Superposition Of Waves Vibration Air Columns
WBBSE Class 11 Vibration of Strings Overview
Musical instruments like flute, organ, etc., are played by vibrating the air columns enclosed within them. Similarly, sound can be produced by blowing the air columns in whistles, thin pipes, etc.
Such an instrument, whatever be its shape, can be considered, as a pipe or tube enclosing an air column within it. In general, there are two types of pipes.
- A pipe having one end open and the other closed is called a closed pipe;
- A pipe having both ends open is called an open pipe.
- In the case of air columns, a closed-end in a column of air is analogous to the fixed end of a vibrating string. That is, at the closed end of an air column, the air is not free to undergo movement.
- Thus it is assumed the nodal position a standing wave, conversely, the air is free to undergo its back-and-forth longitudinal motion at the open end of the air column, and as such, the standing wave pattern will depict antinodes at the open ends of air columns.
- Now, we may hold a vibrating tuning fork or we may whiff at the open end B of a closed or an open pipe. Then a longitudinal sound wave propagates along the pipe towards the end A. This wave is reflected at A.
Vibrating Air Columns: Key Concepts
When this reflected wave comes back towards B, the incident and the reflected waves superpose. As a result, a stationary wave is generated within the pipe. This stationary wave produces the musical note emitted by instruments like flute, organ, etc.
Characteristics Of The Stationary Waves In An Air Column:
- Since sound waves are longitudinal waves, the stationary waves generated in an air column are longitudinal stationary waves. We may recall that the waves in a vibrating stretched string are transverse stationary waves.
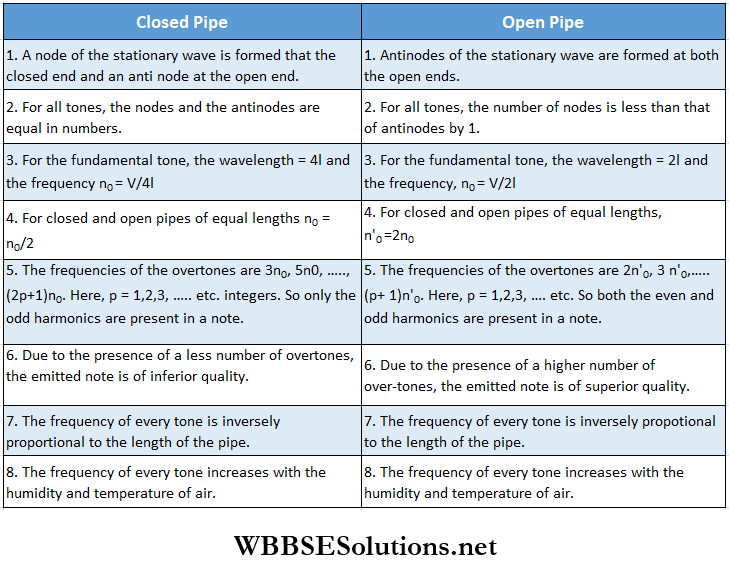
- The air particles at the closed end (end A) of a closed pipe cannot vibrate at all. So, a node is generated at the closed end. On the other hand, the air particles at the open end (end B) of a closed pipe or at both the open ends of an open pipe, can vibrate with maximum amplitude. So, an antinode is generated at each open end.
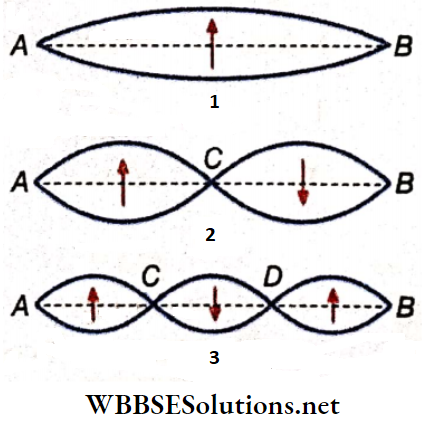
- The distance between two consecutive nodes or between two consecutive antinodes is \(\frac{\lambda}{2}\); here A = wavelength of the sound wave. Likewise, the distance between a node and its adjacent antinode = \(\frac{\lambda}{4}\).
