NEET Chemistry Structure Of Atom MCQs Chapter 4 Structure Of Atom Multiple Choice Question And Answers
Direction: Choose the correct option for each question. There is only one correct response for each question.
Question 1. On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
- Neutrons
- Neutral
- Electron
- Protons
Answer. 4. Protons
Question 2. What should be the pressure inside the discharge tube of the order of 0.001?
- The pressure should be very high.
- Pressure should be normal.
- The pressure should be very low.
- Pressure should be at a certain level.
Answer. 3. Pressure should be very low.
Question 3. Why Bohr’s orbits are called stationary states?
- It has got a fixed value of energy.
- Revolving orbit lose energy continuously.
- It is equal to the number of protons.
- It is equal to the sum of neutrons.
Answer. 1. It has a fixed value of energy.
Question 4. Which atom does not have any neutrons in the nucleus?
- Neutral atom
- Protons atom
- Electron atom
- Hydrogen atom
Answer. 4. Hydrogen atom
NEET Chemistry Structure of Atom MCQs
Question 5. When alpha particles are sent through a thin metal foil, most of them go straight through the foil because
- Alpha particles are much smaller than electrons.
- Alpha particles are positively charged.
- Most part of the atom is space.
- Alpha particles move with low velocity.
Answer. 3. Most part of atom is space.
Read and Learn More NEET Foundation Multiple Choice Questions
Question 6. Rutherford’s scattering experiment showed for the first time that the atom has
- Electrons
- Nucleus
- Protons
- Neutrons
Answer. 2. Nucleus
Question 7. Why does a mica wheel mounted on an axle and placed in the path of cathode rays begin to rotate when cathode rays fall in?
- Cathode rays travel in straight line.
- Particles present in cathode rays are positively charged particles.
- Cathode rays consist of material particles travelling with high velocity.
- Cathode rays consist of material particles travelling with low velocity.
Answer. 3. Cathode rays consist of material particles travelling with high velocity.

Question 8. If the K and L shells of an atom are full, then what would be the total number of electrons in the atom?
- 4
- 10
- 12
- 8
Answer. 2. 10
Question 9. Which of the following statements is not correct?
- An atomic number is alwaysthe whole number.
- All atoms of the same element have same number of protons in the nucleus.
- Atoms of two elements contain the same number of protons in the nucleus.
- Atomic number of the element changes when it undergoes a chemical reaction.
Answer. 4. Atomic number of the element changes when it undergoes a chemical reaction.
Question 10. Which symbols are used to represent different Bohr’s orbit?
- K, L, N, M
- K, M, L, N
- K, M, N, L
- K, L, M, N
Answer. 4. K, L, M, N
Atomic Structure MCQs for NEET
Question 11. What happens in the discharge tube if a high voltage is applied at gas pressure of one atmosphere?
- Current begin to flows between the electrodes.
- Glow disappears.
- No current flows between the electrodes.
- The colour of glow changes.
Answer. 3. No current flows between the electrodes.
Question 12. Atomic number of chlorine is 17. What will be the atomic number of chloride ion (Cl–)?
- 17
- 11
- 10
- 18
Answer. 1. 17
Question 13. What do you mean by Valence electrons?
- Electrons presents in the duplet of the atom.
- Electrons increase in atomic number.
- Electrons presents in the outermost shell of the atom of an element.
- Total number of electrons present in the atom.
Answer. 3. Electrons presents in the outermost shell of the atom of an element.
Atomic Structure MCQs for NEET
Question 14. What is the maximum number of electrons that can be present in the M-Shell?
- 18
- 16
- 12
- 10
Answer. 1. 18
Question 15. What are isobars?
- Atoms of same elements.
- Atomic numbers are same.
- Atoms of different elements having different atomic number.
- Atomic masses taken with average value.
Answer. 3. Atoms of different elements having different atomic number.
Question 16. How do we find the age of fossils i.e., sample of woods or dead animals?
