WBCHSE class 12 physics atom notes
Constituents Of An Atom
Early nineteenth century, John Dalton proposed that all matter is made up of tiny, indivisible particles called ‘atoms! This statement along with other similar statements was later proved to be wrong and discarded
Towards the end of the nineteenth century, William Crookes, Joseph John Thomson, Philipp Lenard, and others, while studying the silent electric discharge in gases at low pressure paved the way for the discovery ofelectrons. Among them, J J Thomson is credited with the discovery of the electron, a subatomic particle. After the discovery of electrons, the myth of the indivisibility of ‘atom’ was dispelled
Electron:
It has been possible to show the emission of electrons from almost all matters through suitable experimental arrangements. Electrons are negatively charged particles carrying a charge which is denoted by e or e-1
Millikan determined the amount of charge of an electron, e = 1.6 × 10-19 coulomb = 4.8 × 10-10 esu J. J Thomson determined the specific charge of an electron, i.e., charge to mass ratio for an electron 1.76 × 1011 C . kg-1.
Therefore, the mass of the electron
Read and Learn More Class 12 Physics Notes
me = \(\frac{\text { amount of charge of an electron }(e)}{\text { specific charge of an electron }\left(\frac{e}{m_e}\right)}\)
9.1 × 10-31 kg-1
= 9.1×10-28 g
Positive charge:
Most materials are electrically neutral. From various electron emission experiments, it was proved that (since matter is neutral) there must be some positive charge also in the atom. Later experiments confirmed the presence of a positive charge on an atom.

WBCHSE class 12 physics atom notes Rutherford’s Atomic Model
Alpha parties scattering experiment:
Rutherford and his co-workers performed the famous alpha par¬ ticle scattering experiment. Alpha particles are emitted from a radioactive source with considerable energy. These a -particles were collimated into a narrow parallel beam which was made incident on a thin foil of a heavy metal like gold (Z = 79), silver (Z = 47), etc.
These metals being ductile, can be easily drawn into a thin foil of width about 107 m. The thinness ensured that eadi a -particle could interact with a single atom in each collision.
Due to collision with the foil, die a -particles were scattered in different directions which were detected by a fluorescent detection screen. Very few a -particles were scattered in a backward direction without penetrating the foil, but being deflected through angles greater than 90°.
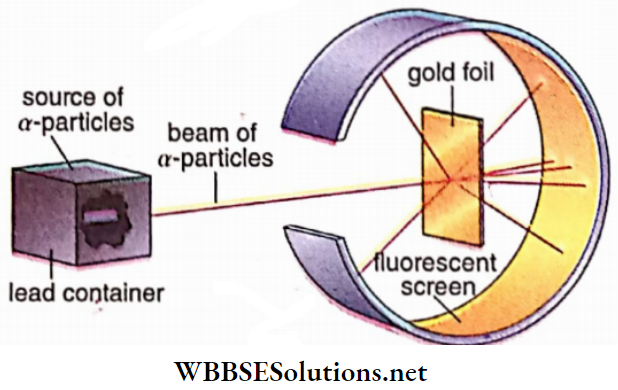
Observation and inference:
- Most of the a -particles passed straight through the metal foil without suffering any deflection. This observa¬ tion leads to the conclusion that most of the space inside the atom is empty.
- Low angle scattering: Some of the alpha particles were scattered through small angles i.e., the scattering angle Here, it is assumed that this scattering
- This takes place due to coulomb attraction between an alpha particle of charge +2e and an electron of charge -e. The deflections of α -particles are so small that an α -particle is about 7000 times heavier than an electron. From this, it is concluded that electrons are embedded discretely inside the atom
- Large angle scattering: Some a -particles, though very few suffered deflection by 90° or larger angles. Some of these a -particles were even deflected through 180° Rutherford quantitatively analyzed the
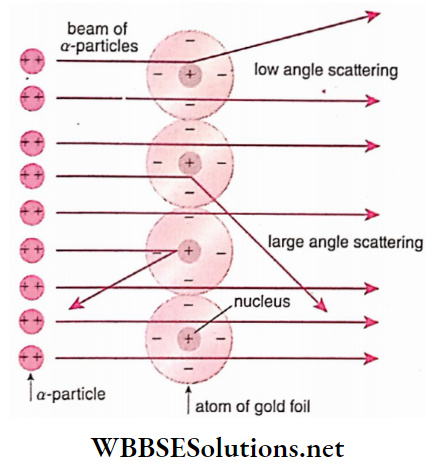
WBBSE Class 12 Atom Notes
A number of these large angle deflections. He argued that, to deflect the a -particle backward, it must experience a large repulsive force which was possible if the entire positive charge and almost total mass of the atom were concentrated in a small space. This confirmed the presence of the nucleus.
Thus the strong electrostatic repulsion between an a -particle of charge +2e and a nucleus of charge +Ze was the cause of these large deflections.
By quantitative analysis, it was also concluded that the diameter ofthe nucleus is about 10-14 m which is about \(\frac{1}{1000}\) times the atomic diameter of 10-10 m. Hence the volume of the nucleus is only about 1 in 1012 parts of the atom
Rutherford’s atomic model is often compared with the solar system, but there are more dissimilarities than resemblances. Similarities are only marginal.
Similarities:

Dissimilarities:

From the observations, Rutherford proposed his atomic model.
The salient features of the model are:
- The nucleus exists at the center of an atom and electrons orbit around the nucleus in circular paths.
- The necessary centripetal force for the orbital motion is provided by the electrostatic attraction between the opposite charges of the nucleus and the electron
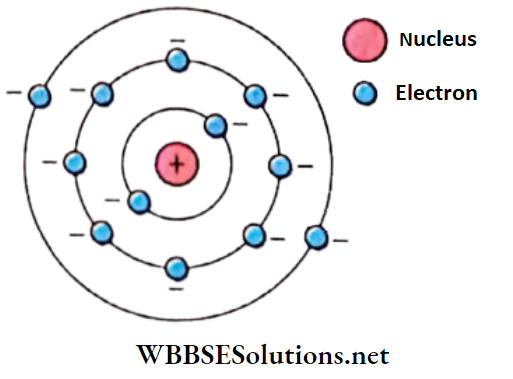
Atoms class 12 notes Drawbacks of the Rutherford model:
InstabilHy of the atom: An orbiting electron in an atom is subjected to centripetal acceleration. According to Max¬ Well’s classical electromagnetic theory, any accelerated charged particle emits electromagnetic radiation.
Since orbiting electrons are accelerated, they should also emit radiation. If this were to happen, the energy of the orbiting electron would keep on decreasing. It would follow a spiral path and ultimately collide with the nucleus.
Theoretical calculations show that under this condition no atom would survive for more than 10-8 s. However, matter is stable implying that atoms too cannot be unstable.
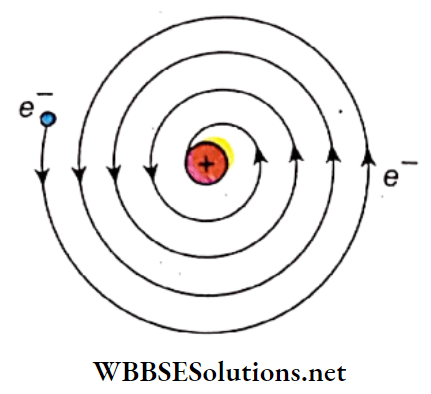
Continuous atomic spectrum: If electron energy in an atom had converted into radiant energy, we would get a con¬ continuous spectrum. Interestingly, atoms of hydrogen, helium, etc. produce a line spectrum instead of a continuous spectrum
Atoms Class 12 Notes
WBCHSE Class 12 Physics Atom Bohr’s Atomic Model
Rutherford’s atomic model was modified by Niels Bohr. The postulates Rutherford model which has no technical difficulties remain intact in the Bohr model of the atom. They are ISM Positively charged nucleus occupies a negligible space at the center of the atom.
Negatively charged electrons revolve around the nucleus in circular orbits. If the mass, speed, and radius of the orbit of an electron are m, v, and r, respectively, the Centripetal force necessary to revolve electrons in a circular orbit,
F = \(\frac{m v^2}{r}\) ……………..(1)
The electrostatic force of attraction acts between the nucleus and electrons. Now, if the atomic number of an atom is Z and the charge of an electron is, the total charge of the nucleus is Ze. So, the electrostatic force of attraction between the nucleus and an electron
F2 = \(\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e \cdot e}{r^2}=\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{r^2}\)
On the other hand, resolving the drawbacks of Rutherford’s model related to the
- Stability of atoms and
- Atomic line spectrum, Bohr introduced some revolutionary ideas that are not consistent with classical physics.
From this, Bohr’s theory of the atom was established and the consequent structure of the atom “is called Bohr’s atomic model
Understanding Atomic Structure for Class 12
Postulates of Bohr’s Theory
Bohr’s model is based on three postulates. These are a[so known as Bohr’s quantum postulates.
- Electrons inside an atom can only revolve in some allowed orbits. When an electron revolves in an allowed orbit, it does not radiate energy.
- Note that, according to classical electromagnetic theory, a rotating (i.e., accelerated) charge radiates energy.
- According to this postulate, the energy of an electron in any allowed orbit remains constant; hence these orbits are called stationary orbits or stable orbits.
- Transition of electrons from one stable orbit to another is possible.
- During the transition, an emission or an absorp¬ tion of radiation occurs.
Its frequency f is determined from the relation, hf = E1~ E2 where h is Planck’s constant and (E1 ~E2) is the energy difference of the electron in the two stable orbits.
When the energy (E1) of an electron in its initial orbit is more than its energy (E2) in its final orbit, i.e., when E1> E2, the difference in energy is converted into the energy of an emitted photon. As a result, the atom radiates energy. In this case,
E1– E2 = hf ……………………………..(1)
Again, if E1 < E2, an external photon should supply the difference in energy. So, in this case, the atom absorbs energy.
