{H}\left(\mathrm{H}_2, \mathrm{~g}\right) \text { and } \bar{H}\left(\mathrm{O}_2, \mathrm{~g}\right)\) are the molar enthalpies of H2O(g) , H2(g) and O2(g) , respectively.
Relation between heat of reaction at constant volume [qv) and heat of reaction at constant pressure (qp)
If a reaction is carried out at a fixed pressure than the heat of the reaction, qp = change in enthalpy in the reaction, AH …[1] If the reaction is carried out at constant volume then the heat of the reaction, qv = change in internal energy in the reaction, AU
If the changes in internal energy and volume in a reaction occurring at constant pressure are AUp and AV, respectively, then according to the relation H = U + PV
⇒ \(\Delta H=\Delta U_P+\Delta(P V)=\Delta U_P+P \Delta V\)
[As pressure (P) is constant, Δ(PV) = PΔV
∴ \(q_P=\Delta H=\Delta U_P+P \Delta V\)
since qp=ΔH
Subtracting equation [2] from [3], we obtain
∴ \(q_P-q_V=\Delta U_P+P \Delta V-\Delta U=\left(\Delta U_P-\Delta U\right)+P \Delta V\)
At a particular temperature, the difference between AUp (change in internal energy at constant pressure) and AU (change in internal energy at constant volume) is very small. Therefore, it is considered that \(\Delta U_P \approx \Delta U.\)
In the case of solids and liquids: For reactions involving only solids and liquids, the change in volume (AV) of the reaction system is negligibly small. So, in such reactions, the heat of reaction at constant pressure (qp or AH) becomes equal to the heat of reaction at constant volume (qV or ΔU).
In case of gases: For a reaction involving gaseous substances [Example \(A(g)+B(g) \rightarrow C(g) \text { or } A(g)+B(g) \rightarrow C(I)\), or \(A(s) \rightarrow B(s)+C(g)\), etc.] the change in volume (A V) of the reaction system may be sufficiently high. We can determine the value of PA V as illustrated below— Let us consider a gaseous reaction that occurs at constant pressure (P) and temperature (T).
Suppose, n1 and V1 are the total number of moles and the total volume of the reactant gases, respectively, and n2 and IA, are the Here, total number of moles and the total volume of the product gases respectively. If the gases are assumed to behave ideally, then for gaseous reactants PV1 = n1RT., and gaseous products PV2 = n2RT.
∴ \(P\left(V_2-V_1\right)=\left(n_2-n_1\right) R T \text { or, } P \Delta V=\Delta n R T\)
Substituting A nRT for PAV into equation [4], we obtain \(q_p-q_V=\Delta n R T \text { and } \Delta H-\Delta U=\Delta n R T\)
∴ \(\left[\boldsymbol{q}_p=\boldsymbol{q}_V+\Delta n \| T\right] \text { and }[\Delta I=\Delta U+\Delta n \ RT] \cdot \cdot \cdot \cdot \cdot [5]\)
Using equation (5), ΔH (or qp) can be calculated from the known value of U (or Δv), and ΔU (or qV) can be calculated from the known value of AII (or ΔyU). If
Δn=0; ΔH=ΔU;Δn>0,ΔH>ΔU;Δn>0,ΔH<ΔU.

Standard State And Standard Reaction Enthalpy
Enthalpy change in a reaction depends on the conditions of temperature, pressure, and physical states of the reactants and products. To compare enthalpies of different reactions, we define a set of conditions called standard state, at which the values of AH for different reactions are calculated.
Standard State: The standard state ofa substance is defined as the most stable and purest state of that substance at the temperature of interest and atm pressure. In the definition of the standard state, temperature is not specified like pressure (1 atm). If the temperature is not mentioned, then 25 °C (298.15 K) is taken as a reference temperature. However, this does not mean 25 °C is the standard temperature. A pure substance can have different standard states depending on the temperature of interest, but in each of these states, the pressure is always 1 atm.
Examples: The standard state of liquid water at a particular temperature means H2O(Z) at that temperature and 1 atm pressure. The standard state of ice at a particular temperature means pure H20(s) at that temperature and atm pressure. The standard state of liquid ethanol at 25 °C means C2H5OH(l) at 25 °C and 1 atm pressure.
Standard enthalpy of reaction: The standard enthalpy change of a reaction is defined as the enthalpy change that occurs when the stoichiometric number of moles of reactants, indicated by the balanced chemical equation of the reaction, is completely converted into products at a particular temperature and l atm (i.e., at the standard state).
The standard enthalpy of reaction at a particular temperature ( T K) is denoted by \(\Delta H_T^0\). The superscript ‘0’ indicates the standard state, and the subscript T indicates the temperature in the Kelvin scale.
Exaplantion: \(\mathrm{C}_3 \mathrm{H}_8(\mathrm{~g})+5 \mathrm{O}_2(\mathrm{~g}) \rightarrow 3 \mathrm{CO}_2(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}(l)\)
For this reaction \(\Delta H_{2 \mathrm{qg}}^0=-2220 \mathrm{~kJ}\) indicating that at. 298 K temperature and atm pressure, if lmol of propane (C3Hs) and mol of O2 react completely to form 3 mol of CO, and 4 mol of water at the same temperature and pressure (i.e, 29S K and atm respectively), then 2220 kj of heat will be evolved. Alternatively, it can be said that at 298K temperature and 1 atm pressure, the change in enthalpy for the following reaction is =-2220 kj.
⇒ \(\begin{array}{|c|}
\hline 1 \underbrace{\text { mol pure } \mathrm{C}_3 \mathrm{H}_8(\mathrm{~g})+5 \mathrm{~mol} \text { pure } \mathrm{O}_2(\mathrm{~g})} \\
\hline 298 \mathrm{~K} \text { and } 1 \mathrm{~atm} \\
\hline \downarrow-2220 \mathrm{~kJ} \text { (heat) } \\
\hline \underbrace{3 \mathrm{~mol} \text { pure } \mathrm{CO}_2(\mathrm{~g})+4 \mathrm{~mol} \text { pure } \mathrm{H}_2 \mathrm{O}(l)} \\
\hline 298 \mathrm{~K} \text { and } 1 \mathrm{~atm} \\
\hline
\end{array}\)
Factors affecting the reaction enthalpy
Physical states of reactants and products: During the change of physical states of a substance (like solid, solid→ liquid, liquid→ vapor, etc.) heat is either absorbed or evolved. Thus, the value of the heat of the reaction or enthalpy of the reaction depends upon the physical states ofthe reactants and products. For example, in the following two reactions, due to the different physical states ofthe products, the values ofthe heat of the reaction are different.
⇒ \(\begin{aligned}
& 2 \mathrm{H}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l) ; \Delta H=-571.6 \mathrm{~kJ} \\
& 2 \mathrm{H}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(g) ; \Delta H=-483.6 \mathrm{~kJ}
\end{aligned}\)
Condition under which the reaction is conducted: At a particular temperature, a chemical reaction can be conducted either at constant pressure or at constant volume. If a reaction occurs at constant pressure, then the heat of reaction (qp) \(=H_{\text {product }}-H_{\text {reactant }}=\Delta H\)
occurs at constant volume, then the heat of reaction (<7v’) — The relation between AH and AH is AH = AH + PAV The quantity PAV indicates pressure-volume work. So, the difference between AH and AH is equal to the pressure-volume work involved during the reaction.
If the volume of the reacting system remains fixed (AV = 0), then pressure-volume work = 0 and ΔH = AH.
If the volume ofthe reacting system changes (ΔV=0), then, the pressure-volume work, PΔV≠0 and ΔH≠ΔU.
Allotropic forms of the reacting elements: As the different allotropic forms have different enthalpies, the value of reaction enthalpy depends upon the allotropic forms of the reactants. For example, the enthalpies of the reaction are different for the oxidation of graphite and diamond (two allotropic forms of carbon) \(\begin{aligned}
& \mathrm{C}(\text { graphitè, } s)+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \Delta H=-393.5 \mathrm{~kJ} \\
& \mathrm{C}(\text { diamond, } s)+\mathrm{O}_2(g) \rightarrow \mathrm{CO}_2(g) ; \Delta H=-395.4 \mathrm{~kJ}
\end{aligned}\)
Amount of the reactants: As enthalpy is an extensive property, the magnitude of enthalpy change in a reaction (AH) is proportional to the amount of reactants undergoing the reaction. For example, in the reactions given below, the magnitude of AH (reaction enthalpy) for the reaction [2] is twice that for the reaction [1], This is because the total number of moles of reactants in the reaction [2] is twice as many as that in the reaction [1]
⇒ \(\begin{array}{ll}
\mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{H}_2 \mathrm{O}(l), \Delta H=-285.8 \mathrm{~kJ} & \cdots[1] \\
2 \mathrm{H}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l), \Delta H=-571.6 \mathrm{~kJ} & \cdots[2]
\end{array}\)
Temperature: Temperature has a significant effect on the reaction enthalpy of a reaction. The extent of temperature dependence of the reaction enthalpy depends on the nature of the reaction.
Numerical Examples
Question 1. The value of AH for the given reaction at 298K is — 282.85 kj. mol-1 . Calculate the change in internal energy: \(\mathrm{CO}(g)+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})\)
Answer: We know, AH = AH + AnRT where, An = (total number of moles of the gaseous products) — (total number of moles of the gaseous reactants)
For the given reaction, ,\(\Delta n=1-\left(1+\frac{1}{2}\right)=-\frac{1}{2}\)
As per given data, AH = -282.85 kj-mol-1 & T = 298 K
∴ \(-282.85=\Delta U+\left[\left(-\frac{1}{2}\right) \times 8.314 \times 10^{-3} \times 298\right]\)
∴ \(\Delta U=-281.61 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Question 2. The bond energy of a diatomic molecule IN given as the change in internal energy due to dissociation of that molecule. Calculate the bond energy of O2. Given: \(\mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{O}(\mathrm{g}) ; \Delta H=498.3 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}, T=298 \mathrm{~K}\)
Answer: In the dissociation reaction of O2 molecules, ΔΔn = 2-1 = 1.
We know, ΔH = A U + ΔnRT
As given, ΔH = 498.3 kj.mol-1 , T = 298 K
∴ \(498.3=\Delta U+\left(1 \times 8.314 \times 10^{-3} \times 298\right)\)
or, ΔU= 495.8J
∴ Bond energy of O2 molecule = 495.8 kj.mol-1
Question 3. Calculate the values of ΔH and ΔH in the vaporization of 90 g of water at 100°C and 1 atm pressure. The latent heat of vaporization of water at the same temperature and pressure = 540 cal g-1.
Answer: \(90 \mathrm{~g} \text { of water }=\frac{90}{18}=5 \mathrm{~mol} \text { of water. }\)
Vaporisation of water: \(\mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{H}_2 \mathrm{O}(\mathrm{g})\)
Therefore, in the vaporization of1 mol of water, An = +1.
Hence, for the vaporization of 5 mol water, Δn = +5.
So, the amount of heat required to vaporize 90 g (5 mol) of water =540 x 90 = 48600 cal.
As the vaporization process occurs at constant pressure (1 atm), the heat absorbed = enthalpy change.
∴ The change in enthalpy in the vaporization of 90g of water, ΔH = 48600 cal
∴ The change in internal energy in the vaporization of 90gofwater, ΔH = ΔH-AnRT
= 48600- 5 X 1.987 X (273 + 100) = 44894.24 cal .
Question 4. Assuming the reactant and product gases obey its ideal gas law, calculate the change in internal energy (AE) at 27°C for the given reaction:
Answer: \(\mathrm{C}_2 \mathrm{H}_4(\mathrm{~g})+3 \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(l) ;\)
In tlui rimdlon, Δn= 2-1=1.
We know, ΔH=ΔU+ΔuRT
As given, ΔH = 498.3 kj. mol-1 , T = 298 K
∴ 498.3 = AH + (1 X 8.314 x 10-3x 298) or, ΔH = 495.8 kj
The bond energy of the O2 molecule = 495.8 kj.mol-1.
In the reaction , Δn=2- (1 + 3)= -2
we know ΔH+ ΔnRT
Or-337= ΔU-2×1.987×10-3×300 or, ΔU =-335.8kcal
Various Types Of Heat Of Reaction Or Enthalpy Of Reaction
Heat or enthalpy of the formation of a compound
Heat or enthalpy of the formation of a compound Definiton: At a given temperature and pressure, the enthalpy of formation of a compound is defined as the enthalpy change for the reaction in which one mole of the compound is formed from its constituent elements
The enthalpy of formation of a compound is denoted by the symbol ΔHf, where subscript ‘f’ stands for formation. To compare the enthalpy of formation for different compounds, the enthalpy of formation at a standard state is calculated for the compounds. The enthalpy of formation for any compound in the standard state is known as the standard enthalpy of formation of that compound.
Standard enthalpy of formation: The standard enthalpy of formation of a compound is defined as the change in enthalpy for the reaction in which one mole of the compound in its standard state (i.e., at a particular temperature and 1 atm pressure) is produced from its constituent elements in their standard states
The standard enthalpy of formation for any compound is denoted by \(\Delta H_f^0\), where subscript ‘f’ stands for formation and superscript ‘0’ indicates the standard state. The value of \(\Delta H_f^0\) may be positive or negative
In the thermochemical equation representing the formation reaction of a compound, one mole of the compound is formed. For this reason, the unit of ΔH0f is expressed in kJ.mol-1 (or J.moI-1) or kcal.mol-1 (or cal. mol-1 ).
Example: \(\mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H_f^0=-285.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Generally, the standard enthalpy of formation \(\Delta H_f^0\) of a compound is quoted at 25°C (or 298 K). If temperature is not mentioned, then 25°C temperature is to be considered. At a given temperature, the standard state of an element or a compound is the most stable and purest state of that element or compound at 1 atm pressure and at that temperature. The most stable forms of some elements at 25°C and 1 atm are given below.
- Dihydrogen: H2(g)
- Dioxygen: O2(g)
- Dinitrogen: N2(g)
- Sodium: Na(s)
- Chlorine molecule: Cl2(g)
- Bromine molecule: Br2(Z)
- Iodine molecule: I2(s)
- Carbon: C (graphite, s)
- Sulfur: S (rhombic, s), etc.
The constituent elements of a compound must be present in their standard states in the equation representing the formation reaction of the compound.
At 25° nC, the standard enthalpy of formation of water =-205.8kI.mol-1: At 25 °C and 1 atm, the heat evolved due to the formation of 1 mol of H2O(/) from the reaction between 1 mol of H2(g) and 1/2 mol of O2(g) is 285.8 kj. Alternatively, it can be said that the change in enthalpy of the given reaction at 25 °C and 1 atm is -285.8 kj
⇒ \(\underbrace{\mathrm{H}_2(g)+5^{\circ} \mathrm{C} \& 1 \mathrm{~atm}}_{25^{\circ} \mathrm{C} \& 1 \mathrm{~atm}} \underbrace{\frac{1}{2} \mathrm{O}_2(\mathrm{~g})} \rightarrow \Delta H_f^0=-285.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
At 25°C, The standard enthalpy of formation of nitric acids \([\mathrm{NO}(g)]=+90.3 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\) At 25°C and 1 atm, the heat evolved due to the formation of 1 mol of NO(g) from the reaction between 1/2 mol of N2(g) and 1/2 mol of 02(g) is 90.3kJ. mol-1 . Alternatively, it is said that the change in enthalpy of the following reaction at 25°C and I atm is +90.3 kj.
⇒ \(\underbrace{\frac{1}{2} \mathrm{~N}_2(g)+\frac{1}{2} \mathrm{O}_2(g)}_{25^{\circ} \mathrm{C} \& 1 \mathrm{~atm}} \rightarrow \underbrace{\mathrm{NO}(g)}_{25^{\circ} \mathrm{C} \& 1 \mathrm{~atm}} ; \Delta H_f^0=+90.3 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Important points about standard enthalpy of formation:
The standard enthalpy of formation of a compound is not always equal to the value of AH° indicated in the thermochemical equation representing the reaction in which the compound in its standard state is formed from its stable constituent elements in their standard states.
Explanation: At 25°C and 1 atm, two chemical equations for the formation of H2O(Z) from their stable constituent elements H2(g) and O2(g) are given below.
⇒ \(\begin{aligned}
& \mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0=-285.8 \mathrm{~kJ} \\
& 2 \mathrm{H}_2(g)+\mathrm{O}_2(g) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0=-571.6 \mathrm{~kJ}
\end{aligned}\)
The AH° in equation [1] indicates the standard enthalpy of the formation of H2O(Z) because, in this reaction, one mole of H2O(l) is formed from its stable constituent elements H2(g) and O2(g). On the other hand, in equation [2], two moles of H2O(Z) are formed from the stable constituent elements H2(g) and O2(g).
Thus, according to the definition of the enthalpy of formation, the value of AH° from equation [2] does not indicate the standard enthalpy of formation of H2O(Z).
The standard enthalpies of formation of all elements in their standard states are conventionally taken as zero. In the case of an element having different allotropes, the standard enthalpy of formation of the most stable allotropes form in the standard state is considered zero.
Explanation: The most stable forms of hydrogen, oxygen, nitrogen, sodium, etc. at 25 °C and 1 atm are H2(g), O2(g), N2(g), Na(s), etc. Thus, the standard enthalpies of formation of H2(g), O2(g), N2(g), Na(s), etc. are zero.
According to the definition, at 25°C and 1at, the standard enthalpy of formation of hydrogen is the same as the standard enthalpy change ofthe following reaction \(\mathrm{H}_2\left(\mathrm{~g}, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm}\right) \rightarrow \mathrm{H}_2\left(\mathrm{~g}, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm}\right)\)
No change has occurred in this process. Hence AH0 = 0. Therefore, at 25°C, for hydrogen \(\Delta H_f^0=0\). For the same reason \(\Delta H_f^0\) is zero for N2(g), O2(g), etc. Diamond and graphite are the two allotropic forms of solid carbon. Between these two forms, graphite is the most stable form at 25°C and 1 atm. Thus the standard enthalpy of formation \(\Delta H_f^0\) of solid graphite [C (graphite, s)] at 25°C is zero. But, for diamond [C (diamond, s)], at 25°C \(\Delta H_f^0 \neq 0\) At 25°C, the standard enthalpy of formation of diamond \(\Delta H_f^0 \neq 0\) is equal to the standard enthalpy of reaction for the following change \(\left.\mathrm{C} \text { (graphite, } s, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm}\right) \longrightarrow \mathrm{C} \text { (diamond, } s, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm} \text { ) }\) In this transformaton, AH° = +1.9kJ. Hence, the standard enthalpy of formation of diamond, \(\Delta H_f^0\) =+1.9kJ.
The most stable form of sulfur at 25°C and 1 atm is solid rhombic sulfur [S (rhombic,s)]. Thus, at 25°C, \(\Delta H_f^0\) for rhombic sulphur is zero.
Determination of the standard enthalpy of reaction (AH0) from the value of the standard enthalpy of formation \(\left(\Delta H_f^0\right)\): At a particular temperature, the standard reaction enthalpy ofa reaction (AH0) = total enthalpy of formation of the products – total enthalpy of formation of the reactants at the same temperature.
∴ \(\Delta H^0=\sum n_i \Delta H_{f, i}^0-\sum n_j \Delta H_{f, j}^0\)
Where \(\Delta H_{f, i}^0\) and \(\Delta H_{f, i}^0\) are the standard enthalpies of formation of i -th product and j -th reactant, respectively, and nt and nj are the number of moles of i -th product and 7 -th reactant respectively in a balanced chemical equation.


