WBCHSE Class 12 Physics Atomic Nucleus
Atomic Nucleus Introduction
Physicist Ernest Rutherford was able to reach two important conclusions from his famous alpha particle scattering experiment, regarding the distribution of charge carriers and mass in an atom:
- The entire positive charge and most of the mass of an atom is concentrated in a very small space of the atom called the nucleus. The volume of the nucleus is only about 1 in 10-12 part of the atom (atomic diameter is about 10-8cm and the diameter of the nucleus is estimated as 10-12 cm).
- The remaining part of the atom contains negatively charged electrons. These electrons are distributed in a regular pattern outside the nucleus, and a large part of the atom is in space. The total mass of the electrons is negligible in comparison to the mass of the atom.
WBCHSE class 12 physics atomic nucleus
Atomic Nucleus Mass-Energy Equivalence
Matter can be viewed as concentrated energy. Max Planck and others had realized the importance of the concept, early in the twentieth century but it was Albert Einstein who first proposed an equivalence of mass and energy. He suggested c² (c = velocity of light) as the conversion factor from mass to energy. The principle of mass-energy equivalence can be stated thus: If the mass m of a body is completely converted to energy, the amount of the energy is
Read and Learn More Class 12 Physics Notes
E = mc² [c = speed of light in vacuum = constant] The relation suggests that the energy E is equivalent to mass m or that the mass m is equivalent to energy E.
Example:
1. In the CGS system
c = \(2.998 \times 10^{10} \mathrm{~cm} \cdot \mathrm{s}^{-1} \simeq 3 \times 10^{10} \mathrm{~cm} \cdot \mathrm{s}^{-1}\)
∴ Equivalent energy contained in 1 g,
E = 1 × (3 ×1010)2 = 9 ×1020 erg = 9 ×1013 J
Again in SI, c = 3×108 m. s-1
∴ Equivalent energy contained in 1 kg,
E =1 kg × (3 × 108 m. s-1)2 = 9 × 1016 J
2. Mass of electron, me = 9.109 × 10-28 g
∴ Equivalent energy of mass of an electron
= 9.109 × 10-28 × (3 × 1010 )2 erg
= \(\frac{9.109 \times 10^{-28} \times 9 \times 10^{20}}{1.602 \times 10^{-12}}\) eV
= 0.511 × 106 eV
= 0.511 MeV
WBBSE Class 12 Atomic Nucleus Notes
Rest mass:
Einstein’s theory of relativity also suggests that the mass of a body is not a constant but depends on the velocity of the body. Especially when speed is comparable to the speed of light in a vacuum, the mass of a body increases considerably. Hence, when mentioning the mass as an innate property of matter, the body should be considered to be at rest. This is called rest mass. For example rest mass of an electron = 9.109 × 10-28 g. Lorentz derived the relation between rest mass ( mQ) and the mass at velocity v close to c as
m = \(\frac{m_0}{\sqrt{1-\frac{v^2}{c^2}}}\)
Unit of mass and energy: As mass and energy are equivalent to each other, their units too are equivalent. Hence, the energy unit is also used to represent mass and vice versa. For example, 1 g energy denotes 9 × 1020 erg of energy or a mass of 9 × 1016 J indicates 1 kg mass. Using this equivalence of mass and energy we can say, the rest mass of an electron, me = 0.511 MeV
Atomic nucleus class 12 notes Law of conservation of mass energy:
When there occurs an interconversion between mass and energy, the law of conservation of mass and the law of conservation of energy cannot be applied separately. Instead, these laws combine to form the law of conservation of mass energy.
In nature, the sum of mass and energy of a system is a con¬ stant. While there may be various changes in the form, energy cannot be destroyed or created.
