Chapter 2 Is Matter Around Us Multiple Choice Question And Answers
Direction: Choose the correct answer for the following questions.
Question 1. Which of these cannot be placed under the category of elements?
- Gold
- Diamond
- Chalk
- Cadmium
Answer. 3. Chalk
Question 2. Identify the heterogeneous mixture:
- Soda water
- Hair spray
- Sea water
- Lemonade
Answer. 2. Hair spray
Question 3. Which of these elements are liquid in the state?
- Bromine
- Chlorine
- Iodine
- Helium
Answer. 1. Bromine
Question 4. The simplest form of matter, which cannot be sub, divided further by chemical/physical method is_______.
- Elements
- Compounds
- Mixtures
- None of these
Answer. 1. Elements
Read and Learn More NEET Foundation Multiple Choice Questions
Question 5. Under which category would you place ‘smoke’?
- Solution of solid in liquid
- Solution of solid in gas
- Solution of gas in solid
- Solution of liquid in gas
Answer. 2. Solution of solid in gas
Question 6. A dilute solution is one in which:
- The amount of solvent is much more than solute.
- The amount of solvent is not much more than solute.
- Solute and solvent are in equal proportion.
- Proportion does not matter at all.
Answer. 1. Amount of solvent is much more than solute.

Question 7. From the given table, choose the correct option
Colloidal solution Dispersion medium Dispersion phase
- Emulsion liquid solid
- Foam liquid-liquid
- Gel solid liquid
- Solid solid gas
aerosol
Answer. 3. Gel solid liquid
Question 8. Which combination of elements is present in ammonium chloride?
- Nitrogen, oxygen, chlorine
- Hydrogen and chlorine
- Nitrogen and hydrogen
- Nitrogen, hydrogen, chlorine
Answer. 4. Nitrogen, hydrogen, chlorine
Question 9. Face cream is an example of___________?
- Foam
- Emulsion
- Aerosol
- Solid foam
Answer. 2. Emulsion
Question 10. In the tincture of iodine, the solute is____
- Water
- Alcohol
- Solid iodine
- Liquid iodine
Answer. 3. Solid iodine
Question 11. Which of these is a perfect example of liquid in solid solution?
- German silver
- Lemonade
- Blue vitriol
- Clouds
Answer. 3. Blue vitriol
Question 12. Which is the correct arrangement concerning the decreasing order of the size of the particles?
- Suspension > true solution > colloid
- Suspension > colloid > true solution
- True solution > suspension > colloid
- Colloid > true solution > suspension
Answer. 2. Suspension > colloid > true solution
Question 13. Sumit wants to separate coloured dye from blue ink. Which separating technique should he opt for?
- Separating funnel method
- Evaporation method
- Sublimation method
- Centrifugation method
Answer. 2. Evaporation method
Question 14. What kind of change is involved in the burning of magnesium wire in the air?
- Only physical
- Only chemical
- Both physical and chemical
- None of these
Answer. 2. only chemical
Question 15. The process of speeding up of sedimentation by adding chemicals like alum is called __________?
- Sedimentation
- Decantation
- Centrifugation
- Coagulation
Answer. 4. Coagulation
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
Question 16. Which of these is not a physical change?
- Dissolution
- Fermentation
- Sublimation
- Condensation
Answer. 2. Fermentation
Question 17. Identify which of these is not an example of a homogeneous mixture:
- Air
- Beach sand
- Blood
- Gun powder
Answer. 2. Beach sand
Question 18. Crystallization is used to separate which of the following combinations?
- Solid – solid (heterogeneous)
- Solid-liquid (heterogeneous)
- Solid-liquid (homogeneous)
- Liquid-liquid (homogeneous)
Answer. 3. Solid – liquid (heterogeneous)
Question 19. Which of these does not exhibit the Tyndall effect?
- The cloud-free sky appears blue.
- A small beam of light from projectors in a cinema hall.