Heat of combustion or enthalpy of combustion
Heat of combustion or enthalpy of combustion Definition: At a Particular temperature and pressure, the change in enthalpy associated with the complete combustion of1 mol of a substance above oxygen is termed the enthalpy of combustion of the substance at that temperature and pressure.
Generally, the enthalpy of combustion is denoted by AHc.
Standard enthalpy Of combustion: The standard enthalpy of combustion of a substance is defined as the enthalpy change for the reaction in which one mole of the substance is completely burnt in oxygen when all the reactants and products are in their standard states.
The standard enthalpy of combustion is denoted by A. As the combustion reactions are exothermic, the values ofthe enthalpy of combustion are always negative.
Examples: The thermochemical equations of the combustion of some substances (elements or compounds) at 25°C and 1 atm pressure are given below.
Combustion of graphite:
⇒ \(\mathrm{C}(\mathrm{s}, \text { graphite })+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \Delta H_c^0=-393.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Equation: Also indicates the formation reaction of CO2(g). So, the standard enthalpy of combustion of graphite is the same as the standard enthalpy of formation of CO2(g).
Combustion of methane:
⇒\(\begin{aligned}
& \mathrm{CH}_4(\mathrm{~g})+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(l) ; \\
& \Delta H_c^0=-890 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
Combustion of sucrose:
⇒ \(\begin{aligned}
\mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}(\mathrm{~s})+12 \mathrm{O}_2(\mathrm{~g}) \rightarrow 12 \mathrm{CO}_2(\mathrm{~g})+11 \mathrm{H}_2 \mathrm{O}(l) ; \\
\Delta H_c^0=-5644 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
At 25°c, the standard enthalpy cf of C4H10(g) is -2878 kj. mol-1: This means that at 25°C and 1 atm pressure when 1 mol of C4H10(g) is completely burnt in the presence of oxygen according to the following reaction, 2878 kj of heat is released.
⇒ \(\begin{aligned}
\mathrm{C}_4 \mathrm{H}_{10}\left(\mathrm{~g}, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm}\right)+\frac{13}{2} \mathrm{O}_2\left(\mathrm{~g}, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm}\right) \\
4 \mathrm{CO}_2\left(\mathrm{~g}, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm}\right)+5 \mathrm{H}_2 \mathrm{O}\left(\mathrm{l}, 25^{\circ} \mathrm{C}, 1 \mathrm{~atm}\right)
\end{aligned}\)

Applications of the enthalpy of combustion:
Calculation of the enthalpy of formation: The enthalpy of formation of many substances cannot be directly measured; for example, CH4 cannot be directly prepared from its constituent elements by the reaction: \(\mathrm{C} \text { (graphite, } s)+2 \mathrm{H}_2(\mathrm{~g}) \rightarrow \mathrm{CH}_4(\mathrm{~g}) \text {. }\)
Similarly, glucose is not formed by the reaction: \(\mathrm{C} \text { (graphite, } s)+6 \mathrm{H}_2(\mathrm{~g})+3 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_6(\mathrm{~s})\) The determination of heat (or enthalpy) of formation of such types of compounds is possible from the known values ofthe heats of combustion different substances.
Example: Calculate the standard enthalpy of formation of CH4(g). Given: The standard heat of combustion of CH4 =- 890 kj.mol-1 and the standard heat of formation of H2O(J) and CO2(g) -285.8 kj.mol-1 and -393.5 kj. mol-1, respectively.
Solution: The thermochemical equation for the combustion of \(\mathrm{CH}_4 \text { is } \mathrm{CH}_4(g)+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(l) \text {, }\) \(\Delta H_c^0=-890 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[1]\)
The formation reactions for H2O(1) and CO2(g) are \(\mathrm{H}_2(\mathrm{~g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{H}_2 \mathrm{O}(l),\) \(\Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(l)\right]=-285.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \quad \cdots[2]\)
⇒ \(\begin{aligned}
& \mathrm{C} \text { (graphite, } s)+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) \\
& \Delta H_f^0\left[\mathrm{CO}_2(\mathrm{~g})\right]=-393.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \quad \cdots[3]
\end{aligned}\)
The formation reactions for CH4(g) is \(\mathrm{C}(\text { graphite, } s)+2 \mathrm{H}_2(\mathrm{~g}) \rightarrow \mathrm{CH}_4(\mathrm{~g})\)
To obtain equation [4], we can write equation [3] + 2 x equation [2]- equation [1]. This given \(\Delta H^0=\Delta H_f^0\left[\mathrm{CO}_2(g)\right]+2 \times\left(\Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(l)\right]\right)-\Delta H_c^0\)
=[- 393.5 + 2(-285.8)- (-890)] kj = -75.1kJ ….[4]
Thus, the standard heat of formation of CH4(g) =-75.1 kj-mol-1.
Fuel efficiency: Based on the data of heat of combustion of various fuels (For example coal, kerosene, petrol, etc.), the efficiency of each fuel with the same amount, the one that liberates a larger amount of heat on considered to be a better fuel.
So, the heat of combustion is very important regarding the selection ofa fuel. The amount of heat produced due to the combustion of l g of a fuel is known as the calorific value of that fuel.
Determination of calorific value of foods: Energy is obtained by the oxidation of carbohydrates and fats present in the food that we consume. These carbohydrates and fats are oxidized into CO4 and H2O along with the liberation of heat This heat of combustion maintains our body temperature and the strength ofthe muscle.
⇒ \(\begin{aligned}
\mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_6(\mathrm{~s})+6 \mathrm{O}_2(\mathrm{~g}) \rightarrow 6 \mathrm{CO}_2(\mathrm{~g})+6 \mathrm{H}_2 \mathrm{O}(l) ; \\
\Delta H^0=-2808 \mathrm{~kJ}
\end{aligned}\)
The amount of heat liberated in the combustion (or oxidation) of 1 g of an edible substance (food) is known as the calorific value of that food. Based on this calorific value, we can easily prepare a balanced food chart. As the calorific values of fats are high, thus in cold countries, foods containing fats are consumed in larger quantities.

Numerical Examples
Question 1. Calculate the standard enthalpy of reaction at 25 temperature for the following reaction: \(\mathrm{C}_6 \mathrm{H}_6(l)+\frac{15}{2} \mathrm{O}_2(g) \rightarrow 6 \mathrm{CO}_2(g)+3 \mathrm{H}_2 \mathrm{O}(l)\) Given: The standard enthalpy of formation of C6H6(f), CO2(l) and H2O(l) -393.5 kj.moH and -285.8 kj. mol-1 respectively.
Answer: \(\mathrm{C}_6 \mathrm{H}_6(l)+\frac{15}{2} \mathrm{O}_2(g) \rightarrow 6 \mathrm{CO}_2(g)+3 \mathrm{H}_2 \mathrm{O}(l)\)
The standard heat of reaction for this reaction is
⇒ \(\begin{aligned}
\Delta H^0=6 \times \Delta H_f^0\left[\mathrm{CO}_2(\mathrm{~g})\right] & +3 \times \Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(l)\right] \\
& -1 \times \Delta H_f^0\left[\mathrm{C}_6 \mathrm{H}_6(l)\right]-\frac{15}{2} \Delta H_f^0\left[\mathrm{O}_2(g)\right]
\end{aligned}\)
⇒ \(\begin{aligned}
& =6 \times(-393.5)+3 \times(-285.8)-1 \times(49.0)-\frac{15}{2} \times 0 \\
& =-3267.4 \mathrm{~kJ}\left[\Delta H_f^0\left[\mathrm{O}_2(g)\right]=0\right]
\end{aligned}\)
Question 2. Calculate standard enthalpy of reaction at 25°C for the reaction: \(\mathrm{CCl}_4(g)+2 \mathrm{H}_2 \mathrm{O}(g) \rightarrow \mathrm{CO}_2(g)+4 \mathrm{HCl}(g) .\) Given: The standard heat of formation of CCl4(g) , H2O(g), CO2(g) and HCl(g) are -25.5, -57.8, -94.1 and -22.1 kcal-mol-1 .
Answer: The standard heat of reaction for the given reaction,
⇒ \(\begin{aligned}
\Delta H^0=\Delta H_f^0[ & \left.\mathrm{CO}_2(g)\right]+4 \times \Delta H_f^0[\mathrm{HCl}(g)] \\
& -\Delta H_f^0\left[\mathrm{CCl}_4(g)\right]-2 \times \Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(g)\right] \\
= & -94.1+4 \times(-22.1)-(-25.5)-2 \times(-57.8) \\
= & -41.4 \mathrm{kcal}
\end{aligned}\)
⇒ \(\begin{gathered}
\mathrm{CH}_4(\mathrm{~g})+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(\mathrm{l}) ; \\
\Delta H^0=-890 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \\
\mathrm{C}(\mathrm{s})+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \Delta H^0=-393.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{gathered}\)
⇒ \(\mathrm{C}(\mathrm{s})+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \Delta H^0=-393.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Reversing equation [1], we get,
⇒ \(\begin{aligned}
& 3 \mathrm{CO}_2(g)+2 \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{C}_2 \mathrm{H}_3 \mathrm{CO}_2 \mathrm{H}(l)+3 \mathrm{O}_2(g) \\
& \Delta H^0=+1368 \mathrm{~kJ} \cdots[4]
\end{aligned}\)
Dividing each of equations [2] and [3] by 2, we obtain
⇒ \(\begin{aligned}
& \mathrm{C}_2 \mathrm{H}_2(g)+\frac{5}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{CO}_2(\mathrm{~g})+\mathrm{H}_2 \mathrm{O}(l) ; \\
& \Delta H^0=-1300 \mathrm{~kJ} \cdots[5] \\
& \mathrm{CO}(\mathrm{g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \Delta H^0=-283 \mathrm{~kJ} \quad \cdots[6]
\end{aligned}\)
Adding equations [4], [5] and [6], we obtain, \(\begin{aligned}
& \mathrm{C}_2 \mathrm{H}_2(g)+\mathrm{CO}(g)+\mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{C}_2 \mathrm{H}_3 \mathrm{CO}_2 \mathrm{H}(l) ; \\
& \Delta H^0=[1368+(-1300)+(-283)] \mathrm{kJ}=-215 \mathrm{~kJ}
\end{aligned}\)
∴ ΔHO for the given reaction =-215 kJ.
Question 3. Calculate the standard enthalpy of formation of CgHg(l) at 25°C temperature using the given data
Answer: Reaction to the formation of C6H6(l):
⇒ \(\begin{aligned}
& 6 \mathrm{C}(s \text {, graphite })+3 \mathrm{H}_2(g) \rightarrow \mathrm{C}_6 \mathrm{H}_6(l) \\
& \text { Given: } \mathrm{C}_6 \mathrm{H}_6(l)+\frac{15}{2} \mathrm{O}_2(g) \rightarrow 6 \mathrm{CO}_2(g)+3 \mathrm{H}_2 \mathrm{O}(l) \text {; }
\end{aligned}\)
⇒ \(\begin{aligned}
\Delta H^0 & =-781 \mathrm{kcal} \\
\mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0 & =-68.32 \mathrm{kcal}
\end{aligned}\)
C(s, graphite) + O2(g)→ CO2(g); AH° = -94.04 kcal [3]
Reversing equation [1], we obtain
⇒ \(\begin{array}{r}
6 \mathrm{CO}_2(g)+3 \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{C}_6 \mathrm{H}_6(l)+\frac{15}{2} \mathrm{O}_2(\mathrm{~g}) ; \\
\Delta H^0=+781 \mathrm{kcal}
\end{array}\)
Multiplying equation [2] by 3 & equation [3] by 6, we get
⇒ \(3 \mathrm{H}_2(g)+\frac{3}{2} \mathrm{O}_2(g) \rightarrow 3 \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0=-204.96 \mathrm{kcal} \quad \cdots[5]\)
⇒ \(\begin{aligned}
& \text { and } 6 \mathrm{C}(s, \text { graphite })+6 \mathrm{O}_2(g) \rightarrow 6 \mathrm{CO}_2(\mathrm{~g}) ; \\
& \Delta H^0=-564.25 \mathrm{kcal}
\end{aligned}\)
Now adding equations [4], [5] and [6], we obtain
⇒ \(\begin{aligned}
& \mathrm{C}(s, \text { graphite })+3 \mathrm{H}_2(g) \rightarrow \mathrm{C}_6 \mathrm{H}_6(l) \\
& \Delta H^0=+781+(-204.96)+(-564.24)=+11.8 \mathrm{kcal} \cdots[7]
\end{aligned}\)
Equation [7] indicates a thermochemical equation for the formation of C6H6(Z) at the standard state. Therefore, the standard heat of formation of C6H6(Z) =+ 11.8 kcal.mol-1
Question 4. At 25°C temperature, the heat of combustion of sucrose, carbon, and hydrogen is -5644 kj. mol-1 , -393.5 kj.mol-1 & -285.8 kj – moM respectively. Determine the heat of the formation of sucrose at 25°C.
Answer: \(\begin{aligned}
\mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}(\mathrm{~s})+12 \mathrm{O}_2(\mathrm{~g}) \rightarrow 12 \mathrm{CO}_2(\mathrm{~g})+11 \mathrm{H}_2 \mathrm{O}(l) ; \\
\Delta H^0=-5644 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[1]
\end{aligned}\)
⇒ \(\begin{aligned}
\mathrm{C}(\text { graphite, } s)+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \\
\Delta H^0=-393.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[2] \\
\mathrm{H}_2(\mathrm{~g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{H}_2 \mathrm{O}(\mathrm{l}) ; \Delta H^0=-285.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[3]
\end{aligned}\)
The formation reaction of sucrose: \(12 \mathrm{C} \text { (graphite, } s)+11 \mathrm{H}_2(g)+\frac{11}{2} \mathrm{O}_2(g) \rightarrow \mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}(s)\)
Reversing Equation [1], we get
⇒ \(\begin{aligned}
& 12 \mathrm{CO}_2(\mathrm{~g})+11 \mathrm{H}_2 \mathrm{O}(\mathrm{g}) \rightarrow \mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}(\mathrm{~s})+12 \mathrm{O}_2(\mathrm{~g}) ; \\
& \Delta H^0=+5644 \mathrm{~kJ} \cdots[4]
\end{aligned}\)
Multiplying equation [2] by 12 and equation [3] by 11.
⇒ \(\begin{aligned}
12 \mathrm{C} \text { (graphite, } s)+12 \mathrm{O}_2(\mathrm{~g}) & \rightarrow 12 \mathrm{CO}_2(\mathrm{~g}) \\
\Delta H^0 & =12 \times(-393.5)=-4722 \mathrm{~kJ} \cdots[5]
\end{aligned}\)
⇒ \(\begin{aligned}
11 \mathrm{H}_2(g)+\frac{11}{2} \mathrm{O}_2(g) & \rightarrow 11 \mathrm{H}_2 \mathrm{O}(l) ; \\
\Delta H^0 & =11 \times(-285.8)=-3143.8 \mathrm{~kJ} \cdots[6]
\end{aligned}\)
By adding equations [4], [5] and [6] and their corresponding AH° values, we obtain
⇒ \(12 \mathrm{C}(s, \text { graphite })+11 \mathrm{H}_2(g)+\frac{11}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}(s)\)
∴ \(\Delta H^0=+5644+(-4722)+(-3143.8)=-2221.8 \mathrm{~kJ} \cdots[7]\)
Equation [7] indicates, the thermochemical equation for the formation of C12H22Ou(s) at standard conditions. Therefore, the standard enthalpy of formation of \(\mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}(\mathrm{~s})=-2221.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Question 5. At 25°C temperature, the standard heat of formation of CH4(g), CO2(g) and H2O(g) are -74.8kJ.mol-1 , -393.5kJ.mol-1 & -241.6kJ.mol-1 respectively. How much heat will be evolved during combustion of lm3 CH4(g) at 25°C temperature and 1 atm pressure? Consider CH4(g) behaves like an ideal gas
Answer: Combustion reaction of CH4(g) is given by: \(\mathrm{CH}_4(g)+2 \mathrm{O}_2(g) \rightarrow \mathrm{CO}_2(g)+2 \mathrm{H}_2 \mathrm{O}(l)\)
∴ Standard heat of combustion of CH4(g) \(\begin{gathered}
\Delta H_c^0=\left[1 \times \Delta H_f^0\left[\mathrm{CO}_2(g)\right]+2 \times \Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(l)\right]\right] \\
-\left[1 \times \Delta H_f^0\left[\mathrm{CH}_4(\mathrm{~g})\right]+2 \times \Delta H_f^0\left[\mathrm{O}_2(\mathrm{~g})\right]\right] \\
=-393.5+2 \times(-241.6)-1 \times(-74.8)+2 \times 0=-801.9 \mathrm{~kJ}
\end{gathered}\)
since In standard state for stable and pure element \(\left.\Delta H_f^0=0 \text {; thus, } \Delta H_f^0\left[\mathrm{O}_2(g)\right]=0\right)\)
lm3 CH4(g) = 103L CH4(g)
Number of moles of 103L CH4(g) at 25°C and 1 atm pressure \(n=\frac{P V}{R T}=\frac{1 \times 10^3}{0.0821 \times 298}=40.87\) \(\text { pressure, } n=\frac{P V}{R T}=\frac{1 \times 10^3}{0.0821 \times 298}=40.87\)
∴ Heat evolved in combustion = 40.87 x 801.9 = 32773.6 kj
Question 6. Calculate the value of enthalpy of combustion of cyclopropane at 25°C and 1 atm pressure. Given: Standard enthalpy of formation of CO2(g), H2O(I) & propene (g) at 25°C are -393.5 kj-mol-, -285.8 kj.mol-1 & 20.4 kj.mol-1 respectively. Also, the standard enthalpy change for isomerization reaction: Cyclopropane(g)y=yPropene(g) is -33.0kJ.mol-1.
Answer: Given: \(\begin{aligned}
\mathrm{C}(\text { graphite, } s)+\mathrm{O}_2(\mathrm{~g}) & \rightarrow \mathrm{CO}_2(\mathrm{~g}) \\
\Delta H_f^0 & =-393.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[1]
\end{aligned}\)
⇒ \(\mathrm{H}_2(\mathrm{~g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H_f^0=-285.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[2]\)
⇒ \(\begin{aligned}
&3 \mathrm{C} \text { (graphite, } s)+3 \mathrm{H}_2(\mathrm{~g}) \rightarrow \mathrm{C}_3 \mathrm{H}_6(\mathrm{~g}) \text { (propene); } \\
& \Delta H_f^0=+20.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \ldots[3]
\end{aligned}\)
Reversing equation [3], we get,
⇒ \(\begin{aligned}
& \left.\mathrm{C}_3 \mathrm{H}_6 \text { (propene, } g\right) \rightarrow 3 \mathrm{C}(\text { graphite, } s)+3 \mathrm{H}_2(g) ; \\
& \Delta H_f^0=-20.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[5]
\end{aligned}\) \begin{aligned}
& \left.\mathrm{C}_3 \mathrm{H}_6 \text { (propene, } g\right) \rightarrow 3 \mathrm{C}(\text { graphite, } s)+3 \mathrm{H}_2(g) ; \\
& \Delta H_f^0=-20.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \cdots[5]
\end{aligned}
Multiplying equations [1] and [2] by 3, we get,
⇒ \(\begin{aligned}
& 3 \mathrm{C} \text { (graphite, } s)+3 \mathrm{O}_2(\mathrm{~g}) \rightarrow 3 \mathrm{CO}_2(\mathrm{~g}) \text {; } \\
& \Delta H^0=-1180.5 \mathrm{~kJ} \cdots[6] \\
& 3 \mathrm{H}_2(g)+\frac{3}{2} \mathrm{O}_2(g) \rightarrow 3 \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0=-857.4 \mathrm{~kJ} \\
&
\end{aligned}\)
⇒ \(3 \mathrm{H}_2(\mathrm{~g})+\frac{3}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow 3 \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0=-857.4 \mathrm{~kJ}\)
Adding equations [4], [5], [6] and [7] we obtain,
⇒ \(\begin{gathered}
\left.\mathrm{C}_3 \mathrm{H}_6 \text { (cyclopropane, } g\right)+\frac{9}{2} \mathrm{O}_2(g) \rightarrow 3 \mathrm{CO}_2(g)+3 \mathrm{H}_2 \mathrm{O}(l) \\
\Delta H^0=-33+(-20.4)+(-1180.5)+(-857.4)=-2091.3 \mathrm{~kJ}
\end{gathered}\)
So, the standard enthalpy of combustion of lmol cyclopropane (g) at 25°C and 1 atm pressure = -2091.3 kj.mol-1.
Question 7. At 25°C, the heat evolved due to the complete combustion of 7.8g of C6H6(Z) is 326.4 kj. Calculate the heat evolved due to complete combustion of the same amount of CgHg(/) at the same temperature and constant pressure of 1 atm.
Answer: 7.8 \(\mathrm{~g} \mathrm{C}_6 \mathrm{H}_6=\frac{7.8}{78}=0.1 \mathrm{~mol}\left[ \mathrm{M}_{\mathrm{C}_6 \mathrm{H}_6}=78\right]\)
The combustion reaction of C6H6(Z):
⇒ \(\mathrm{C}_6 \mathrm{H}_6(l)+\frac{15}{2} \mathrm{O}_2(g) \rightarrow 6 \mathrm{CO}_2(g)+3 \mathrm{H}_2 \mathrm{O}(l) \cdot \cdot\cdot\cdot\cdot [1]\)
∴ \(\Delta n=6-\frac{15}{2}=-\frac{3}{2}\)
From the given information, the heat evolved in complete combustion of 7.8 g or 0.1 mol of C6H6(Z) at 25°C temperature and at constant volume =-326.4 kj.
Therefore, at 25°C and constant volume, the heat evolved due to the complete combustion of lmol C6H6(Z) =-30264 kj.
Now, the heat of reaction at constant volume = ΔU.
∴ ΔU = -3264 kj.mol-1
Again we know, ΔH = ΔU + ΔnRT
For reaction [1]
⇒ \(\Delta H=\left[-3264+\left(-\frac{3}{2}\right) \times\left(8.314 \times 10^{-3}\right)(298)\right]=3267.71 \mathrm{~kJ}\)
Therefore, at 25°C and 1 atm pressure, the heat evolved due to the complete combustion of1 mol C6H6(l) =-3267.71kj.
∴ At 25°C and 1 atm pressure, the heat evolved due to complete combustion of 7.8 g or 0.1 mol of C6H6(l) =- 326.771 kj.
Enthalpy change due to phase transition
A physical process in which a substance undergoes a change from one physical state to another, but its chemical identity remains the same is called phase transition.