Atomic energy:
Conversion of mass into energy can take place only in nuclear phenomena within atoms. Energy from this conversion is the source of atomic energy. Atomic energy is used in making nuclear weapons like atomic bombs, and hydrogen bombs, in generating electricity in nuclear power stations, etc

Short Notes on Nuclear Structure
Atomic Nucleus Mass-Energy Equivalence Numerical Examples
Example 1. In any nuclear reaction \(\frac{1}{1000}\) part ofthe mass of a particular substance is converted into energy. If 1 g of that substance takes part in a nuclear reaction then determine the energy evolved in kilowatt-hour
Solution:
Energy converted = \(\frac{1}{1000}\) × 1
= 0.001 g
∴ Energy involved
E = mc² = 0.001 × (3 × 1010)2
= 9 × 1017erg
= 9 × 1010 J
= 9 × 1010 kW
= \(\frac{9 \times 10^{10}}{1000 \times 3600}\) kW. h.
h = 25000 kW.h
Example 2. If a metal is completely converted into energy, calculate how much of this metal would be required as fuel for a power plant in a year. The power plant, let us suppose, generates 200 MW on average.
Solution:
200 MW = 200 × 1016 W = 2 × 108 W,
1 year = 365 × 24 × 60 × 60 J
In 1 year the energy generated,
E= 2 × 108 × 365 × 24 × 60 × 60 J
Equivalent mass m = \(\frac{E}{c^2}=\frac{2 \times 10^8 \times 365 \times 24 \times 60 \times 60}{\left(3 \times 10^8\right)^2}\)
= 0.070 kg
= 70 kg
Atomic nucleus class 12 notes
Atomic Nucleus Nuclear Structure
Constituents of the Nudeus
Proton:
It is an elementary particle that carries a charge equal to the charge of an electron. Unlike an electron, a proton is positively charged. Its rest mass is about 1836 times that of an electron. Thus rest mass of a proton,
mp = 1836 × (9.109 × 10-28 ) =1.672 × × 10-24 g
And equivalent energy of mp = 938.8 MeV (approx.)
Neutron:
An uncharged or electrically neutral elementary particle of mass slightly greater than that of a proton and equal to 1839 times the rest mass of an electron.
∴ mn = 1839 × (9.109 × 10-28 ) = 1.675× 10-24 g
And equivalent energy of mn = 939.6 MeV (approx.)
Protons and neutrons occupy the space in the nucleus and hence are commonly called nucleons.
Atoms have two parts, a nucleus at the center and electrons that revolve in orbits surrounding the nucleus
- Electron,
- Proton and
- Neutron
Are the constituents of the atom of any element though relative abundance differs from element to element shows the constituents of neutral atoms of few elements
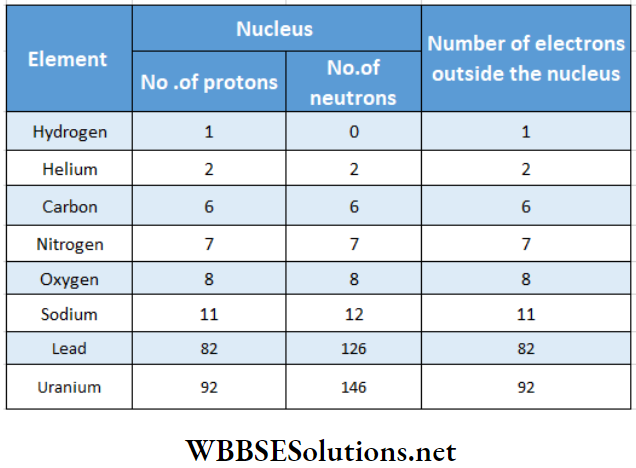
Common Questions on Atomic Nucleus
The following points must be remembered:
- There is no neutron in the hydrogen nucleus.
- Only one proton forms its nudes. As electrons and protons have equal and opposite charges so for a neutral atom, several protons in nudes is equal to the number of electrons outside the nucleus.
- Only neutrons cannot form a nucleus.
Nuclear force
A strong force of attraction keeps the neutrons and the protons bound together within the nucleus. This interaction is called strong interaction and the force thus produced and acting between the neutrons is called nuclear force. Neither the law of gravitation nor Coulomb’s law can explain the intensity or properties of this nuclear force.