- Geological dating
- Carbon dating
- Helium
- Isoelectronic
Answer. 2. Carbon dating
Question 17. If an element has 5 electrons in the outermost M-shell, what element is this?
- Iodine
- Cobalt
- Methane
- Phosphorus
Answer. 4. Phosphorus
Atomic Structure MCQs for NEET
Question 18. Covalency is the number of electrons
- Sharing with other atoms
- Lost by an atom
- Gain by an atom
- Compound by an atom
Answer. 1. Sharing with other atoms
Question 19. Why helium does not take part in chemical reaction?
- Outermost shell has 8 electrons.
- Only one shell containing 2 electrons.
- Most of the elements have isotopes.
- Complete octet in the outermost shell.
Answer. 2. Only one shell containing 2 electrons.
Question 20. What is Radioisotopes?
- Some isotopes have unstable nuclei.
- Some isotopes have stable nuclei.
- Elements have different atomic numbers.
- Large amount of radiation is emitted.
Answer. 1. Some isotopes have unstable nuclei.
Question 21. Three isotopes of oxygen are known with mass number 16, 14 and 18. How do they differ from each other in term of electron, proton and neutrons?
- Same number of protons and neutrons but different number of electrons.
- Same number of electrons and neutrons but different number of protons.
- Same number of protons and electrons but different number of neutrons.
- None of above
Answer. 3. Same number of protons and electrons but different number of neutrons.
Question 22. How are positive ions formed?
- Gain of electrons in their outermost shell.
- Anion is equal to the number of electrons.
- Equal to the units of charger present on the ion.
- Loss of electrons by neutral atom.
Answer. 4. Loss of electrons by neutral atom.
Atomic Structure MCQs for NEET
Question 23. If an atom contains one electron and one proton, will it carry any charge or not?
- The net charge on the atom will be more than 1.
- Yes, atom will carry the charge.
- No, atom will not carry any charge.
- None of above
Answer. 3. No, atom will not carry any charge.
Question 24. On the basis of Thomson’s model of an atom how the atom is neutral as a whole?
- Negative charge on the electrons is equal to positive charge of the sphere.
- Positive charge on the electrons is equal to positive charge of the sphere.
- Negative charge on the electrons is equal to negative charge of the sphere.
- None of above
Answer. 1. Negative charge on the electrons is equal to positive charge of the sphere.
Question 25. What is proton?
- An electron is that subatomic particle which carries one-unit negative charge.
- A smallest indivisible particle made up of electrons.
- A subatomic particle which carries one unit positive charge.
- Number of elements with low atomic number.
Answer. 3. A subatomic particle which carries one unit positive charge.
Structure of Atom NEET Questions
Question 26. How do you determine the charge on the cathode rays?
- Atoms absorb energy
- Electric field deflected towards the positive plates
- Mass is equal to the mass of the atom of the gas taken
- None of above
Answer. 2. Electric field deflected towards the positive plates
Question 27. What is the origin of the anode rays?
- Atoms forming positive icons start moving towards cathode rays.
- Ratio is always same irrespective of the gas taken in the discharge tube.
- Atoms of different elements having different atomic number.
- Electrons presents in the duplet of the atom.
Answer. 1. Atoms forming positive icons start moving towards cathode rays.
Question 28. Helium atom has an atomic mass of 4 u and two protons in its nucleus. How many neutrons does it have?
- 4
- 8
- 2
- 6
Answer. 3. 2
Question 29. Oxygen atom has 8 electrons, 8 protons and 8 neutrons whereas sulphur atom has 16 electrons, 16 protons and 16 neutrons. Find out the mass number of oxygen and sulphur atoms.
- 16 and 32
- 8 and 32
- 32 and 16
- 8 and 16
Answer. 1. 16 and 32
Question 30. If K and L-shells of an atom are full, then what would be the total number of electrons in the atom?
- 18
- 12
- 08
- 10
Answer. 4. 10
Question 31. What are canal rays?
