Heat Engine Refrigerator
Question 1. The efficiency of a Carnot engine operating between the temperatures of 100°C and -23°C will be
- \(\frac{100-23}{273}\)
- \(\frac{100+23}{373}\)
- \(\frac{100+23}{100}\)
- \(\frac{100-23}{100}\)
Answer: 2. \(\frac{100+23}{373}\)
Temperature of source = T1 = (273 + 100) K
= 373 K,
Temperature of sin k = T2 = (273 – 23 K)
= 250 K.
The efficiency of the Carnot engine is
⇒ \(\eta=1-\frac{T_2}{T_1}\)
= \(\frac{T_1-T_2}{T_1}\)
= \(\frac{123}{373}\)
= \(\frac{100+23}{373}\)

Question 2. An engine takes heat from a reservoir and converts \(\frac{1}{6}\) of it into work. If the temperature of the sink is reduced by 62 °C, the efficiency of the engine becomes double. The temperatures of the source and sink must be
- 90°C, 37°C
- 99°C,37°C
- 372°C,37°C
- 206°C, 37°C
Answer: 2. 99°C,37°C
Efficiency = \(\frac{\text { output }}{\text { input }}=\frac{\text { work done }(W)}{\text { heat absorbed }(Q)}\)
⇒ \(1-\frac{T_2}{T_1}=\frac{1}{6}\)…..(1) [∵ W = \(\frac{Q}{6}\)]
When the temperature T2 of the sink is reduced by 62°C, the new temperature T2 = T2– 62, and
⇒ \(\eta^{\prime}=1-\frac{T_2^{\prime}}{T_1}\)
= \(2 \eta=2\left(\frac{1}{6}\right)\)
= \(\frac{1}{3}\)
⇒ \(1-\left(\frac{T_2-62}{T_1}\right)\)
= \(\frac{1}{3}\)…(2)
From(1), \(\frac{T_2}{T_1}=\frac{5}{6}\) and from(2), \(\frac{T_2}{T_1}=\frac{2}{3}+\frac{62}{T_1}\)
difference between heat engine heat pump and refrigerator
Equating \(\frac{T_2}{T_1}\) as mentioned above, \(\frac{5}{6}=\frac{2}{3}+\frac{62}{T_1}\)
T1 = 372K
= 99°C,
and \(T_2=\frac{5}{6} T_1\)
= 310K
= 37°C.
∴ The required temperatures are 99 °C and 37°C.
Question 3. The temperatures of the source and sink of a heat engine are 127°C and 27°C respectively. A technician claims its efficiency to be 26%.
- It is impossible.
- It is possible with high probability.
- It is possible with low probability.
- The data is insufficient.
Answer: 1. It is impossible.
Given that T1 = (273 + 127) K
= 400 K
T2 = (273 + 27) K
= 300 K.
The efficiency of an ideal engine (Carnot engine),
⇒ \(\eta=1-\frac{T_2}{T_1}\)
= \(1-\frac{300}{400}\)
= \(\frac{1}{4}\)
= 25%
No engine can have an efficiency more than that of a Carnot engine.
Hence, the claim of 26% efficiency is impossible.
Question 4. The efficiency of a Carnot engine is 50% and the temperature of the sink is 500 K. If the temperature of the source is kept constant and the efficiency of the engine is to be raised to 60% then the required temperature of the sink will be
- 600 K
- 500 K
- 400 K
- 100 K
Answer: 3. 400 K
Efficiency \(\eta=1-\frac{T_2}{T_1}\)
Given that T2 = 500 K and \(\eta=50 \%=\frac{1}{2} \Rightarrow \frac{1}{2}=1-\frac{500}{T_1}\)
hence T1 = 1000 K.
For 60% efficiency, let the temperature of the sink be T2
difference between heat engine heat pump and refrigerator
⇒ \(\frac{60}{100}=1-\frac{T_2^{\prime}}{T_1}=1-\frac{T_2^{\prime}}{1000}\)
∴ T2 = 400 K
Question 5. An ideal gas heat engine operates as a Carnot cycle between 227°C and 127°C. It absorbs 6 kcal of heat at a higher temperature. The amount of heat (in kcal) converted into work is equal to
- 1.6
- 1.2
- 4.8
- 3.5
Answer: 2. 1.2
Temperature of source = T1 = (227 + 273) K
= 500 K.