Characteristics of nuclear force:
- The nuclear force is stronger than gravitational or coulombia force.
- It Is only an attractive force
- Nuclear force Is Independent of charge.
- So the magnitude of the nuclear force of proton and neutron Is the same l.e., neutron-proton, proton-proton, and neutron-neutron pairs experience die same force.
- It Is a very short-range force limited to a distance of about 10 -12 cm. So only closer nucleons are bound together by this force, not the distant ones.
- Protons, neutrons, and some other fundamental particles take part in nuclear interaction.
Atomic Nucleus Atomic Mass And Numar Mass
Atomic mass unit Definition:
\(\frac{1}{12}\) th of the mass of a C¹² atom is called 1 atomic mass unit(u)
Unified atomic mass unit
The mass of an atom is so small that It is not expressed In kilogram or grams. Instead, a special unit called imified atomic mass unit has been designed for this and is expressed as u. This unit is often called Dalton or Da. Earlier the atomic mass was represented by amu or atomic mass unit, it was expressed in terms of the mass of hydrogen or oxygen atom. Presently, carbon Is taken as the standard as C¹² atoms can be obtained free from its Isotopes, in nature.
1 mol of carbon-12 has a mass of 12 g and contains 6.023× 10 23 (Avogadro’s number) of atoms
Hence, mass of 1 atom of C¹² = \(\frac{12}{6.023 \times 10^{23}} \mathrm{~g}\)
∴ As per definition.
1 u = \(\frac{1}{12} \times \frac{12}{6.023 \times 10^{23}}\)g
= 1.66 × 10 -24 g
= 1.66 × 10 -27 kg
Equivalent energy of the unified atomic mass:
According to E = mc². equivalent energy of
1 u of mass = \(1.66 \times 10^{-24} \times\left(2.998 \times 10^{10}\right)^2\) erg
= \(=\frac{1.66 \times 10^{-24} \times\left(2.998 \times 10^{10}\right)^2}{1.6022 \times 10^{-12}} \mathrm{eV}\)
= \(931.2 \times 10^6 \mathrm{eV}=931.2 \mathrm{MeV}\)
It is important to remember the value 931.2 MeV for several mathematical calculations. In nuclear physics.
Atomic moss: Relative atomic men
The atomic mass of an element Is the mass of an Isotope of that element (discussed later).
Depending upon the abundance of Isotopes of an element present on the earth’s surface or in the atmosphere, an average mass of the button of an element Is calculated. This average atomic mass is called the relative atomic mass of that element. It is sometimes referred to as atomic weight though it expresses the mass and not the weight.
It has been possible to measure precisely the atomic mass or relative atomic mass of elements using mass spectroscopes.