(1) They are anode rays
(2) They pass through the holes or canals in the cathode
(3) They travel in straight lines
(4) None of above
- (1) and (2)
- (1) and (3)
- None of above
- (1), (2), and (3)
Answer. 4. (1), (2), and (3)
Question 32. What is mass number?
(1) Sum of number of neutrons
(2) Average relative mass of its atoms
(3) Sum of number of protons
(4) Whole number of electrons
- (1) and (4)
- (1) and (2)
- (1) and (3)
- (1), (2), and (3)
Answer. 3. (1) and (3)
Question 33. How can we check that electrons are material particles?
(1) Electric field is applied on the cathode rays.
(2) Mica wheels mounted on an axle.
(3) Mica wheels placed in the path of cathode rays.
(4) Cathode rays deflected towards electric fields.
- (1) and (2)
- (2) and (4)
- (2) and (3)
- (1), (2), and (4)
Answer. 2. (2) and (4)
Question 34. Which of the following is correct about cathode rays?
(1) They are deflected towards the positive plate of the electric field.
(2) The nature of cathode rays does not depend upon the nature of the material of the cathode.
(3) The nature of cathode rays depends upon the nature of the gas taken in the discharge tube.
(4) Cathode rays are made up of electrons.
- (1) and (2)
- (2) and (4)
- (2) and (3)
- (1), (2), and (4)
Answer. 4. (1), (2), and (4)
Structure of Atom NEET Questions
Question 35. How many of the following are correct observation/conclusion from Rutherford’s scattering experiment?
(1) Nucleus is small but heavy.
(2) Nucleus always carries positive charge.
(3) The number of α-particles hitting the nucleus is very large.
(4) Atom is nearly 105 times greater than the size of the nucleus.
- (1) and (2)
- (2) and (4)
- (1), (2) and (4)
- (1), (2) and (3)
Answer. 3. (1), (2) and (4)
Question 36. Neutron is present in all atoms
(1) Protium
(2) Deuterium
(3) Tritium
(4) Helium
- (2), (3) and (4)
- (3) and (4)
- (1), (2) and (3)
- (1), (2) and (4)
Answer. 1. (2), (3) and (4)
Question 37. Isobars differ in number of
(1) Protons
(2) Electrons
(3) Neutrons
(4) Nucleons
- (1), (3) and (4)
- (2) and (4)
- (1), (2) and (3)
- (1), (2) and (4)
Answer. 3. (1), (2) and (3)
Question 38. Which of the following isotopes are not used in the treatment of cancer?
(1) P-32
(2) I-131
(3) Co-60
(4) U-235
- (1) and (2)
- (2) and (4)
- (1), (2) and (4)
- (2), (3) and (4)
Answer. 4. (2), (3) and (4)
Structure of Atom NEET Questions
Question 39. The valency of which of the following elements is not zero?
(1) Hydrogen
(2) Helium
(3) Gold
(4) Silver
- (1) and (4)
- (1) and (2)
- (1) and (3)
- (1), (3) and (4)
Answer. 4. (1), (3) and (4)
Question 40. Which of the following are true for an element?