Closed Pipe: Let the length of a closed pipe be l and the velocity of sound be V. In a closed pipe, incident waves, reflected waves from closed ends and the stationary wave produced are longitudinal waves. But for convenience, the waves in the pipe are drawn.

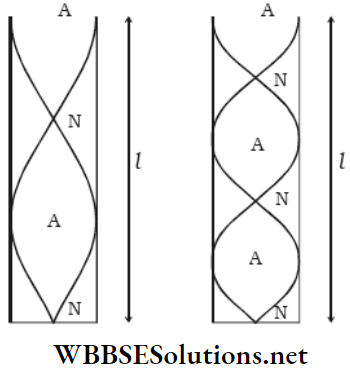
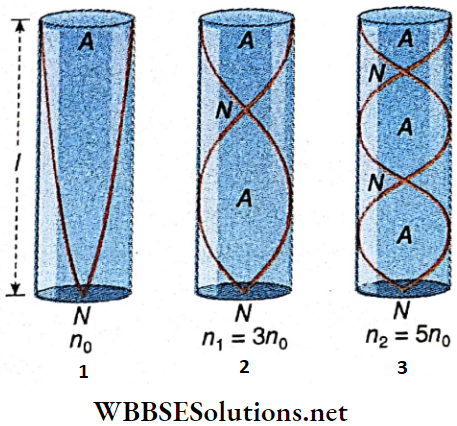
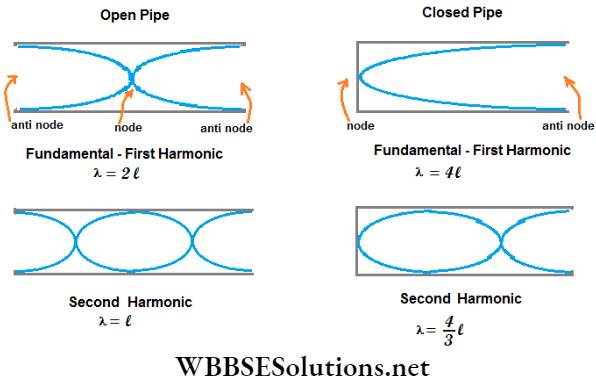
Fundamental Tone In A Dosed Pipe: For the simplest stationary wave in a closed pipe, the node is at the closed end and the antinode is at the open end. There is no other node or antinode between them. So, the tone produced in the pipe is called the fundamental tone or the fundamental.
Fundamental Frequency of a Vibrating String
If λ0 is the wavelength of the sound wave, the length of the pipe, l = the distance between a node and its adjacent antinode = \(\frac{\lambda_0}{4}\)
or, λ0 = 41.
So, the frequency of the emitted fundamental tone, \(n_0=\frac{V}{\lambda_0}=\frac{V}{4 l}\)…..(1)
This fundamental tone is also known as the 1st harmonic. Equation (1) implies that n0 increases when l decreases. As a result, the pitch of the emitted tone will be higher for shorter pipes.
Overtones In A Closed Pipe: When there is another node-antinode pair between the node at the closed end and the antinode at the open end, the next higher tone or the 1st overtone is produced.
If λ1 is the wavelength of the sound wave, the length of the pipe, l = distance between the node at the closed end and the second antinode from it = \(\frac{3 \lambda_1}{4}\)
∴ \(\lambda_1=\frac{4 l}{3}\)
So, the frequency of the emitted tone, \(n_1=\frac{V}{\lambda_1}=3 \cdot \frac{V}{4 l}\)
Applications of String Vibration in Musical Instruments
Clearly, n1 = 3n0 …(2)
This means that the frequency of the 1st overtone is three times that of the fundamental. For this reason, the 1st overtone is called the 3rd harmonic.