Represents the values In u of a few elements. Data sources are
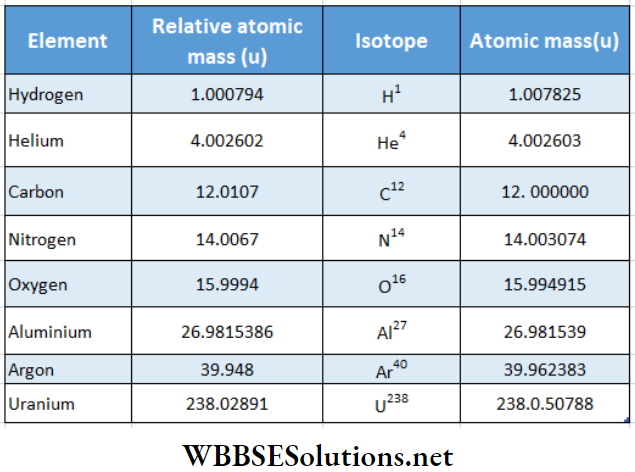
Nuclear mass
Nuclear mass = atomic mass- a mass of electrons in the atom ofthe element Mass of one electron I is taken as 0.00055 u . Except for very precise measurements, mass of a the nucleus and the atomic mass are taken to be the same
Mass number
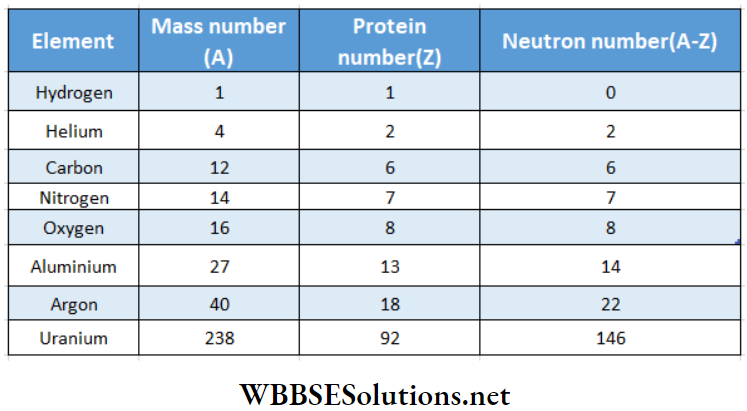
Mass number Definition:
The whole number nearest to the atomic mass of an element expressed in atomic mass unit, is the mass number of the element Mass number is equal to the number of protons and neutrons in the nucleus of that atom.
Example: The mass number of hydrogen (H¹) is 1 and that of uranium (U238) is 238.
A mass number is simply a number and has no unit It is usually expressed by the letter A.
The number of protons present in the nucleus of an element is called the atomic number ofthe element and is represented by Z. The difference (A-Z) represents the number of neutrons in the nucleus. The mass number, atomic number, and neutron number of some elements are listed in
Important Definitions in Nuclear Physics
Notation for mass number
To denote or express the mass number (A) of any element the symbol of the element is used then the mass number is written as a superscript either to the left or to the right side of the symbol
Example: H1, C12, N14,- or, 1H, 4He, 12C, 14N
Isotopes
Atoms having the same atomic number but different mass numbers are called isotopes.
Isotopes have identical chemical properties because they are the same element, but differ in physical properties because they differ in mass. All the isotopes of an element should in principle occupy the same place as the parent element in the periodic table. The name has originated from this property. Isotopes are formed due to the difference in number of neutrons in the nucleus of an element. lists some important isotopes of a few elements.
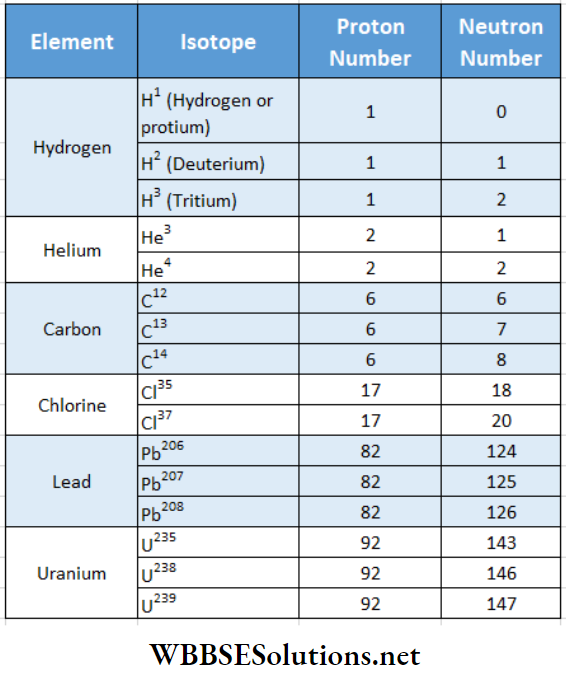
Carbon has three isotopes C12, C13, and C14. Therefore, when defining relative atomic mass one should write carbon- 12 atom and not carbon atom.
Heavy water
The isotope of hydrogen having mass number 2 is called deuterium. Its symbol is H2. Often it is also expressed as D . Deuterium has one proton and one neutron in its nucleus.