(1) Atomic number = number of protons + number of electrons
(2) Mass number = number of protons + number of neutrons
(3) Atomic mass = number of protons + number of neutrons
(4) Atomic number = number of protons + number of electrons
- (1) and (2)
- (1) and (3)
- (2) and (3)
- (2) and (4)
Answer. 4. (2) and (4)
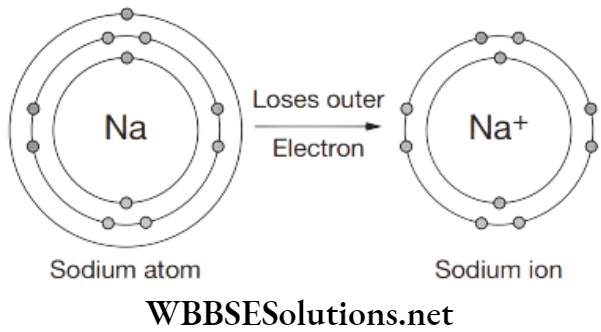
Question 41. Positive ions are formed from the neutral atom by the
- Increase of nuclear charge
- Gain of protons
- Loss of electrons
- Loss of protons
Answer. 3. Loss of electrons
Question 42. A neutral atom (Atomic no. > 1) consists of
- Only protons
- Neutrons + protons
- Neutrons + electrons
- Neutrons + protons + electrons
Answer. 4. Neutrons + protons + electrons
NEET Foundation Chemistry Chapter 4
Question 43. The mass of electron is
- 9.1083 × 10-31 kg
- 9.1083 × 10-24 kg
- 9.1083 × 10-28 kg
- 1.67 × 10-24 kg
Answer. 1. 9.1083 × 10-31 kg
Question 44. Size of the nucleus is of the order
- 10-12 m
- 10-8 m
- 10-15 m
- 10-10 m
Answer. 3. 10-15 m
Question 45. In a given atom no two electrons can have the same values for all the four quantum numbers.
- Hund’s rule
- Aufbau principle
- Uncertainty principle
- Pauli’s exclusion principle
Answer. 2. Aufbau principle
NEET Foundation Chemistry Chapter 4
Question 46. An explanation of the presence of three unpaired electrons in the nitrogen atom is given by
- Pauli’s principle
- Hund’s rule
- Aufbau principle
- uncertainty principle
Answer. 2. Hund’s rule
Question 47. The \(\frac{e}{m}\) ratio of an electron for all type of substances is
- constant
- less than one
- greater than one
- zero
Answer. 1. constant
Question 48. The ratio of charge and mass would be greater for
- Proton
- Electron
- Neutron
- Alpha
Answer. 2. Electron
Structure of Atom Multiple Choice Questions
Question 49. A proton is identical to
- the nucleus of helium
- the nucleus of a hydrogen atom
- a molecule of a hydrogen
- an atom of hydrogen
Answer. 4. an atom of hydrogen
Question 50. The electron is
- α-ray particle
- β-ray particle
- Hydrogen ion
- Positron
Answer. 2. β-ray particle
Question 51. An α-particle is
- a hydrogen molecule
- a helium nucleus
- an electron
- a proton
Answer. 2. a helium nucleus
Question 52. A natural phenomenon that supports the experimental conclusion that atoms are divisible is
- allotropy
- radioactivity
- cracking
- None of these
Answer. 2. radioactivity
Question 53. Designation of an orbital with n = 6 and l = 4, is
- 6s
- 6d
- 6f
- 6p
Answer. 4. 6p
Structure of Atom Multiple Choice Questions
Question 54. Which of the following nuclear particles is responsible for holding nucleons together in a nucleus?
- Protons
- Mesons
- Positrons
- Neutrons
Answer. 2. Mesons
Question 55. Though three fundamental particles are present in almost all elements. One element does not have
- neutron
- proton
- electron
- nucleons
Answer. 1. neutron
Question 56. The nucleus of the atom consists of
- Proton and neutron
- Proton and electron
- Neutrons and electron
- Proton, neutrons and electron
Answer. 1. Proton and neutron
Question 57. The size of nucleus is of the order of
- 10-12 m
- 10-8m
- 10-15 m
- 10-10 m
Answer. 3. 10-15 m
Question 58. A completely filled and half filled orbit is spherically symmetrical. Point out which has spherical symmetry.
- Na
- C
- Cl–
- S
Answer. 1. Na
Structure of Atom Multiple Choice Questions
Question 59. Mosley’s name is related with the discovery of
- Atomic mass
- Atomic number
- Neutrons
- Effective atomic number
Answer. 2. Atomic number
Question 60. Radius of first Bohr’s orbit of hydrogen atom is
- 0.529 Å
- 5.29 Å
- 2.59 Å
- 9.25 Å
Answer. 1. 0.529 Å
Question 61. If the value of En = –0.85 eV for hydrogen in which of the following excited state electron is present?