Temperature ofsink = T2 = (127 + 273) K
= 400 K.
∴ efficiency \(\eta=1-\frac{T_2}{T_1}\)
= \(1-\frac{400}{500}\)
= \(\frac{1}{5}\)
But \(\eta=\frac{\text { output }(W)}{\text { input }(Q)}\)
= \(\frac{W}{6 \mathrm{kcal}}\)
= \(\frac{1}{5}\)
∴ work W = \(\frac{6kcal}{5}\)
= 1.2 kcal
Question 6. An engine has an efficiency of \(\frac{1}{6}\) When the temperature of the sink is reduced by 62°C, its efficiency is doubled. The temperature of the source is
- 120°C
- 35°C
- 99°C
- 65°C
Answer: 3. 99°C
Let T1 = temperature of the source.
Given that efficiency n = \(\frac{1}{6}\)
⇒ \(\eta=1-\frac{T_2}{T_1} \Rightarrow \frac{1}{6}=1-\frac{T_2}{T_1}\)….(1)
When T2 is reduced to T2– 62, efficiency gets doubled. So,
difference between heat engine heat pump and refrigerator
⇒ \(\frac{2}{6}=1-\frac{T_2-62}{T_1}\)…..(2)
Solving (1) and (2), T1 = 372 K = 99°C.
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
Question 7. A Carnot engine whose sink is at 300 K has an efficiency of 40%. By how much should the temperature of the source be increased so as to increase its efficiency by 50% of the original efficiency?
- 250 K
- 370 K
- 270 K
- 390 K
Answer: 1. 250 K
Efficiency of Carnot engine is \(\eta=1-\frac{T_2}{T_1}\)
Given that \(\eta=40 \%=\frac{4}{10}\), the temperature of sink = T2 = 300 K.
Temperature of source = T1.
∴ \(\frac{4}{10}=1-\frac{300}{T_1}\)
⇒ T1 = 500 K
Let the temperature T1 of the source be increased by ΔT1 so that the increased efficiency becomes
⇒ \(\eta^{\prime}=40 \%+50 \% \text { of } \eta=\frac{40}{100}+\frac{50}{100} \times \frac{40}{100}=\frac{60}{100}\)
Hence,
⇒ \(\eta^{\prime}=\frac{60}{100}\)
= \(1-\frac{T_2}{T_1+\Delta T_1}\)
⇒ \(\frac{60}{100}=1-\frac{300}{500+\Delta T_1}\)
⇒ \(\Delta T_1=250 \mathrm{~K}\)
Question 8. Two Carnot engines A and B are operated in series. Engine A receives heat from the source at temperature T1, and rejects heat to the sink at temperature T. The second engine receives heat at temperature T and rejects a part of it to its sink at temperature T2. For what value of T are the efficiencies of the two engines equal?
- \(\frac{T_1+T_2}{2}\)
- \(\frac{T_1-T_2}{2}\)
- \(\sqrt{T_1 T_2}\)
- \(\frac{\sqrt{T_1 T_2}}{2}\)
Answer: 3. \(\sqrt{T_1 T_2}\)
For Carnot engine A, efficiency is \(\eta_{\mathrm{A}}=1-\frac{T}{T_1}\) and for Carnot engine B,
it is \(\eta_{\mathrm{B}}=1-\frac{T_2}{T}\)
Given that \(\eta_{\mathrm{A}}=\eta_{\mathrm{B}}\)
⇒ \(1-\frac{T}{T_1}=1-\frac{T_2}{T}\)
⇒ \(\frac{T}{T_1}=\frac{T_2}{T}\)
Hence, \(T=\sqrt{T_1 T_2}\)
Question 9. The coefficient of performance of a refrigerator is 5. If the temperature inside the freezer is -20°C, the temperature of the surroundings to which it rejects heat is
- 42 °C
- 31 °C
- 21 °C
- 15°C
Answer: 2. 31 °C
The coefficient of performance (k) of a refrigerator is expressed as
⇒ \(k=\frac{T_2}{T_1-T_2}\)
where T2 = temperature of the refrigerated space,
and T1 = temperature of surroundings.