E2– E2 = hf ……………………………..(2)
Equations (1) and (2) are called Bohr’s frequency conditions
The orbit. where the angular momentum of the electron Is an integral multiple of \(\frac{k}{2 \pi}\) , is known as a stationary or stable orbit
Now, if the radius of any stable orbit is rn, the speed of
electron in drat orbit be vn, angular momentum,
Ln = momentum x radius of the circular path
So, according to this postulate,
⇒ \(m v_n r_n=\frac{n h}{2 \pi}\)
where, n = 1.2.3,…… This equation is called Bohr’s quantum condition, n is called the principal quantum number of an electron or its orbit. These stable orbits are called Bohr orbits.
It is dear that, none of Bohr’s postulates is consistent with classical physics.
- Yet, the success of Bohr’s theory is borne out by die analysis of the atomic spectrum of hydrogen and some other elements.
- Moreover, a dear qualitative picture related to the atomic structure of any element can be obtained from Bohr’s theory.
- Hence, in spite, of some inconsistency relating to some classical experiments, the Bohr model is considered the basis of the atomic structure of all elements.
Atoms class 12 notes Bohr’s Quantum Condition from de Broglie’s Hypothesis
- According to de Broglie’s hypothesis, a stream of any parties can be considered to be matter waves
- If the stream of particles advances freely, the corresponding matter wave behaves as a progressive wave
- On the other hand, if the stream of particles is confined within a definite region, then naturally the corresponding matter wave behaves as a stationary wave
- From these considerations, de Broglie assumed that,
- A matter wave of electrons confined within an atom is a stationary wave.
- The electronic orbits in an atom should be such that, an integral number of electron waves is present in a complete orbit.
- Otherwise, after a complete revolution, the
- Wave will reach n different point mul It once a Stationary wave will not be formed.
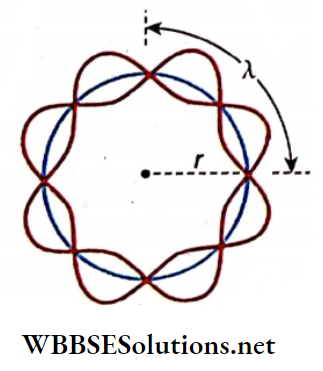
If the radius of the die circular path is r, circumference = 2πr. So, if the wavelength of the electron wave is λ, then
2πr = nλ [n = 1,2,3, ……… ]
Here, four complete waves arc shown In n complete
orbit i.e., n = 4.
Again, according to de Broglie’s hypothesis, if the mass of electron = m and its speed = vn, then
= \(\lambda=\frac{h}{m v}\)
h = planks constant
So, 2πr = \(n \frac{h}{m v}\)
Where n = 1,2,3…….
Or mvr = \(n \frac{h}{2 \pi}\)
For different values of n, the values of r and v will be different; if these values are taken as rn and vn for the definite value of n, then
⇒ \(m v_n r_n=n \frac{h}{2 \pi}\)
Where n = 1,2,3 ……………
This is Bohr’s quantum condition. So the quantum condition is consistent with the idea of de Broglie’s matter wave. But it should be kept in mind that, as an explanation of unstable orbits of an atom, the above-mentioned discussion is a kind of oversimplification. A true explanation can only be made with quantum mechanics.
Common Questions on Atomic Structure
Application of Bohr’s Theory
According to this condition, the radii of different Bohr orbits and orbital speeds of electrons are different. In the case of n -th orbit
⇒ \(m v_n r_n=\frac{n h}{2 \pi} ; \text { where } n=1,2,3 \ldots\)
Accroding \(v_n^2=\frac{n^2 h^2}{4 \pi^2 m^2 r_n^2}\) …………………….(1)
The radius of the n-th Bohr orbit (r„): Electrostatic force of attraction between electron and nucleus of nth orbit
⇒ \(\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{r_n^2}\)
This force of attraction supplies the necessary centripetal force \(\frac{m v_n^2}{r_n}\)
So, \(\frac{m v_n^2}{r_n}=\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{r_n^2}\)
Or, \(v_n^2=\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{m r_n}\) …………………….(2)
Comparing equation (1) and (2), we get
⇒ \(\frac{n^2 h^2}{4 \pi^2 m^2 r_n^2}=\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{m r_n}\)
Or, \(r_n=\frac{\epsilon_0 n^2 h^2}{\pi m Z e^2}\) ……………………………… (3)
This is the expression of the radius of n -th Bohr orbit
Total energy of the electron in n-th orbit (En):
The kinetic energy of the electron in n -th orbit,
⇒ \(E_k=\frac{1}{2} m \nu_n^2=\frac{1}{2} \cdot \frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{r_n}=\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{2 r_n}\)
Now, we know that the potential energy of an electron in n -th orbit due to the electrostatic force of attraction
⇒ \(E_p=-\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{r_n}\)
So, the total energy of the electron in n -th orbit
⇒ \(E_n=E_k+E_p=\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{r_n}\left(\frac{1}{2}-1\right)\)
⇒ \(-\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2}{2 r_n}\) ………………………. (4)
Hence, En= -En and Ep = 2En
Substituting the value of rn from equation (3) into equation (4), we get
⇒ \(-\frac{1}{4 \pi \epsilon_0} \cdot \frac{Z e^2 \cdot \pi m Z e^2}{2 \cdot \epsilon_0 n^2 h^2}=-\frac{m e^4 Z^2}{8 \epsilon_0^2 n^2 h^2}\) …………………….(5)
Hydrogen atom:
The structure of the hydrogen atom is the simplest of all. For hydrogen Z = 1, i.e., the electric charge of its nucleus is +e, and only one electron having charge -e revolves around it.
All the information obtained from Bohr’s theory for this atom is given below
The radius of n – th Bhor orbit:
Substituting Z = 1 in the equation (3) we get,
rn = \(\frac{\epsilon_0 n^2 h^2}{\pi m e^2}\) ……………………………… (6)
Here, ∈0 = permittivity of vacuum
= 8.854 × 10-12C2 . N-1 . m-2
h = Planck’s constant = 6.63 × 10-34 J.s
m = mass of electron = 9.1 × 10-31 kg
e = charge of an electron = 1.6 × 10-19 C
Hence, rn α n²
The information obtained about Bohr orbits from equation (6) is:
1. For n = 1, r becomes minimum, i.e., the orbit lies closest to the nucleus. This is known as the first Bohr orbit or K – shell of the atom. The radius of this orbit is called the first
Bohr radius is denoted by the sign a0, i.e., r1 = a0.
Putting n = 1 in equation (6), we get,
a0 = \(\frac{\epsilon_0 h^2}{\pi m e^2}\)………………..(7)
Substituting the values of the constants in this expression we get
a0 = 0.53 × 10-10 m
= 0.53Å
= 0.053 nm
Angstrom and nanometre units:
1 angstrom (Å) = 10-10m; 1 nanometre (nm) = 10-9m
So, 1nm = 10Å or, 1Å = 0.1 nm. Nowadays nm unit Is more commonly used instead of Å in SI.
2. Using the value of aQ in equation (6), we get, rn = n²a0
Substituting n = 2, 3, 4, in this equation, the radii of the orbits, L, M, N,….. are obtained, respectively.
Since rn the ratio of the radii of the orbits K, L, M, N…. is 1: 4:9:16………………………….
For example,
The radius of the L -orbit, rn = 2². a0 = 4 × 0.53 = 2.12Å;
The radius of the M -orbit, r3 = 3² . a0 = 9 × 0.53 = 4.77Å ; etc.
3. The orbits having radii a0, 4a0, 9a0, ……………………….are permissible and no orbit exists in between them.
For example, the radii of the first and the second Bohr orbits are 0.53 Å and 2.12 Å, respectively. So, no electron can revolve along any circular path having a radius >0.53 A but <2.12 Å. So, the Bohr orbits are discrete, due to integral values of n. Thus n is called the principal quantum number
The energy of the electron to the n-th orbit (En):
Substituting the value of rn from equation (8) in equation (4), we get for hydrogen Z = 1
(En) = \(E_n=-\frac{1}{4 \pi \epsilon_0} \frac{e^2}{2 n^2 a_0}\) ………………. (9)
This is the expression for the total energy of the electron revolving in the n-th orbit of a hydrogen atom.
The inferences made from equation (9) are:
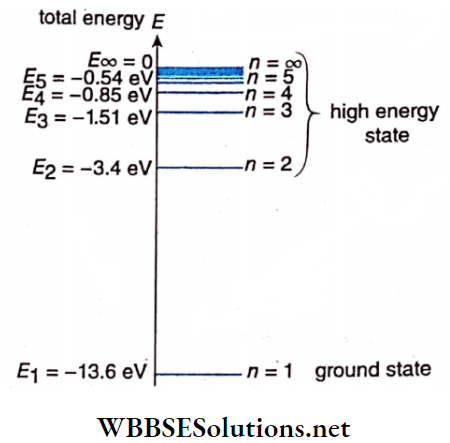
The value of En is negative; hence higher the value of n, i.e., the greater the distance greater the energy of the electron. So in the first Bohr orbit or the K-orbit, the energy of the electron is the least This is called the ground state of the hydrogen atom. Putting n = 1 in equation (9), we get
⇒ \(E_1=-\frac{1}{4 \pi \epsilon_0} \cdot \frac{e^2}{2 a_0}\) ………………………(10)
Using the values of the constants we get, E1 = -13.6eV; hence, the ground state energy of the hydrogen atom = -13.6eV.