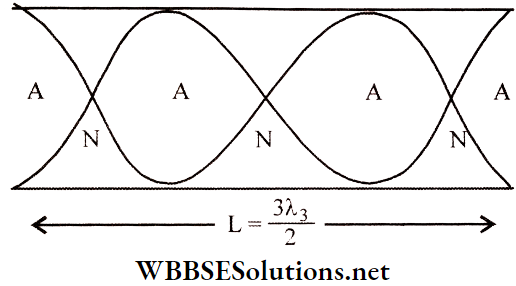
Similarly, for two node-antinode pairs between the open and the closed ends, the 2nd overtone is produced. The same calculations show that n2 = 5 n0; the frequency of the 2nd overtone is five times that of the fundamental.
So, it is the 5th harmonic. Clearly, the next overtones will have frequencies, n3 = 7n0, n4 = 9n0, ………., np = (2p+ 1)n0, etc.
The following two features are evident in this case:
- The frequency of each overtone is an integral multiple of the fundamental frequency. So, each overtone is a harmonic.
- The harmonics with frequencies \(2 n_0, 4 n_0, 6 n_0, \cdots\) are absent. So, a closed pipe can emit the fundamental tone and its odd harmonics only.
Open Pipe: Let the length of an open pipe be l and the velocity of sound be V. In an open pipe, incident waves, reflected waves from open ends and the stationary wave produced are longitudinal waves. But, for convenience, the waves in the pipe are drawn in as transverse waves.
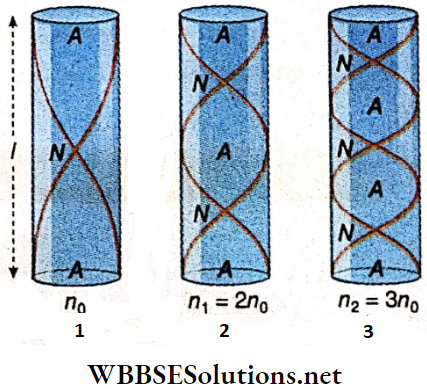
Fundamental Tone In An Open Pipe: For the simplest stationary wave in an open pipe, two antinodes are formed at the two ends. There is only one node just at the middle of the pipe. This is called the fundamental tone.
If λ0 is the wavelength of the sound wave, the length of the pipe, l = the distance between two consecutive antinodes = \(\frac{\lambda_0}{2}\)
or, λ0 = 21
So, the frequency of the emitted fundamental tone, \(n_1=\frac{V}{\lambda_1}=\frac{V}{l}=2 \cdot \frac{V}{2 l}\)…(2)
This fundamental tone is also known as the 1st harmonic.
Overtones In On Open Pipe: The 1st overtone is formed when two nodes lie between the two antinodes of the two ends and a third antinode is at the mid-point.
If λ1 is the wavelength of the sound wave, the length of the pipe, l = distance between three consecutive antinodes = λ1
i.e., λ1 = 1
So, the frequency of the emitted tone, \(n_1=\frac{V}{\lambda_1}=\frac{V}{l}=2\cdot \frac{V}{2 l}\)
Clearly, \(n_1=2 n_0\)
This means that the frequency of the 1st overtone is twice that of the fundamental. For this reason, the 1st overtone is called the 2nd harmonic.
Similarly, for three nodes between the antinodes at the open ends, the 2nd overtone is produced. The same calculations show that n2 = 3n0; the frequency of the 2nd overtone is 3 times that of the fundamental frequency. So, it is the 3rd harmonic. Clearly, the next overtones will have frequencies, \(n_3=4 n_0, n_4=5 n_0, n_5=6 n_0, \cdots, n_p=(p+1) n_0 \text {, etc. }\).
The Following Two Features Are Evident In This Case:
- The frequency of each overtone is an integral multiple of the fundamental frequency. So, each overtone is a harmonic.
- An open pipe can emit the fundamental tone and all its even and odd harmonics. No harmonic is absent in this case.
A closed pipe does not emit the even harmonics, but all the even and odd harmonics are emitted from an open pipe. So, in general, notes emitted from an open pipe have a greater number of overtones.