Fusion, sublimation, and vaporization are endothermic processes. So, these processes involve the absorption of heat. On the other hand, solidification, condensation, and deposition are exothermic processes, involving the evolution of heat.
Enthalpy of fusion:
Enthalpy of fusion Definition: At constant pressure, the amount of heat required by one mole of a solid substance for its complete liquefaction at its melting point is called the enthalpy of fusion of that solid.
The enthalpy of fusion of a substance is the same as its molar latent heat of fusion. As fusion is an endothermic process, the value of enthalpy of fusion of a substance is positive.
Example: Heat absorbed during the transformation of 1 mole of ice into 1 mole of water at 0°C is 6.02 kj. Therefore, the enthalpy of fusion of ice at 0°C and 1 atm pressure,
Significance of the enthalpy of fusion: The enthalpy of fusion (AHfus) of a solid substance is a measure of the interparticle forces of attraction in the solid. The stronger the interparticle forces of attraction in a solid, the larger the value of its \(\Delta H_{\text {fus }}\). The interparticle forces ofattraction in ionic solids [like— NaCl, MgCl2, etc.] are stronger than those in the molecular solids [like ice, I2(s) etc.]. This is why the values of ionic solids are found to be greater than those of molecular solids.
For example, the values of \(\Delta H_{f u s}\) for Nacl, which is an ionic solid, is +28.8kJ. mol-1 where for .ice, which is a molecular solid it is +6.02kj. mol-1.
Solidification or freezing is the reverse process of fusion because during solidification liquid phase is transformed into the solid phase, while the solid phase is transformed into the liquid phase during the fusion process. Hence, at a particular temperature and pressure, the enthalpy of solidification ofa substance =(-) its enthalpy of fusion. For example, at 0°C temperature and 1 atm pressure, the enthalpy of fusion of ice = + 6.02 kj.mol-1 and the enthalpy of solidification of water = -6.02kj. mol-1
Enthalpy of vaporisation:
Enthalpy of vaporization Definition: At constant pressure, the amount of heat required for the complete vaporization of 1 mole of a liquid at its boiling point is termed the enthalpy of vaporization of that liquid.
The enthalpy of vaporization of a liquid is the same as its molar latent heat of vaporization. The enthalpy of vaporization is a positive quantity because vaporization is an endothermic process.
Example: At 100°C and 1 atm pressure, 40.4 kj of heat is required to completely convert 1 mol of water into 1 mol of water vapor. Thus, at 100°C and 1 atm, the enthalpy of vaporization of water \(\left(\Delta H_{\text {vap }}\right)=+40.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{H}_2 \mathrm{O}(\text { vap }), \Delta H_{v a p}=+40.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Significance of the enthalpy of vaporization: The value of the enthalpy of vaporization of a liquid is a measure of the intermolecular forces of attraction in the liquid. The stronger the intermolecular forces of attraction the larger the values of ΔHvap. For example, ΔHvap for water is +40.4 kj.mol-1, while that for benzene is +30.5 kj.mol-1, indicating that the intermolecular forces of attraction are stronger in water than in benzene. Thus, the amount of heat required to vaporize 1 mol of benzene is less than that required to vaporize 1 mol of water.
Condensation is die reverse process of vaporisation because during condensation vapour phase transforms into a liquid phase whereas during vaporisation liquid phase
transforms into a vapor phase. Thus, the magnitude of the enthalpy of vaporization \(\left(\Delta H_{\text {vap }}\right)\) and enthalpy of condensation \(\left(\Delta H_{\text {condensation }}\right)\) are the same but the opposite in sign ., i., e ΔH For example: At 100°C temperature and 1 atm pressure, for water ⇒ \(\begin{aligned}
& \Delta H_{\text {vap }}=+40.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \text { and } \\
& \Delta H_{\text {condensation }}=-40.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
Enthalpy of sublimation:
Enthalpy of sublimation Definition: Enthalpy of sublimation is defined as the amount of heat required by lmol of a solid substance for its complete vaporization at a given condition of temperature and pressure.
The value of the enthalpy of sublimation is always positive because sublimation is an endothermic process.
Example: At ordinary temperature and pressure, 62.3 kj heat is required to convert 1 mol of solid I2 to I2 vapor. Thus, at ordinary temperature and pressure, the enthalpy of sublimation of iodine \(\left(\Delta H_{s u b}\right)=+62.3 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
As the enthalpy is a state function, the change in enthalpy in the following two processes (1 and 2) will be the same.
⇒ \(\text { (I) Solid } \xrightarrow{\Delta H_{\text {sub }}} \text { Vapour, (II) Solid } \xrightarrow{\Delta H_{\text {fus }}} \text { Liquid } \xrightarrow{\Delta H_{\text {vap }}} \text { Vapour }\)
The total change in enthalpy \(=\Delta H_{\text {fus }}+\Delta H_{\text {vap }}\)

Numerical Examples
Question 1. Calculate The enthalpy change in the fusion of 100 g ice at 0°C temperature and 1 atm pressure The enthalpy change in the vaporization of 10 g water at 100°C temperature and 1 atm pressure. Given: Latent heat of ice at 0°C temperature and 1 atm pressure = 6.02 kJ.mol-1 and latent heat of vaporisation of water at 100°C temperature = 40.4 kj.mol-1
Answer: Latent heat of fusion of ice at 0°C temperature and 1 atm pressure = 6.02 kj. mol-1. Therefore, the enthalpy of fusion of 18g ice = 6.02 kj.
∴ Enthalpy of fusion 100 ice \(=\frac{6.02}{18} \times 100=33.44 \mathrm{~kJ}\)
∴ Change in enthalpy of fusion of 100 g ice = + 33.44 kj
Latent heat of vaporization of water at 100°C = 40.4 kj. mol-1 . Therefore, the enthalpy of vaporization of 18g water = 40.4 kj
∴ Enthalpy of vapourisation of 10g water \(=\frac{40.4}{18} \times 10=22.44 \mathrm{~kJ}\)
Question 2. The heat required to completely vaporize 7.8 g of benzene at 1 atm pressure and 80°C temperature (boiling point of benzene) is 3.08 kj. What is the value of the enthalpy of vaporization of benzene? What will be the change in enthalpy if 54.6 g of benzene vapor is condensed at 1 atm pressure and 80°C temperature?
Answer: \(7.8 \mathrm{~g} \text { benzene }=\frac{7.8}{78}=0.1 \mathrm{~mol} \text { benzene }\) since \(\mathrm{M}_{\mathrm{C}_6 \mathrm{H}_6}=78\)
∴ Change in enthalpy of fusion of100 g ice = + 33.44 kj
∴ Latent heat of vaporization of water at 100°C = 40.4 kj.mol-1. Therefore, the enthalpy of vaporization of18g water = 40.4 kj
∴ Enthalpy of vapourisation of 10g water \(=\frac{40.4}{18} \times 10=22.44 \mathrm{~kJ}\)
Question 2. The heat required to completely vaporize 7.8 g of benzene at 1 atm pressure and 80°C temperature (boiling point of benzene) is 3.08 kj. What is the value of the enthalpy of vaporization of benzene? What will be the change in enthalpy if 54.6 g of benzene vapor is condensed at 1 atm pressure and 80°C temperature?
Answer: \(7.8 \mathrm{~g} \text { benzene }=\frac{7.8}{78}=0.1 \mathrm{~mol} \text { benzene }\)
First part: As per the given data, the heat required for complete vaporization of 0.1 mol of benzene at 1 atm pressure and 80°C temperature is 3.08 kj. So, at the same temperature and pressure, the heat required for complete vaporization of1 mol of benzene is 30.8 kj.
Hence, according to the definition, enthalpy of vaporization of benzene at atm pressure and 80°C temperature = + 30.8 kj.
Second part: 54.6g of benzene \(=\frac{54.6}{78}=0.7 \mathrm{~mol}\)
Therefore, enthalpy of vaporization of 0.7 mol benzene at 1 atm pressure and \(80^{\circ} \mathrm{C}=\frac{0.7 \times(+3.08)}{0.1}=+21.56 \mathrm{~kJ}\)
1 atm pressure and 80°C \(=\frac{0.7 \times(+3.08)}{0.1}=+21.56 \mathrm{~kJ} \text {. }\)
Again, enthalpy of condensation =(-) enthalpy of vaporization. So, the change in enthalpy of condensation of 54.6g benzene at 1 atm pressure and 80°C temperature =-21.56 kf.
Heat or enthalpy of neutralization
Heat or enthalpy of neutralization Definition: The change in enthalpy that occurs when 1 gram equivalent of an acid is completely neutralized by 1 gram equivalent of a base or vice-versa in a dilute solution at a particular temperature is called the enthalpy (or heat) of neutralization.
The change in enthalpy that occurs when 1 mol of H+ ions reacts completely with lmol of OH- ions in a dilute solution to form 1 mol water at a particular temperature is known as the enthalpy (or heat) of neutralization.
The enthalpy of neutralization is denoted as AHN, where subscript TV ‘indicates ‘neutralization’.
Neutralization of strong acid and strong base: if both the acid and base are strong, then the value of heat of neutralization constant, is found to be almost and this value is -57.3 kj.
⇒ \(\begin{gathered}
\text { Examples: } \mathrm{HCl}(a q)+\mathrm{NaOH}(a q) \rightarrow \mathrm{NaCl}(a q)+\mathrm{H}_2 \mathrm{O}(l) ; \\
\Delta H_N=-57.3 \mathrm{~kJ} \\
\mathrm{HNO}_3(a q)+\mathrm{KOH}(a q) \rightarrow \mathrm{KNO}_3(a q)+\mathrm{H}_2 \mathrm{O}(l) ; \\
\Delta H_N=-57.3 \mathrm{~kJ}
\end{gathered}\)
Explanation: Consider the neutralization reaction involving HC1 and NaOH in a dilute aqueous solution. \(\mathrm{HCl}(a q)+\mathrm{NaOH}(a q) \rightarrow \mathrm{NaCl}(a q)+\mathrm{H}_2 \mathrm{O}(l)\) As HC1, NaOH and NaCl all are strong electrolytes, they completely dissociate in aqueous solution.
Hence, the above neutralization reaction can be written as:
⇒ \(\begin{array}{r}
\mathrm{H}^{+}(a q)+\mathrm{Cl}^{-}(a q)+\mathrm{Na}^{+}(a q)+\mathrm{OH}^{-}(a q) \longrightarrow \\
\mathrm{Na}^{+}(a q)+\mathrm{Cl}^{-}(a q)+\mathrm{H}_2 \mathrm{O}(l)
\end{array}\)
Cancelling the species that appear on both sides, we have
⇒ \(\mathrm{H}^{+}(a q)+\mathrm{OH}^{-}(a q) \rightarrow \mathrm{H}_2 \mathrm{O}(l)\)
Therefore, the neutralisation of strong acid and strong base is essentially, the combination of H+ ions and OH- ions to form water. This is the only reaction that occurs during the neutralization of strong acid and strong base.
This is why the heat of neutralization of all strong acids and strong bases is virtually constant and equal to -57.3 kj.mol-1
Neutralization of a strong acid by the weak base, weak acid by the strong base, and weak acid by weak base: if either acid or base is weak or both are weak, then the heat of neutralization value will be different in each case
Examples: \(\begin{aligned}
& \mathrm{CH}_3 \mathrm{COOH}(a q)+\mathrm{NaOH}(a q) \longrightarrow \\
& \quad \mathrm{CH}_3 \mathrm{COONa}(a q)+\mathrm{H}_2 \mathrm{O}(l) ; \Delta H_N=-55.9 \mathrm{~kJ}
\end{aligned}\)
⇒ \(\begin{aligned}
\mathrm{HCN}(a q)+\mathrm{NaOH}(a q) \rightarrow \mathrm{NaCN}(a q)+\mathrm{H}_2 \mathrm{O}(l) ; \\
\Delta H_N=-12.1 \mathrm{~kJ}
\end{aligned}\)
Explanation: A weak acid or weak base partially ionizes in aqueous solution. The ionization of a weak acid or base keeps on going during its neutralization process. Heat is absorbed in the ionization process. A part of the heat evolved during the reaction between H+ and OH- ions is utilized for the ionization of weak acid or weak base.
Hence, the value of heat of neutralization of strong acid and strong base is numerically greater than that associated with a neutralization process in which either acid or base or both are weak.
As the heats of ionization of different weak acids or weak bases are different, the value of the heat of neutralization of a weak acid by a strong base is different for a different weak acid. Similarly, the heat to be liberated in the neutralization of a weak base by a strong acid depends on the nature ofthe weak base.
In an acid-base neutralization, if the acid or base is weak, then the heat of neutralization = the heat of ionization of the weak acid (or weak base) + the heat of reaction for the reaction;\(\left[\mathrm{H}^{+}(a q)+\mathrm{OH}^{-}(a q) \rightarrow \mathrm{H}_2 \mathrm{O}(l)\right]\) Using this equation the heat of ionization of the weak acid or weak base can be determined.
Numerical Examples
Question 1. Determine the heat of neutralization for the following neutralization reactions: 100 mL of 0.2 (M) HCl solution is mixed with 200 mL of 0.15 (M) NaOH solution. 200 mL of 0.4 (M) H2SO4 solution is mixed with 300 mL of 0.2 (M) KOH solution.
Answer: The amount of HCI in 100 mL of 0.2 (M) HCI solution \(=\frac{0.2}{1000} \times 100=0.02\) mol and the amount of NaOH in 200 of 0.15 (M) NaOH solution \(=\frac{0.15}{1000} \times 200=0.03 \mathrm{~mol}.\) 0.02 mol of H+ ions are produced in the ionization of 0.02 (M) aqueous HCl solution & 0.03 mol OH ions are produced in the ionization of0.03(M) aqueous NaOH solution.
Therefore, if 100 mL of 0.2 (M) HC1 is mixed with 200 mL of 0.15 (M) NaOH, then 0.02 mol of H2O(/) will be formed from a die reaction between 0.02 mol H+ ions and 0.02 mol OH- ions. Thus heat evolved (i.e., heat of neutralisation) for tills reaction = 0.02 x 57.3 = 1.146 kJ.
⇒ \(\text { (2) } 200 \mathrm{~mL} \quad 0.4 \text { (M) } \mathrm{H}_2 \mathrm{SO}_4=\frac{0.4}{1000} \times 200=0.08 \mathrm{~mol}\)
⇒ \(\mathrm{H}_2 \mathrm{SO}_4 \& 300 \mathrm{~mL} 0.2(\mathrm{M}) \mathrm{KOH}=\frac{0.2}{1000} \times 300=0.06 \mathrm{~mol} \mathrm{KOH.}\)
The amount of H+ ions formed from the complete ionisation of O.OOmol of aqueous H2SO4 = 2 x 0.00 =0.16mol
since 2 mol H+ is formed from 1 mol of h2SO4] and that of OH- ions formed due to dissociation of 0.06 (m) KOH =0.06 mol since 2 mol H+ is formed from 1 mol of h2So4] and that of OH- ions formed due to dissociation of 0.06(M) KOH=0.06 mol
Therefore, if 200 mL of 0.4 (M) H2SO2 solution is mixed with 300 mL of 0.2 (M) KOH solution then effectively 0.06 mol H2O(Z) will be formed from the reaction between 0.06 mol OH- ions and the same amount of H+ ions.
Thus the amount of heat produced (i.e., the heat of neutralization) of this reaction = 0.06 x 57.3 = 3.438 kj.
Question 2. The heat of neutralization of acetic acid and NaOH is 55.9 kj. If the heat of neutralization of all strong acids and strong bases is 57.3 kj, then calculate the heat of ionization of acetic acid.
Answer: Neutralisation reaction of the strong acid-strong base in an aqueous solution is: \(\mathrm{H}^{+}(a q)+\mathrm{OH}^{-}(a q) \rightarrow \mathrm{H}_2 \mathrm{O}(l)\) The change in enthalpy in this reaction =-57.3 kj. The heat of neutralization in the reaction of a weak acid (acetic acid) and strong base = heat of ionization of acetic acid + heat of reaction for \(\left[\mathrm{H}^{+}(a q)+\mathrm{OH}^{-}(a q) \rightarrow \mathrm{H}_2 \mathrm{O}(l)\right].\). Given, -55.9kJ=heat of ionization of acetic acid -57.3 kj. Therefore, heat of ionisation of acetic acid = 57.3- 55.9 = 1.4 kj.mol-1
Heat or enthalpy of solution
Generally, when a solute is dissolved in a solvent, heat is evolved or absorbed. At a given temperature the amount of heat absorbed or evolved depends upon the amount and nature of both solvent and solute.
Heat or enthalpy of solution Definition: At a given temperature, the heat (or enthalpy) change associated with the dissolution of 1 mol of a solute in a specified amount of solvent so that (further addition of solvent will not produce any significant thermal effect) is termed as heat (or enthalpy) of solution of that solute at that temperature.
Therefore, if 100 mL of 0.2 (M) HC1 is mixed with 200 mL of 0.15 (M) NaOH, then 0.02 mol of H2O(/) will be formed from a die reaction between 0.02 mol H+ ions and 0.02 mol OH- ions.
Thus heat evolved (i.e., heat of neutralisation) for tills reaction = 0.02 x 57.3 = 1.146 kJ.
Explanation: The addition Of More Solvent to the solution (i.e., alter dissolution of solute) causes dilution of the solution and enthalpy changes In a continuously decreasing manner. At 25°G, the heat absorbed for the dissolution of 1 mol of KC1 in 50 mL of water Is 171191 J. Addition of another 50 mL of water to the tills solution caused absorption of 401. 1J of heat.
If we again add 50 mL of water to the solution, then 142.3 of heat is absorbed. Further addition of 50 mL of water leads to an absorption of 100.4 J of heat. If water is again added to the solution, no change in enthalpy is found to occur. Therefore, the heal of solution of KC1 at 25°C = (17091 +401.1 + 142.2+ 100.4) f =10614.7 J.
The dissolution of I mol of KC1 in a sufficient amount of water can be expressed by the thermochemical equation as \(\mathrm{KCl}(s)+a q \rightarrow \mathrm{KCl}(a q); \Delta H=+18.61 \mathrm{~kJ}\) In this equation, ‘ aq ‘ indicates a large amount of water and KCl(ag) indicates infinitely diluted aqueous solution of KC1.
⇒ \(\text { Similarly, } \mathrm{CuSO}_4(s)+a q \rightarrow \mathrm{CuSO}_4(a q) ; \Delta H=-66.5 \mathrm{~kJ}\)
⇒ \(\begin{aligned}
& \mathrm{H}_2 \mathrm{SO}_4(l)+a q \rightarrow \mathrm{H}_2 \mathrm{SO}_4(a q) ; \Delta H=-96.2 \mathrm{~kJ} \\
& \mathrm{HCl}(g)+a q \rightarrow \mathrm{HCl}(a q) ; \Delta H=-74.8 \mathrm{~kJ}
\end{aligned}\)
Generally, the heats of the solution are found to be positive for the hydrated salts (like CuSO4-5H2O, MgSO4-7H2O, FeSO4-7H2O, etc.) and the salts which cannot form stable hydrates (like NaC1, KC1, NH4C1, etc.). For anhydrous salts such as CuSO4, CaCl2, MgCl2, etc., the heat of the solution is normally negative.
Integral heat Of solution: At a specified temperature, the change in enthalpy associated with the dissolution of one mole ofa solute in a specified amount of a solvent is known as the integral heat solution. For example,
⇒ \(\text { (1) } \mathrm{KCl}(s)+12 \mathrm{H}_2 \mathrm{O}(l) \xrightarrow{25^{\circ} \mathrm{C}} \mathrm{KCl}\left(12 \mathrm{H}_2 \mathrm{O}\right) ; \Delta H=+15.8 \mathrm{~kJ}\)
⇒ \(\text { (2) } \mathrm{KCl}(s)+100 \mathrm{H}_2 \mathrm{O}(l) \xrightarrow{25^{\circ} \mathrm{C}} \mathrm{KCl}\left(100 \mathrm{H}_2 \mathrm{O}\right) ; \Delta H=+18.4 \mathrm{~kJ}\)
The values of AH in equations (1) and (2) indicate the integral heats of the solution when the amounts of water are 12 mol and 100 mol respectively. As the integral heat of the solution depends upon the amount of solvent, it is necessary to specify the amount of solvent while reporting the integral heat of the solution.
Heat or enthalpy of dilution
The dilution of a solution increases with the addition of more and more solvent to the solution, and consequently, heat is either absorbed or evolved in the process.
Integral heat of dilution
Integral heat of dilution Definition: It Is defined as the change In heat (or enthalpy) when the concentration of a solution containing one one mole of the solute is changed (diluted) by adding more solvent. The heat (or enthalpy) of dilution is equal to the difference between the integral heats of the solution at the two concentrations.
Integral heat of dilution Explanation: At 25°C when 1 mol of HCl(g) is dissolved In 25 mol of water, 72.3 kj of heat is evolved,\(\mathrm{HCl}(\mathrm{g})+25 \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{HCl}\left(25 \mathrm{H}_2 \mathrm{O}\right); \Delta H=-72.3 \mathrm{~kJ} \cdots[1]\)
Again, if 1 mol of HCl(g) is dissolved in 40 mol of water, then 73.0 kj of heat is evolved \(\mathrm{HCl}(g)+40 \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{HCl}\left(40 \mathrm{H}_2 \mathrm{O}\right); \Delta H=-73.0 \mathrm{~kJ} \cdots[2]\)
⇒ \(\begin{aligned}
& \mathrm{HCl}\left(25 \mathrm{H}_2 \mathrm{O}\right)+15 \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{HCl}\left(40 \mathrm{H}_2 \mathrm{O}\right) ; \\
& \Delta H=(-73.0+72.3)=-0.7 \mathrm{~kJ}=-700 \mathrm{~J}
\end{aligned}\)
Thus, the addition of 15 mol of water to the solution containing 1 mol of dissolved HC1 in 25 mol of water evolves 700J of heat. Therefore, for the process \(\mathrm{HCl}\left(25 \mathrm{H}_2 \mathrm{O}\right) \rightarrow \mathrm{HCl}\left(40 \mathrm{H}_2 \mathrm{O}\right) \text {, }\) the heat of dilution = -700 J.
Hess’s Law Of Constant Heat Summation
Hess’s Law Of Constant Heat Summation Law: If the reaction is carried out in one step or a series of steps, then the change in enthalpy in both cases will be the same provided that the initial and final states are the same in both cases

Explanation: Let us consider, for example, the reaction A → D, which is carried out by two different processes. In process 1, A Is directly converted into D, and the enthalpy change in the process is A. In process 2, which consists of two steps, A is first converted into C, and then C is converted into D . If the enthalpy changes in two steps (A→C) and (C→D) of process 2 are ALL, and AH3, respectively, then according to the Hess’s law, AH2 = A H2 + AH3.
Examples: CO2 can be prepared from carbon in two ways: Direct oxidation of graphite into CO2 (one step): C(graphite, s) + O2(g)→CO2(g); A = -393.5 kj
C is first oxidized to CO, and then CO is oxidized to CO2 (two steps):
⇒ \(\begin{aligned}
& \mathrm{C}(\text { graphite, } s)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{CO}(g) ; \Delta H_2^0=-110.5 \mathrm{~kJ} \\
& \mathrm{CO}(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{CO}_2(g) ; \Delta H_3^0=-283.0 \mathrm{~kJ}
\end{aligned}\)
According to the Hess’s law \(\Delta H_2^0+\Delta H_3^0=[-110.5+(-283.0)] \mathrm{kJ}=-393.5 \mathrm{~kJ}=\Delta H_1^0\)