Again, putting the value of a0 from equation (7) into equation (10), we get,
E1 = \(-\frac{1}{4 \pi \epsilon_0} \cdot \frac{e^2 \pi m e^2}{2 \epsilon_0 h^2}\)
= \(-\frac{m e^4}{8 \epsilon_0^2 h^2}\)
Where c is the speed of light in a vacuum
Where, r = \(\frac{m e^4}{8 \epsilon_0^2 c h^3}\) = constant
Here, R is a constant called the Rydberg constant Substituting the accepted values of the physical constants, the value of
R comes out to be 1.09737 × 107m–
From equations (9) (10) and (11), we get
⇒ \(E_n=-\frac{R c h}{n^2}=-\frac{13.6 \mathrm{eV}}{n^2}\)
Or, \(E_n \propto \frac{1}{n^2}\)
Thus the energies in the orbits K, L, M, N.. are as \(1: \frac{1}{4}: \frac{1}{9}: \frac{1}{16}\)
So, in the orbits, of hydrogen atoms, the energies are E1 = -13.6eV, E2 = -3.4eV, E3 = -1.51eV, and E4 = -0.85eV, respectively.
Hence, these energy levels are discrete, i.e., the electron cannot exist in any intermediate energy state.
Clearly, as n increases, En becomes less negative i.e., the energy increases. The energy levels of the hydrogen atoms are represented in the energy level diagram the highest energy state corresponds to n = and has energy
⇒ \(E_{\infty}=\frac{13.6}{\infty^2}=0 \mathrm{eV}\)
Note that an electron can have any total energy above QeV. In such a situation, the electron is free and there is a continuum of energy state above
E∞ = \(\frac{13.6}{\infty^2}\)
= 0eV
Class 12 Physics Atoms Chapter Notes
Speed of the electron in n th orbit (v):
In the case of the hydrogen atom, putting Z = 1, we get from equation (2)
⇒ \(v_n^2 \propto \frac{1}{r_n} \text { or, } v_n \propto \frac{1}{\sqrt{r_n}}\)
Again from equation (3), we get
⇒ \(r_n \propto n^2 \text { so, } v_n \propto \frac{1}{n}\)
Hence, the ratio of the speeds of electrons in K, L, M, N … orbit is
rn ∝ n²
So, rn ∝ 1/ n
Hence speed of the electron is highest in the first Bohr orbit
In the case of the hydrogen atom, putting Z = 1 in equation (2), we get
⇒ \(v_n^2=\frac{1}{4 \pi \epsilon_0} \cdot \frac{e^2}{m r_n}\)
Or, \(\frac{1}{4 \pi \epsilon_0} \cdot \frac{e^2}{m v_n r_n}=\frac{1}{4 \pi \epsilon_0} \cdot \frac{e^2}{n \cdot\left(\frac{h}{2 \pi}\right)}\)
= \(=\frac{c}{n}\left[\frac{e^2}{4 \pi \epsilon_0 c\left(\frac{h}{2 \pi}\right)}\right]=\frac{c}{n} \alpha\)
c is the speed of light in a vacuum
Where = \(\alpha\left(=\frac{e^2}{4 \pi \epsilon_0 c\left(\frac{h}{2 \pi}\right)}\right)\) is a dimensionless constant
a is called Sommerfeld’s fine structure constant. Substituting the accepted values of the physical constants, the value of a comes out to be a
⇒ \(\frac{1}{137}\) approximately.
For the first Bohr orbit n = 1, the speed of an electron,
⇒ \(v_1=\frac{c}{137}\) = 2.18 × 106 m.s-1
This speed is approximately part of the speed of light (c) in a vacuum.
So, the speed of an electron in the next orbit is:
⇒ \(v_2=\frac{1}{2} v_1=\frac{c}{274}\)
= \(v_3=\frac{1}{3} v_1=\frac{c}{411}\)
Orbital angular momentum of the electron in n-th orbit (Ln):
From Bohr’s quantum condition, we directly obtain
Ln= nh/2π
Naturally, Ln oc n, i.e., L1: L2: L3: …………….. = 1: 2: 3: ………………….
So, the greater the distance of the orbits, the greater will be the value of
For example,
L1= \(\frac{h}{2 \pi}=\frac{6.63 \times 10^{-34}}{2 \pi}\)
= 1.06 × 10-34m. s-1
L1= 2 L1= 2.12 ×10-34 J.s : etc
Hydrogen Spectrum
1. Atomic spectrum:
Line spectrum:
If electric discharge occurs inside any elementary gas or vapor kept in a discharge tube at a few mm of mercury pressure, the tube acts as a bright source of light. It is generally called a discharge lamp. Discharge lamps of neon, sodium, mercury, and halogen gases are used in our daily lives.
With the help of a prism or other instruments, different fundamental colors of different wavelengths are obtained in the dispersion of light emitted from a discharge lamp. This is known as the atomic spectrum. In this spectrum, there are ultraviolet rays and infrared rays along with visible light.
With the help of a special experimental arrangement, the wavelength of each fundamental ray can be determined. Atomic spectra for different elements are different
The characteristics of the atomic spectra are that these are usually a combination of some thin, bright and discrete lines; i.e., in between two bright lines there is a dark space. This spectrum is known as line spectrum

On the other hand, the spectrum obtained from a heated solid (e.g., an incandescent tungsten lamp) is a continuous spectrum; the different colored lights present in it form continuous illumination on the screen and there is no dark space in between them
Again, the molecular spectrum is usually a band spectrum. Instead of discrete lines, closely spaced groups of lines are so formed that each group appears to be a band. Between two consecutive bands, there is a dark space
2. Balmer series of hydrogen spectrum:
In the visible region of the atomic spectrum of hydrogen, there are four bright lines. The experimental values of their wavelengths are 6563A, 4861 Å, 4341 Å, and 4102 Å. These four lines constitute the Balmer series of the hydrogen spectrum. A Swiss mathematics teacher, Balmer tried to express these wavelengths by a simple relation in 1884, many years before the proposal of the Bohr model, which is
⇒ \(\frac{1}{\lambda}=A\left(\frac{1}{2^2}-\frac{1}{n^2}\right)\) …………………… (1)
Here, λ = wavelength of spectral line,
A = constant and n = 3, 4, 5, …………………………..
The number of complete waves present in unit length is denoted by the reciprocal of wavelength, I; hence j is called the number and is sometimes expressed by the symbol.
Substituting A = 1.09678 × 107 m-1 in equation (1), the experimental values of the wavelength of the spectral lines can be found.
For example,
For n = 3 , λ = 6563 Å; for n = 4, λ = 4861 Å
For n = 5, λ = 4341 Å ; for n = 6, λ = 4102 Å
Moreover, substituting n = 7, 8, 9, different values of /I are obtained and these also belong to the Balmer series. But these wavelengths lie in the ultraviolet region, not in the visible region
Balmer could arrange the spectral lines of hydrogen in a definite pattern, but could not determine relation (1) theoretically
Important Formulas in Atomic Physics
3. Other series of hydrogen spectrum:
Lyman series:
The relation denoting this series is
⇒ \(\frac{1}{\lambda}=A\left(\frac{1}{1^2}-\frac{1}{n^2}\right) ; n=2,3,4, \cdot \cdot\)
Using the same value of A, the values of A obtained from this relation, resemble that of the lines obtained in the ultraviolet region of the hydrogen spectrum. For example, if n
If n = 2, then λ = 1216 Å; if n = 3, then λ = 1026Å
Paschen series:
The relation denoting this series is
⇒ \(\frac{1}{\lambda}=A\left(\frac{1}{3^2}-\frac{1}{n^2}\right) ; n=4,5,6, \cdots\)
From this relation, the wavelength of some spectral lines of the infrared region of the hydrogen spectrum is obtained. For example,
If n = 4 then λ = then 18751 Å
Class 12 physics atoms chapter notes
Brackett series and Pfund series:
In the infrared region of the hydrogen spectrum, some more series are present except the Paschen series; these are the Brackett series and the Pfund series. But in this case, the brightness (or intensity) ofthe corresponding spectral lines is negligibly small.
The relation denoting the Brackett series is,
⇒ \(\frac{1}{\lambda}=A\left(\frac{1}{4^2}-\frac{1}{n^2}\right)\) : n = 5,6,7 ………………..
The relation denoting the Pfund series is
⇒ \(\frac{1}{\lambda}=A\left(\frac{1}{5^2}-\frac{1}{n^2}\right)\) : n = 6,7,8 ………………..
4. Rydberg formula:
Just after the discovery of the Balmer series, Rydberg expressed the following general equation related to the series of spectrums. This is known as the Rydberg formula
⇒ \(\frac{1}{\lambda}=\frac{R}{(m+a)^2}-\frac{R}{(n+b)^2}\) …………………… (2)
Where, R is constant (Rydberg constant) for a particular element, a and b are the characteristic constants for a particular series, m is a whole number that is constant for a given series and n is a varying whole number whose different values indicate the different lines of the series. With the help of equation (2), the series of spectra of most of the elements can be expressed accurately.
Explanation of Hydrogen Spectrum from Bohr’s Theory
Rydberg constant:
The discrete energy levels of the hydrogen atom
En = \(-\frac{R c h}{n^2}\) ……………………. (1)
Here, R = \(\frac{m e^4}{8 \epsilon_0^2 c h^3}\) ……………………. (2)
Where, c = speed of light in vacuum = 3 × 108 m .s-1 and
n = 1,2,3………………..
∴ Ground state energy, E1 = -Rch
Substituting the values of different constants in equation (2), we get, R ≈ 1.09737 × 107m-1
Note that, in the analysis of the Balmer series of the hydrogen spectrum, the value of the constant A obtained (1.09678 × 107 m -1 ) is slightly less than the above value of R.