- As a result, this note is of a higher quality and is more pleasant to hear as compared to that emitted from a closed pipe. For this reason, open pipes are used in all good-quality flutes, organs, etc.
- The fundamental frequency is inversely proportional to the length of a closed or open pipe. In musical instruments like flute, some holes are made along its length. A good instrumentalist can open or close the holes according to the need and can change the effective length of the pipe. In this way, he can change and control the frequency of the emitted tones.
Comparison Of Frequencies Of The Tones Emitted By Closed And Open Pipes Of Equal Length: Let l be the length of each of a closed and an open pipe and V be the velocity of sound in air.
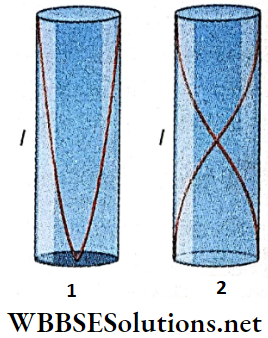
Comparison Of Fundamental Frequencies: For the fundamental tone emitted by the closed pipe, a node is formed at the closed end and an antinode at the open end.
There is no other node or antinode between them.
If λ0 is the length of the stationary wave,
l = distance from a node to its adjacent antinode = \(\frac{\lambda_0}{4}\)
or, λ0 = 4l
So, the fundamental frequency, \(n_0=\frac{V}{\lambda_0}=\frac{V}{4 l}\)…..(1)
For the fundamental tone emitted by an open pipe, two antinodes are formed at the two open ends. There is only a single node in between them.
If λ’0 is the length of the stationary wave, l = distance between two consecutive antinodes = \(\frac{\lambda_0^{\prime}}{2}\)
or, λ’0 = 2l
- So, the fundamental frequency, \(n_0^{\prime}=\frac{V}{\lambda_0^{\prime}}=\frac{V}{2 l}\)….(2)
- Comparing the relations (1) and (2), we get \(\frac{n_0}{n_0^{\prime}}=\frac{\frac{V}{4 l}}{\frac{V}{2 l}}=\frac{1}{2} \quad \text { or, } n_0^{\prime}=2 n_0\)…(3)
- So, the fundamental frequency of an open pipe is twice that of a closed pipe of the same length. We know that the pitch of a musical sound increases with its frequency. While whiffing at one end of an open pipe, if the opposite end is suddenly closed with a finger, the pipe becomes a closed one.
- As a result, the pitch of the sound falls abruptly. Conversely, when the closed end of a closed pipe is suddenly opened, the sound emitted from the pipe becomes abruptly sharper.
Comparison Of The Frequencies Of Overtones: In the note emitted from a closed pipe, the frequencies of the fundamental and the overtones are \(n_0, 3 n_{o^{\prime}} 5 n_0, \ldots\), etc.
- On the other hand, the fundamental and the overtone frequencies in a note emitted from an open pipe are \(n_0^{\prime}, 2 n_0^{\prime}, 3 n_0^{\prime}, \cdots\), etc. If the closed pipe and the open pipe are of equal length, then n’0 = 2n0. So for the open pipe, the frequencies are \(2 n_0, 4 n_0, 6 n_0, \cdots, \text { etc. }\).
- So, it is seen that if one end of a pipe can be opened and closed as required, it can emit the fundamental tone as well as all the even and odd harmonics.
End Error In Closed And Open Pipes: Some amount of air is trapped inside a closed or an open pipe. Accurate experiments show that a small layer of air in close vicinity outside an open end also behaves like a trapped layer. So, this layer should also be considered as a part of the air column enclosed in the pipe.
- As a result, the antinode of a stationary wave is formed not exactly at the open end; rather it is formed at a point slightly outside every open end. This means that the effective length of the pipe is slightly greater than its actual length.
- This increase in the effective length, due to the open end of a closed pipe or due to both the open ends of an open pipe, is called the end error. If c is the magnitude of the end error for an open end of a pipe of length l then, effective length of a closed pipe, L = l+c; effective length of an open pipe, L = 1 + 2c. The relations of Sections 4.6.1 and 4.6.2 should be modified accordingly.