In water (H2O) hydrogen chemically combines with oxygen. Deuterium too, can form a similar molecule, D20 in combination with oxygen and is called deuterium oxide or heavy. Natural water and heavy water have the same chemical properties but they differ in physical properties as shown
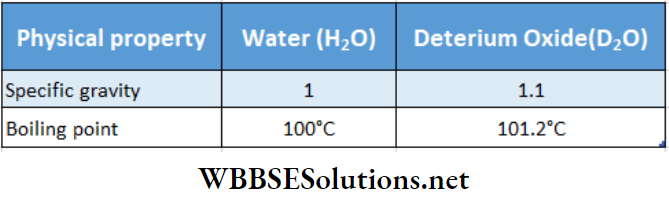
In any sample of common water, the abundance of heavy water is 1 in 5000 parts. Heavy water is used in nuclear reactors as ‘moderators’ that slow down the fast-moving neutrons formed.
Isobars
The atoms having the same mass number are called isobars. However, isobars differ in their neutron and pro¬ ton numbers.
Example:
C14 and N14 are isobars but the neutron number and proton number in C14 are 8 and 6 while those in N14 are 7 and 7 respectively.
Isotones
Atoms having the same number of neutrons in their nucleus are isotones. They differ in mass number and proton number.
Example: C14 and O16 are mutual isotones. In C14, neutron number = 8, proton number = 6, mass number = 14. In O16 the corresponding numbers are 8, 8, and 16 respectively.
Atomic number
Atomic nucleus class 12 notes Definition:
Taking hydrogen as the first element, the serial number of an element, arranged according to gradual changes in chemical properties in the periodic table is called the atomic number of that element
1. Importance of atomic number in an atom:
The number of protons in the nucleus of an atom of an element determines the characteristics of that element. For instance, if there are 6 protons it is a carbon atom, if there are 8 protons it is an oxygen atom. Neutrons inside the nucleus and electrons outside the nucleus cannot be used to identify the element
If the number of neutrons in an atom changes an isotope is formed and when there is a change in the number of electrons an ion is formed. Interestingly, if the proton number is altered, the element itself changes into another element For example, all atoms of oxygen invariably have 8 protons in their nucleus. While the proton number remains constant, the electron and neutron numbers may vary in an atom.
We know, that elements are arranged in the periodic table according to the change in their chemical properties, taking hydrogen as the first element. In the periodic table, we observe that a change in chemical property results in a corresponding change in its atomic number (or number of protons). This observation led to the definition of atomic number.
Modem definition of atomic number is as follows:
Definition:
The atomic number or proton number (Z) of an element is the number of protons present in the nucleus of an atom of that element.
From the position of the element in the periodic table its atomic number can be determined.
Since, an atom is electrically neutral and the number of electrons is equal to the number of protons so, the number of electrons in a neutral atom can also be taken as the atomic number of that element.
2. Representation of atomic number:
An atomic number of an element is represented by Z. The Electric charge in the nucleus of an atom is +Ze where e is died magnitude of the charge of an electron. Similar to the method of denoting the mass number of an atom, we can express the atomic number of an element
In this case, we write the symbol of the element, and then either on the left or on the right side of the symbol we write the atomic num¬ ber as a subscript. When writing the symbol of an element we don’t need to write the atomic number But the mass number needs to be mentioned.
For example, we can write H¹ instead of 1H1 but not H as it could mean either H¹ or H² and we will not be able to differentiate between the two.
Atomic Nucleus Binding Energy Of A Nucleus
Binding Energy Definition:
The energy that keeps protons and neutrons confined to the nucleus, is called nuclear binding energy. If an amount of energy equal to the nuclear binding energy is supplied from outside then the nucleus disintegrates and the protons and neutrons exist as free particles. Hence, the binding energy of a nucleus is also defined as the external energy required to separate the constituents of the nucleus.