This difference becomes negligible if the mass of the hydrogen nucleus is corrected. So, we can say that, the constant A is the Rydberg constant, and equation (2) denotes its expression
Calculation of Rydberg constant in the CGS system:
The expression for the Rydberg constant in the CGS system can be obtained by substituting in place of e0 in equation (2). Using the CGS values of the constants, we get
R = \(\frac{2 \pi^2 m e^4}{c h^3}\)
= 109737 cm -1
The wavelength of the emitted radiation:
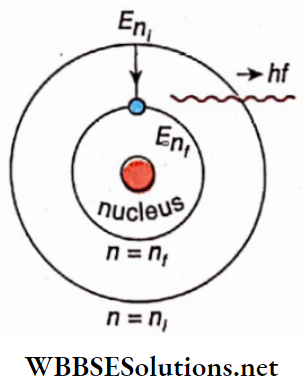
Let the electron in a hydrogen atom make a transition from a higher energy state Eni to a lower energy state Eni According to Bohr’s postulate, a photon will be emitted from the hydrogen atom due to this transition.
If the frequency of this photon is f (wavelength \(\) ) then
⇒ \(E_{n_i}-E_{n_f}=h f=\frac{h c}{\lambda}\) …………….(3)
Substituting n = ni and n = nf respectively in equation (1), we get
⇒ \(E_{n_i}=-\frac{R c h}{n_i^2} \text { and } E_{n_f}=-\frac{R c h}{n_f^2}\)
So, from equation (3), we get,
⇒ \(\frac{h c}{\lambda}=-\frac{R c h}{n_i^2}+\frac{R c h}{n_f^2}\)
= \({Rch}\left(\frac{1}{n_f^2}-\frac{1}{n_i^2}\right)\)
Or \(\frac{1}{\lambda}=R\left(\frac{1}{n_f^2}-\frac{1}{n_i^2}\right)\) ……………………….. (4)
1. Balmer series:
If the electron in a hydrogen atom jumps from any one of the energy states E3, E4, E5,……………. etc. to the energy state ,E2 then putting ni = 3,4, 5, …………… and nf= 2 in equation (4), we get
⇒ \(\frac{1}{\lambda}=R\left(\frac{1}{2^2}-\frac{1}{n^2}\right) ; n=3,4,5, \cdots\) …………. (5)
This relation indicates the Balmer series of the atomic spectrum of hydrogen
2. Other series:
Similarly, substituting ni = 2, 3, 4, and nf = 1 in equation (4), we get the Lyman series :
⇒\(\frac{1}{\lambda}=R\left(\frac{1}{1^2}-\frac{1}{n^2}\right)\): n =2,3,4 ………………. (6)
Again, substituting nt = 4, 5, 6— and nÿ= 3 , we get Paschen series
⇒\(\frac{1}{\lambda}=R\left(\frac{1}{3^2}-\frac{1}{n^2}\right)\): n = 4,5,6 ………………. (7)
Substituting ni = 5, 6, 7, ……….. and nf= 4, we get Brackett series
⇒\(\frac{1}{\lambda}=R\left(\frac{1}{4^2}-\frac{1}{n^2}\right)\): n = 5,6,7 ………………. (8)
Substituting ni = 6,7,8,…….. and nf= 5, Pfund series is obtained:
⇒\(\frac{1}{\lambda}=R\left(\frac{1}{5^2}-\frac{1}{n^2}\right)\): n = 6,7,8 ……………… (9)
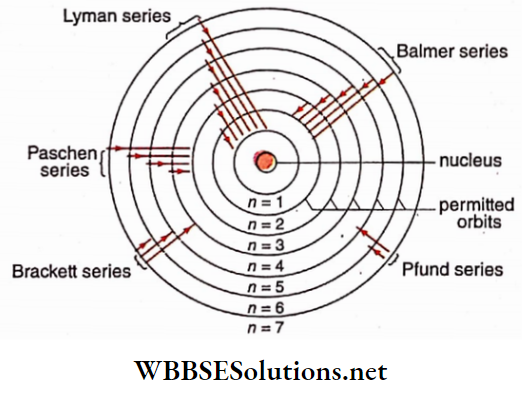
So the relations, shown by Balmer and other scientists for different wavelengths of the atomic spectrum of hydrogen, can be established accurately from Bohr’s theory. The basis of the success of Bohr’s theory lies in the accurate explanation of the hydrogen spectrum, although it has deviations from classical physics Different series of the atomic spectrum of hydrogen are
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
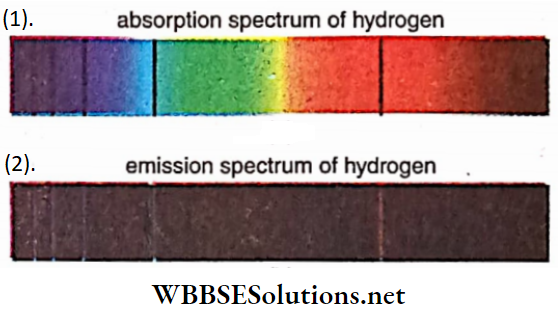
Absorption and emission spectrum of hydrogen:
We have seen that if an electron jumps from a higher energy state to a lower one, energy equal to the difference between the states is emitted where AE = hc/ λ. So the reverse process, if the atom absorbs a photon of wavelength A, the electron will jump from the lower energy state to the higher energy state. Since the difference in energy states is fixed, the wavelength of the absorbed photon and that of the emitted photon are exactly equal.
Thus, if the light coming from a source (e.g., an incandes¬ cent tungsten lamp) passes through hydrogen gas, due to absorption, some dark lines are formed in its spectrum. These dark or black lines form an absorption spectrum The bright lines present in the emission spectrum obtained from the hydrogen gas discharge tube, become dark in the continuous spectrum of other sources while absorbed by hydrogen
Class 12 physics atoms chapter notes
WBCHSE Class 12 Physics Atom Bohr’s Atomic Model Numerical Examples
Example 1. If the value of the Rydberg constant of hydrogen is 109737 cm-1, determine the longest and the shortest Paschen wavelength of the Balmer series.
Solution:
If the wavelength of the Balmer series is λ, then
⇒ \(\frac{1}{\lambda}=R\left(\frac{1}{2^2}-\frac{1}{n^2}\right)\) n = 3,4,5 ………….
R = Rydberg constant
Substituting the minimum value of n, i.e., n = 3, we get
⇒ \(\frac{1}{\lambda}=R\left(\frac{1}{2^2}-\frac{1}{3^2}\right)=R \times \frac{5}{36}\)
Or, \(\lambda=\frac{36}{5 R}=\frac{36}{5 \times 109737}\)
= \(\frac{36 \times 10^8}{5 \times 109737}\)
= 6561 Å
This is the longest wavelength.
Again, by substituting the maximum value of n, i.e n = we get
⇒ \(\frac{1}{\lambda}=R\left(\frac{1}{2^2}-\frac{1}{\infty}\right)=R \times \frac{1}{4}\)
Or, \(\) cm-1
= \(\lambda=\frac{4}{R}=\frac{4}{109737} \mathrm{~cm}\)
= \(\frac{4 \times 10^8}{109737}\)
= 3645 Å
This is the shortest wavelength
Example 2. As a result of a collision with a 20 eV energy, a hydrogen atom is excited to the higher energy state, and the electron is scattered with a reduced velocity. Subsequently, a photon with a length of 1216 Å is emitted from the hydrogen wave atom Determine the velocity of the electron after collision
Solution:
The wavelength of the emitted photon,
λ = 1216 Å = 121 6 × 10-8 cm
∴ The amount of energy gained by the hydrogen atom from the electron
E1 – E2 = hf
= \(\frac{h c}{\lambda}=\frac{6.625 \times 10^{-27} \times 3 \times 10^{10}}{1216 \times 10^{-8}}\)
= 1.634 × 10-11 erg
According to the questions, the initial kinetic energy of the electron
= 20 eV = 20 × 1.6 × 10-12 erg
= 3.2 × 10-11erg.
∴ The remaining kinetic energy of the electron after its collision with a hydrogen atom
⇒ \(\frac{1}{2} m v^2=(3.2-1.634) \times 10^{-11}\) = (3.2 – 1.634)
= 1.566 × 10-11 erg
Or, v = \(\sqrt{\frac{2 \times 1.566 \times 10^{-11}}{9.1 \times 10^{-28}}}\)
= 1.855 × 10 cm .s-1
Real-Life Applications of Atomic Models
Example 3. Light from a discharge tube containing hydrogen atoms is incident on the surface of a piece of sodium. The maximum kinetic energy of the photoelectrons emitted from sodium is 0.73 eV. The work function of sodium is 1.82 eV. Find
- The energy of photons responsible for the photoelectric emission
- The quantum numbers of the two orbits in the hydrogen atom involved in the emission of photons and
- The change in angular momentum ofthe electron of a hydrogen atom in the two orbits
Solution:
1. According to the photoelectric equation, energy of the photon, hf = Emax + W0 = 0.73 + 1.82 = 2.55 eV
2. The energy difference between the two orbits is 2. 55 eV. Now in case of the hydrogen atom
Energy in the ground state (n= 1) , E1 = -13.6 eV
Energy in n = 2 state E2 = \(\frac{E_1}{2^2}=-\frac{13.6}{4}\)
= -3.4 eV
Energy in n = 3 state E3 = \(\frac{E_1}{3^2}=-\frac{13.6}{9}\)
= 1.51 eV
Energy in n = 4 state E3 = \(\frac{E_1}{2^2}=-\frac{13.6}{16}\)
E4– E2 = 2.55 eV
So, the quantum numbers of the two orbits are 4 and 2.