- A beam of light enters a room through a hole.
- All of these exhibit the Tyndall effect.
Answer. 1. Cloud-free sky appears blue.
Question 20. Which non-metal has a lustrous appearance?
- Bromine
- Iodine
- Phosphorous
- Sulphur
Answer. 2. Iodine
Question 21. The colloidal particles always move in _________ motion
- Straight
- Circular
- Zig zag
- Parallel
Answer. 3. Zig zag
Question 22. As a part of a homework assignment, a student was asked to prepare a molecular solution. Which of the following solutions he must not prepare?
- Magnesium hydroxide in water
- Alcohol in water
- CuSO4 in water
- Ethanoic acid in water
Answer. 1. Magnesium hydroxide in water
Question 23. In which of the following will the technique of chromatography be used?
- To separate sand from water.
- Water treatment plants.
- Distilling pure water from a salt solution.
- Separate different colour pigments from leaf extracts.
Answer. 4. Separate different colour pigments from leaf extracts.
Question 24. In a colloidal system, which combination of dispersed phase and dispersed medium is not possible?
- Solid–solid
- Solid–gas
- Liquid-liquid
- Gas–gas
Answer. 4. Gas-gas
Question 25. In which group will Germanium occupy its place?
- Metals
- Non-metals
- Gases
- Metalloids
Answer. 4. Metalloids
Question 26. Taking fog into consideration, which is the correct option?
- Solute–liquid; solvent–gas
- Solute–gas; solvent–solid
- Solute–gas; solvent–gas
- Solute–solid; solvent–gas
Answer. 1. Solute-liquid; solvent-gas
Question 27. Which of the given statements is incorrect about suspensions?
- The sediments of particles settle down in a suspension.
- It is a type of heterogeneous mixture.
- The size of the particles is less than 100 nm.
- The suspension is translucent or opaque.
Answer. 3. The size of the particles is less than 100 nm.
Question 28. An alloy is a mixture of two or more elements. Bronze is an alloy of___________?
- Copper and tin
- Copper and zinc
- Copper and silver
- Copper and nickel
Answer. 1. Copper and tin
Question 29. Which is the incorrect pair?
- Growth of a plant: chemical change
- Boiling of an egg: chemical change
- Cloud formation :chemical change
- Burning of paper: chemical change
Answer. 3. Cloud formation: chemical change
Question 30. Oil and water are separated bya sea separating funnel. What could be the justification?
- Difference in temperature
- Difference in pressure
- Difference in density
- All of the above
Answer. 3. Difference in density
Question 31. Given below is the characteristic of mixtures. Which of these is/are not true?
1. A mixture has no definite melting point
2. A mixture is always homogeneous
3. Water is an example of mixture
4. A mixture has a definite boiling point
- All are correct
- 1 and 4 are correct
- Only 1 is incorrect
- Only 1 is correct
Answer. 4. Only 1 is correct
Question 32. Which of these can be classified as elements?
Salt, water, hydrogen, sodium, oxygen
- Salt , water
- Hydrogen, sodium, oxygen
- All of these
- None of these. All are mixtures
Answer. 2. Salt , water
Question 33. Identify what will happen if wheat flour is added to a glass of water and stirred properly?
- A suspension is formed
- The suspension particles settle down after some time
- Both of (a) and (b)
- None of the above because it is not a suspension but a true solution
Answer. 3. Both of (a) and (b)
Question 34. Solid – solid homogeneous mixture can be separated by:
- Distillation
- Crystallization
- Evaporation
- All of the above
Answer. 4. All of the above
Question 35. Air is a mixture, why, choose proper reasons:
1. It has no fixed chemical formula
2. It varies in its composition
3. It involves energy change
- All are correct
- Only 1 and 2 are correct
- Only 3 is correct
- Only 2 and 3 are correct
Answer. 2. Only 1 and 2 are correct
Question 36. Which of the following are not physical changes?