Relation between binding energy and mass defect:
The binding energy of a nucleus can be explained using mass-energy equivalence. When protons and neutrons exist freely, the sum of their masses gives the ‘mass energy of the system. But when these very protons and neutrons form a nucleus, both nuclear binding energy and a nuclear mass exist. Hence, from the law of conservation of mass energy,
(Sum of masses of protons and neutrons) × c² = mass of nucleus × c² + nuclear binding energy
If Z = atomic number and A = mass number of the nucleus and mp and mn, mass of proton and mass of neutron independently, the conservation condition can be mathematically expressed as
Zmpc² + (A- Z)mnc²= MzA c² > AC² + ΔE
Where Mz A = mass of the nucleus and AE = binding energy
Hence, AE = {Zmz + (A-Z)mn MZ,A}c² ………………………………………………. (1)
This can be written as
ΔE = Am c²………………………………………………. (2)
The expression within the second bracket in equation (1) represents Δm. When a nucleus is formed from its nucleons, the mass of the nucleus is less than the masses of the nucleons taken together. This means that {Zmp + (A-Z)mn} is greater than MZ, A. This difference is called mass defect, Am. In fact, this reduced mass is transformed into binding energy to form the nucleus
Example: Mass of proton mp = 1.0073 u and that of neutron
mn = 1.0087 u. Since the nucleus of He4 consists of 2 protons and 2 neutrons, their total mass
The experimental value of the mass of He nucleus, M2 > 4 = 4.0015.
Hence, mass defect in the He4 nucleus
Δm =[2mp + 2mn]-M2,4 = 4.0320-4.0015 = 0.0305 u
The binding energy of the He4 nucleus
ΔE = 0.0305 × 931.2 MeV = 28.4016 MeV
Hence, the binding energy per nucleon or the average binding energy of the He4 nucleus
⇒ \(\frac{\Delta E}{4}=\frac{28.4016}{4}\)= 7.1004 MeV
Stability of a nucleus:
The more the binding energy of a nucleus, more is the energy required to separate its nucleons and hence the nucleus is more stable. In this respect, a nucleus can be compared with a liquid drop. A very large liquid drop tends to break up into smaller drops whereas a large number of small drops tend to join to form a large drop. Similarly, a large nucleus (like that of U238 ) and a small nucleus (like H² ) both are unstable.
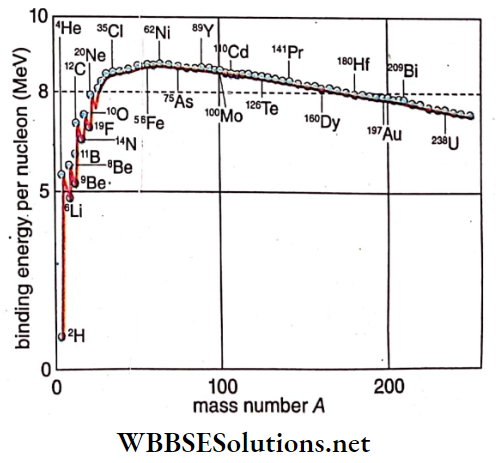
The stability of a nucleus depends upon the binding energy per nucleon. The graph of binding energy per nucleon of different elements vs mass number is.
Significance of the binding energy curve:
The nearly constant value of the binding energy per nucleon for nuclei of about A
= 25 to A = 170 shows a
- Saturation, since no further increase occurs if extra nucleons are added.
- The saturation means that nuclear force is a short-range force—any extra nucleon, when added, resides on the surface, not affect the nucleons deep inside the pre¬ existing nucleus.
- The binding energy per nucleon for A higher than about 220 is less than that at the middle region of the periodic table.
- So a heavy nucleus tends to break up into two mid-range nuclei to attain higher stability this is nuclear fission. A significant amount of energy is released in this process.
- Nuclei with say, A < 4 to 6 have a relatively low value of binding energy per nucleon. So two or more of them tend to unite into one nucleus with a higher value of binding energy per nucleon, to attain stability. This is nuclear fusion. In this pro¬ cess also, a significant amount of energy is released.