- 1st
- 2nd
- 3rd
- 4th
Answer. 4. 4th
Question 62. The atomic number of an element is 11 and its mass number is 23. The prespective number of electrons, protons and neutrons in this atom will be
- 11, 11, 12
- 11, 12, 11
- 12, 11, 11
- 23, 11, 23
Answer. 1. 11, 11, 12
Question 63. Number of electrons in the outer shell of the most stable or inert atoms is
- 1
- 4
- 6
- 8
Answer. 4. 8
Atomic Structure MCQs for NEET
Question 64. Number of electrons in an element with atomic number X and atomic mass Y will be
- X – Y
- Y – X
- X + Y
- X
Answer. 4. X
Question 65. Which of the following pairs are isotopes?
- Oxygen and ozone
- Ice and steam
- Nitric oxide and nitrogen dioxide
- Hydrogen and deuterium
Answer. 4. Hydrogen and deuterium
Atomic Structure MCQs for NEET
Question 66. The energy needed for sending electron from 1st Bohr orbit (n = 1) to second orbit (n = 2) of hydrogen atom is
- 13.6 eV
- 3.4 eV
- 10.2 eV
- 6.8 eV
Answer. 3. 10.2 eV
Question 67. Total number of neutrons in dipositive Zinc ion (Zn2+) with mass number 70 is
- 30
- 40
- 50
- 20
Answer. 2. 40
Question 68. Correct set of four quantum number for valence shell electron of Rubidium (at No. 37) is
- \(0,0,5, \pm \frac{1}{2}\)
- \(5,0,0,+\frac{1}{2}\)
- \(6,0,0,-\frac{1}{2}\)
- \(5,1,1,+\frac{1}{2}\)
Answer. 2. \(5,0,0,+\frac{1}{2}\)
Question 69. An atom of an element has 26 electrons and has a mass number 56. The nucleus of this atom contains _______ neutrons.
- 26
- 36
- 30
- 56
Answer. 3. 30
Atomic Structure MCQs for NEET
Question 70. For an element with atomic number 19, the 19th electron will occupy
- L-shell
- M-shell
- N-shell
- K-shell
Answer. 2. M-shell
Chapter 4 Structure Of Atom Practice Exercies
NCERT Examples
Question 1. Which of the following correctly represent the electronic distribution in the Mg atom?
- 3, 8,
- 2, 8, 2
- 1, 8, 3
- 8, 2, 2
Answer. 2. 2, 8, 2
Question 2. Rutherford’s ‘alpha (α) particles scattering experiment’ resulted in to discovery of
- Electron
- Proton
- Nucleus in the atom
- Atomic mass
Answer. 3. Nucleus in the atom
Question 3. The number of electrons in an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of the element?
- 3115X
- 3116X
- 1615X
- 1516X
Answer. 1. 3115X
Structure of Atom Multiple Choice Questions
Question 4. Dalton’s atomic theory successfully explained
(1) Law of conservation of mass
(2) Law of constant composition
(3) Law of radioactivity
(4) Law of multiple proportion
- (1), (2) and (3)
- (1), (3) and (4)
- (2), (3) and (4)
- (1), (2) and (4)
Answer. 4. (1), (2) and (4)
Question 5. Which of the following statements about Rutherford’s model of atom are correct?
(1) considered the nucleus as positively charged
(2) established that the a–particles are four times as heavy as a hydrogen atom
(3) can be compared to solar system
(4) was in agreement with Thomson’s model
- (1) and (3)
- (2) and (3)
- (1) and (4)
- only (1)
Answer. 1. (1) and (3)
Structure of Atom Multiple Choice Questions
Question 6. Which of the following is true for an element?
(1) Atomic number = number of protons + number of electrons
(2) Mass number = number of protons + number of neutrons
(3) Atomic mass = number of protons = number of neutrons
(4) Atomic number = number of protons = number of electrons
- (1) and (2)
- (1) and (3)
- (2) and (3)
- (2) and (4)
Answer. 4. (2) and (4)
Question 7. In the Thomson’s model of atom, which of the following statements are correct?