3. According to Bohr’s postulate, change in angular momentum
= \(4 \cdot \frac{h}{2 \pi}-2 \cdot \frac{h}{2 \pi}=\frac{h}{\pi}\)
= \(\frac{6.625 \times 10^{-27}}{\pi}\)
= 2.11 × 10-27 erg. s
Atomic Structure Class 12 Notes
Example 4. When ultraviolet light of wavelengths 800Å and 700Å are incident on the hydrogen atom at ground state, electrons are emitted with energies 1.8 eV and 4 eV, respectively. Determine the value of Planck’s constant
Solution:
Let the ground state energy of the hydrogen atom = -E0. Hence, the minimum amount of energy E0 is required to liberate its electron, i.e., the work function of the hydrogen atom, W0 = E0.
So, if the incident photon can provide E amount of kinetic energy to the electron, then
hf = E + E0 or, hc/λ = E + E0
In the first case,
λ1 = 800 Å = 800 × 10-8 cm ,
E1 = 1.8 eV = 1.8 × 1.6 × 10-12 erg
In the second case,
λ2 = 700 A = 700 × 10-8 cm ,
E2 = 4.0 eV = 4.0 × 1.6 × 10-12 erg
From equation (1), we get,
⇒ \(\frac{h c}{\lambda_1}-\frac{h c}{\lambda_2}=E_1-E_2\)
Or, \(h c\left(\frac{1}{\lambda_1}-\frac{1}{\lambda_2}\right)=E_1-E_2\)
= E1 – E2
Or, \(h=\frac{E_2-E_1}{c} \cdot \frac{\lambda_1 \lambda_2}{\lambda_1-\lambda_2}\)
∴ h = \(\frac{(4.0-1.8) \times 1.6 \times 10^{-12}}{3 \times 10^{10}} \times \frac{800 \times 700 \times 10^{-16}}{(800-700) \times 10^{-8}}\)
= 6.57 × 10-27 erg.s
Examples of Electron Configuration
Example 5. In absorbing 10.2 eV energy, the electron of a hydrogen atom Jumps from Its Initial orbit to the next higher energy orbit. As the electron returns to the former orbit, a photon Is emitted. What Is the wavelength of this photon?
Solution:
According to Bohr’s postulate,
hν = Ei – Ef
Or, \(\frac{h c}{\lambda}=E_l-B_f\)
Or,\(\lambda=\frac{h c}{E_l-E_f}\)
The difference between the two energy levels,
Ei – Ef = 10.2 eV = 10.2 × 1.6 × 10-12 erg
λ = \(\frac{6.55 \times 10^{-27} \times 3 \times 10^{10}}{102 \times 16 \times 10^{-12}}\)
= 1204 × 108cm
= 1204 Å
Atomic structure class 12 notes
WBCHSE Class 12 Physics Atom Ionisation Energy And Ionisation Potential
Ionization energy:
The minimum amount of energy required to ionize an atom of an element at its ground state is called the ionization energy of that element.
Example:
The ground state energy of hydrogen atoms = -13.6 eV. In a H+ ion, the only electron of the corresponding hydrogen atom has been removed. In this state, the electron is no longer attracted by the atom, i.e., its potential energy becomes zero.
Again, the condition for minimum energy to be possessed by the electron is that its kinetic energy is zero; as a result its total energy = kinetic energy + potential energy = 0. Naturally, if the electron is brought from a -13.6 eV energy state to a zero energy state, the atom can be converted into an ion. So, the minimum amount of energy supplied = 0- (-13.6) = 13.6 eV
Hence, the ionization energy of hydrogen = 13.6 eV.
Ionization potential:
The minimum potential to be applied to an atom of an element in its ground state to convert it into a positive ion is called the ionization potential of that element.
Example:
According to the definition, if 1 V potential is applied to an electron having charge -e, the gain in energy of the elec¬ tron = 1 eV.
Ionization energy of hydrogen = 13.6 eV; so, to supply this 13.6 eV energy to the electron of the hydrogen atom, a minimum 13.6V potential should be applied to it.
Hence, the ionization potential of hydrogen = 13.6 V
Atomic Structure Class 12 Notes
Atom Ionisation Energy And Ionisation Potential Numerical Examples
Example 1. A hydrogen atom in its ground state, is excited using monochromatic radiation of wavelength 975 Å. How many different lines are possible in the resulting spectrum? Calculate the longest wavelength amongst them. Given, ionization energy of the hydrogen atom is 13.6 eV
Solution:
Wavelength of incident radiation,
A = 975 Å = 975 × 10-8cm
∴ Energy of this photon
hf = \(\frac{6.625 \times 10^{-27} \times 3 \times 10^{10}}{975 \times 10^{-8}}\)
= \(\frac{6.625 \times 10^{-27} \times 3 \times 10^{10}}{975 \times 10^{-8}} \mathrm{erg}\)
= \(\frac{6.625 \times 10^{-27} \times 3 \times 10^{10}}{975 \times 10^{-8} \times 1.6 \times 10^{-12}} \mathrm{eV}\)
= 12.47 eV
The ionization energy of the hydrogen atom = 13.6 eV, i.e., the ground state energy of this atom = -13.6 eV. So, the energy ofthe excited state in which the atom is raised under the influence of incident radiation = – 13.6 + 12.74 = -0.86 eV.
The quantum number in the ground state = 1. So, if the quantum number in the excited state be n, then
– 0.86 = \(-\frac{13.6}{n^2}\)
Or = \(\frac{13.6}{0.86}\)
= 16 (approx)
So, n = 4, i.e., the excited state is the fourth Bohr orbit. During the transition from the fourth Bohr orbit to the ground state, the decrease in energy ofthe atom may occur in 6 different ways. As a result, 6 lines will be obtained in the spectrum
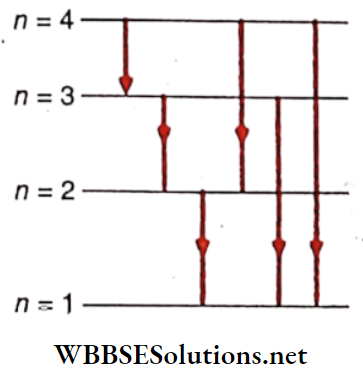
Of them, during the transition from n = 4 to n = 3, the energy difference is the least and hence in this case, the wavelength of the emitted spectral line will be the maximum
Energy of the third Bohr orbit = – \(\frac{13.6}{3^2}\)
= -1.51 eV
∴ Decrease in energy due to transition from n = 4 to n = 3
=-0.86 -(-1.51) = 0.65 eV
Hence, the relation hf = hc/ λ
= E4 – E3 gives
= \(\frac{h c}{E_4-E_3}\)
= \(\frac{6.625 \times 10^{-27} \times 3 \times 10^{10}}{0.65 \times 1.6 \times 10^{-12}}\)
= 19110 × 10-8 cm
= 19110 Å
The relation between the energy of photon E and wavelength A after substituting the values of different constants is E
eV =12400/λ (in Å)
In any calculation, this relation can be used directly.
Success and Failure of the Bohr’s Theory
1. With the help of Bohr’s theory, the spectrum of hydrogen or a hydrogen-like atom can be explained almost accurately. QQ With the help of Bohr’s theory, the main characteristics of the atomic spectrum of alkali metals can be explained.
2. Bohr’s theory cannot analyze the energy of atoms having more than one electron, nor can it explain the wavelengths of spectral lines of such atoms quantitatively. Still Bohr model is the most suitable model supporting the electronic configuration of any atom.
3. Actually, every spectral line of the spectrum of hydrogen or other atoms consists of several spectral lines; they remain so close to each other that with the help of an ordinary prism disperser, they cannot be identified separately.
4. This fine structure of every primary spectral line cannot be explained with the help of Bohr’s theory. This can be called the limitation of Bohr’s theory instead of its failure.
5. If a charged particle possesses acceleration, it radiates electromagnetic radiation. This theory of classical physics has been proved by different experiments. However, Bohr’s theory fails to explain why no electromagnetic radiation is emitted from the electron having centripetal acceleration while revolving in a Bohr orbit.
WBCHSE Class 12 Physics Atom X-rays
Roentgen’s discovery:
In 1895, German physicist Wil helm Roentgen observed that, when high-velocity cathode rays were obstructed by any solid target, an invisible ray came out from that target.
The characteristics of this ray that he observed are. The rays:
- Can produce fluorescence,
- Can affect photographic plates,
- Can penetrate thin sheets of light materials like paper, glass, wood, aluminum, etc.
- Cannot penetrate heavy metals like iron, lead, etc. Rather. it casts a shadow behind them
At that time, the nature of this ray was unknown, and hence eV = hfx Amin Roentgen called this ray X-ray
Nature of X-rays
X-rays are not deflected by electric or magnetic fields; from this, we can conclude that X-rays are not streams of charged particles.
So, an X-ray is either a stream of high-velocity uncharged particles or a kind of wave
If an X-ray is considered to be a kind of wave, it should show properties like interference, diffraction, etc. But in any traditional experimental setup, these properties were not observed, until finally Max von Laue and others after him observed the diffraction of X-ray by passing it through three-dimensional crystals: Thus it was proved that X-ray is a kind of wave. The wavelength of this ray is so small that its diffraction is possible only by crystals of regular intermolecular space (10-8 cm approximately).
Later on, it was possible to observe the wave properties of X-rays like reflection, refraction, interference, and polarisation.