Thermodynamic explanation of Hess’s law: As enthalpy is a state function, for any chemical reaction, the change in enthalpy (AH) depends only on the states of the reactants (initial state) and the products (final state), and not on how the change is brought about. This means that the change in enthalpy does not depend upon the number of intermediate states in a process.
Therefore, if a reaction is carried out by a process involving one step or by a process involving multiple steps, the change in enthalpy will be the same in either case. Hess’s law is simply a corollary of the first law of thermodynamics: Let us consider the reaction A + B→+D, which is carried out by two alternative processes I and II as given below.
⇒ \(\begin{aligned}
& \text { Process-I: } \quad A+B \rightarrow D ; \Delta H_1=-x \mathrm{~kJ} \\
& \text { Process II: } A+B \rightarrow C ; \Delta H_2=-y \mathrm{~kJ} \\
& C \rightarrow D ; \Delta H_3=-z \mathrm{~kJ} \\
&
\end{aligned}\)
According to Hess’s law, if the initial state and final state are the same, then x = y + z. Let (y + z) > x. This means that preparing D from A and B by process 2 and converting D back into A and B by following the process will lead to an evolution of [(y + z)- x] amount of heat without any input of energy from outside. This contradicts the first law of thermodynamics. So, (y + z) & x must be equal, which corroborates the Hess’s law.
So, Hess’s law regarding heat change in a chemical reaction is a corollary of the first law of thermodynamics.
Applications of Hess’s law
The importance of Hess’s law lies in the fact that thermochemical equations can be treated as algebraic equations. They can be added or subtracted as we do with algebraic equations.
There are many reactions whose heats of reaction are not possible to be measured directly by experiment. However, we can determine the heat of reaction for their reactions in an indirect way by making use of Hess’s law. A few applications of Hess’s law are given below.
Calculation of the heats of formation of the compounds whose heats of formation cannot be determined directly: The formation of many compounds such as CH4(g), C2H2(g), and C2H5OH(Z) from their constituent elements is practically impossible.
So, we cannot directly determine the heat of formation of these compounds. However, we can do so indirectly by applying Hess’s law.
Example: Determination of the standard heat of formation of C2H2(g) from the following information:
⇒ \(\begin{aligned}
& \mathrm{C}_2 \mathrm{H}_2(\mathrm{~g})+\frac{5}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{CO}_2(\mathrm{~g})+\mathrm{H}_2 \mathrm{O}(l) ; \\
& \Delta H^0=-1300 \mathrm{~kJ} \quad \cdots[1]
\end{aligned}\)
⇒ \(\begin{aligned}
& \mathrm{C}(\text { graphite, } s)+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \Delta H^0=-393.5 \mathrm{~kJ} \cdots[2] \\
& \mathrm{H}_2(\mathrm{~g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0=-285.8 \mathrm{~kJ} \quad \cdots[3]
\end{aligned}\)
Reversing equation [1] and multiplying equation [2] by 2,
⇒ \(\begin{aligned}
2 \mathrm{CO}_2(g)+\mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{C}_2 \mathrm{H}_2(\mathrm{~g})+\frac{5}{2} \mathrm{O}_2(\mathrm{~g}) ; \\
\Delta H^0=+1300 \mathrm{~kJ}
\end{aligned}\)
⇒ \(\text { and } 2 \mathrm{C}(\text { graphite, } s)+2 \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{CO}_2(\mathrm{~g})\)
⇒ \(\Delta H^0=2 \times(-393.5)=-787 \mathrm{~kJ} \quad \cdots[5]\)
Adding equations [3], [4] and [5], we obtain, \(\begin{aligned}
& 2 \mathrm{C} \text { (graphite, } s)+\mathrm{H}_2(\mathrm{~g}) \rightarrow \mathrm{C}_2 \mathrm{H}_2(\mathrm{~g}) \\
& \Delta H^0=[-285.8+1300-787] \mathrm{kJ}=+227.2 \mathrm{~kJ} \quad \cdots[6]
\end{aligned}\)
Since equation [6] is the formation reaction of C2H2(g), the AH° of this reaction represents the standard heat of formation of C2H2(g). Hence, the standard enthalpy of
formation \(\mathrm{C}_2 \mathrm{H}_2(\mathrm{~g})=+227.2 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \text {. }\)
Calculation of heat of reaction for the reactions whose heats of reaction cannot be determined directly: The heats of reaction for many chemical reactions cannot be determined directly. \(\mathrm{C}(\text { graphite, } s)+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}(\mathrm{g}),\) cannot be determined directly because when 1 mol of solid graphite reacts with 0.5 mol of O2(g), CO2(g) along with CO(g) is produced. However, it is possible to calculate the heat of the reaction indirectly by applying Hess’s law
Example: Let us consider (the determination of heat of a reaction in which 1 mol of CO(g) is formed from the reaction of solid graphite with oxygen;
⇒ \(C(g r a p h i t e, s)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow CO(g); \Delta H^0=? \quad \cdots[1]\)
CO2(g) he prepared by the complete combustion of both solid graphite and CO(g). From the heats of reactions of the following two reactions, the heat of reaction for the reaction can easily be calculated.
⇒ \(\begin{aligned}
& \text { C(graphite, s) }+\mathrm{O}_2(g) \rightarrow \mathrm{CO}_2(g) ; \Delta H^0=-393.5 \mathrm{~kJ} \cdots[2] \\
& \mathrm{CO}(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{CO}_2(g) ; \Delta H^0=-283.0 \mathrm{~kJ} \quad \cdots[3]
\end{aligned}\)
Adding equation [2] to the reverse of equation [3] we get \(\begin{gathered}
\mathrm{C}(\text { graphite, } s)+\mathrm{O}_2(\mathrm{~g})+\mathrm{CO}_2(\mathrm{~g}) \longrightarrow \\
\mathrm{CO}_2(\mathrm{~g})+\mathrm{CO}(\mathrm{g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) ; \\
\Delta H^0=[+283.0-393.5] \mathrm{kJ}=-110.5 \mathrm{~kJ}
\end{gathered}\)
∴ \(\mathrm{C}(\text { graphite, } s)+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}(\mathrm{g}) ; \Delta H^0=-110.5 \mathrm{~kJ}\)
Therefore, the heat of reaction for reaction = -110.5kj
Determination of heat of transition: Some elements (like C, S, P, etc,) exist in two or more allotropic forms. During the allotropic transformation of such elements, heat is generally absorbed or evolved. The enthalpy change that occurs in an allotropic transformation is known as heat of transition.
At a given temperature and pressure, the heat or enthalpy change that occurs when 1 mol of an allotropic form of an element transforms into another form is called the heat of transition (AHtrn).
Example: The two important allotropes of sulfur are rhombic sulfur and monoclinic sulfur. By applying Hess’s law, the heat of transition of these two allotropes can easily be calculated from their heats of combustion data.
⇒ \(\mathrm{S}(\text { monoclinic }, s) \rightarrow \mathrm{S}(\text { rhombic }, s) ; \Delta H^0=?\)
The heats of combustion of rhombic and monoclinic sulphur at 25°C temperature and 1 atm pressure are, -296.9 and -297.2 kj – mol-1, respectively.
⇒ \(\begin{aligned}
\mathrm{S}(\text { monoclinic, } s)+\mathrm{O}_2(\mathrm{~g}) & \rightarrow \mathrm{SO}_2(\mathrm{~g}) ; \\
\Delta H^0 & =-297.2 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \quad \cdots[1]
\end{aligned}\)
⇒ \(\begin{aligned}
\mathrm{S}(\text { rhombic, } s)+\mathrm{O}_2(\mathrm{~g}) & \rightarrow \mathrm{SO}_2(\mathrm{~g}) \\
\Delta H^0 & =-296.9 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \quad \cdots[2]
\end{aligned}\)
Subtracting equation [2] from equation [1], we obtain, S(monoclinic, s) -+ Sfrhombic, s) \(\Delta H_{t r n}^0=[-297.2-(-296.9)] \mathrm{kJ} \cdot \mathrm{mol}^{-1}=-0.3 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Therefore, at 25°C temperature and 1 atm pressure 0.3 kj heat is evolved due to the transformation of lmol monoclinic sulfur to 1 mol rhombic sulfur.
Determination of heat of hydration: Hess’s law can be applied to calculate the enthalpy changes associated with the hydration of several salts to form their corresponding hydrates.
Heat Of Hydration: At a given temperature and pressure, the change in enthalpy accompanying the formation of l mol of a hydrate from the anhydrous form of the compound is called heat of hydration of that anhydrous compound.
For example, at 25°C temperature and 1 atm pressure, the combination of1 mol of anhydrous MgSO4(s), and 7 mol H2O(Z) produces lmol of MgSO4-7H2O(s). It is accompanied by the liberation of 105 kj of heat. Therefore, the heat of hydration of MgS04(s) = -105 kj-mol-1.
⇒ \(\begin{aligned}
\mathrm{MgSO}_4(s)+7 \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{MgSO}_4 \cdot 7 \mathrm{H}_2 \mathrm{O}(s) & ; \\
\Delta H_{\text {hyd }}^0 & =-105 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
The heat of hydration of a salt cannot be determined directly. However, it can be determined by applying Hess’s law from the known values of the heat of the solution of the hydrate and the anhydrous form of the salt.
Example: \(\mathrm{CuSO}_4(s)+5 \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{CuSO}_4 \cdot 5 \mathrm{H}_2 \mathrm{O}(s) ; \Delta H_{\text {hyd }}^0=?\)
At 25°C temperature and 1 atm pressure, the heat of the solution of CuS04(s) and CuS04-5H20(s) are -66.1 kj.mol-1 and +11.5 kj.mol-1, respectively.
⇒ \(\mathrm{CuSO}_4(s)+n \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{CuSO}_4(a q) ;\) [n is a very large number]
⇒ \(\Delta H_{\text {sol }}^0=-66.1 \mathrm{~kJ}\)
⇒ \(\begin{gathered}
\mathrm{CuSO}_4+5 \mathrm{H}_2 \mathrm{O}(s)+(n-5) \mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{CuSO}_4(a q) ; \\
\Delta H_{\text {sol }}^0=+11.5 \mathrm{~kJ}
\end{gathered}\)
Subtracting equation [2] from equation [1], we obtain, \(\begin{aligned}
\mathrm{CuSO}_4(s)+5 \mathrm{H}_2 \mathrm{O}(l) & \rightarrow \mathrm{CuSO}_4 \cdot 5 \mathrm{H}_2 \mathrm{O}(s) ; \\
\Delta H_{\text {hyd }}^0 & =[-66.1-(+11.5)] \mathrm{kJ}=-77.6 \mathrm{~kJ}
\end{aligned}\)
Therefore, at 25°C temperature and 1 atm pressure, the heat of hydration of anhydrous CuSO2(s) = -77.6kJ.mol-1
Standard Heat Of Formation Of An Ion
Heat or enthalpy change occurs when an ionic compound dissociates into ions in solution. So, like a compound, an ion also has the heat of formation.
Standard Heat Of Formation Of An Ion Definition: Enthalpy or heat change associated with the formation of one mole of an ion in an infinitely dilute solution at standard state (i.e., at 1 atm pressure and a specified temperature) is termed as the standard heat of formation of that ion The determination of standard heat of formation of an ion is usually done at 25°C temperature. By convention, the standard heat of formation (or enthalpy of formation) of H+ ion in aqueous solution is taken to be zero.
Therefore \(\frac{1}{2} \mathrm{H}_2(g) \rightarrow \mathrm{H}^{+}(a q) ; \Delta H_f^0\left[\mathrm{H}^{+}(a q)\right]=0\left[25^{\circ} \mathrm{C}\right]\)
Example: \(\text { (1) } \mathrm{H}^{+}(a q)+\mathrm{OH}^{-}(a q) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H^0=-57.3 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\text { (2) } \begin{aligned}
\mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow & \mathrm{H}_2 \mathrm{O}(l) ; \\
& \Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(l)\right]=-285.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
By writing an equation in a reverse manner and then adding it to an equation, we obtain,
⇒ \(\mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{H}^{+}(a q)+\mathrm{OH}^{-}(a q)\)
⇒ \(\Delta H^0=-285.8+57.3=-228.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Therefore \(\begin{aligned}
& \Delta H^0=\Delta H_f^0\left[\mathrm{H}^{+}(a q)\right]+\Delta H_f^0\left[\mathrm{OH}^{-}(a q)\right] \\
& -\Delta H_f^0\left[\mathrm{H}_2(g)\right]-\frac{1}{2} \Delta H_f^0\left[\mathrm{O}_2(g)\right]
\end{aligned}\)
⇒ \(\text { or, }-228.5=0+\Delta H_f^0\left[\mathrm{OH}^{-}(a q)\right]-0-0\)
[In standard state, the heat of formation of any stable pure dement is taken as 0. So \(\left.\Delta H_f^0\left[\mathrm{H}_2(g)\right]=\Delta H_f^0\left[\mathrm{O}_2(g)\right]=0\right]\)
∴ \(\Delta H_f^0\left[\mathrm{OH}^{-}(a q)\right]=-228.5 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Alternative method: From the equation we get,
⇒ \(\begin{aligned}
& \Delta H^0=\Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(l)\right]-\Delta H_f^0\left[\mathrm{H}^{+}(a q)\right]-\Delta H_f^0\left[\mathrm{OH}^{-}(a q)\right] \\
& \text { or, }-57.3 \mathrm{~kJ}=-285.8 \mathrm{~kJ}-0-\Delta H_f^0\left[\mathrm{OH}^{-}(a q)\right]
\end{aligned}\)
∴ \(\Delta H_f^0\left[\mathrm{OH}^{-}(a q)\right]=-228.5 \mathrm{~kJ}\)
Heat Of Solution Of Ionic Compounds
The dissolution of an ionic compound (like NaCl, KC1, etc.) in a polar solvent (like water) can be considered as the combination ofthe following two processes: Breaking of the crystal lattice ofthe compound into gaseous constitutes ions Interaction of resulting ions with the solvent molecules i.e., solvation of the ions (or hydration if water is used solvent). Process (1) is endothermic, while the process (2) is exothermic.
In an ionic compound [MX(s)], the energy required to transform one mole of the ionic crystal into its gaseous constituent ions (M+ and X-), separated by infinite distance, is equal to the reaction enthalpy of the following reaction and is called lattice enthalpy (AHl).
⇒ \(\mathrm{MX}(\mathrm{s}) \rightarrow \mathrm{M}^{+}(\mathrm{g})+\mathrm{X}^{-}(\mathrm{g}) \text {; reaction enthalpy }=\Delta H_L \quad \cdots[1]\)
The change in enthalpy accompanying the hydration (AHhy(J) ofthe cations [M+(g)] and anions [X'(g)], that formed on dissociation of1 mol of MX(s), is equal to the change in enthalpy of the following hydration process.
⇒ \(\begin{aligned}
& \mathrm{M}^{+}(g)+\mathrm{X}^{-}(g)+\text { water } \rightarrow \mathrm{M}^{+}(a q)+\mathrm{X}^{-}(a q) ; \\
& \text { Change in enthalpy }(\Delta H)=\Delta H_{h y d} \cdots[2]
\end{aligned}\)
The change in enthalpy accompanying the dissolution of 1 mol of an ionic compound such as MX(s) is termed the enthalpy of solution (AI1sol) ofthe compound.
⇒ \(\mathrm{MX}(s)+\text { water } \rightarrow \mathrm{M}^{+}(a q)+\mathrm{X}^{-}(a q)\) \(\text { Enthalpy change }(\Delta H)=\Delta H_{\text {sol }} \cdots \text { [3] }\)
Adding equations [1] and [2] results in equation [3]. Thus, A Hsol equals the sum of AH2 and AHhyd. This is per Hess’s law.
The value of ΔHl is always positive and that of ΔHhyd is always negative. Depending upon the magnitude of these two, the sign of the ΔHsol will be either positive or negative. In the case of most ionic salts, the ΔHsol is positive This is why the solubilities of such salts, increase with temperature rise.

Lattice Energy (Or Lattice Enthalpy) And Born-Haber Cycle
Lattice Energy (Or Lattice Enthalpy) And Born-Haber Cycle Definition: The lattice energy of a solid ionic compound it The. the energy required to break 1 mol of the compound (at a particular temperature and pressure) into its gaseous ions, separated by an infinite distance.
Example: The lattice enthalpy of NaCl at 25°C and ] atm pressure = +788 kj .mol-1 means that at 256C temperature and 1 atm pressure, 788 kj of heat is required to break mol of NaCl(s) into lmol of Na+(g) & I mol of Cl-(g) ions, separated by infinite distance.
Therefore, the value of the lattice enthalpy for MX(s) type of the compound is equal to the value of the enthalpy change for the process \(\mathrm{MX}(\mathrm{s}) \rightarrow \mathrm{M}^{+}(\mathrm{g})+\mathrm{X}^{-}(\mathrm{g})\)
Determination of lattice enthalpy of an ionic compound a standard state by applying Hess’s law: To illustrate how to determine the lattice energy of a solid ionic compound, let us consider, for example, the determination of lattice energy of NaCl. NaCl can be prepared in a single step by reacting to its constituent elements or by an indirect process involving multiple steps.
Preparation of 1 mol of NaCl in a single step: 1 mol of metallic Na is reacted with 0.5 mol of Cl2 gas to form lmol of solid NaCl. The enthalpy change in the reaction is equal to the standard enthalpy of the formation of solid sodium chloride (NaCl).
⇒ \(\mathrm{Na}(s)+\frac{1}{2} \mathrm{Cl}_2(g) \rightarrow \mathrm{NaCl}(s) ; \Delta H_f^0(\mathrm{NaCl})=-411.2 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)


According to Hess’s law, if the initial and final states remain fixed, then the change in enthalpy for the transformation from the initial state to the final state is the same, regardless of whether the reaction is completed in one step or several steps.
Hence \(\Delta H_f^0(\mathrm{NaCl})=376.5-\Delta H_L^0 \text { or, }-411.2=376.8-\Delta H_L^0\)
∴ \(\Delta H_L^0=+788.0 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Thus, the lattice enthalpy of solid NaCl at standard state = + 788 kj. mol-1 .
Born-Haber cycle: Bom-Haber cycle is a thermochemical cycle consisting of a series of steps that describe the formation of an ionic solid from its constituents elements. It is based on Hess’s law and establishes a relation between the lattice energy of an ionic compound and the enthalpy changes that occur in various steps associated with the formation ofthe compound. This cycle is very useful for calculating lattice enthalpy and electron gain enthalpy (electron affinity), which are difficult to measure experimentally.

As enthalpy is a state function, the total change in enthalpy for a cyclic process is zero. Thus, \(+411.2+108.4+121+496-348.6-\Delta H_L^0=0\)
∴ \(\Delta H_L^0=+788 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Atomisation Enthalpy
Standard enthalpy of atomation of an element: The enthalpy change associated with the formation of 1 mol of gaseous atoms from a stable element when all substances are in their standard states is called the enthalpy of atomization \(\left(\Delta H_{\text {atom }}^0\right)\) of the element.
The atomization of any substance is an endothermic process. So, for any substance, \(\Delta \boldsymbol{H}_{\text {atom }}^0\) is always positive. The unit of \(\Delta H_{\text {atom }}^0\) kj mol-1 or kcal- mol-1.
Explanation: The standard enthalpy of atomization of hydrogen at 25°C \(\frac{1}{2} \mathrm{H}_2(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g}); \Delta H^0{ }_{\text {atom }}=+218 \mathrm{~kJ}\)
The standard enthalpy of atomization of graphite at 25°C, AH°fom = +717 kj-mol-1: This means that at 25°C and 1 atm pressure, +717 kj heat (or enthalpy) is required to produce 1 mol of carbon vapor [C(g)] from graphite. Thus the change in enthalpy for the process, C(graphite, s) -C(g) at 25°C and 1 atm pressure, AH° = +717 kj-mol-1.
At a particular temperature, for homonuclear diatomic gases, the heat (or enthalpy) of atomization is equal to half the heat of dissociation, i.e., the standard enthalpy of atomization \(=\frac{1}{2} \times \text { enthalpy of dissociation. }\)
At a particular temperature, for a monoatomic solid, the heat (or enthalpy) of atomization is equal to its heat of sublimation at that temperature.
Bond Bond Dissociation Energy (Or energy enthalpy)(Or Enthalpy)
Chemical reactions are associated with the formation and dissociation of bond(s). During dissociation of a bond heat or energy is required, so it is an endothermic process. On the other hand, bond formation is associated with the evolution of heat. Hence it is an exothermic process. At a particular temperature and pressure, the amount of heat required to break a bond is equal to the amount of heat released to form that bond at the same conditions.
Bond Dissociation Enthalpy
Bond Dissociation Enthalpy Definition: The Amount of energy required to dissociate one mole of a specific type of bond in a gaseous covalent compound to form neutral atoms or radicals is called bond dissociation energy (ΔHbond) of that bond. As bond dissociation is an endothermic process, the value of the bond dissociation energy is always positive. The bond dissociation energy is generally expressed in kJ. mol-1 or kcal- mol-1. Here the term ‘mol’ indicates per mole of bond.
The bond dissociation energy of a diatomic gaseous molecule:
The change in standard enthalpy (AH0) for the process \(\mathrm{Cl}_2(\mathrm{~g}) \rightarrow 2 \mathrm{Cl}(\mathrm{g}) \text { at } 25^{\circ} \mathrm{C}\) and 1 atm pressure = 248 kj.mol-1. In this process, the gaseous Cl is formed by the dissociation of1 mol of Cl—Cl bonds atoms in a gaseous state.
Therefore, this standard enthalpy change is equal to the bond dissociation energy (or enthalpy) of the Cl—Cl bond \(\left(\Delta H_{\mathrm{Cl}}^0-\mathrm{Cl}\right)\) The change in standard enthalpy (AH0) for the process 02(g) → 20(g) at 25°C temperature and 1 atm pressure = 498 kj. mol-1.
In this process, the gaseous O atoms are formed by the dissociation of lmol of 0=0 bonds in the gaseous state at 25°C and 1 atm pressure. Therefore, this standard enthalpy change is equal to the bond dissociation energy of O=0 bond \(\left(\Delta H_{\mathrm{O}}^0=0\right)\).
The bond dissociation energy of the bond in a multi-atomic molecule: If a molecule has more than one bond of a particular type, then the stepwise dissociation of these bonds requires different amounts of energy. For example, in the NH3 molecule, although three N—H bonds are equivalent stepwise dissociation of these bonds requires different amounts of energy.
⇒ \(\mathrm{NH}_3(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{NH}_2(\mathrm{~g}) ; \Delta H_{\text {bond }}^0=+431 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\mathrm{NH}_2(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{NH}(\mathrm{g}) ; \Delta H_{\text {bond }}^0=+381 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\mathrm{NH}_2(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{NH}(\mathrm{g}) ; \Delta H_{\text {bond }}^0=+381 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
If more than one similar type of bond is present in a molecule, then the average bond dissociation energy of those bonds is expressed as the bond energy of that bond.
Therefore, the bond energy of the N—H bond in NH3 molecule \(=\frac{431+381+360}{3}=391 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \text {. }\) = 391 KJ. mol-1
⇒ \(\mathrm{CH}_4(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{CH}_3(\mathrm{~g}) ; \Delta H_{\text {bond }}^0=+427 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\mathrm{CH}_3(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{CH}_2(\mathrm{~g}) ; \Delta H_{\text {bond }}^0=+439 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\mathrm{CH}_2(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{CH}(\mathrm{g}) ; \Delta H_{\text {bond }}^0=+452 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\mathrm{CH}(\mathrm{g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{C}(\mathrm{g}) ; \Delta H_{\text {bond }}^0=+347 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\mathrm{CH}(\mathrm{g}) \rightarrow \mathrm{H}(\mathrm{g})+\mathrm{C}(\mathrm{g}) ; \Delta H_{\text {bond }}^0=+347 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Therefore, the bond energy ofthe C —H bond in CH4(g) , molecule \(\text { molecule }=\frac{427+439+452+347}{4}=416.25 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \text {. }\)
Definition Of Bond Energy
The average value of the dissociation energies of all the similar types of bonds present in a gaseous compound is called the bond energy of that type of bond
![Chemical Thermodynamic Standard Enthalpy Of Dissociation [Standard Bond dissovstion energy] diatomic Molecules at 25C.](https://wbbsesolutions.net/wp-content/uploads/2024/03/Chemical-Thermodynamic-Standard-Enthalpy-Of-Dissociation-Standard-Bond-dissovstion-energy-diatomic-Molecules-at-25C-1.png)