So, the fundamental frequency for a closed pipe, \(n_0=\frac{V}{4 L}=\frac{V}{4(l+c)}\)
and the fundamental frequency for an open pipe, [lat6ex]n_0=\frac{V}{2 L}=\frac{V}{2(l+2 c)}[/latex]
- This is known as end correction.
- Scientists Helmholtz and Rayleigh experimentally showed that, c = 0.58 ≈ 0.6 r; where r = radius of the tube.
- This means that narrower tubes correspond to fewer end errors. The theory also proposes that the end error increases with the wavelength of the sound emitted.
- For higher harmonics, the frequencies are very high and the wavelengths are very low. In that case, the end error becomes negligible.
Effect of Different Physical Quantities on the Frequencies of Air Columns: Considering the end errors, the fundamental frequencies of the notes emitted by a closed and an open pipe are, respectively,
⇒ \(n_0=\frac{V}{4(l+0.6 r)} \text { and } n_0^{\prime}=\frac{V}{2(l+2 \times 0.6 r)}\)
where, l = length of the pipe, r = radius of the pipe and V = velocity of sound in air.
These expressions show that the frequency becomes higher when
- l is less,
- r is less,
- V is higher due to rise in temperature and
- V is higher due to an increase in humidity.
The pitch of the emitted sound increases with the increase in frequency. So, the pitch becomes higher for,
- Shorter pipes,
- Narrower pipes,
- The higher temperature of air and
- Higher humidity in the air.
Comparison Between The Vibrations Of Air Columns In A Closed And An Open Pipe:
Unit 10 Oscillation And Waves Chapter 4 Superposition Of Waves Vibration Air Columns Numerical Examples
Harmonics in Vibrating Strings
Example 1. Find out the frequency of the first overtone emitted by a1.25 m long organ pipe closed at one end. Given, the velocity of sound in air = 320 m · s-1.
Solution:
Mathematical Formulas for Vibration of Strings
Given, the velocity of sound in air = 320 m · s-1
For the 1st overtone, a node-antinode pair is formed in between a node at the closed end and an antinode at the open end.
So, the distance between the node at the closed end and the 2nd antinode from it = \(\frac{3 \lambda}{4}\) = l = length of the pipe.
∴ \(\lambda=\frac{4 l}{3}\)
So, the frequecny of this 1st overtone is \(n_1=\frac{V}{\lambda}=\frac{3 V}{4 l}=\frac{3 \times 320}{4 \times 1.25}=192 \mathrm{~Hz}\)
Example 2. Find out the frequencies of the fundamental and its nearest harmonic emitted by a 1m long closed organ pipe. Given, the velocity of sound in air =332 m · s-1.
Solution:
Given, the velocity of sound in air =332 m · s-1
Fundamental frequency, \(n_0=\frac{V}{4 l}=\frac{332}{4 \times 1}=83 \mathrm{~Hz}\)
A closed organ pipe can produce the odd harmonics only. So the frequency of the next harmonic,
n1 = 3n0 = 3 x 83 = 249 Hz.
Example 3. The length of an organ pipe, closed at one end is 90 cm. Find out the frequency of the harmonic next to the fundamental. Velocity of sound in air = 300 m · s1
Answer:
Given
The length of an organ pipe, closed at one end is 90 cm.
A closed pipe emits the odd harmonics only. So, the frequency of the harmonic next to the fundamental is
n1 = 3 x fundamental frequency
= \(3 \cdot \frac{V}{4 l}=3 \times \frac{(300 \times 100)}{4 \times 90}=250 \mathrm{~Hz} .\)
Example 4. The length of an organ pipe open at two ends is twice that of another organ pipe closed at one end. If the fundamental frequency of the open pipe is 100 Hz, find out the frequency of the 3rd harmonic emitted by the closed pipe.