Given that T2 = -20°C = 253 K.
difference between heat engine heat pump and refrigerator
For k = 5,
⇒ \(5=\frac{253}{T_1-253}\)
⇒ \(T_1=303.6 \mathrm{~K} \approx 31^{\circ} \mathrm{C}\)
Question 10. A Carnot engine having an efficiency of \(\frac{1}{10}\) as a heat engine is used as a refrigerator. If the work done on the system is 10 J, the amount of energy absorbed from the reservoir at a lower temperature is
- 110 J
- 90 J
- 95 J
- 10 J
Answer: 2. 90 J
When used as a Carnot engine, efficiency \(\eta=1-\frac{T_2}{T_1}\)
Given that n = \(\frac{1}{10}\)
Hence, \(\frac{T_2}{T_1}=\frac{9}{10}\)……(1)
When used as a refrigerator, its coefficient of performance is
⇒ heat extracted from the reservoir (Q2) / work done on the system
Question 11. A refrigerator works between 4°C and 30°C, It is required to remove 600 calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is
- 24.65 W
- 236.5 W
- 230 W
- 2.365 W
Answer: 2. 236.5 W
For the refrigerator, T2 = (4 + 273) K = 277 K.
T1 = (30 + 273) K
= 303 K.
The coefficient of performance is
⇒ \(\frac{Q_2 \text { (heat absorbed) }}{W(\text { Work done })}=\frac{Q_2}{Q_1-Q_2}\)
= \(\frac{1}{\frac{Q_1}{Q_2}-1}\)
⇒ \(\frac{Q_2}{W}=\frac{1}{\frac{T_1}{T_2}-1}\)
= \(\frac{T_2}{T_1-T_2}\)
Given that Q2 = 600 cal s-1 = 600 x 4.2 J s-1.
∴ required power is,
⇒ \(W=Q_2 \frac{\left(T_1-T_2\right)}{T_2}\)
= \(\frac{\left(600 \times 4.2 \mathrm{~J} \mathrm{~s}^{-1}\right)(26 \mathrm{~K})}{(277 \mathrm{~K})}\)
= 236.5 W.
Question 12. The temperature inside a refrigerator is t2°C and the room temperature is t1°C. The amount of heat delivered to the room for each joule of electrical energy consumed ideally will be
- \(\frac{t_1+t_2}{t_1+273} \mathrm{~J}\)
- \(\frac{t_1+273}{t_1-t_2} \mathrm{~J}\)
- \(\frac{t_1}{t_1-t_2} \mathrm{~J}\)
- \(\frac{t_1+273}{t_1-273} \mathrm{~J}\)
Answer: 2. \(\frac{t_1+273}{t_1-t_2} \mathrm{~J}\)
For a refrigerator, the ratio
⇒ \(\frac{Q_1 \text { (heat delivered to the room) }}{Q_2 \text { (heat absorbed) }}=\frac{T_1}{T_2}\)
But Q1– Q2 = W, so Q2 = Q1– W, where W = electrical energy is consumed.
∴ \(\frac{Q_1}{Q_1-W}=\frac{T_1}{T_2} \Rightarrow \frac{1}{1-\frac{W}{Q_1}}=\frac{T_1}{T_2} \Rightarrow Q_1=\frac{W T_1}{T_1-T_2}\)
Given that T1 = 273 + t1, T2 = 273 + t2, W = 1 J.
∴ \(Q_1=\frac{(1 \mathrm{~J})\left(273+t_1\right)}{\left(t_1-t_2\right)}=\frac{273+t_1}{t_1-t_2} \mathrm{~J}\)
Question 13. Determine the efficiency of a Carnot engine if, during its adiabatic expansion, the volume is increased to 3 times the initial volume and y = 1.5.