Determination of bond enthalpy or bond energy
The standard bond enthalpy of a bond in a compound can be calculated from the known value of the standard enthalpy of formation of that compound and the value of the standard enthalpy of atomization of the elements constituting the compound. For example, consider the determination of bond enthalpy ofthe C—H bond in methane molecule.
In 1 mol of CH4(g) molecule, the energy required to break 4 mol of C— H bonds is equal to the standard enthalpy of reaction for the following change (AHO). CH4(g) C(graphite, s) + 4H(g); AHO The value of AH° can be calculated from the value of standard enthalpy of formation of CH4(g) and the standard enthalpy of atomization of C(graphite.s) and Ha(g). The formation reaction of CH4(g) and the atomisation reaction of C(graphite.s) and Ha(g) are as follows;
⇒ \(\begin{aligned}
& \mathrm{C}(\text { graphite, } s)+2 \mathrm{H}_2(\mathrm{~g}) \rightarrow \mathrm{CH}_4(\mathrm{~g}) ; \\
& \left.\Delta \mathrm{H}_f^0\left[\mathrm{CH}_4(\mathrm{~g})\right]=-7.8 \mathrm{k}\right] \cdot \mathrm{mol}^{-1} \quad \ldots[2]
\end{aligned}\)
⇒ \(\begin{aligned}
& \mathrm{C}(\text { graphite, } s) \rightarrow \mathrm{C}(\text { graphite, } g): \\
& \left.\Delta H_{\text {atom }}^0=717 \mathrm{k}\right] \cdot \mathrm{mol}^{-1} \quad \ldots|3|
\end{aligned}\)
⇒ \(\frac{1}{2} \mathrm{H}_2(\mathrm{~g}) \rightarrow \mathrm{H}(\mathrm{g}) ; \Delta H_{\text {atom }}^0=218 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
Equation[3] + 4 x equation [4]- equation [2] gives, CH4(g)—>C(graphite, g) + 4H(g); AH° = [717 + 4 X 218- (-74.8)]kJ = 1663.8k] Therefore, in 1 mol of CH4(g) molecule, the energy required to break 4 mol of C —H bonds = 1663.8k).
Energy required to break1 mol of C — H bonds \(=\frac{1}{4} \times 1663.8 \mathrm{~kJ}=415.95 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\) So, the bond energy of the C— H bond = 415.95 kJ. mol-1
Determination of the standard enthalpy of a gaseous reaction from the values of bond enthalpies of the reactant(s) and product(s)
In general, chemical reactions are associated with the breaking of bonds (on the reactant side) and the formation of new bonds (on the product side). When a chemical bond is formed energy is released.
On the other hand, energy is required to break a bond. The standard enthalpy change in a reaction in the gaseous state is related to the bond energies of the reactants and products.
If the total bond energy of all the bonds in reactant molecules is reactant tie clane in enthalpy for breaking all the bonds in reactant molecules \(\Delta H_1^0=\sum(\mathrm{BE})_{\text {reactant }} \text {. }\) Similarly if the total bond energy of the product(s) be \(\text { (BE) product, }\), then the change in enthalpy for the formation of bonds in product molecules p\(\Delta H_2^0=-\sum(\mathrm{BE})_{\text {product }} \) [-ve sign indicates that heat is released during the formation.
⇒ \(\Delta H^0=\Delta H_1^0+\Delta H_2^0=\sum(\mathrm{BE})_{\text {reactant }}-\sum(\mathrm{BE})_{\text {product }}\)
∴ \(\Delta H^0=\sum(\mathrm{BE})_{\text {reactant }}-\sum(\mathrm{BE})_{\text {product }}\)
Using equation [1], it is possible to determine the standard reaction enthalpy of a gaseous reaction from the bond energy data ofthe reactants and products. \(\text { If } \sum(\mathrm{BE})_{\text {reactant }}>\sum(\mathrm{BE})_{\text {product }} \text {, then } \Delta H^0>0 \text {. }\)
This means that if the total bond energy of all the bonds in reactant molecules are greater than the total bond energy of all the bonds in product molecules, then the reaction will be endothermic.
On the other hand, if \(\sum(\mathrm{BE})_{\text {reactant }}<\sum(\mathrm{BE})_{\text {product, }} \text { then } \Delta \boldsymbol{H}^{\mathbf{0}}<\mathbf{0}\) \(\Delta H^0<0\) and the willl be exothedrmic.
Numerical Examples
Question 1. Calculate The bond energy of O-H Bond In H2O(g) at the standard state from the following data;
⇒ \(\mathrm{H}_2(g) \rightarrow 2 \mathrm{H}(\mathrm{g}) ; \quad \Delta H^0=436 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\frac{1}{2} \mathrm{O}_2(g) \rightarrow O(g) ; \quad \Delta H^0=249 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
⇒ \(\begin{aligned}
& \mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{H}_2 \mathrm{O}(g) \\
& \Delta H_f^0\left[\mathrm{H}_2 \mathrm{O}(g)\right]=-241.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
Answer: Equation + equation (2)- equation gives,
⇒ \(\begin{array}{r}
\mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g)-\mathrm{H}_2(g)-\frac{1}{2} \mathrm{O}_2(g) \rightarrow 2 \mathrm{H}(g)+\mathrm{O}(g)-\mathrm{H}_2 \mathrm{O}(g) ; \\
\Delta H^0=(436+249+241.8) \mathrm{kJ}
\end{array}\)
or, \(\mathrm{H}_2 \mathrm{O}(\mathrm{g}) \rightarrow 2 \mathrm{H}(\mathrm{g})+\mathrm{O}(\mathrm{g}) ; \Delta H^0=+926.8 \mathrm{~kJ}\)
The equation indicates the dissociation of O—H bonds Present in 1 mol of H20(g) . The standard enthalpy change for this process (AH0) = +926.8kJ
No. of O— H bonds present in lmol H20 =2mol. Thus, the energy required to break 2mol O—H bonds =+926.8 kj.
∴ The energy required to break 1 mol O — H = + 463.4 kj So, bond energy of O—H bond = + 463.4 kj.mol-1.
Question 2. Calculate the bond energy of the S—F bond in the SF6 molecule. Given: Enthalpy of formation for SF6(g) , S(g),F(g) are -1100, 275, 80 kj.mol-1 respectively.
Answer: \(\begin{aligned}
& \frac{1}{8} \mathrm{~S}_8(s) \rightarrow \mathrm{S}(g) ; \Delta H_f^0[\mathrm{~S}(g)]=275 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} \\
& \frac{1}{2} \mathrm{~F}_2(g) \rightarrow \mathrm{F}(g) ; \Delta H_f^0[\mathrm{~F}(g)]=80 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
⇒ \(\begin{aligned}
& \frac{1}{8} \mathrm{~S}_8(s)+3 \mathrm{~F}_2(g) \rightarrow \mathrm{SF}_6(g) \\
& \Delta H_f^0\left[\mathrm{SF}_6(g)\right]=-1100 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
No. of moles of S—F bonds in lmol SF6 molecule = 6. In 1 mol of SF6, the breaking of S- F bonds is given by the reaction: SF6(g) -S(g) + 6F(g) Standard enthalpy for this reaction is \(\begin{aligned}
\Delta H^0=\Delta H_f^0[\mathrm{~S}(g)] & +6 \Delta H_f^0[\mathrm{~F}(g)]-\Delta H_f^0\left[\mathrm{SF}_6(g)\right] \\
& =[275+6 \times 80-(-1100)] \mathrm{kJ}=1855 \mathrm{~kJ}
\end{aligned}\)
Thus 1855 kj energy is required to break 6 mol of S — F bonds. Hence, the energy required to break lmol of S —F bond \(=\left(\frac{1}{6} \times 1855\right)=309.166 \mathrm{~kJ} \text {. }\)
So, the bond energy of the S—F bond = 309.166 kj.mol-1
Question 3. Determine the standard enthalpy of formation of isoprene(g) at 298 K temperature. Given:
⇒ \(\begin{aligned}
& \Delta H^0(\mathrm{C}-\mathrm{H})=413 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}, \\
& \Delta H^0(\mathrm{H}-\mathrm{H})=436 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}, \\
& \Delta H^0(\mathrm{C}-\mathrm{C})=346 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}, \\
& \Delta H^0(\mathrm{C}=\mathrm{C})=611 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}, \\
& \mathrm{C}(\text { graphite }, s) \rightarrow \mathrm{C}(\text { graphite }, \mathrm{g}) ; \\
& \Delta H^0=717 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
Answer: The formation reaction of isoprene [C4Hg(g)]:
⇒ \(5 \mathrm{C} \text { (graphite, } s)+4 \mathrm{H}_2(g) \rightarrow \mathrm{H}_2 \mathrm{C}=\mathrm{C}-\mathrm{CH}=\mathrm{CH}_2(g) \quad \cdots[1]\)
This reaction can be considered as the sum of the following two reactions:
According to Hess’s law, the total change in enthalpy of these two reactions is equal to the change in enthalpy of the reaction [1],
Therefore; \(\Delta H^0=\Delta H_1^0+\Delta H_2^0\)
⇒ \(\Delta H_1^0\) = Enthalpy of atomization of 5 mol C(g) + Bond energy of4 mol H —H bonds = (5 x 717 + 4 x 436) = 5329kj \(\Delta H_2^0=(-)\) Bond energy of 8 mol (C—H) + Bond energy of2mol (C=C) bonds + Bond energy of2 mol (C —C) bonds] [Here, – ve sign indicates the bond is formed in step (2)] =-[8 x 413 + 2 X 611 + 2 X 346]kJ =-5218 kj
Therefore \(\Delta H^0=(5329-5218) \mathrm{kJ}=111 \mathrm{~kJ} .\)
Determination Of The Value Of Δu And ΔH: Calorimetry
The process of measuring the amount of heat transferred during any physical or chemical transformation Is called calorimetry. The device by which the amount of heat trans¬ ferred is measured is called a calorimeter. The heat change at constant pressure {qp) and that at constant volume (qv) are determined by calorimetry.
The heat change at constant pressure is equal to the change In enthalpy (AH) of the system, and the heat change at constant volume is equal to the change in internal energy (AH) of the system, Among the different types of calorimeters we will discuss here only the bomb calorimeter. The heat of the reaction at constant volume can be measured by using a bomb calorimeter.
Bomb calorimeter: In general, a bomb calorimeter is used to determine the heat of combustion of a reaction at a constant volume. The bomb is a rigid closed steel container that can resist high pressures and the inside portion of the container is coated with platinum metal.
A known amount ofa substance (whose heat of combustion is to be determined) is taken in a platinum crucible and placed inside the bomb. The bomb is then filled with excess O2 by passing pure O2 at 20-25 atm pressure through a valve.
The bomb is now immersed in an insulated, water-filled container fitted with a mechanical stirrer and thermometer. The sample present in the crucible is then ignited electrically in the presence of oxygen.
During combustion, heat is evolved. As the calorimeter is insulated, heat evolved during combustion cannot escape. Evolved heat is absorbed by the bomb, water, and other parts of the calorimeter. As a result, the temperature of the calorimeter increases, which is recorded from a thermometer.

Calculation If the heat capacity of the calorimeter is cal and the Increase in temperature due to absorption of heat by the calorimeter is ΔT, then the beat of reaction, \(\begin{aligned}
q_{\text {reaction }} & =(-) \text { heat absorbed by the calorimeter } \\
& =-q_{\text {calorimeter }}=-C_{c a l} \times \Delta T
\end{aligned}\)
Determination of Using die same bomb calorimeter and die same amount of water, a substance of known heat of combustion is burnt In the calorimeter, and AT is measured. From the amount of the substance of known heat of combustion, we calculate the reaction Putting the values of reaction end AT into the above relation, we get the value of C cal.
As the wall of the bomb calorimeter is very rigid, the volume of the die system remains unchanged (AV = 0) during a reaction, ffence a/ [=- PΔ P =0 and according to the first law of thermodynamics, All = \(\Delta U=q+w=q+0=q_V.\) Thus, the heat of reaction for a reaction occurring In a bomb calorimeter ~ changes in internal energy of the reaction system.
Numerical Examples
Question 1. When 1.0 g of a compound (molecular weight = 28) Is burnt In a bomb calorimeter, the temperature of the calorimeter rises from 25′-C to 25.45’T). Calculate the heat that evolves when 1 mol of this compound is completely burnt (Ccal = 2.5 kj – K-1 ).
Answer: ΔT = [(273 + 2545)-(273 + 25)] = 0.45 K
Therefore, heat evolved due to combustion of 1 g of the compound \(=C_{c a l} \times \Delta T=2.5 \mathrm{~kJ} \cdot \mathrm{K}^{-1} \times 0.45 \mathrm{~K}=1.125 \mathrm{~kJ}.\)
∴ Feat produced due to combustion of1 moJ or 28 g of that compound J.125 X 28 = 31.5 kj.
Question 2. At 25-C, the heat of combustion at a constant volume of J mol of a compound Js 5J50 of. The temperature of a bomb calorimeter rises from 25-C to 30.5°C when a certain amount of the compound Is burnt In It. If the heat capacity of the calorimeter Is 9.76 kj .K-1 then how much of the compound was taken for combustion? (Molar mass of the substance = 128
Answer: As given in the question, the grain-molecular weight of
that compound = 128 g- mol-1.
Therefore, the amount of heat that evolves in the combustion of 128 g ofthe substance = 5150 kJ.
Again \(\text { Again, } \Delta T=[(273+30.5)-(273+25)] \mathrm{K}=5.5 \mathrm{~K} \text { and }\)
Thus, die amount of heat evolves when a certain amount of compound is burnt = Ccal x AT = 9.76 x 5.5 = 53.68 kj.
Now 5150 kj of heat is liberated due to the combustion of the 128g compound. Hence, the amount ofthe compound required for the evolution of53.68 kj of heat \(=\frac{128}{5150} \times 53.68=1.334 \mathrm{~g} .\)
The Second Law Of Thermodynamics
The second law of thermodynamics can be stated in various ways. Some common statements ofthe law are given below:
Clausisus statement: It is impossible to construct a device, operating in a cycle, that will produce no effect other than the transfer of heat from a lower-temperature reservoir to a higher-temperature reservoir.
Planck -Kelvin statement: It is impossible to construct an engine, operating in a cycle, that will produce no effect other than the extraction of heat from a reservoir and the performance of an equivalent amount of work.
Spontaneous And Non-Spontaneous Processes
A process that occurs on its own accord under a given set of conditions, without any outside assistance, is called a spontaneous process.
Examples:
- The flow of heat from a hotter body to a colder body.
- Evaporation of water kept in an open container at normal temperature.
- Conversion of water to ice at a temperature below 0°C.
- Rusting of iron.
Many processes require to be initiated by some external assistance. However, once initiated, they continue to occur under the prevailing conditions without any assistance from outside.
Examples:
- No reaction occurs when a piece of coal is kept in an open atmosphere. However, once it is ignited, its combustion starts and continues spontaneously till it is completely burnt out with the formation of CO2 and H2O.
- A candle when enkindled in air bums spontaneously till the end. Here, the combustion of candles (hydrocarbon) produces CO2 and H2O.
- When an electric spark is created for once in a mixture of hydrogen and oxygen, the reaction between these two gases starts and goes on spontaneously at room temperature to produce water.
Non-spontaneous processes
A process that needs external assistance for its occurrence is called a non-spontaneous process.
Examples:
- Underground water is lifted to the roof with the help of an engine. As long as the engine is in operation, water continuously goes up. But the upward flow of water ceases at the moment when the engine is stopped.
- O2 gas is produced when KC1O3 is heated with MnO2. However, the evolution of oxygen ceases when the source of heat is removed.
- Energy must be supplied from the external source to recoil a spring.
Spontaneity Of Physical Processes And Chemical Reactions
There are many physical processes or chemical reactions which occur spontaneously in preferred directions under certain conditions. For this type of process, energy is not required from any external agency.
A large number of processes, both exothermic and endothermic, are found to occur spontaneously. The question that comes into our mind is: ‘What is the driving force that makes such processes spontaneous
Factors affecting the spontaneity of a process
The tendency of a system to attain stability through lowering its energy or enthalpy: Our experience shows that every system has a natural tendency to attain stability by lowering its intrinsic energy.
Examples:
- When water falls from a high region, its potential energy goes on decreasing gradually and when it reaches the earth’s surface, its potential energy gets converted completely into a different form of energy.
- Heat always flows from a hot body to a cold body. As a result, the internal energy of the hot body decreases.
- From the above examples, we see that a process occurring spontaneously is accompanied by a decrease in the energy of the system.
- In the case of chemical reactions, the decrease or increase in energy is usually in terms ofthe change in enthalpy (AH) in the reaction.
- In a reaction, if the total enthalpy of the products Is less than that of the reactants, then heat is evolved, and hence AH = -ve, indicating that the reaction is exothermic. It has also been observed in actual experiments that most of the exothermic reactions occur spontaneously. For example
⇒ \(\begin{gathered}
\mathrm{C}(\mathrm{s})+\mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{CO}_2(\mathrm{~g}) ; \Delta H=-393.5 \mathrm{~kJ} \\
\mathrm{H}_2(\mathrm{~g})+\frac{1}{2} \mathrm{O}_2(\mathrm{~g}) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H–285.83 \mathrm{~kJ}
\end{gathered}\)
The fact that the decrease in energy of the reaction system in an exothermic reaction led the chemists to believe that only exothermic reactions would occur spontaneously. This means that the condition of spontaneity Is AH = -ve.
To put it in another way the decrease in enthalpy in a reaction is the driving force for the reaction to occur spontaneously. However, there are quite some reactions that do not occur spontaneously even though AH is negative for these reactions.
Spontaneous processes in which energy (enthalpy) of the system increases: The endothermic reactions are associated with the increase in energy of the reaction system. It is probably not wrong to believe that such reactions would be nonspontaneous.
But, there are some processes, both physical and chemical, which occur spontaneously even though they are associated with the increase in enthalpy. Here are a few examples.
Although the evaporation of water at room temperature Is an endothermic process, It occurs spontaneously \(\mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{H}_2 \mathrm{O}(\mathrm{g}); \Delta H=+44.1 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
The dissolution of NH4Cl in water Is a spontaneous process although this process is endothermic.
⇒ \(\mathrm{NH}_4 \mathrm{Cl}(s) \rightarrow \mathrm{NH}_4^{+}(a q)+\mathrm{Cl}^{-}(a q) ; \Delta H=+15.1 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
The reaction of Ba(OH)2. 8H2O(s) with NH4NO3(s) is so endothermic that it decreases the temperature of the reaction system to a very low value.
⇒ \(\begin{aligned}
& \mathrm{Ba}(\mathrm{OH})_2 \cdot 8 \mathrm{H}_2 \mathrm{O}(s)+2 \mathrm{NH}_4 \mathrm{NO}_3(s)- \\
& \mathrm{Ba}\left(\mathrm{NO}_3\right)_2(a q)+2 \mathrm{NH}_3(a q)+10 \mathrm{H}_2 \mathrm{O}(l)
\end{aligned}\)
Spontaneous process and tendency to increase the randomness of a system: Our experience tells us that the natural tendency of any spontaneous process is that it tends to occur in a direction in which the system associated with the process moves from an order to a disordered state.
Examples: Some red marbles are kept on one side of a tray, and on the other side it the same number of blue marbles, identical in mass and size, are kept perfectly in order.
If the tray is shaken properly, the marbles of both types will be mixed (a disordered condition), and the system will be in a state of disorderliness, which is considered to be a more stable state of the system.
Because if that tray is shaken several times, the marbles will never return to their previous orderly arrangement i.e., the natural tendency of the system is to achieve a state of randomness from a well-ordered state. In ice, water molecules exist in an ordered state as their motions are restricted.
But, in liquid water, molecules have more freedom of motion as intermolecular forces of attraction in water are not as strong as in ice. Thus, the melting of ice leads to an increase in molecular disorder in the system.
⇒ \(
\mathrm{H}_2 \mathrm{O}(s) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H=+v e
(ice) (water)\)
⇒ \(
\mathrm{H}_2 \mathrm{O}(l) \rightarrow \mathrm{H}_2 \mathrm{O}(\mathrm{g}) ; \Delta H=+v e
(water) (vapour)\)
When salts such as NH4C1, NaCl, etc., are added to water, they undergo dissolution spontaneously. In the crystal structures of the salts, ions are held in an ordered arrangement.
On dissolution of the salts, the ordered arrangement of the ions is lost as the ions get dispersed throughout the solution in a disordered way. Thus, the dissolution ofa salt in water leads to a disordered state.
Thus from the above examples, it is clear that spontaneous processes occur with an increase in the randomness of the system. Hence we can say, that the primary condition of the spontaneity of any process is the increase in randomness in the system.
Spontaneous processes involving a decrease in randomness: It is true that in most of the spontaneous processes, the degree of randomness of the system gets increased. However many spontaneous processes are accompanied by the decrease in randomness in the system.
Examples: Water molecules in the clouds exist in extremely disordered states. But, when they fall in the form of rain, their freedom of movement decreases.
Molecules in water are in a state of randomness. However, when water undergoes freezing, the motion of the molecules becomes restricted, leading to a decrease In disorder in the system. Thus, the increase in randomness of the constituent particles of a system is not an essential condition for the spontaneity ofthe process that the system undergoes.
The driving force in a spontaneous process
From the above discussion, we see that neither the decrease in energy alone nor the increase in randomness alone can determine the spontaneity of a process. This is because many processes occur with an increase in energy, and any processes occur with a decrease in randomness.
Thus, we can conclude the effects of both of these factors have a role in determining the spontaneity of a process. To put it in another way we can say that the combined effect of these two factors is the driving for a process to occur spontaneously.
In this regard, it is important to note that these factors are not dependent on each other, and they may work in the same or opposite direction. Hence, we may have the following combinations of these two factors, the results of which may be favorable or unfavorable about spontaneity.