Solution:
Given
The length of an organ pipe open at two ends is twice that of another organ pipe closed at one end. If the fundamental frequency of the open pipe is 100 Hz
The fundamental frequency of the open pipe, \(n_0^{\prime}=\frac{V}{2 l}; \text { so, } V=2 \ln n_0^{\prime}\); so, V = 2ln0‘ [V = velocity of sound in air, l = length of the open pipe]
The length of the closed pipe is \(\frac{l}{2}\). So the frequency of the 3rd harmonic is
n = 3x fundamental frequency
= \(3 n_0=3 \cdot \frac{V}{4\left(\frac{l}{2}\right)}=\frac{3 \cdot 2 \mathrm{~V}}{4 l}\)
= \(\frac{3 \times 2 \times 2 l n_0^{\prime}}{4 l}=3 n_0^{\prime}\)
= \(3 \times 100=300 \mathrm{~Hz} .\)
Example 5. Find out the fundamental frequency of a 125 cm long organ pipe closed at one end. Given, the velocity of sound in air = 350 m s-1.
Solution:
Given, the velocity of sound in air = 350 m s-1.
If the length of the closed pipe is l and the wavelength of the fundamental is λ,
l = \(\frac{\lambda}{4}\) or, λ= 4l
If V is the velocity of sound in air, fundamental frequency, \(n_0=\frac{V}{4 l}=\frac{350 \times 100}{4 \times 125}=70 \mathrm{~Hz}.\)
Example 6. A 20 cm long closed pipe emits a tone of frequency 400Hz. Find out the length of an open pipe emitting a tone of frequency 600 Hz at the same
Solution:
Given
A 20 cm long closed pipe emits a tone of frequency 400Hz.
The fundamental frequency for the dosed pipe, \(n_0=\frac{V}{4 l}\) [l = length of closed pipe]
The fundamental frequency for the open pipe, \(n_0^{\prime}=\frac{V}{2 L}\)
[L = length of open pipe]
∴ \(\frac{n_0}{n_0^{\prime}}=\frac{V}{4 l} \cdot \frac{2 L}{V}=\frac{1}{2} \cdot \frac{L}{l}\)
or, \(L=2 l \cdot \frac{n_0}{n_0^{\prime}}=2 \times 20 \times \frac{400}{600}=26.67 \mathrm{~cm}\)
Example 7. The length of an open organ pipe is twice the length of a closed organ pipe. If the fundamental frequency of the open pipe is 100 Hz, what is the frequency of the third harmonic of the closed pipe?
Solution:
Given
The length of an open organ pipe is twice the length of a closed organ pipe. If the fundamental frequency of the open pipe is 100 Hz,
Let the length of the closed organ pipe = l
Length of the open organ pipe =2l.
Frequency of the fundamental of the open pipe, \(n_0=\frac{V}{2 \cdot 2 l}=\frac{V}{4 l}\) [V= velocity of sound in air]
or, l = \(\frac{V}{4 n_0}=\frac{V}{4 \times 100}=\frac{V}{400}\)
Frequency of the fundamental of the closed pipe, \(n_c=\frac{V}{4 l}=\frac{V}{4} \cdot \frac{400}{V}=100 \mathrm{~Hz}\)
Only the odd harmonics are produced from a closed pipe; so the frequency of the third harmonic = 100 x (3 x 2- 1) = 500 Hz.
Question 8. One end of an open pipe is suddenly closed. It is the closed pipe is 100 Hz higher than the fundamental frequency of the open pipe. Find out this fundamental frequency when both ends are open.
Solution:
Given
One end of an open pipe is suddenly closed. It is the closed pipe is 100 Hz higher than the fundamental frequency of the open pipe.
If V is the tire velocity of sound and l is the length of the pipe, the fundamental frequencies in the open and closed conditions are respectively,
⇒ \(n_0^{\prime}=\frac{V}{2 l} \text { and } n_0=\frac{V}{4 l}=\frac{n_0^{\prime}}{2}\)
The frequency of the 3rd harmonic for the closed pipe,
n = \(3 n_0=\frac{3}{2} n_0^{\prime}\)
∴ \(n-n_0^{\prime}=100 \text { or, } \frac{3}{2} n_0^{\prime}-n_0^{\prime}=100 \text { or, } n_0^{\prime}=200 \mathrm{~Hz}\).