So, it has been observed, that an X-ray is not a stream of any high-velocity uncharged particles, but an electromagnetic wave like visible light. Though compared to visible light, the wavelength of X-ray is very small, almost \(\frac{1}{1000}\) parts or even less than that of the former. It was not until 1923 when A. H. Compton, using his X-ray scattering experiment, established the particle nature of photons, and hence of X-rays
Frequency and Wavelength of X-ray
According to quantum theory, electromagnetic radiation is a stream of massless and chargeless photons which travels
With the speed of light (c = 3 × 108 m . s-1). The energy of each photon,
E = hf = hc/λ ………………(1)
Here, h = Planck’s constant =6.63 × 10-34J . s;
f = frequency and λ = wavelength of the electromagnetic wave
When an electron (charge = e ) overcomes a potential difference V, energy gained by it = eV. If this energy is spent entirely to eject an X-ray photon, the energy and frequency of the photon become maximum. If the corresponding wavelength is taken as λmin, then
eV = \(h f_{\max }\) = \(h \frac{c^{\circ}}{\lambda_{\min }}\)
Or, \(\lambda_{\min }=\frac{h c}{e V}\) ………………(2)
In Sl, e = 1.6 × 10-19 C, c = 3 × 10-8 m.s-1 and
h = 6.63 × 10-34J . s
If the potential difference between the anode and the cathode is V, putting these values in equation (2) we get
= \(\frac{1.243 \times 10^{-6}}{V} \mathrm{~m}\)
= \(\frac{12430}{V}\) Å
= \(\frac{1243}{V}\) ……………(3)
For example,
If V = 50 kV = 50000V, then λmin = 0.2486 Å
If V = 10 kV = 10000 V, then λmin = 1.243 Å
Discussions:
1. When the whole energy of an electron (eV) belonging to cathode rays is utilized to bombard the target to produce a photon, only then the wavelength of the X-ray becomes equal to λmin as given in equation (2). Generally, the whole energy is not converted into the energy of an X-ray photon; hence the wavelength becomes more than λminin For this, is called the minimum wavelength of X-ray.
2. The higher the value of the potential difference Vi smaller the wavelength of X-rays and hence greater its penetrating power. From equation (3), we see that V should be more than 106 volts to produce hard X-rays equivalent to the wavelength 0.01 Å
3. The wavelength of X-ray \(\frac{1}{1000}\) Is part of the wavelength of visible light. Hence, from the equation, E = \(\frac{h c}{\lambda}\), we can say that, the energy of an X-ray photon is about 1000 times greater.
4. Soft and hard X-rays:
The wavelength range of X-rays is from 0.01 Å to 10Å. If λ ≈ 10 A, according to the relation E = hc/λ, the energy of the X-ray photon becomes 1240 eV or 1.24 keV (approx.); this kind of X-ray has less penetrating power and is called soft X-ray. On the other hand, if λ =; 0.01 Å, the energy of the X-ray photon becomes 1.24 MeV (approx.). Due to this high energy, the penetrating power of X-ray becomes very high. It is known as a hard X-ray
Uses of X-rays:
- Important uses of X-rays in medical science:
- Radiography: X-rays are used for the detection of fractured bones and kidney stones.
- Radiotherapy: Hard X-rays are used in radiotherapy to destroy the cancer-affected cells.
- Fluoroscopy: Fluoroscopy is an imaging technique in which X-rays are used to take real-time moving images of the internal structures of a living body.
Important uses of x rays in other fields:
- X-rays are used to analyze the structures of different crystals.
- In metallurgy, X-rays are used to determine the defects in metallic castings
WBCHSE Class 12 Physics X-rays And Atomic Numbers
When a solid target (made of solid copper or tungsten) is bombarded with a stream of high-velocity electrons having a kinetic energy of some keV or higher X-rays are emitted from the target. Sometimes, this kind of material absorbs X-rays. Analyzing the emitted or absorbed X-rays, we come to know many aspects of the atomic structure of these elements
Consider a molybdenum (Mo) target being bombarded by a stream of electrons having a kinetic energy of 35 keV. The spectrum of the wavelengths of X-rays is thus produced.
This spectrum is formed due to the superposition of two kinds of spectra: O continuous spectrum, characteristic or line spectrum. A continuous X-ray spectrum superimposed with some brighter lines is formed on the photographic plate. The origins of the two spectra are different and explained in the next article.
Continuous X-ray Spectrum
During the discussion of continuous spectrum, the characteristic spectrum consisting of two sharp peaks will be overlooked.
Suppose an electron traveling with kinetic energy K0 undergoes collision with an atom of molybdenum Assume that the electron loses Δk amount of energy by this collision. This energy is converted into the energy of an X-ray photon. It should be mentioned here that, the atom being much heavier than an electron, the amount of energy transferred to the atom is neglected.
The electron may collide again with another atom after its colli¬ sion with the first atom. In this case, the electron collides with (K0-ΔK) amount of energy. The X-ray photon thus emitted, generally possesses less energy than that of the previous photon.
In this way, the electron may suffer successive collisions with different atoms until it comes to rest. The photons thus emitted during these collisions having different energies, form a part of the continuous spectrum of X-rays. But in actual practice, a tar
Get is hit with innumerable electrons. Hence, the entire X-ray spectrum looks like that. This kind of spectrum has an important characteristic. It has a definite cut-off wavelength (say, λmin). Below this cut-off
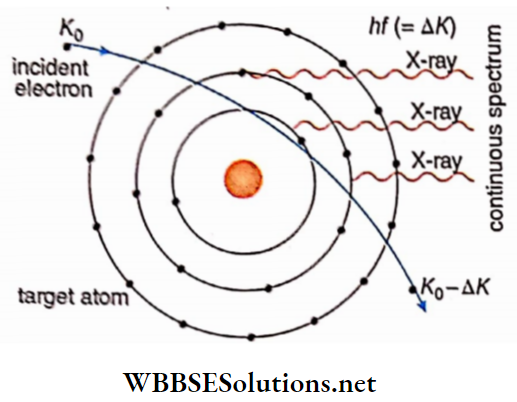
Wavelength, there is no existence ofthe spectrum. If the electron loses its whole kinetic energy (XQ) in the first impact, an X-ray of wavelength is emitted. If/ be the frequency ofthe emitted X-ray photon, then
⇒ \(K_0=h f=\frac{h c}{\lambda_{\min }}\)
⇒ \(\lambda_{\min }=\frac{h c}{K_0}\)………………(1)
So, the die value does not depend on the nature ofthe solid used as the target. So, if copper is used instead of molybdenum as the die target material, the continuous X-ray spectrum changes but the die value of Am;n remains the same
WBCHSE Class 12 Physics Atom X-rays And Atomic Numbers Numerical Examples
Example 1. If a stream of electrons of kinetic energy 36 keV is bombarded on a molybdenum target, what will be the cut-off wavelength of the emitted X-ray? nucleus
Solution:
We know that, \(\lambda_{\min }=\frac{h c}{K_0}\)
Where h = 4.14 × 10-15 eV. s, c = 3 × 108 m .s1
Given. K0 = 36 keV= 3.6 × 10-4 eV
∴ λmin = \(\frac{4.14 \times 10^{-15} \times 3 \times 10^8}{3.6 \times 10^4}\)
= 3.45 × 10-11 m
= 0.0345 nm
Example 2. What minimi terminal potential difference of a Coolidge tube should be maintained to produce an X-ray of wavelength 0.8 Å? [h = 6.62 × 10-34 J.s, e = 1.60 × 10-19 r= C = 3 × 108 m. s -1]
Solution:
The energy of X-ray photons,
E = hc/λ
[here, λ = 0.8 Å = 0.8 × 10-10 m]
If electrons are accelerated in an X-ray tube by a potential difference V, the energy of an electron = eV; if this energy is completely converted into the energy of an X-ray photon, the value of V will be the minimum
So, eVmin = \(\frac{h c}{\lambda}\)
Vmin = \(\frac{h c}{e \lambda}=\frac{6.62 \times 10^{-34} \times 3 \times 10^8}{1.60 \times 10^{-19} \times 0.8 \times 10^{-10}}\)
= 15516 V
WBCHSE physics class 12 atomic notes Characteristic X-ray Spectrum
The two sharp peaks in the spectrum of characteristic X-rays are named as Kα and Kβ These two peaks mainly form the spectrum of the characteristic X-rays of molybdenum.
In an X-ray tube (Coolidge tube), the target is bombarded by high-energy cathode rays. The electrons present in the rays are of very high energy, their effect is not limited to the outer electron levels; the levels adjacent to the nucleus are also affected.
If an electron from any of these levels comes out of the atom, a deficit of electrons occurs in that orbit. If this deficit occurs in the X-orbit (the orbit closest to the nucleus), an elec¬ tron from a higher energy state will transit to this X-orbit.
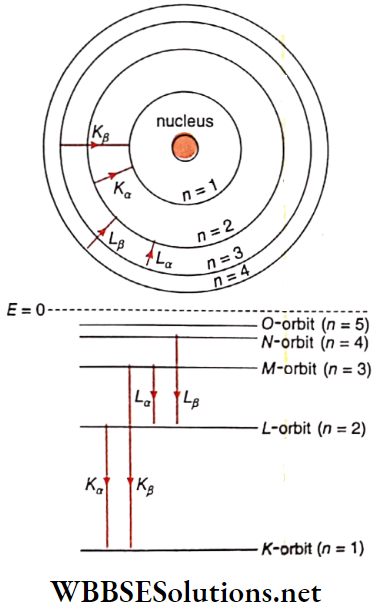
Now, during the transition of an electron from L -orbit to Korbit, it radiates energy as a photon. This radiation forms the Kα -peak of the characteristic X-ray spectrum of molybdenum. The energy level diagram of molybdenum is How different peaks of the spectrum are formed has been shown in this diagram. Again, during the transition of the electron from M orbit to K-orbit, the radiation thus emitted forms the Kβ-peak of the spectrum.