Concept Of Entropy
We have already seen that enthalpy is not the ultimate criterion of spontaneity. Another factor such as the randomness of the constituting particles (molecules, atoms, or ions) of the system may also be responsible for determining the spontaneity of a process.
Rudolf Clausius introduced a new thermodynamic property or state function known as entropy, from the Greek word ‘trope’ meaning transformation. It is denoted by the letter ‘S’. The entropy of a system is a measure of the randomness or disorderliness of its constituent particles.
The more disordered or random state of a system, the higher the entropy it has. Thus, from the molecular point of view, the entropy of a system can be defined below.
The entropy of the system is a thermodynamic property that measures the randomness or disorderliness of the constituent particles making up the system.
According to the above definition, it may seem that entropy is related to the individual constituent particle of the system. However, thermodynamics, whose framework is based on a macroscopic approach, is not concerned with the existence and the nature of the constituent particles of the system.
Entropy, which is a macroscopic property, is in no way related to the behavior of the individual atoms or molecules of a system, instead, it reflects the average behavior of a large collection of atoms or molecules by which a system usually consists.
Mathematical interpretation of entropy: Since heat (q) is not a state function, the exchange of heat (5q) i.e., the amount of heat absorbed or rejected by a system during a process is not an exact differential.
But in a reversible process, the ratio of the heat exchanged between the system and surroundings \(\frac{\delta q_{r e v}}{T}\) to an absolute temperature at which the heat exchanger takes place is an exact differential. Hence, the quantity indicates the change ofa state function. This function is called entropy (S). Therefore, the change in entropy,
⇒ \(d S=\frac{\delta q_{\text {rev }}}{T}=\frac{\begin{array}{c}
\text { Reversible heat transfer between } \\
\text { system and its surroundings }
\end{array}}{\begin{array}{c}
\text { Temperature (K) at which } \\
\text { heat is transferred }
\end{array}}\)
If, in a reversible process, the state of a system changes from state 1 (initial state) to state 2 (final state), then the change in entropy (AS) in the process can be determined by integrating the equation
⇒ \(\int_1^2 d S=\int_1^2 \frac{\delta q_{r e v}}{T} \text { or, } S_2-S_1=\int_1^2 \frac{\delta q_{r e v}}{T} \text { or, } \Delta S=\int_1^2 \frac{\delta \tilde{q} q_{r e t}}{T}\)
For the process occurring at a constant temperature, the change in entropy, \([\Delta S=\frac{1}{T} \int_1^2 \delta q_{r e v}=\frac{q_{r e v}}{T} .\)
It is not possible to define entropy; however, we can define the change in entropy of a system ( dS or AS) undergoing a reversible process. It is defined as the ratio of reversible heat exchange between the system and its surroundings to the temperature at which the heat exchange takes place.
The relation tells us for a given input of heat into a system, the entropy of the system increases more at a lower temperature than at a higher temperature.
The randomness in a system is a measure of its entropy. The more randomness the more entropy. Therefore, for a given input of heat into a system, the randomness of the system increases more when heat is added to the system at a lower temperature than at a higher temperature.
Characteristics of entropy: Entropy ofa system is a measure of the randomness of its constituent particles. Entropy is a state function because its value for a system depends only on the present state of the system, and its change (AS) in a process depends only on the initial and final states of the system in the process, not on the path followed to carry out the process.
As it is a state function, it is a path-independent quantity. Entropy is an extensive property because its value for a system depends on the amount of matter the system consists of.
The entropy of the universe increases in a spontaneous process (ASuniv > 0) and (ASuniv < 0) for a non-spontaneous process. At equilibrium, ASys = 0. At the absolute zero temperature, the entropy of a pure and perfect crystalline substance is zero.
Physical significance of entropy: There exists a relationship between the entropy of the system and the randomness of its constituent particles (atoms, ions, or molecules).
The entropy of a system increases or decreases with the increase or decrease in randomness of the particles constituting the system. Therefore, entropy is the measure of the randomness of the constituent particles in a system. this is the physical significance of ewntropy.
The change in entropy is defined in terms of a reversible process, for which it is defined as \(d S=\frac{\delta q_{r e v}}{T}\) where 8qrev) Is the reversible exchange of heat between a system and its surroundings at 7’K. in case of an irreversible process, the change in entropy \(d S \neq \frac{\delta q_{i r r}}{T}\) where 5<](rr represents irreversible exchange of heat between a system and its surroundings at 7’K.
As entropy is a state function, its change in a particular process does not depend on the nature of the process. Thus, the change in entropy in a process carried out reversibly is the same as the change in entropy that occurs if the same process is carried out irreversibly.
Unit of entropy: In the CGS system, the unit of entropy = cal.deg-1, while the unit of entropy in SI = J. K-1.
Change In Entropy Of The System In Some Processes
When a system undergoes a process, its change in entropy in the process is AS = S2- S1; where S1 and S2 are the entropies of the initial and final states of the system respectively, in a process.
In a process, if S2 > S1, then AS is positive. This means that the entropy of a system increases in the process. For example, the melting of ice or vaporization of water is associated with an increase in the entropy of the system, so AS is involved in these processes.
If S2 < S1, then AS is negative, indicating that the entropy of the system decreases in the process. For example, when ice is formed from liquid water or water is formed from water vapor, the entropy of the system decreases i.e., AS =-ve.
Change in entropy in a chemical reaction: in any chemical reaction, the initial entropy (S1) of the system means the total entropy of the reactants, and the final entropy of the system (S2) means the total entropy of the products.
Hence, the change in entropy in a chemical reaction, ΔS = \(S_2-S_1^{3 i}=\sum S_{\text {products }}-\sum S_{\text {reactants }} \text {, where, } \sum S_{\text {reactants }}\text { and } \sum S_{\text {products }}\)

Change in entropy of the system in a cyclic process: The change in entropy of the system in any process, AS = S2– S1 where S1 and S2 are the initial and final entropies of the system, respectively. Because entropy is a state function, and in a cyclic process the initial and the final states of a system are the same, = S2, and the change in entropy ofthe system, AS = 0.
Change in entropy of the system in a reversible adiabatic process: In an adiabatic process, heat exchange does not occur between a system and its surroundings. Therefore, in a reversible adiabatic process, qrev = 0 and the change in entropy ofthe system in this process.
⇒ \(d S=\frac{\delta q_{r e v}}{T}=\frac{0}{T}=\mathbf{0} \text { or, } d s=0 \text { or, } \Delta S=0\)
Therefore, the change in entropy of a system undergoing a reversible adiabatic process is zero, i.e., the entropy of a system remains the same in an adiabatic reversible change. Owing to this a reversible adiabatic process is sometimes called an isentropic process.
Change in entropy of the system in an irreversible adiabatic process: Like reversible adiabatic process, heat exchange does not also occur in an irreversible adiabatic process. However, it can be shown that in an irreversible adiabatic process, the entropy change of a system is always positive, i.e., AS > 0.
Change in entropy of the system in an isothermal reversible process: Let us consider, a system changes from state 1 to state 2 in an isothermal reversible process. Therefore, in this process, the change in entropy of the system.
⇒ \(\int_1^2 d S=\int_1^2 \frac{\delta q_{r e v}}{T} \text { or, } S_2-S_1=\frac{1}{T} \int_1^2 \delta q_{r e v} \text { or, } \Delta S=\frac{q_{r e v}}{T}\)
[since T= constant as the process is isothermal]
Change in entropy during a phase transition: Melting of a solid, vaporization of a liquid, solidification of a liquid, condensation of vapor, etc. are some examples of phase transition.
At a particular temperature, a phase transition occurs at a constant pressure. The temperature remains unaltered during the transition although heat is exchanged between the system and the surroundings.
The phase transition can be considered as a reversible process. If qrev of heat is absorbed during a phase transition at constant pressure and 7’K, then the change in entropy, of the system. As the process is occurring at constant pressure
\(q_{\text {re }}=q_p=\Delta H.\).
hence \(\Delta s=\frac{\Delta n}{r}\) \(\Delta s=\frac{\Delta n}{r}\)
Entropy of fusion: It In defined as the change in entropy associated with the transformation of one mole of a solid substance into its liquid phase at its melting point
where ΔHvap= the enthalpy of fusion = die heat required for the transformation of 1 mol of a solid at its melting point into 1 ml of liquid and T1= melting point (K) of the given solid As the fusion of a solid substance is an endothermic process (Le, A> 0 ), the change in entropy due to fusion (ASvap) is always positive.
In the solid phase of a substance, the constituent particles are held in an ordered state. The degree of orderliness is less in the liquid phase as the particles in the liquid have freedom of motion. This is why, when a solid melts, the randomness within the system increases, causing an increase in the entropy of the system.
Example: The enthalpy of fusion of ice at 0°C and 1 atm. Therefore, the change in entropy during the transformation of 1 mol of ice into 1 mol of water at 0°C and 1 atm is-
Entropy of vaporization: It is defined as the change in entropy when one mole of a liquid at its boiling point changes to its vapor phase.
Where AH = the enthalpy of vaporization = the heat required for the transformation of 1 mol of liquid at its boiling point into 1 mol of vapor and Tb = boiling point ofthe liquid (K).
As the vaporization of a liquid is an endothermic process [i.e., \(\Delta H_{v a p}>0\)), the change in entropy in a vaporization process (ASvap) is always positive. When a liquid vaporizes, the molecular randomness in the system increases as the molecules in the vapor phase have more freedom of motion thus they have in the liquid phase. As a result, the vaporization of a liquid always leads increase in the entropy ofthe system.
Example: The enthalpy of vaporization of water at 100°C and 1 atm (AHvap) = 40.4 kj .mol-1. Thus, the change in entropy due to the transformation of 1 mol of water into 1 mol of water vapor at 0°C temperature and 1 atm pressure is
⇒ \(\Delta S_{\text {vap }}=\frac{\Delta H_{\text {vap }}}{T_b}=\frac{40.4 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}}{373 \mathrm{~K}}=108.3 \mathrm{~J} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}\)
Entropy change in an isothermal reversible expansion or compression of an ideal gas
Let, n mol of an ideal gas undergoes an isothermal reversible expansion from its initial state (P1 V1 to the final state (P2, V2). The equation showing the change in entropy of the gas in the process can be derived.
The result of this derivation gives the relation—
⇒ \(\Delta S=n R \ln \frac{V_2}{V_1}=2.303 n R \log \frac{V_2}{V_1}=2.303 n R \log \frac{P_1}{P_2}\)
As the gas expands, V2 > V2 (or, P1> P2 ), so according to the equation [1], the change in entropy (AS) of the gas due to its expansion is positive i.e., the entropy of the system increases.
If the isothermal reversible compression of the same amount of gas causes a change in the state of the gas form (P1 V1 to (P2, V2), then the change in entropy of the gas is given by
⇒ \(\Delta S=n R \ln \frac{V_2}{V_1}=2.303 n R \log \frac{V_2}{V_1}=2.303 n R \log \frac{P_1}{P_2}\)
As the gas is compressed, V2 < V1 (or P2> P1 ), According to equation [2], the change in entropy (AS) of the gas due to its compression is negative, i.e., the entropy of the system decreases.
If the volume of a gas is increased, the gas molecules will get more space for their movement i.e., the gas molecules will move in greater volume. As a result, the randomness of the gas molecules as well as the entropy ofthe system (gas) will increase.
Thus, the entropy of a gas increases with the increase of its volume. On the other hand, the entropy of a gas decreases with the decrease of its volume.
Change in entropy of the surroundings
When a system exchanges heat with its surroundings, the entropy of the system as well as its surroundings changes.
To calculate the change in entropy of the surroundings, the given points are to be considered: Surroundings are so large compared to one system that they serve as a heat reservoir without undergoing any temperature change.
Surroundings absorb or release heat reversibly, and during these processes, the temperature and pressure of the surroundings remain almost the same In a process at TK, if the amount of heat released by the system to the surroundings is sys, then the amount of heat absorbed by tire surroundings = -guys (the sign of q is -ve).
Therefore, in this process, the change in entropy of the surroundings \(\Delta S_{s u r r}=-\frac{q_{s y s}}{T}\)
Hence, the entropy of the surroundings increases if heat is released by the system to the surroundings.
In a process at T K, if the amount of heat absorbed by the system from the surroundings is q, then the amount of heat released by the surroundings will be -qsys (the sign of qsys is +ve ). So, in this process, the change in entropy of the surroundings, \(\Delta S_{\text {surr }}=-\frac{q_{s y s}}{T}.\)
Hence, the entropy of the surroundings decreases if heat is absorbed by the system from the surroundings
Standard entropy change in a chemical reaction
Standard molar entropy of a substance: Entropy of 1 mol of a pure substance at a given temperature (usually 25°C) &1 atm pressure is termed as the standard molar entropy of that substance.
It is denoted by S° and its unit is J. K-1.mol-1.
Standard entropy change in a chemical reaction (ASO): In a chemical reaction, the change in standard entropy, AS0 = total standard entropies of the products – total standard entropies of the reactants i.e., \(\Delta s^0=\sum n_i s_i^0-\sum n_j s_j^0\)
where S0i and Sj0 are the standard entropy of the i -tit product and j -th reactant, respectively. n1 and n2 are the number of moles of the Mil product and i-th reactant, respectively In the balanced equation. In the case of the reaction. \(a A+b B \rightarrow c C+a D\Delta S^0=\left(c S_C^0+d S_b^0\right)-\left(a S_A^0+b S_B^0\right)\)
Change in entropy of the surroundings in a chemical reaction: The change In entropy of the surroundings in a given process,\(\Delta S_{s t u r}=\frac{-q_{s y s}}{T},\) where qÿ is the heat absorbed by the system at 7’K.
In case of chemical reactions occurring at constant pressure \(q_{s y s}=q_P=\) change in enthalpy of the reaction system \(=\Delta H . \mathrm{So}_1, \Delta \mathrm{S}_{\text {surr }}=-\frac{\Delta H}{T}\)
For exothermic reactions, \(\Delta H<0. \text { So, } \Delta S_{\text {surr }}=+v e \text {. }\) I-Ience, the entropy of the surroundings increases In an exothermic reaction.
For endothermic reaction \(\Delta H>0 \text {. So, } \Delta S_{\text {surr }}=-v e \text {. }\) Hence, the entropy of the surroundings decreases in an endothermic reaction.
Change In Entropy And Spontaneity Of A Process
We have seen that there are many spontaneous processes (like the melting of ice above 0°C and 1 atm) in which the entropy ofthe system increases (ASurr > 0). Again, there are some spontaneous processes (such as the transformation of water into ice below 0°C and 1 atm) in which the entropy of the system decreases (A < 0).
Hence, the change in entropy ofthe system alone cannot predict the spontaneity of a process; instead, we must consider the change in entropy of the system as well as that of the surroundings. In a process, if the change in entropy of the system and its surroundings are AS and ASsurr respectively, then the total change in entropy in the process, \(\Delta S_{\text {total }}=\Delta S_{\text {sys }}+\Delta S_{\text {surr }}\)
The system and its surroundings constitute the universe. So, \(\Delta S_{\text {total }}=\Delta S_{\text {univ }}=\Delta S_{s y s}+\Delta S_{\text {surr }}\)
All spontaneous processes occur irreversibly, and in any irreversible process, the entropy of the universe increases. So, for a spontaneous process, \(\Delta S_{\text {univ }}=\left(\Delta S_{\text {sys }}+\Delta S_{\text {surr }}\right)>0\)
In a reversible process, any change in the entropy of the system is exactly balanced by the entropy change in the surroundings. Therefore, in a reversible process.
⇒ \(\Delta S_{\text {sys }}=-\Delta S_{\text {surr }} \text { or, }-\Delta S_{\text {sys }}=\Delta S_{\text {surr }}\)
So, for a reversible process, \(\Delta S_{u n i v}=\Delta S_{s y s}+\Delta S_{s u r r}=0\) When the spontaneous process reaches equilibrium, the value of \(\Delta S_{u n i v}\left(=\Delta S_{s y s}+\Delta S_{\text {surr }}\right)=0.\).
For any spontaneous process, \(\Delta S_{u n i v}\left(=\Delta S_{s y s}+\Delta S_{s u r r}\right)>0\)
For any reversible process, \(\Delta S_{u n i v}\left(=\Delta S_{s y s}+\Delta S_{s u r r}\right)>0\)
At equilibrium ofa process, \(\Delta S_{u n i v}\left(=\Delta S_{s y s}+\Delta S_{s u r r}\right)=0\)