Comparison of Open and Closed Air Columns
Question 9. In winter, the frequency of a tone emitted at 10°C by an open organ tube is 400 Hz. What will be the frequency of this tone at 40°C In summer?
Solution:
Given
In winter, the frequency of a tone emitted at 10°C by an open organ tube is 400 Hz
Let V0 be the velocity of sound in air at 0°C.
So, the velocity at 10°C , V1 = V0(1 + 0.00183 x 10) = 1.0183 V0;
The velodty at 40°C, V2 = VQ(1 + 0.00183 x 40) = 1.0732 V0
The frequency of a tone is proportional to the velocity of sound.
∴ \(\frac{n_1}{n_2}=\frac{V_1}{V_2}\)
or, \(n_2=n_1 \cdot \frac{V_2}{V_1}=400 \times \frac{1.0732 V_0}{1.0183 V_0}=421.57 \mathrm{~Hz}\).
Example 10. A 1 m long uniform cylindrical container Is closed by two thin vibrating membranes A and B at Its two ends. A third thin vibrating membrane C divides the container Into two equal parts. The parts AC and BC arc are filled with hydrogen and oxygen gases, respectively. The membranes A and B are vibrated with the same frequency. Find out the minimum frequency of this vibration so that a node is formed at C. Given, velocities of sound In hydrogen and oxygen gases arc 1100 m – s1 and 300 m · s-1, respectively.
Solution:
Given
A 1 m long uniform cylindrical container Is closed by two thin vibrating membranes A and B at Its two ends. A third thin vibrating membrane C divides the container Into two equal parts. The parts AC and BC arc are filled with hydrogen and oxygen gases, respectively. The membranes A and B are vibrated with the same frequency.
AC = BC = 0.5 m = l (say).
If membranes A and B are vibrated, antinodes are produced at points A and B. Given, a node is produced at point C.
For an antinode at A and its adjacent node at C, the frequency is \(n_H=\frac{V_H}{4 l}=\frac{1100}{4 \times 0.5}=550 \mathrm{~Hz}\)
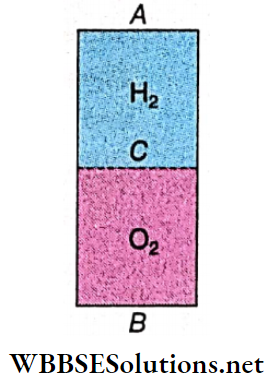
Similarly, for the part BC \(n_O=\frac{V_O}{4 l}=\frac{300}{4 \times 0.5}=150 \mathrm{~Hz}\)
Now, \(\frac{n_H}{n_O}=\frac{550}{150}=\frac{11}{3} or, 3 n_H=11 n_O\)
So, the frequency of the 3rd harmonic in AC matches exactly with that of the 11th harmonic in BC.
∴ The minimum frequency = 3 x 550 = 1650 Hz.
Example 11. Find out the coefficient of linear expansion of the material of an open pipe so that the frequency of any tone emitted from it does not vary with temperature.
Solution:
The fundamental frequencies at 0°C and t°C are respectively,
⇒ \(n_1=\frac{V_0}{2 l_0} \text { and } n_2=\frac{V_t}{2 l_t}\)
Here, V0 = velocity of sound at 0°C, l0 = length of the pipe at 0°C, Vt = velocity of sound at t°C, lt = length of the pipe at t°C.