(1) Juice freezes in refrigerator
(2) Gasoline is ignited
(3) Rusting of iron rod
(4) Water evaporates from the river
- (1) and (3)
- (1) and (4)
- (2) and (3)
- All of the above
Answer. 3. (2) and (3)
Question 37. Which of the given alloys does not have only metals as its constituents?
(1) Amalgam
(2) Invar
(3) Type metal
(4) Steel
- (1), (2), (3)
- (3) and (4)
- (1) and (2)
- (2), (3), (4)
Answer. 2. (3) and (4)
Question 38. Which statement is/are correct for mixtures?
1. Mixture may contain only solids
2. Mixture may contain only liquids
3. Mixture cannot contain only gases
4. Substances in different states cannot make a mixture
- 1 and 2
- 1 and 3
- 2 and 3
- 1, 2, 4
Answer. 1. 1 and 2
Question 39. The colloidal solution of water in air is/are____?
- Clouds
- Fog
- Mist
- All of the above
Answer. 4. All of the above
Question 40. Why water is supposed to be a compound and not a mixture? Choose the correct statements:
1. It has a definite formula
2. The properties of water are same as its constituents Hydrogen and oxygen
3. It has a fixed boiling point
- Only 1
- Only 2
- Only 3
- 1 and 3
Answer. 4. 1 and 3
Question 41. A mixture of sodium chloride and sulphur can be separated by the following set of processes. Identify the correct one.
- Addition of water, filtration, evaporation of water
- Addition of carbon disulphide, filtration, evaporation of carbon disulphide
- Sublimation
- Both (a) and (b)
Answer. 4. Both (a) and (b)
Question 42. Which of the following gives a true solution in water?
- Starch
- Sugar
- Chalk powder
- Egg albumin
Answer. 2. Sugar
Question 43. Oxygen is separated from nitrogen by fractional distillation. Which of the following statements gives the reason for choosing the above method of separation?
- Oxygen is more dense than nitrogen
- Oxygen is more reactive than nitrogen
- The boiling points of the liquefied gases are different
- Oxygen is more soluble in water than nitrogen
Answer. 3. The boiling points of the liquefied gases are different
Question 44. Which of the following gives a colloidal solution in water?
- Common salt
- Alum
- Sand
- Starch.
Answer. 4. Starch.
Question 45. Which of the following is not a compound?
- Common salt
- Water
- Iron filings
- Copper sulphate
Answer. 3. Iron filings
Question 46. Potassium nitrate crystals can be separated from sand using one of the following set of processes. Identify which one?
- Filter, dissolve, evaporate, crystallise
- Dissolve, evaporate, crystallise, filter
- Dissolve, evaporate, filter, crystallise
- Dissolve, filter, evaporate, crystallise
Answer. 4. Dissolve, filter, evaporate, crystallise
Question 47. Which of the following is not a mixture?
- Soil
- Air
- Steam
- Milk
Answer. 3. Steam
Question 48. From which of the following mixtures can the underlined substance be obtained by adding water, stirring and filtering?
- Calcium carbonate and sodium chloride
- Iron and magnesium
- Copper sulphate and potassium chloride
- Ethanol and ethanoic acid
Answer. 1. Calcium carbonate and sodium chloride
Question 49. Which of the following is not a physical change?
- Freezing of water
- Mixing or iron filings and sulphur
- Cooking of food
- Evaporation of alcohol
Answer. 4. Evaporation of alcohol
Question 50. Odd one among the following, which does not belong to the set is
- Graphite
- Gold
- Germanium
- Ice
Answer. 4. Ice
Question 51. The process used to remove solids from water during water treatment is
- centrifugation
- filtration
- sublimation
- all the above
Answer. 2. filtration
Question 52. Which of the following is not a chemical change?
- Electrolysis of water
- Boiling of water
- Digestion of food
- Burning of magnesium ribbon in oxygen to form magnesium oxide
Answer. 2. Boiling of water
Question 53. Which of the following is a liquid metal?