At normal atmospheric pressure, water spontaneously converts into ice below o°C although the entropy change of the system (ASsys) in v.n is negative: H2O molecules in ice are held orderly at fixed positions, which make them unable to move about within ice. On the other hand, H2O molecules in water are not held at fixed positions as in ice and are capable of moving throughout the water.
Therefore, the molecular randomness in water is quite greater than that in ice. Thus entropy ofthe system decreases when water transforms into ice. So, in this process, A < 0.
As this transformation is an exothermic process, heat released by the system is absorbed by the surroundings. As a result, the randomness as well as the entropy ofthe surroundings increases. So, ASys> 0.
However, in this process, the increase in entropy of the surroundings is greater than the decrease in entropy of the system. Consequently, the total change in entropy or the entropy ofthe universe (ASuntv) becomes positive. This favours the spontaneous conversion of water into ice below 0°C temperature and at normal atmospheric pressure.
Change in entropy and condition of spontaneity of a process in an isolated system
As an isolated system does not interact with its surroundings the total energy of sucj a system always remains constant is any process in it.
Therefore, the driving force for any spontaneous process in an isolated system is the change in entropy of the system. In such type of system, since surroundings remain unchanged, \(\Delta S_{\text {surr }}=0\) hence for any spontaneous process occurring in an isolated system,
⇒ \(\Delta S_{s y s}+\Delta S_{s u r r}>0 \text { or }, \Delta S_{s y s}\)
With the progress of a spontaneous process occurring in an isolated system, the entropy of the system gradually increases. When the process reaches equilibrium, the entropy
ofthe system gets maximised, and no further change in its value takes place.
Hence, at equilibrium, of a process occurring in an isolated system, \(S_{\text {sys }}=\text { constant or, } d S_{\text {sys }}=0 \text { or, } \Delta S_{\text {sys }}=0 \text {. }\).
Change in entropy and spontaneity of exothermic and endothermic reactions
Change in entropy and spontaneity of an exothermic reaction: In an exothermic reaction, heat is released by the reaction system. The released heat is absorbed by the surroundings, causing the randomness as well as entropy of the surroundings to increase. Thus, ASsurr is always positive for exothermic reactions. However, the entropy of the system may decrease or increase for such type of reactions.
If the total entropy ofthe products is greater than the total entropy of the reactants in an exothermic reaction, then \(\Delta S_{s y s}>0\) In this case the value \(\Delta S_{u n i v}\) is greater or less than \(\Delta S_{\text {surr }}\). As result, the reaction occurs sponataneouslty.
In an exothermic reaction if the total entropy of the reactants is greater than the total entropy of the products then \(\Delta S_{s y s}<0\). Such type of reactions will be spontaneous if the numerical value of \(\Delta S_{\text {surr }}\) is graeter than \(\Delta S_{s y s}\) because only in this condition \(\Delta S_{\text {univ }}>0\).
Change in entropy and spontaneity of an endothermic reaction: In an endothermic reaction, the heat is absorbed by the system from surroundings. Causing the randomness as well as entropy of the surroundings to decrease. So, in an endothermic reaction, ASJurr < 0. Hence, an endothermic reaction will be spontaneous only when \(\Delta S_{s y s}\) is +ve and its magnitude is greater than that of \(\Delta S_{\text {surr }}\)
Entropy and the second law ot thermodynamic
The second law of thermodynamics is expressed in various ways. One statement of this law is—”All spontaneous processes occur irreversibly and proceed in a definite direction We know that, for any spontaneous or natural changes, the total change in entropy of the system and the surroundings is positive, i.e., for any spontaneous change \(\Delta S_{s y s}+\Delta S_{s u r r}>0.\).
Therefore, in all natural or spontaneous processes, the entropy of the universe continuously increases In other words all natural or spontaneous processes move that direction which leads to the increase in entropy of the universe. Conversely, any process, which does not increase the entropy ofthe universe, will not occur spontaneously.
Second law of thermodynamics in view of entropy For all the natural processes, the entropy of the universe is gradually increasing and approaching to a maximum.
Process occurring in nature are spontaneous. In all these process, energy may be transformed into different forms although the total energy of the universe remains constant But the entropy of the universe does not remain constant It is always increasing due to natural processes.
Numerical Examples
Question 1. Latent heat offusion ofice at 0°C is 6025.24 J-mol-1 Calculate molar entropy of the process at 0°C.
Answer: The change in entropy due to melting of lmol of ice \(=\frac{\text { Molar latent heat (or enthalpy) of fusion of ice }}{\text { Melting point of ice }}\)
⇒ \(=\frac{6025.24 \mathrm{~J} \cdot \mathrm{mol}^{-1}}{(273+0) \mathrm{K}}=22.07 \mathrm{~J} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}\)
Question 2. Enthalpy change for the transformation of water into vapour at the standard boiling point is 40.8 kl . mol-1 . Calculate the entropy change for the process.
Answer: Change in entropy for the process \(\begin{aligned}
& =\frac{\text { Molar enthalpy of vaporisation of water }}{\text { Boiling point of water }} \\
& =\frac{40.8 \times 10^3 \mathrm{~J} \cdot \mathrm{mol}^{-1}}{(273+100) \mathrm{K}}=109.38 \mathrm{~J} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
Question 3. The enthalpy of vaporization of benzene at 80°C (boiling point) is Slkl-mol-1. What will be the change in entropy for the transformation of 31.2 g of benzene vapor into liquid benzene at 80°C?
Answer: \(31.2 \mathrm{~g} \text { benzene }=\frac{31.2}{78}=0.4 \mathrm{~mol}[\text { Molar mass }=78] .\)
Enthalpy of condensation for lmol benzene vapor =(-)x enthalpy of vaporization of lmol liquid benzene =-31 kj. Therefore, the enthalpy of condensation of 0.4 mol of benzene =-31 x 0.4 =-12.4 kj.
∴ The change in entropy for the transformation of0.4 mol of benzene vapor into liquid benzene at 80°C,
⇒ \(\Delta S=-\frac{12.4 \times 10^3 \mathrm{~J}}{(273+80) \mathrm{K}}=-35.127 \mathrm{~J} \cdot \mathrm{K}^{-1}\)
Question 4. 1 mol or on Ideal gas If. expanded from lu Initial: volume of II, lo (lie Hind volume of 1(H) t, ul 25C. What will be (be change In enthalpy for this process?
Answer: \(\Delta S=2.303 n R \log \frac{V_2}{V_1}\)
⇒ \(=2.303 \times 8.314 \log \frac{100}{1}=38.29 \mathrm{~J} \cdot \mathrm{K}^{-1}\)
So, the change in entropy for this process = +398.29j
Question 5. The pressure of 1 mol of an Ideal gas confined In a cylinder fitted with a piston is 50 atm. The gas is expanded reversibly when the cylinder Is kept in contact to a thermostat at 25°C. During expansion, the pressure of the gas is decreased from 90 to 9 atm. Calculate the change in entropy in (Ids process, ff the heat absorbed by the gas during expansion he 9709 f, then calculate the change in entropy of the surroundings?
Answer: we know \(\Delta S=2.303 n R \log \frac{p_1}{p_2}\)
Given, Px = 50 atm, P2 – 5 atm and n = 1
∴ The change in entropy of the system (i.e., gas)
⇒ \(\Delta S=2.303 \times 8.314 \log \frac{50}{5}=19.15 \mathrm{~J} \cdot \mathrm{K}^{-1}\)
Here surroundings are at a fixed temperature (25°C). During expansion, the heat absorbed by the gas from the surroundings = 5705 J. Therefore, at 25’C, the heat released by the surroundings =-5705 j.
∴ Change in entropy of the surroundings,
⇒ \(\Delta S_{\text {surr }}=-\frac{5705}{(273+25)} \mathrm{J} \cdot \mathrm{K}^{-1}=-19.14 \mathrm{~J} \cdot \mathrm{K}^{-1}\)
Question 6. At 1 atm and 298 K, entropy change of the reaction, \(4 \mathrm{Fe}(s)+3 \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{Fe}_2 \mathrm{O}_3(s) \text { is }-549.4 \mathrm{~J} \cdot \mathrm{K}^{-1} \text {. }\) In this reaction, if A// = -1648 kj, then predict whether the reaction is spontaneous or not.
Answer: As the reaction enthalpy is negative so the reaction is exothermic. Therefore, the heat released by the given reaction will be equal to the heat absorbed by the surroundings Consequently the entropy of the surroundings will increase. Heat absorbed by the surroundings =(-) x heat released by the system =-AH = -(-1648)kJ = +1648 kj.
∴ Change in entropy of the surroundings
⇒ \(\Delta S_{\text {surr }}=-\frac{\Delta H}{T}=\frac{1648 \times 10^3}{298} \mathrm{~J} \cdot \mathrm{K}^{-1}=5530.2 \mathrm{~J} \cdot \mathrm{K}^{-1}\)
Thus, in this reaction \(\Delta S_{\text {untv }}=\Delta S_{\text {sys }}+\Delta S_{\text {surr }}\)
= (-549.4 + 5530.2)J-K-1 = +4980.8 J-K-1.
Since \(\Delta S_{u n I v}>0\) the reaction will occur spontaneously.
Question 7. At 1 atm and 298 K, AH0 value for the reaction \(2 \mathrm{H}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{H}_2 \mathrm{O}(l) \text { is }-572 \mathrm{~kJ}\) . Calculate the change in entropy ofthe system and surroundings for this reaction. Is this reaction spontaneous at that temperature and pressure? Given: Standard molar entropies of H2(g), O2(g) & H2O(f) at 298K are 130.6, 205.0, and 69.90 J. K-1. mol-1 respectively
Answer: The change in entropy ofthe given reaction
⇒ \(\begin{aligned}
& \Delta S_{s y s}^0=\left[2 S^0\left(\mathrm{H}_2 \mathrm{O}, l\right)\right]-\left[2 \times S^0\left(\mathrm{H}_2, g\right)+S^0\left(\mathrm{O}_2, g\right)\right] \\
&=[2 \times 69.9-(2 \times 130.6+205)] \mathrm{J} \cdot \mathrm{K}^{-1} \\
&=-326.4 \mathrm{~J} \cdot \mathrm{K}^{-1}
\end{aligned}\)
Change in entropy ofthe surroundings in the reaction
⇒ \(\Delta S_{\text {surr }}=-\frac{\Delta H^0}{298}=\frac{572 \times 10^3}{298} \mathrm{~J} \cdot \mathrm{K}^{-1}=+1919.4 \mathrm{~J} \cdot \mathrm{K}^{-1}\)
∴ Total change in entropy,
⇒ \(\begin{aligned}
\Delta S_{\text {univ }}=\Delta S_{\text {sys }}+\Delta S_{\text {surr }} & =-326.4+1919.4 \\
& =+1593 \mathrm{~J} \cdot \mathrm{K}^{-1}
\end{aligned}\)
Since \(\Delta S_{u n i v}>0,\) then the reaction will occur spontaneously at 298 K and 1 atm.
Question 8. The molar enthalpy of fusion and the molar entropy of fusion for ice at 0°C and 1 atm are 6.01 kj- mol-1 and 22.0 J K-1 .mol-1, respectively. Assuming AH and AS are independent of temperature, show that the inciting of ice at 1 atm is not spontaneous, while the reverse process is spontaneous.
Answer: A process is spontaneous when the change in entropy of the universe \(\left(\Delta S_{u n i v}=\Delta S_{s y s}+\Delta S_{s u r r}\right)\) is positive. The transformation ofice into water involves the process.
⇒ \(\begin{aligned}
\mathrm{H}_2 \mathrm{O}(s) & \rightarrow \mathrm{H}_2 \mathrm{O}(l) \\
\Delta H & =6.01 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} ; \Delta S=22.0 \mathrm{~J} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
⇒ \(\begin{aligned}
\text { Now, } \Delta S_{\text {sys }} & =22.0 \mathrm{~J} \cdot \mathrm{mol}^{-1} \\
\Delta S_{\text {surr }} & =\frac{\Delta H}{T}=\frac{-6010}{271}=-22.17 \mathrm{~J} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
⇒ \(\text { Thus, } \begin{aligned}
\Delta S_{\text {univ }}=\Delta S_{\text {sys }}+\Delta S_{\text {surr }} & =22.0+(-22.17) \mathrm{J} \cdot \mathrm{mol}^{-1} \\
& =-0.17 \mathrm{~J} \cdot \mathrm{mol}^{-1}
\end{aligned}\)
Hence, the total entropy change in the process is negative at 271 K. Therefore, the transformation of ice into water at -2°C is not spontaneous. The reverse process i.e., the conversion of water to ice at -2°C is spontaneous. This is because at -2°C ans1 atm pressure the overall entropy (ASuniv) is +0.17 J . mol-1.
Gibbs Free Energy Or Free Energy
In the previous section, we have seen that entropy can be used as a criterion for the spontaneity of a process. In an isolated system, a process will be spontaneous if ASÿ is positive during this process.
But natural processes seldom occur in isolated systems. In other systems, a process will be spontaneous if \(\Delta S_{\text {universe }}\left(=\Delta S_{s y s}+\Delta S_{s u r t}\right)\) is positive. There are many are quite inconvenient.
It is, thus, very useful to reformulate the spontaneity criterion in such away that only the system is to be considered. For us purpose, J. ‘Willard Gibbs introduced a new thermodynamic function called Gibbs free energy” or free energy, denoted by‘ G —immerse processes in which determination of ASt At constant temperature and pressure, the spontaneity ofa process can be determined from the value ofthe change in Gibbs free energy of the system.
As most of the physical or chemical changes occur at constant pressure, it is convenient to use the concept of free energy to determine the spontaneity of that change.
Definition Of Gibbis free energy It is the thermodynamic property of a system, whose decrease in a spontaneous process at constant temperature and pressure, measures the maximum useful energy obtainable in the form of work from the process.
In any spontaneous process occurring at constant temperature and pressure, the decrease in Gibbs free energy (-ΔG) = maximum useful or network performed bythe system on the surroundings.
Mathematical form of Gibbs free energy: Of the total energy of a system, one part is free for doing useful work, and another part is unavailable, which cannot be converted into work. Ifthe value of entropy of a system is S at T K, then the amount of unavailable energy of the system is TxS.
Therefore, the total energy of the system = G (free energy) + TS (unavailable energy) Generally enthalpy (H) is considered as the total energy of the system. Thus, H = G+TS or, [G = H-TS]
Where G, H, and S are Gibbs free energy, enthalpy, and entropy ofthe system respectively. T is the temperature of the system in the Kelvin scale. Equation [1] is the mathematical form of Gibbs free energy or free energy.
Gibbs free energy is a state function: Gibbs free energy (G), enthalpy (H) and entropy (S) of a system are related by the equation, G = H-TS. As H. S and T are state functions, Gibbs free energy (G) is also a state function.
Thus, the value of Gibbs free energy (G) of any system depends only on the present state of the system and not on how the system has reached its present state. Hence, the change in Gibbs free energy (AG) of any process doesn’t depend upon the nature of the process but depends only on the initial and final states ofthe process.
Gibbs free energy is an extensive property: Enthalpy (H) and the product of entropy and absolute temperature (fxS) of the system depends on the amount of the substance present in the system.
With increasing amounts of the substance, values ofthese quantities are increased. Hence Gibbs free energy (G = H- TS) depends on the amount of the substance present in the system. Therefore, Gibbs free energy (G) is an extensive property of the system.
Change in Gibbs free energy in a process occurring at constant temperature and pressure: The change in Gibbs free energy in a process occurring at a constant temperature and pressure, ΔG = Δ(H-TS) = ΔH-A(TS)
∴ ΔG = ΔH- TAS) [Δ(TS) = TAS as T = constant] The above equation represents the relation between the entropy change (AS), enthalpy change (AH), and absolute temperature (T) for a physical or chemical change occurring at a particular temperature and pressure. Using this equation, it is possible to predict the spontaneity of a process at constant temperature and pressure.
Change in Gibbs free energy for a physical or chemical change and process
At a particular temperature (T) and pressure (P), if the change in enthalpy and change in entropy for a physical or chemical process are AH and AS, respectively, then the change in Gibbs free energy, ΔG = ΔH -TAS…[1]
If ASys and ASsrr are the changes in entropies of the system and its surroundings, respectively, then the total change in entropy for the process \(\Delta S_{\text {total }}=\Delta S_{\text {univ }}=\Delta S_{\text {sys }}+\Delta S_{\text {surr }}\) The heat gained by the system at fixed pressure and temperature, qsys= change in enthalpy (ΔH). So sys=ΔH
Therefore, the heat lost by the surroundings at a fixed pressure and temperature \(\left(q_{\text {surr }}\right)=-q_{\text {sys }}=-\Delta H.\)
So, the change in entropy ofthe surroundings for a physical or chemical change at a particular temperature and pressure, \(\Delta S_{\text {surr }}=\frac{q_{\text {surr }}}{T}=-\frac{\Delta H}{T}\) and the total change in entropy at a particular temperature and pressure,
⇒ \(\begin{aligned}
& \Delta S_{\text {total }}=\Delta S_{\text {sys }}+\Delta S_{\text {surt }}=\Delta S-\frac{\Delta H}{T} \quad\left[\text { where } \Delta S=\Delta S_{\text {sys }}\right] \\
& \text { or, } T \Delta S_{\text {total }}=T \Delta S-\Delta H \text { or, } \Delta H^{\prime}-T \Delta S=-T \Delta S_{\text {total }} \quad \cdots[2]
\end{aligned}\)
Comparing equations [1] and [2], \(\Delta G=-T \Delta S_{\text {total }} \cdots\)
Equation [3] represents the relation between changes in Gibbs free energy (AG) for a physical or chemical change at a fixed temperature and pressure and the total change in entropy of the system and surroundings \(\left(\Delta S_{\text {total }}\right)\) for that process.
We know that a process will be spontaneous if the total change in entropy of the system and surrounding in the process is positive; i.e., \(\Delta S_{\text {total }}\left(=\Delta S_{\text {sys }}+\Delta S_{\text {surr }}\right)>0.\) According to equation [3], \(\text { if } \Delta S_{\text {total }}>0 \text {, then } \Delta G<0 \text {. }\)
Thus, a physical or chemical change at a fixed temperature and pressure will be spontaneous ifthe change in Gibbs free energy (AG) is negative i.e., AG < 0.
If in a process, the total change in entropy ofthe system and its surroundings is -ve, i.e., \(\Delta S_{\text {total }}\left(=\Delta S_{s v s}+\Delta S_{s u r r}\right)<0,\) then the process will be non-spontaneous, but the reverse
process will be spontaneous. From equation [3], if \(\Delta S_{\text {total }}<0 \text {, then } \Delta G>0 \text {. }\) Thus, for a physical or chemical
change at a fixed pressure and temperature, if the Gibbs free
change is positive (AG > 0), then the process will be nonspontaneous but the reverse one will be spontaneous.
Total change in entropy of the system and surroundings will be zero, i.e., \(\Delta S_{\text {total }}\left(=\Delta S_{\text {sys }}+\Delta S_{\text {surr }}\right)=0\) for a process at equilibrium. According to equation [3], if \(\Delta S_{\text {total }}=0 \text { then } \Delta G=0\). Therefore, the Gibbs free energy change will be zero, i.e., AG = 0 for a physical change or chemical reaction at equilibrium under the condition ofa fixed pressure and temperature.
Reaction; A→ B (at constant and pressure)
If &G(=GB- GA) <0 at a fixed temperature and pressure; then the transformation of A to B will be spontaneous.
If AG(= GB- Ga) > 0 at a fixed temperature and pressure; then the transformation of A to B will be non-spontaneous. But the transformation of B to A will be spontaneous as GB > GA and for the reverse process the value of AG(= GA- GB) is negative.
If AG(=Gb- Ga) = 0, then A and B will be in equilibrium. In this condition, the rate of transformation of B into A or A into B will be the same. So no net change will occur either in the forward or in the reverse direction.


Effect of temperature on the change in Gibbs free energy and the spontaneity of a process
We know that AG = AH- TAS. In this relation, the values of both AH and AS may be either positive or negative, but the temperature (in the Kelvin scale) is always positive. Again TAS is a temperature-dependent quantity.
With the increase or decrease in temperature, the magnitude of TAS increases or decreases, and the value of AH almost remains unchanged. So, AG depends on temperature. From the signs of AH and AS, we can predict the effect of temperature on AG. The different possibilities have been discussed in the following table.