- \(1-\frac{1}{\sqrt{2}}\)
- \(1-\frac{1}{\sqrt{3}}\)
- \(1-\frac{1}{\sqrt{2}}\)
- \(1-\frac{1}{\sqrt{3}}\)
Answer: 2. \(1-\frac{1}{\sqrt{3}}\)
During the adiabatic expansion,
⇒ \(T_1 V_1^{\gamma-1}=T_2 V_2^{\gamma-1}\)
⇒ \(\frac{T_2}{T_1}=\left(\frac{V_1}{V_2}\right)^{\gamma-1}\)
difference between heat engine heat pump and refrigerator
⇒ \(\frac{T_2}{T_1}=\left(\frac{1}{3}\right)^{1.5-1}\)
= \(\frac{1}{\sqrt{3}}\)
Hence, efficiency is \(\eta=1-\frac{T_2}{T_1}=1-\frac{1}{\sqrt{3}}\)
Question 14. In a refrigerator, the heat absorbed from the source is 800 J and the heat supplied to the sink is 500 J. The coefficient of performance is
- \(\frac{5}{8}\)
- \(\frac{8}{5}\)
- \(\frac{5}{3}\)
- \(\frac{3}{5}\)
Answer: 3. \(\frac{5}{3}\)
The coefficient of performance of a refrigerator is defined as
⇒ \(k=\frac{\text { heat absorbed }\left(Q_2\right)}{\text { work done on the coolant }(W)}\)
⇒ \(\frac{Q_2}{W}=\frac{Q_2}{Q_1-Q_2}\)
Given that Q1 = 800J,
Q2 = 500J,
Hence k = \(\frac{5}{3}\)
Question 15. A Carnot engine works between 27°C and127°C. The heat supplied by the source is 500 J. The heat delivered to the sink is
- 100 J
- 667 J
- 375 J
- 500 J
Answer: 3. 375 J
In a Carnot engine,
⇒ \(\frac{Q_2}{Q_1}=\frac{T_2}{T_1}\)
= \(\frac{27+273}{127+273}\)
= \(\frac{3}{4}\)
∴ heat delivered to the sink,
Q2 = \(\frac{3}{4}\)
Q1 = \(\frac{3}{4}\) x 500J
= 375J.
Question 16. A Carnot engine has an efficiency of \(\frac{1}{6}\). When the temperature of the sink is reduced by 62°C, its efficiency is doubled. The temperatures of the source and the sink are respectively
- 99°C, 37°C
- 124°C, 62°C
- 37°C, 99°C
- 62°C, 124°C
Answer: 1. 99°C,37°C
The efficiency of the Carnot engine is
⇒ \(\eta=\frac{1}{6}=1-\frac{T_2}{T_1}\)
⇒ \(\frac{T_2}{T_1}=\frac{5}{6}\)
When T2 is reduced by 62°C, the efficiency becomes
⇒ \(\eta^{\prime}=2 \eta\)
= \(\frac{1}{3}\)
difference between heat engine heat pump and refrigerator
= \(1-\frac{T_2-62}{T_1}\)
⇒ \(\frac{T_2-62}{T_1}=\frac{2}{3}\)
Dividing (2) by (1),
⇒ \(\frac{T_2-62}{T_2}=\frac{2}{3} \times \frac{6}{5}\)
= \(\frac{4}{5}\)
=> 5T2 – 310 = 4T2
=> T2 = 310 K
= (310- 273) °C
= 37°C.
⇒ \(T_1=\frac{6}{5} T_2\)
= \(\frac{6}{5}(310 \mathrm{~K})\)
= 372 K
= (372- 273) °C
= 99°C
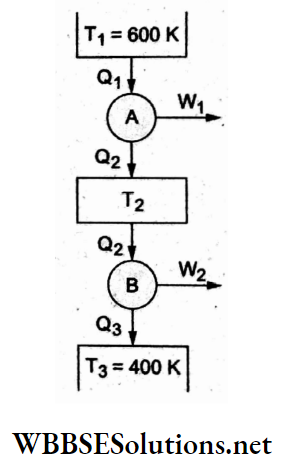
Question 17. TwoCarnotenginesAand B areoperatedinseries. EngineAreceives heat at T1 (= 600 K) and rejects it to a reservoir at temperature T2. Engine B receives heat rejected by Engine A and in turn, rejects heat to a heat reservoir at T3 (= 400 K). If the work outputs by the two engines are equal, the temperature T2 is equal to
- 600 K
- 500 K
- 400 K
- 300 K
Answer: 2. 500 K
The diagram shows the working of Carnot engines A and B in series.
For A: Q1 = Q2 + W1,
So, W1 = Q1– Q2.
For B: Q2 = Q3 + W2,
So W2 = Q2-Q3.
Given that work, the output is the same for both A and B, hence W1 = W2
Q1 – Q2 = Q2-Q3
=> Q1 + Q3 = 2Q2
⇒ \(\frac{Q_1}{Q_2}+\frac{Q_3}{Q_2}=2\)
⇒ \(\frac{T_1}{T_2}+\frac{T_3}{T_2}=2\)
Substituting, \(\frac{600 \mathrm{~K}}{T_2}+\frac{400 \mathrm{~K}}{T_2}=2\)
⇒ T2 = 500 K.