(1) the mass of the atom is assumed to be uniformly distributed over the atom
(2) the positive charge is assumed to be uniformly distributed over the atom
(3) the electrons are uniformly distributed in the positively charged sphere
(4) the electrons attract each other to stabilise the atom
- (1), (2) and (3)
- (1) and (3)
- (1) and (4)
- (1), (3) and (4)
Answer. 1. (1), (2) and (3)
Structure of Atom Multiple Choice Questions
Question 8. Rutherford’s α–particle scattering experiment showed that
(1) electrons have negative charge
(2) the mass and positive charge of the atom is concentrated in the nucleus
(3) neutron exists in the nucleus
(4) most of the space in atom is empty Which of the above statements are correct?
- (1) and (3)
- (2) and (4)
- (1) and (4)
- (3) and (4)
Answer. 2. (2) and (4)
Question 9. The ion of an element has 3 positive charges. Mass number of the atom is 27 and the number of neutrons is 14. What is the number of electrons in the ion?
- 13
- 10
- 14
- 16
Answer. 2. 10
Structure of Atom Multiple Choice Questions
Question 10. Identify the Mg2+ ion from the where, n and p represent the number of neutrons and protons respectively




Answer.
4. 
Question 11. In a sample of ethyl ethanoate (CH3COOC2H5) the two oxygen atoms have the same number of electrons but different number of neutrons. Which of the following is the correct reason for it?
- One of the oxygen atoms has gained electrons
- One of the oxygen atoms has gained two neutrons
- The two oxygen atoms are isotopes
- The two oxygen atoms are isobars.
Answer. 3. The two oxygen atoms are isotopes
Structure of Atom Multiple Choice Questions
Question 12. Elements with valency 1 are
- always metals
- always metalloids
- either metals or non-metals
- always non-metals
Answer. 3. either metals or non-metals
Question 13. The first model of an atom was given by
- N. Bohr
- E. Goldstein
- Rutherford
- J.J. Thomson
Answer. 4. J.J. Thomson
Question 14. An atom with 3 protons and 4 neutrons will have a valency of
- 3
- 7
- 1
- 4
Answer. 3. 1
Question 15. The electron distribution in an aluminium atom is
- 2, 8, 3
- 2, 8, 2
- 8, 2, 3
- 2, 3, 8
Answer. 1. 2, 8, 3
Atomic Structure MCQs for NEET
Question 16. Which of the following do not represent Bohr’s model of an atom correctly?

- (1) and (2)
- (2) and (3)
- (2) and (4)
- (1) and (4)
Answer. 3. (2) and (4)
Question 17. Which of the following statement is always correct?
- An atom has equal number of electrons and protons.
- An atom has equal number of electrons and neutrons.
- An atom has equal number of protons and neutrons.
- An atom has equal number of electrons, protons and neutrons.
Answer. 1. An atom has equal number of electrons and protons.
Question 18. Atomic models have been improved over the years. Arrange the following atomic models in the order of their chronological order
(1) Rutherford’s atomic model
(2) Thomson’s atomic model
(3) Bohr’s atomic model
- (1), (2) and (3)
- (2), (3) and (1)
- (2), (1) and (3)
- (3), (2) and (1)
Answer. 3. (2), (1) and (3)
Atomic Structure MCQs for NEET
Chapter 4 Structure Of Atom Track Your learning Question And Answers
Question 1. First time the cathode ray experiment was done by:
- Rutherford
- Goldstein
- Chadwick
- Dalton
Answer. 2. Goldstein
Question 2. Complete mass of an atom is concentrated in the __________.
Answer. Molecule
Structure of Atom NEET Questions
Question 3. Who discovered neutron?
- Chadwick
- Dalton
- Bohr
- Rutherford
Answer. 1. Chadwick
Question 4. __________ is present outside the nucleus.
Answer. Electron
Question 5. The term electron was coined by GJ Stoney. (True/False)
Answer. True