- Copper
- Mercury
- Bromine
- Silver
Answer. 2. Mercury
Question 54. A sample of a pure solid compound is heated until it melts completely. The compound is then allowed to cool until it solidifies again. The graph shows how the temperature of the compound changes with time.
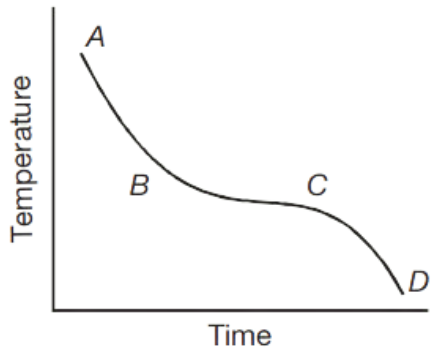
In which region of the graph both the liquid and the solid are present?
- A to B
- C to D
- B to C
- A to B and C to D
Answer. 3. B to C
Question 55. Which of the following pairs does not contain both elements?
- Carbon, silicon
- Helium, nitrogen
- Bronze, zinc
- Copper, silver.
Answer. 3. Bronze, zinc
Question 56. Brass contains
- Gold and copper
- Copper and zinc
- Zinc and silver
- Copper and silver
Answer. 2. Copper and zinc
Question 57. Fog and smoke are colloids of the type aerosol. Which of the following is different in both?
- Dispersed phase
- Dispersion medium
- Both (a) and (b)
- None of the above
Answer. 1. Dispersed phase
Question 58. Which of the following is not a pure substance?
- Mercury
- Sugar
- Blood
- Salt
Answer. 3. Blood
Question 59. Which pairs of substance are both mixtures?
- Air, sea water
- Sea water, ethanol
- Iron, water
- Lime water, marble
Answer. 1. Air, sea water
Question 60. Gases can be obtained from air by the process of
- Fractional distillation
- Condensation
- Crystallisation
- Evaporation
Answer. 1. Fractional distillation
Question 61. Which method cannot be used for separating solution of solid in liquid?
- Distillation
- Separating funnel
- Crystallisation
- Evaporation
Answer. 2. Separating funnel
Question 62. Sodium chloride is a considered as a pure substance because
- it is a solid at room temperature
- it contains only one type of particles
- its composition remains the same throughout
- Both (b) and (c)
Answer. 4. Both (b) and (c)
Question 63. Which of the following is a mixture?
- Calcium oxide
- Carbon dioxide
- Soil
- Coal
Answer. 3. Soil
Question 64. A mixture consists of a soluble solid in a liquid. Which method is most suitable for obtaining both the compounds?
- Fractional distillation
- Sublimation
- Distillation
- Evaporation
Answer. 3. Distillation
Question 65. In coloured gemstones, the dispersing medium and dispersed phase are
- solid, gas
- solid, liquid
- gas, solid
- solid, solid
Answer. 4. solid, solid
Question 66. Which of the following procedures will helpto determine whether a colourless liquid is a solution of a salt or pure water?
- Filtration
- Sedimentation and decantation
- Evaporation
- Chromatography
Answer. 3. Evaporation
Question 67. Odd one among the following which does not belong to the set is?
- Gun powder
- Air
- Graphite
- Brass
Answer. 3. Graphite
Question 68. Which of the following is a liquid non-metal?
- Copper
- Mercury
- Bromine
- Silver
Answer. 3. Bromine
Question 69. Tyndall effect is observed when
- sunlight passes through a small hole in a closed room
- a beam of light from a torch is directed on a starch solution
- sunlight enters a dense forest through spaces between the branches of trees
Answer. 3. sunlight enters a dense forest through spaces between the branches of trees
Question 70. Match the methods of separation with the physical properties of the substances on which they are based.