According to the question, n1 = n2
So, \(\frac{V_0}{2 l_0}=\frac{V_t}{2 l_t}\)
or, \(V_0 l_t=V_t l_0 \quad \text { or, } V_0 l_0(1+\alpha t)=V_0(1+0.00183 t) l_0 \)
(\(\alpha=\text { coefficient of linear expansion }\))
or, \(\alpha=0.00183^{\circ} \mathrm{C}^{-1} \text {. }\)
Determination of the Velocity of Sound in Air by Resonant Air Column: Consider a uniform glass tube P which has its upper end open. The tube is partially filled with water; so the glass tube effectively behaves as a pipe closed at one end.
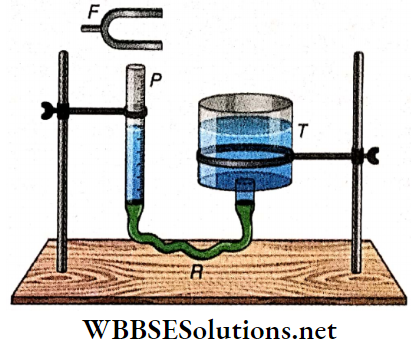
- The tube P is connected to a big water container T through a rubber tube R. The length of the air column in the closed pipe P can be increased or decreased according to the need by raising or lowering the container T. The length of the air column can also be measured from a scale marked on the tube P.
- Now, a tuning fork F vibrating with a frequency n is held over the open end of the tube P. As a result, forced vibration is produced in the air column of P. The air column gradually rises from a very low value until the tuning fork and the air column are in unison.
Factors Affecting Frequency of Vibrating Strings
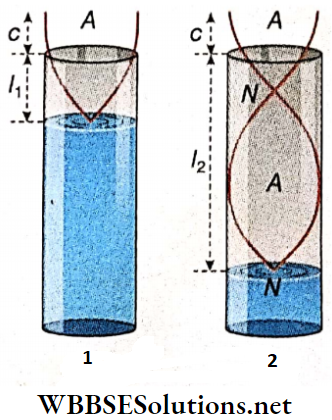
Under these circumstances, the frequencies become equal and a loud sound is heard due to resonance. This corresponds to the fundamental tone emitted from the tube P. So, the fundamental frequency of the pipe is also n. If l1 is the length of the resonant air column, then neglecting the end error we get,
⇒ \(l_1=\frac{\lambda}{4} \quad \text { or, } \lambda=4 l_1\)
So, \(n=\frac{V}{\lambda}=\frac{V}{4 l_1} \quad or, \quad V=4 n l_1\)…(1)
Here, λ is the wavelength of the sound emitted from the tuning fork and V is the velocity of sound in air.
So, V can be calculated from equation (1) by knowing the frequency n of the tuning fork and by measuring the length l1 of the air column in P.
End Correction: Due to the end error, equation (1) cannot provide the accurate value of V. If c is the end error corresponding to the open upper end of P, equation (1) is modified as
V = \(4 n\left(l_1+c\right) \quad \text { or, } \quad l_1+c=\frac{V}{4 n}\)…(2)
Now, the length of the air column in P can be gradually increased further until the next overtone, which is the 3rd harmonic, comes in unison with the tuning fork. Here again, a loud sound is heard due to resonance. If l2 is the length of the air column in this case, taking the end error into consideration we have,
n = \(3 \cdot \frac{V}{4\left(l_2+c\right)} \quad \text { or, } \quad l_2+c=\frac{3 V}{4 n}\)…(3)
Subtracting (2) from (3), \(l_2-l_1=\frac{V}{2 n} \quad \text { or, } \quad V=2 n\left(l_2-l_1\right)\)…(4)
Equation (4) has been obtained by eliminating the end error c effectively. So it provides the accurate value of the velocity of sound V in air.
Estimation Of End Error: From equations (2) and (3), \(3\left(l_1+c\right)=\frac{3 V}{4 n}=l_2+c\)
or, \(3 l_1+3 c=l_2+c \quad \text { or, } 2 c=l_2-3 l_1\)
or, c = \(\frac{1}{2}\left(l_2-3 l_1\right)\)…(5)
So, the end error can be estimated by measuring the values of l1 and l2. It is observed that c is nearly equal to 0.6r, where r is the radius of the tube P.