This spectrum contains several smaller peaks and the brightness of these lines in the spectrum is very low. Formation of Lα and Lβ peaks among the small peaks is als
Moseley’s Law
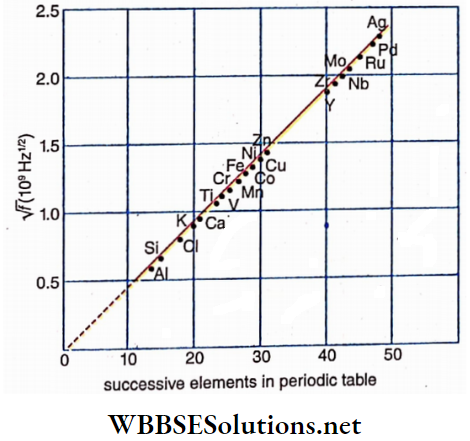
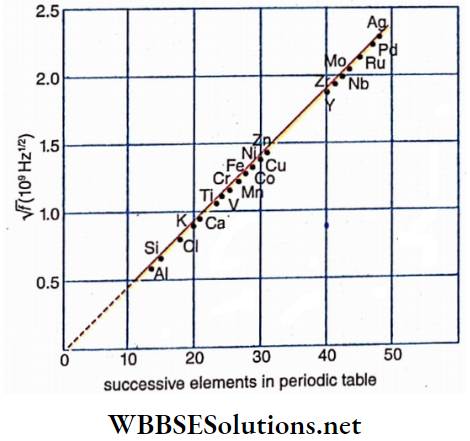
In 1913, British physicist H. G. I. Moseley analyzed the spectrum of characteristic X-rays of all the elements he knew as targets. He observed that the spectra of different elements, particularly the Kα -peaks follow a particular rule. According to the position of different elements in the periodic table, he drew a graph of the square root of the frequency of Kα and obtained a straight line.
A part of the graph based on which, Moseley concluded that “there was a fundamental quantity which increases by regular steps as we pass from one element to the next”. In 1920 Rutherford identified this quantity as the atomic number Z of the element which denotes the number of positive charges present in the nucleus. So, only from the atomic number of an element can its identity be known, not from its atomic weight.
Statement of Moseley’s law:
The square root of the frequency of a peak of the characteristic X-ray spectrum of
Any element is directly proportional to the atomic number of that element.
If the frequency of Kα -line of any element having atomic num¬ ber Z be f. Then according to Moseley’s law,
√f ∝ Z
Explanation of Moseley’s plot from Bohr’s theory:
With the help of the equation of the n-th energy state of an atom obtained from Bohr’s theory, this plot can be explained.
Equation (13)
⇒ \(E_n=-\frac{R c h}{n^2}=-\frac{13.6}{n^2} \mathrm{eV}\)………………(1)
Where, n = 1,2,3,
We know that in an atom containing two or more electrons, only two are in the K-orbit Let any of these come out of the atom. Then the electrons present in other orbits like L, and M, would experience not only the effect of the positive charge of the nucleus but also the influence of the negative charges of the remaining electron in the kT-orbit.
This is because the radius of the kT-orbit in an atom is the least compared to that of other orbits. So, we can assume that, relative to the surface of the sphere on which the electron of the K-orbit lies, the other electrons lie outside.
So, the effective amount of positive charge that attracts an electron of L, M, orbits is (Z- 1)e, where Z is the atomic number. The above equation (1) is applicable for hydrogen atoms. In the case of an atom having atomic number Z, its changed form is applicable. The equation for the n-th energy of the atom is
En = \(E_n=-\frac{13.6(Z-1)^2}{n^2}(\text { in } \mathrm{eV})\) ……………. (2)
The earlier discussion shows that Ka of the spectrum is produced due to the transition of an electron from the L(n =2) orbit to the K(n = 1) orbit. Due to this transition, if the frequency of the emitted X-ray is f, then
⇒ \(h f=\left(E_n\right)_{n=2}-\left(E_n\right)_{n=1}=E_2-E_1\)
= \(-\frac{13.6(Z-1)^2}{2^2}+\frac{13.6(Z-1)^2}{1^2}\) (in eV)
∴ hf = 10.2(Z- 1)²
√f ∝ (Z – 1)…………………………………. (3)
Equation (3) is the equation of a straight line. Hence, the graph of the square root of the frequency of the peak Kα concerning the atomic number of the atom is a straight line. In this way, the theoretical basis of Moseley’s plot is established in the light of Bohr’s theory.
Importance of Moseley’s work:
1. According to Moseley’s law, the characteristic X-ray spectrum is regarded as the identifying character of an element.
2. Before 1913, the elements were arranged in the periodic table according to the increasing order of their atomic weights.
3. Despite that, according to the basis of chemical tests, some elements were placed before the elements hav¬ ing comparatively lower atomic weights in the periodic table. The reason for this exception was not understood before Moseley’s analysis.
4. Moseley showed that there would not be any exception to the arrangements of the periodic table if we arranged the elements according to their increasing atomic numbers.
5. There were many vacant places in the periodic table in 1913. After the discovery of Mosley’s law, it has been possible to fill those gaps accurately.
6. Lanthanide elements are more or less identical in respect of their chemical properties. Thus, it was difficult to identify
WBCHSE Class 12 Physics Atom Conclusion
Bhor model of an atom is based on three postulates
1. For the revolutions of electrons inside an atom, there are some allowed orbits. When an electron revolves in an allowed orbit, it does not radiate energy. Since the energy of an electron in any allowed orbit is constant, die orbits are called stable orbits.
2. An electron can transit from one stable orbit to another. During this transition, homogeneous absorp¬ tion or emission of radiation occurs whose frequency is determined from the relation, hf = E1~ E2. [h = Planck’s constant, E1~ E2 energy difference of the electron in the two stable orbits]
3. The orbits, where the angular momentum of the electron is an integral multiple of h/2λ are the only allowed orbits and are called Bohr orbits.
4. In the visible region of the atomic spectrum of hydrogen, four bright lines can be observed. Their wavelengths, as obtained from the experiment are 6563 Å, 4861 Å, 4341 Å, and 4102 Å. These four lines are known to form the Balmer series of hydrogen spectra.
5. The number of complete waves of radiation present in unit length is 1/λ, and hence 1/λ (=f) is called wave number.
6. In the first Bohr orbit, Le., in K-orbit, the energy electron becomes minimum. This is known as the ground state energy or the lowest energy level of the atom.
7. The minimum amount of energy required to ionize an atom of an element in its ground state, is called the ionization energy of that element.
The ionization energy of hydrogen = 13.6 eV
8. The minimum potential applied to an atom of an element at its ground state to convert it into a positive ion, is called the ionization potential of that element Ionisation potential of hydrogen = 13.6 V
9. X-ray is not a stream of fast-moving charged or uncharged particles, rather it is an electromagnetic wave like light
10. The energy of X-ray is more than that oflight Spectrum of X-rays is formed due to die superposition of two spectra
- Continuous spectrum and
- Characteristic tic spectrum.
A continuous X-ray spectrum has a definite cut-off wavelength below which no radiation exists.
Applications of Atomic Concepts in Real Life
11. Moseley’s law: The square root of the frequency of a peak of the characteristic X-ray spectrum of an element is directly proportional to the atomic number of that element
12. Energy difference of an electron in two stable orbits,
E1~ E2 = hf
13. According to Bohr’s quantum condition, if rn = radius of n -th orbit and vn = velocity of the electron in n -th orbit, then

Here, the principal quantum number, n = 1, 2, 3,…………
14. First Bohr radius of ground state (rt = 1)
⇒ \(a_0=\frac{\epsilon_0 h^2}{\pi m e^2}\)
= 0.53 Å
= 0.053 nm
And \(E_1=\frac{m e^4}{8 \epsilon_0^2 c h^3}\) . ch = -Rch
Where , R = \(\frac{m e^4}{8 \epsilon_0^2 c h^3}\)
Rydberg constant = 1.09737 × 107 m-1
15. Ground state energy of hydrogen atom =-13.6 eV
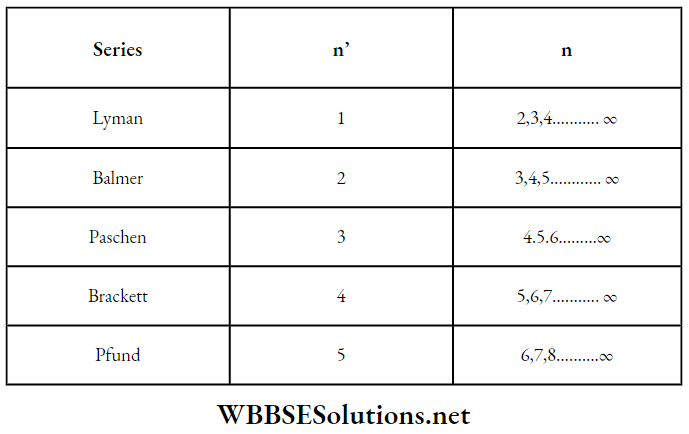
16. For the hydrogen spectrum, if A is the wavelength of the spectral line then
⇒ \(\frac{1}{\lambda}=R\left[\frac{1}{\left(n^{\prime}\right)^2}-\frac{1}{n^2}\right]\)
17. According to Bohr’s postulate, when the electron in a hydrogen atom transits from a higher energy state lower energy state Enÿ, a photon is emitted from the hydrogen atom. If the frequency of this photon is (wavelength, λ = then
⇒ \(E_{n_i}-E_{n_f}=h f=\frac{h c}{\lambda}\)
The minimum wavelength of X-ray
⇒ \(\lambda_{\min }=\frac{h c}{e V} .\)
In SI, e = 1.6C, c = 3× 108m. s-1 , and
h = 6.63 × 10-34 J.s-1
⇒ \(\frac{1.243 \times 10^{-6}}{V} \mathrm{~m}\)
= 12340/v Å
18. If h, f, and c are Planck’s constant, the frequency of the emitted X-ray photon, and the speed of the X-ray respectively, then the kinetic energy of the incident electron
⇒ \(K_0=h f=\frac{h c}{\lambda_{\min }}\)
Or,\(\lambda_{\min }=\frac{h c}{K_0}\)
λmin = cut-off wavelength
19. If the electron in a hydrogen atom is excited to the n -th state, then the number of possible spectral lines it can emit in transition to the ground state is
⇒ \({ }^n C_2=\frac{1}{2} n(n-1)\)
20. Speed of electron in the n -th orbit of the hydrogen atom
⇒ \(v_n=\frac{c}{137 n}\)
WBCHSE physics class 12 atomic notes
WBCHSE Class 12 Physics Atom Very Short Question And Answers
Question 1. In the Bohr model of the hydrogen atom, what Is the ratio of the kinetic energy to the total energy of the electron in any quantum state
Answer:
According to the Bohr model, if the kinetic energy of the electron in any quantum state is E, total energy becomes -II. So, the ratio of these two energies is
Question 2. How are X-rays produced?