Numerical Examples
Question 1. \(\mathrm{Br}_2(l)+\mathrm{Cl}_2(\mathrm{~g}) \rightarrow 2 \mathrm{BrCl}(\mathrm{g})\); Whether the reaction is spontaneous or not at a certain pressure & 298 K ? [Aff=29.3 kj.mol-1, AS=104.1 J. K-1.mol-1 ]
Answer: We know, ΔG = ΔH- ΔTS
∴ ΔG =29.3 xl03-(298x 104.1) =-1721.8 J- mol-1
As ΔG is negative, this process is spontaneous.
Question 2. At a certain pressure and 27°C, the values of ΔG and ΔH ofa the process are- 400 kj and 50 kj respectively. Is the process exothermic? Is it spontaneous? Determine the entropy change of the process.
Answer: As per given data AH = 50 kj. As the value of ΔH is positive, it is not an exothermic process.
For the process ΔG =-400 kj at a certain pressure and 27°C. As AG is negative, it is a spontaneous process.
ΔG = ΔH- TΔS
∴ -400 = 50-300xΔS
or, ΔS=1.5Kj.K-1=1500j.k-1
Question 3. Values of ΔH to ΔS for the given reaction are -95.4kJ and -198.3 J-K_1 respectively: \(\mathrm{N}_2(g)+3 \mathrm{H}_2(g) \rightarrow 2 \mathrm{NH}_3(g)\) State whether the reaction will be spontaneous at 500 K or not. Consider AH and AS are independent of temperature.
Answer: We know, ΔG = ΔH- TΔS
Given, ΔH = -95.4 kj , ΔS = -198.3 J.K-1 and T = 500 K.
∴ ΔG= [-95.4 X 103- 500 X (-198.3)]J = 3750 J = 3.75 kj
As the value of ΔG is positive at constant pressure and 500 K temperature, it is not a spontaneous process.
Question 4. In and the ASreaction,= + 35 JA(s). K-1+. State B(g)-C(g)whether+ D(g)the reaction, AH =31 will k-1 be spontaneous at 100°C and 1100°C or not? Consider AH and AS are independent of temperature.
Answer: We know, AG = AH- TAS. Now at 100°C,
ΔG = ΔH-TAS =31 x 103- (273 + 100) X 35 =+ 17945J
∴ At 1100°C, =31 X 103-(273 + 1100) X 35 =-17055 J
At 100°C, AG for the given reaction is positive, so the reaction will be non-spontaneous. On the contrary, at 1100°C, AG for the given reaction is negative, so it will be spontaneous.
Question 5. Is the vaporization of water at 50°C and 1 atm spontaneous? Given: For vaporization of water at that temperature and pressure, AH = 40.67 kj.mol-1 and AS = 108.79 J.K-1.mol-1.
Answer: We know, ΔG = ΔH- TΔS
∴ ΔG=ΔH- TΔS = 40.67 X 103- (323 X 108.79)
= + 5530.83 J.mol-1
As ΔG = +ve, so vaporization will be non-spontaneous.
Question 6. At 25°C and 1 atm, the heat of formation of 1 mol of water is -285.8 kj. mol-1. State whether the formation reaction will be spontaneous at that temperature and pressure or not. Given: The molar entropies of H2(g) , 02(g) & H20(Z) at 25°C and 1 atm are 130.5, 205.0 and 69.9 J.K1.mol-1 respectively.
Answer: Equation for the formation reaction of water:
⇒ \(\mathrm{H}_2(g)+\frac{1}{2} \mathrm{O}_2(g) \rightarrow \mathrm{H}_2 \mathrm{O}(l) ; \Delta H=-285.8 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
The change in entropy for this reaction, \(\Delta S=S_{\mathrm{H}_2 \mathrm{O}(l)}-\left(S_{\mathrm{H}_2(g)}+\frac{1}{2} S_{\mathrm{O}_2(g)}\right)\)
\(=\left[69.9-130.5-\left(\frac{1}{2} \times 205\right)\right] \mathrm{J} \cdot \mathrm{K}^{-1}=-163.1 \mathrm{~J} \cdot \mathrm{K}^{-1}\)
Therefore, the change in Gibbs free energy at 25°C and 1 atm for the formation of1 mol of H20(Z) from 1 mol of H2(g) and \(\text { and } \frac{1}{2} \mathrm{~mol} \text { of } \mathrm{O}_2(\mathrm{~g}), \Delta G=\Delta H-\mathrm{T} \Delta S\) = -285.8 X 103- 298 X (-163.1) =-237.196 kj As AG is negative at 25°C and 1 atm, so the formation of water at this temperature and pressure will be spontaneous.
Determination of temperature at which equilibrium is established in a physical or chemical change
We know that at a given temperature and pressure the change in free energy (AG) for a reaction is zero when the reaction is at equilibrium. Therefore, at equilibrium,
⇒ \(\Delta G=\Delta H-T \Delta S=0 \text { or, } \mathbf{T}=\frac{\Delta \boldsymbol{H}}{\Delta S} \cdots[1]\)
Applying equation no. [1], we can determine the temperature at which equilibrium is established in a physical or chemical change.
Example: The values of the enthalpy and entropy changes ofthe system are + 40.7 kj- mol-1 and 109.1 J. K-1.mol-1 respectively for the process, H2O(Z) H2O(g) at 1 atm pressure. At which temperature equilibrium will be established between water and water vapour?
Answer: At equilibrium AG = 0 and T \(=\frac{\Delta H}{\Delta S} .\)
Given: ΔH = 40.7 kj.mol-1 = 40.7 x 103 J.mol-1 and AS = 109.11 -K-1- mol-1
∴ \(T=\frac{40.7 \times 10^3 \mathrm{~J} \cdot \mathrm{mol}^{-1}}{109.1 \mathrm{~J} \cdot \mathrm{K}^{-1} \cdot \mathrm{mol}^{-1}}=373 \mathrm{~K}\)
373 K (100°C) is the normal boiling point of water. So, at 1 atm and 100°C, water and its vapor will remain in equilibrium.
The temperature at which a physical or chemical change becomes spontaneous We have seen. that in any physical or chemical change, ifthe signs of AH and AS are the same (either + or – ), then the sign of AG as well as the spontaneity of that change depends on temperature.
Suppose, for a physical or chemical change at a particular temperature and pressure, ΔH > Δ and AS > Δ. According to the relation, ΔG = ΔH- TΔS the process is spontaneous because when TΔS > ΔH, AG<Δ This means that ΔH- TΔS < 0 or TΔS > ΔH thus, \(T>\frac{\Delta H}{\Delta S}\)
Hence, the process will be spontaneous when \(T>\frac{\Delta H}{\Delta S}\), if \(T<\frac{\Delta H}{\Delta S}\) AS the process will be non-spontaneous, but the reverse process will be spontaneous. For a physical or chemical change if AH < 0 and AS < 0, then it can be shown that the process will be spontaneous when \(T<\frac{\Delta H}{\Delta S}. \text { If } T>\frac{\Delta H}{\Delta S}\) the process will be non-spontaneous, but the reverse process will be spontaneous.
Numerical Examples
Question 1. In the reaction, A-B+ C, ΔH = 25 kj. mol-1 and AS = 62.5 J.K-1. At which temperature the reaction will occur spontaneously at constant Pressure?
Answer: The condition for the spontaneity of a reaction at a given temperature & pressure is ΔG < 0. We know ΔG = ΔH- TΔS For spontaneous reaction, ΔH- TΔS < 0 or, \(\Delta H<T \Delta S \quad \text { or, } T \Delta S>\Delta H \quad \text { or, } T>\frac{\Delta H}{\Delta S}\) Given: ΔH = 25 x 103 J and AS = 62.5 J . K-1
therefore \(T>\frac{25 \times 10^3 \mathrm{~J}}{62.5 \mathrm{~J} \cdot \mathrm{K}^{-1}} \quad \text { or, } T>400 \mathrm{~K} \text {. }\)
Question 2. H2O(g) H2O(l) ; Δh = -40.4 kj.mol-1 ΔS = -108.3 J.K-1.mol-1. At which temperature the process will be spontaneous at a constant 1 atm?
Answer: In this process ΔH<0 and ΔS <0. For such type of process, the temperature at which the reaction will occur spontaneously \(T<\frac{\Delta H}{\Delta S}\)
Given: ΔH=-40.4 x 103 J mol-1, ΔS=-108.3 J.K-1.mol-1
∴ \(T<\frac{\Delta H}{\Delta S} \quad \text { or, } T<\frac{40.4 \times 10^3}{108.3} \quad \text { or, } T<373 \mathrm{~K}\)
∴ The process will be spontaneous below 373 K (100°C).
Question 3. H2(g)+Br2(Z)-+2HBr(g); AH=-72.8kJ (1 atm, 25°C) If molar enterpoies of H2(g), Br2(l),HBr(g) are 130.5, 152.3 and 198.3j.k-1.mol-1 respectively then at which temperature the reaction will be spontaneous?
Answer: Change in entropy for the given reaction,
⇒ \(\begin{aligned}
\Delta S= & 2 S_{\mathrm{HBr}(g)}-\left[S_{\mathrm{H}_2(g)}+S_{\mathrm{Br}_2(l)}\right] \\
& =[2 \times 198.3-(130.5+152.3)] \mathrm{J} \cdot \mathrm{K}^{-1}=113.8 \mathrm{~J} \cdot \mathrm{K}^{-1}
\end{aligned}\)
As AH < 0 and AS > 0 for the given reaction, the reaction will be spontaneous at any temperature.
Question 4. For the reaction A(g) + B(g)→ C(s) +D(l); AH =-233.5 kj and AS = -466.1 J. K-1. At what temperature, equilibrium will be established? In which directions the reaction will proceed above and below that temperature?
Answer: At constant temperature and pressure, the equilibrium temperature ofa reaction \(T=\frac{\Delta H}{\Delta S}.\)
Given: AH = -233.5 x 103 J and AS = -466.1 J .K-1
Therefore \(T=\frac{233.5 \times 10^3}{466.1} \mathrm{~K}=500.9 \mathrm{~K}=227.9^{\circ} \mathrm{C}\)
∴ At 227.9°C, the reaction will attain equilibrium.
When T > 500.9 K, the magnitude of TAS is greater than that of AH. Then according to the equation, AG = AH- TAS, the value of AG will be positive. So the reaction will be non-spontaneous above 500.9 K.
When T < 500.9 K, the magnitude of TAS is less than that of AH. According to the equation, AG = AH- TAS, AG will be negative. Therefore, the reaction will be spontaneous below 500.9 K
The standard free energy of formation of a substance and the standard free energy change in a chemical reaction
Standard free energy of formation: The standard free energy of formation of a compound is denoted by \(\Delta G_f^0\) and its unit is kj.mol-1 (or kcal-mol-1).
Definition: The free energy change associated with the formation of 1 mol of a pure compound from its constituent elements present at standard state is termed as the standard free energy of formation of that compound.
The value of standard free energy of formation of any element at 25°C and 1 atm pressure is taken as zero. For the elements having different allotropic forms, the standard free energy of formation of the most stable allotrope at 25°C and 1 atm pressure is taken as zero. For example,\(\left(\Delta G_f^0\right)\) [C (graphite)] = 0 but \(\left(\Delta G_f^0\right)\) [C dimond] ≠0.
Standard free energy change in a chemical reaction: In any reaction, the change in free energy (AG) depends on temperature, pressure, and concentration. Thus to compare the AG of different reactions, the standard free energy change is calculated.
Definition: it is defined as the change in free energy when the specified number of moles of reactions (indicated by the balanced chemical equation] in their standard states are completely converted to the products in their standard states.
It is expressed by ΔG0. At a particular temperature, the standard free energy change in a reaction is calculated from the standard free energy of formation ofthe reactants and products at that temperature.
The change in standard free energy in a reaction, AG = the sum of the standard ‘free energy of formation of the products – the sum of the standard free energy of formation.
Wham is the number of moles of t -th product and j-th reactant In the balanced chemical equation whereas formation of the i-th product and j-th reactant respectively.
Example: in the case of the reaction aS+bB-cC+dD:
⇒ \(\begin{aligned}
& \Delta G^0=\left[c \times \Delta G_f^0(C)+d \times \Delta G_f^0(D)\right]- \\
& \quad\left[a \times \Delta G_f^0(A)+b \times \Delta G_f^0(B)\right]
\end{aligned}\)
The standard free energy change (ΔG°) in a reaction can also be determined from the values of the standard change enthalpy (ΔH0) and standard change in entropy (ΔS0), using the following equation,
⇒ \(\Delta G^0=\Delta H^0-T \Delta S^0\)
ΔG0 =ΔH0-TΔS0 …[1]
ΔG0 and the spontaneity of a physical or chemical change: If ΔG° < 0 for any physical or chemical change, then the process will be spontaneous under standard conditions.
If the value of ΔG° > 0 for any physical or chemical change, then the process will be nonspontaneous under standard conditions. However, the reverse process will be spontaneous under standard conditions.
Free energy change in a chemical reaction, reaction equilibrium, and equilibrium constant
ΔG° in a reaction can be determined either from the values of the standard free energies of formation of the participating reactants and products or from the equation, ΔG° ΔAH0- FΔS0. But if a reaction occurs in a condition other than standard condition then the free energy change of the reaction can be calculated using the following relation.
⇒ \(\Delta G=\Delta G^0+R T \ln Q\)
where Q = reaction quotient [a detailed discussion on reaction quotient has been made, T = temperature in Kelvin scale, I? = the universal gas constant, ΔGs the free energy change in a reaction at constant pressure and a constant temperature FK, AG°= the standard free energy change in the reaction at TK
Relation between standard free energy change (AG°) and equilibrium constant (K The relation between AG° and K can be derived from the above equation [1] For a reaction at equilibrium, AG = 0 at constant temperature and pressure. Also at equilibrium, the reaction quotient (Q) = equilibrium constant (JC). Therefore, according to the equation [1], at equilibrium
∴ 0=ΔG0 +RT in K-1
or, ΔG-RT in k……[2]
Or, ΔG0= -2.03 RT Log K ……[3]
Equations [2] and [3] show the relationship between the standard free energy change in a reaction (ΔG0) and the equilibrium constant (K) of the reaction at a particular temperature (T). Hence, using these equations [2 and 3], the value of the equilibrium constant (K) of a reaction at a particular temperature (F) can be determined from the value of the standard free energy change of the reaction (ΔG0) at the same temperature (T). Alternatively, the value of ΔG° can be determined from the value of K by using equations [2] and [3]. According to equation [2] (or [3]);
- If AG° is negative, then In K (or logic) will be positive. Thus K > 1
- If AG° is positive, then In K (or logic) will be negative Thus K < 1.
- If AG° is equal to zero, then In K (or logic) will be equal to zero. Thus K = 1.
Numerical Examples
Question 1. In the given reaction, calculate the standard free energy change at 25°C: \(\mathrm{N}_2(g)+3 \mathrm{H}_2(g) \rightarrow 2 \mathrm{NH}_3\) [Given that, AH0 = -91.8 kj and AS0 = -198 J. K-1 ]
Answer: We know, AG° = AH0- TAS°
AH° = -91.8 kJ , AS°=-198 J.K-1 , F = (273 +25) = 298K
AG° =-91.8 xl03- 298 x (-198) =-32796J=-32.796kJ
Question 2. In the given reaction, calculate standard free energy change at 25°C: 2NO(g) + 0,(g) 2NO2(g). Is the reaction spontaneous under standard conditions?
Answer: \(\left[\right. Given: At 25^{\circ} \mathrm{C}, \Delta G_f^0[\mathrm{NO}(\mathrm{g})]=86.57 \mathrm{~kJ} \cdot \mathrm{mol}^{-1} and \Delta G_f^0\left[\mathrm{NO}_2(g)\right]=51.30 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}.\)
Answer: \(\left.\Delta G^0=\sum \Delta G_f^0 \text { (products }\right)-\sum \Delta G_f^0(\text { reactants })\)
In the given case,
⇒ \(\begin{aligned}
& \Delta G^0=2 \Delta G_f^0\left[\mathrm{NO}_2(g)\right]-2 \Delta G_f^0[\mathrm{NO}(g)]-\Delta G_f^0\left[\mathrm{O}_2(g)\right] \\
& \text { Given: } \left.\Delta G_f^0[\mathrm{NO}(g)]=86.57 \mathrm{k}\right] \cdot \mathrm{mol}^{-1},
\end{aligned}\)
⇒ \(\Delta G_f^0\left[\mathrm{O}_2(\mathrm{~g})\right]=0 \text { and } \Delta G_f^0\left[\mathrm{NO}_2(\mathrm{~g})\right]=51.30 \mathrm{~kJ} \cdot \mathrm{mol}^{-1}\)
[∴ Standard free energy of formation of an element =0]
Since the value of ΔG° is negative, so the reaction will be spontaneous under standard conditions.
Question 3. At 25°C, the standard free energy change for a reaction is 5.4 kj. Calculate the value of the equilibrium constant of the reaction at that temperature.
Answer: We know, AG° = -RTlnK.
∴ We have, AG° = 5.4 kJ = 5.4 x 103 J and T = 298 K
or, log k = 0.95
∴ K= 0.113
Therefore, at 25°C the value of the equilibrium constant for the given reaction will be 0.113.
Question 4. The equilibrium constant for a reaction is 1.6 x 10-G at 298K. Calculate the standard free energy change (AG°) and standard entropy change (AS0) of the reaction at that temperature. Given, at 298 K, AH0 = 25.34 kj.
Answer: We know. AG° = -RTinK
∴ AG° =-8.314 X 298 ln(1.6 x 10-6) = 33064J = 33.064 kj
∴ The standard free energy change at 298 K = + 33.064 kj
Again we know, ΔG°=ΔH°- TAS0. Here, ΔH0=25.34 kj
∴ +33.064 = 25.34- 298 X ΔS0
So, the standard entropy change for the reaction = 25.9j. k-1.
Question 5. Al 298 K, the standard free energy of formation of H2O(/) 8-287.19 kt mol. calculate the value of equilibrium constant temperature for the following reaction: \(2 \mathrm{H}_2 \mathrm{O}(t) \rightarrow 2 \mathrm{H}_2(g)+\mathrm{O}_2(g)\)
Answer: standard free energy change for the given reaction,
⇒ \(\Delta G^0=2 \Delta G^0\left[\mathrm{H}_2(g)\right]+\Delta G^9\left[\mathrm{O}_2(g)\right]-2 \Delta G^9\left\{\left[\mathrm{H}_2 \mathrm{O}(l)\right]\right.\)
The standard free energy of formation of an element Is
taken as zero, so \(\Delta G_f^0\left[\mathrm{O}_2(g)\right]=0 \text { and } \Delta G_j^9\left[\mathrm{H}_2(g)\right]=0 \text {. }\)
Therefore \(\left.\left.\Delta G^0=[0+0-2 \times(-237.13)] \mathrm{k}\right]=+474.25 \mathrm{k}\right]\)
∴ +474.26×103=-8.314×298 in k or k = -191.42
∴ K= 7.36×10-84
So, the equilibrium constant for the reaction =7.36×10-84
The Third Law Of Thermodynamics
Statement of the third law of thermodynamics Entropy of a pure and perfectly crystalline substance at 0 K temperature is equal to zero.
Explanation: In a perfect crystalline solid, all the constituent particles are perfectly arranged in a well-ordered manner. Defects like point defects, line defects, etc., that can generally be observed in crystal lattices, are found to be absent in a perfect crystal.
The constituent particles become motionless and attain the lowest energy in such as substance at absolute zero. As a result, the randomness of those constituent particles becomes zero. For this reason, the value of entropy of a pure and perfectly crystalline substance is zero.
Explanation of thermodynamic probability: A system can achieve a particular thermodynamic state in various ways or configurations. The number of ways in which a particular state can be achieved is called thermodynamic probability and is designated by the symbol ‘W’.
With increasing thermodynamic probability the randomness as well as the entropy of the system increases. So we can expect that there exists a relationship between the two quantities S and W. Lauding Boltzmann introduced a relation between S and W. The relation is S = k InW; where k- Boltzmann constant.
At absolute zero, all the constituent particles would occupy the minimum energy state, and hence there is only one way of arranging the constituent particles in different energy levels i.e., W = 1. This gives S = 0. So, for a pure and perfect crystalline substance, the value of entropy is zero at zero kelvin. This is the statement of the third law of thermodynamics.
Application of the third law of thermodynamics: The absolute value of entropy can be determined by making use of this law. If the increase in entropy of a substance is AS due to an increase in temperature from OK to TK then AS = ST- SQ; where, ST and SQ are the entropies of that substance at TK, and 0 K, respectively. According to the third law of thermodynamics, SQ = 0. Therefore, S = ST. So by measuring AS, it is possible to determine the absolute value of entropy of a substance at T K.
Heat or enthalpy of neutralization
Heat or enthalpy of neutralization Definition: The change in enthalpy that occurs when 1 gram equivalent of an acid is completely neutralized by 1 gram equivalent of a base or vice-versa in a dilute solution at a particular temperature is called the enthalpy (or heat) of neutralization.
The change in enthalpy that occurs when 1 mol of H+ ions reacts completely with mol of OH- ions in a dilute solution to form 1 mol water at a particular temperature is known as the Enthalpy (or heat) of neutralization.
The enthalpy of neutralization is denoted as AHN, where subscript TV ‘indicates ‘neutralization’.
Neutralization of strong acid and strong base: if both the acid and base are strong, then the value of heat of neutralization constant, is found to be almost and this value is ~57.3 kj.









































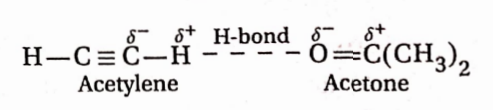
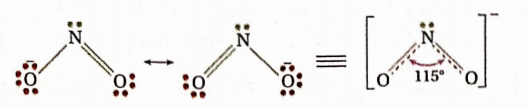
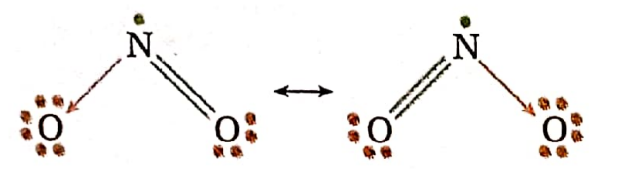
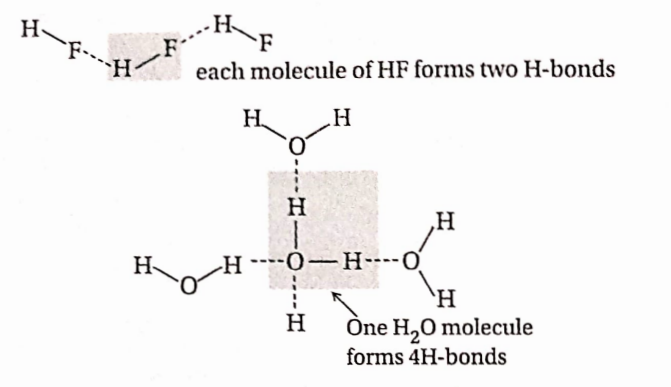
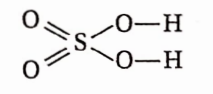
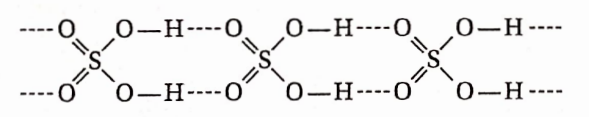
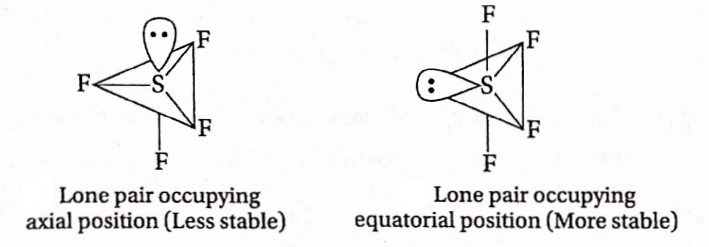
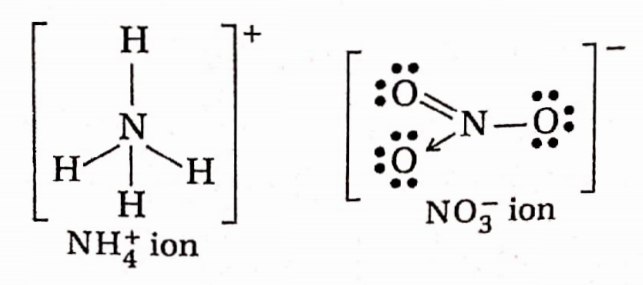
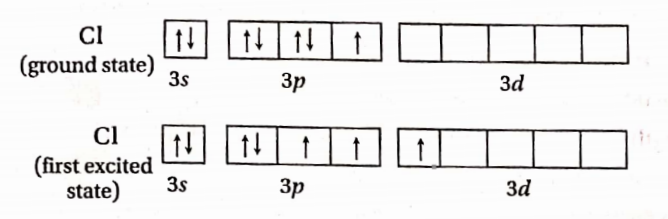
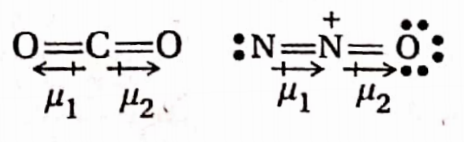
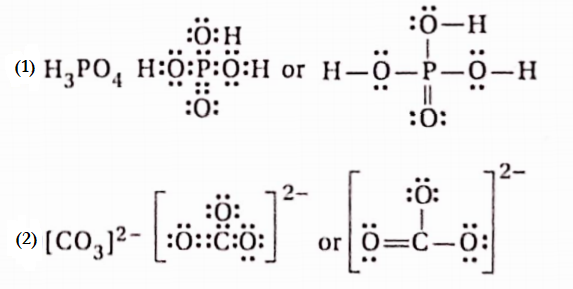
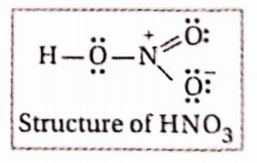


































![Chemical Thermodynamic Standard Enthalpy Of Dissociation [Standard Bond dissovstion energy] diatomic Molecules at 25C.](https://wbbsesolutions.net/wp-content/uploads/2024/03/Chemical-Thermodynamic-Standard-Enthalpy-Of-Dissociation-Standard-Bond-dissovstion-energy-diatomic-Molecules-at-25C-1.png)