(1) Distillation (P) Difference in density
(2) Separation using a separating funnel (Q) Difference in solubility
(3) Filtration (R) Difference in boiling point
- (1) – P, (2) – R, (3) – Q
- (1) – Q, (2) – P, (3) – R
- (1) – Q, (2) – R, (3) – P
- (1) – R, (2) – P, (3) – Q
Answer. 4. (1) – R, (2) – P, (3) – Q
Question 71. Which of the following is not an emulsion?
- Mayonnaise
- Egg Yolk
- Shaving foam
- Cold Cream
Answer. 2.Egg Yolk
Question 72. Stirring Sulphur in carbon disulphide produces:
- Colloidal Solution
- Suspension
- True Solution
- Carbon trisulphide
Answer. 3. True Solution
Question 73. A solution with water as the solvent is called a/an ___________ solution.
Answer. Aqueous
Question 74. A solution in which the solute particles have a diameter below 1 nm is called a/an _________ solution.
Answer. True
Question 75. Which of the following statements are true for pure substances?
(1) Pure substances contain only one kind of particles
(2) Pure substances may be compounds or mixtures
(3) Pure substances have the same composition throughout
(4) Pure substances can be exemplified by all elements other than nickel
- (1) and (2)
- (1) and (3)
- (3) and (4)
- (2) and (3)
Answer. 2. (1) and (3)
Question 76. Rusting of an article made up of iron is called
- corrosion and it is a physical as well as chemical change
- dissolution and it is a physical change
- corrosion and it is a chemical change
- dissolution and it is a chemical change
Answer. 3. corrosion and it is a chemical change
Question 77. A mixture of sulphur and carbon disulphide is
- heterogeneous and shows Tyndall effect
- homogeneous and shows Tyndall effect
- heterogeneous and does not show Tyndall effect
- homogeneous and does not show Tyndall effect
Answer. 4. homogeneous and does not show Tyndall effect
Question 78. Tincture of iodine has antiseptic properties. This solution is made by dissolving
- iodine in potassium iodide
- iodine in vaseline
- iodine in water
- iodine in alcohol
Answer. 4. iodine in alcohol
Question 79. Which of the following are homogeneous in nature?
(1) ice
(2) wood
(3) soil
(4) air
- (1) and (3)
- (2) and (4)
- (1) and (4)
- (3) and (4)
Answer. 3. (1) and (4)
Question 80. Which of the following are physical changes?
(1) Melting of iron metal
(2) Rusting of iron
(3) Bending of an iron rod
(4) Drawing a wire of iron metal
- (1), (2) and (3)
- (1), (2) and (4)
- (1), (3) and (4)
- (2), (3) and (4)
Answer. 3. (1), (3) and (4)
Question 81. Which of the following are chemical changes?
(1) Decaying of wood
(2) Burning of wood
(3) Sawing of wood
(4) Hammering of a nail into a piece of wood
- (1) and (2)
- (2) and (3)
- (3) and (4)
- (1) and (4)
Answer. 1. (1) and (2)
Question 82. Two substances, A and B were made to react to form a third substance, A2B according to the following reaction: 2 A + B → A2 B Which of the following statements concerning this reaction are incorrect?
(1) The product A2B shows the properties of substances A and B
(2) The product will always have a fixed composition
(3) The product so formed cannot be classified as a compound
(4) The product so formed is an element
- (1), (2) and (3)
- (2), (3) and (4)
- (1), (3) and (4)
- (2), (3) and (4)
Answer. 3. (1), (3) and (4)
Question 83. Two chemical species X and Y combine together to form a product P which contains both X and YX + Y → PX and Y cannot be broken down into simpler substances by simple chemical reactions. Which of the following concerning the species X, Y and P are correct?
(1) P is a compound
(2) X and Y are compounds
(3) X and Y are elements
(4) P has a fixed composition
- (1), (2) and (3)
- (1), (2) and (4)
- (2), (3) and (4)
- (1), (3) and (4)
Answer. 4. (1), (3) and (4)