Answer:
When a stream ofelectrons, having a kinetic energy of a few keV or more, hits a solid target (like copper, tungsten, molybdenum, etc.), X-rays are emitted from that target
Question 3. In which part of the electromagnetic do the spectral lines of a hydrogen atom given by the Balmer series occur?
Answer: Visible range.
Question 4. What is Bohr’s quantum condition for the angular momentum of an electron in a hydrogen atom?
Answer:
Angular momentum = nh/ 2: n = 1,2,3,……………..
Question 5. Name the different types of X-ray spectrum
Answer: Continuous spectrum and characteristics spectrum
Question 6. The nucleus contains the entire ____________ charge and nearly the entire
_______ of an atom
Answer: Positive And Mass
Question 7. In Rutherford’s experiment, which particle is responsible for the low-angle scattering of a -particles?
Answer: Electron
Question 8. The total energy of the electron in the first Bohr orbit of a hydrogen atom is -13.6 eV. What are the kinetic and potential energies of the electron?
Answer: 13.6 eV, -27.2 eV
Question 9. The wavelength of a spectral line found in the atomic spectrum of hydrogen is 4861 Å. Between which two quantum states does the transition of electrons occur to generate this line? Rydberg constant = 1.097 × 108. m-1
Answer: From the fourth to the second
Question 10. What is the order of magnitude of the ratio between the volume of? an atom and that of its nucleus?
Answer: 1012: 1
Question 11. The energy of an electron in the first excited state of a hydrogen atom is -3.4 eV what is the kinetic energy of this electron
Answer: + 3.4 eV
Question 12. What is the ratio of the areas of the first and the second orbits of a hydrogen atom?
Answer: 1:16
Question 13. What is the approximate diameter of a hydrogen atom
Answer: 1.06A
Question 14. If the radius of the first Bohr orbit is r, what would be the radius of the second?
Answer: 4r
Question 15. The radius of the first electron orbit of a hydrogen atom is 5.3 × 10-11m. What is the radius of its second orbit?
Answer: 21.2 m
Question 16. An electron beam hits a target to produce a continuous X-ray spectrum. If E is the kinetic energy of each electron in the beam, what would be the lowest wavelength of the emitted X-rays?
Answer: hc/E
WBCHSE Class 12 Physics Atom Assertion Type
Direction: These questions have statement 1 and statement 2 Of the four choices given below, choose the one that best describes the two statements.
- Statement 1 is true, statement 2 is true, and statement II is a correct explanation for statement 1.
- Statement 1 is true, and statement 2 is true; statement 2 is not a correct explanation for statement 1.
- Statement 1 is true, and statement 2 is false.
- Statement 1 is false, and statement 2 is true.
Question 1.
Statement 1: The total positive charge and almost all the mass of an atom are confined in the nucleus.
Statement 2: In Rutherford’s a -particle scattering experiment, the majority of the a -particles penetrate the target without any deflection.
Answer: 2. Statement 1 is true, and statement 2 is true; statement 2 is not a correct explanation for statement 1.
Question 2.
Statement 1: The circular orbit of the electron as stated in Rutherford’s atomic model can never be a stable orbit
Statement 2: Any accelerated charged particle radiates energy
Answer: 1. Statement 1 is true, statement 2 is true, and statement II is a correct explanation for statement 1.
Question 3.
Statement 1: The distance of the electron from the nucleus is minimal when a hydrogen atom is in the ground state.
Statement 2: According to Bohr’s theory the radius of circular motion of an electron in n -th energy state, rn oc n.
Answer: 3. Statement 1 is true, and statement 2 is false.
Question 4.
Statement 1: The kinetic energy of an electron in the first excited state of a hydrogen atom is 6.8 eV.
Statement 2: The total energy of the first excited state of a hydrogen atom is -3.4 eV.
Answer: 4. Statement 1 is false, and statement 2 is true.
Question 5.
Statement 1: All lines in the Balmer series of the hydrogen spectrum are in the visible region.
Statement 2: Balmer series is formed due to the transition of electrons from 2, 3, 4- – permitted energy levels to the ground level.
Answer: 3. Statement 1 is true, and statement 2 is false.
Question 6.
Statement 1: The ionization potential of a hydrogen atom is 13.6 eV.
Statement 2: The Ground state energy of the hydrogen atom is 13.6 eV.
Answer: 3. Statement 1 is true, and statement 2 is false.
Question 7.
Statement 1: Magnetic moment of election In the n-th orbit of the hydrogen atom
Statement 2: The magnetic moment of a particle of charge rotating in an orbit of radius r with velocity v is given by \(\mu=\frac{1}{2} e v r\)
Answer: 1. Statement 1 is true, statement 2 is true, and statement II is a correct explanation for statement 1.
WBCHSE Class 12 Physics Atom Match The Columns
Question 1. Match the columns for the hydrogen atom.
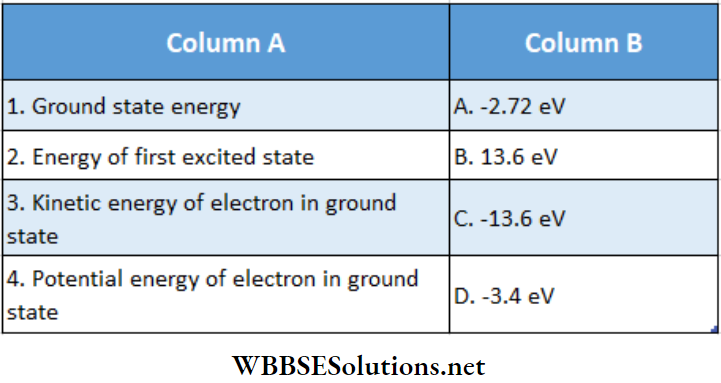
Answer: 1- C, 2-D, 3- C, 4- A
Question 2. Match the columns for an electron rotating in the n-th orbit of an atom
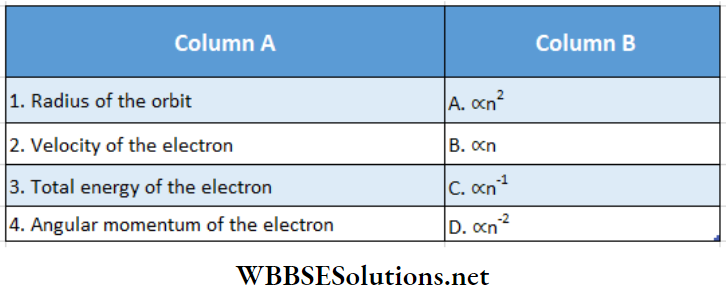
Answer: 1- A, 2 – C, 3 – D, 4 – B
Question 3.
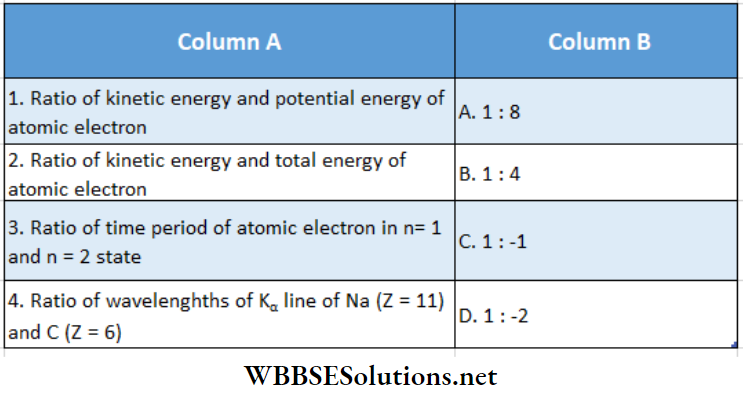
Answer: 1- D, 2 – C, 3 – A, 4- B
Question 4. Match Column A (fundamental experiment) with Column B (its 1 conclusion) and select the correct option from the choices given below the list:
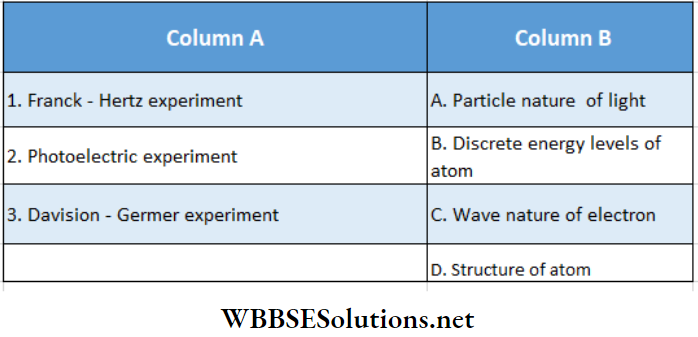
- 1- A, 2- D, 3 – C
- 1- B, 2- D, 3 – C
- 1- B, 2- A, 3 – C
- 1- D, 2- C, 3 – B
Answer: 3. 1- B, 2- A, 3 – C