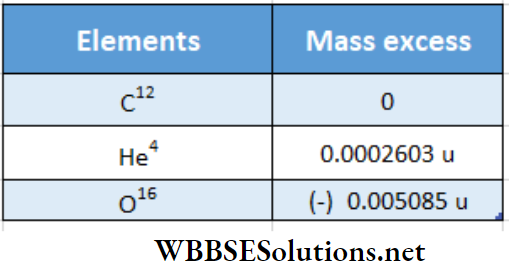
Atomic nucleus class 12 notes Mass excess
Mass excess Definition:
If mass number = A and atomic mass = M of a nuclide then mass excess of that nucleus, ΔM = M- A.
For the C12 atom, A = 12, and in this case, | according to the definition of atomic mass the actual mass of the C12 atom, M = 12 u. However, for all other elements, the values of A and M are different. For example, for He4, A = 4 but Af
= 4.002603 u, for O16, A = 16 but M = 15.994915 u. This difference in the value A and the experimental value of M is called mass excess. Mass excess can be either positive or negative for different nuclei
From the above examples, the mass excess ofthe elements is listed below
Atomic Nucleus Volume And Density Of A Nucleus
Different nuclei are similar to a drop of liquid of constant density. The volume of a liquid drop is proportional to its mass, which is proportional to the number of molecules contained in it. Similarly, the nuclear density is also a constant quantity. So the nuclear volume is directly proportional to the mass number and is independent of the separate values of the proton number and the neutron number.
Radius of the nucleus
Experimentally it has been found that a proton or a neutron has a radius,
r0 = 1.2 × 10 -13 cm
= 1.2 × 10 -15 m
So the volume of each proton or neutron, V’ = \(\frac{4}{3} \pi r_0^3 A\)
Let the total number of protons and neutrons in the nucleus = mass number =A
Then the volume of the nucleus V = \(\frac{4}{3} \pi R^3\)
Let the total number of protons and neutrons in the nucleus = mass number =A
Hence, R³ = r ³0 A or, R = r0 A1/3
For A = 216 , we get R = 6r0 = 7.2 × 10 -13 cm
.
Hence even the radius of a heavy nucleus is less than 10-12cm.
Calculation of nuclear density:
Estimated mass M of a nucleus of mass number A,
M = A u = A × 1.66 × 10 -24 g
Also, volume of this nucleus, V = \(\frac{4}{3} \pi r_0^3 \times\) × A
Hence, nuclear density
V = \(\frac{M}{V}=\frac{A \times 1.66 \times 10^{-24}}{\frac{4}{3} \pi r_0^3 \cdot A}\)
= \(\frac{3 \times 1.66 \times 10^{-24}}{4 \pi \times\left(1.2 \times 10^{-13}\right)^3}\)
= 2.3 × 10 -14 g. cm-3
Generally, nuclear density Is taken as 2 × 10 14 g.cm-3 or 2 × 10 17 kg. m-3, which is very high and represents the presence of a lot of mass concentrated within a very small space. So, nuclear density is more than 1014 times the density of water
Class 12 physics nucleus chapter notes
Atomic Nucleus Volume And Density Of A Nucleus Numerical Examples
Example 1. For a nearly spherical nucleus-, r =r0 A1/3, where r is the radius A is the mass number and rQ is a constant of value 1.2 × 10 -15 m. If the mass of the neutrons and protons are equal and equal to 1.67 × 10 -27 kg, prove that the density of the nucleus is 10 14 times the density of water.
Solution:
Mass of A -number of neutrons and protons « mass of nucleus (M) = 1.67 × 10 -27 A kg
Again, the volume of the nucleus
V = \(\frac{4}{3} \pi r^3=\frac{4}{3} \pi r_0^3 A\)
= \(\frac{4}{3} \times 3.14 \times\left(1.2 \times 10^{-15}\right)^3\)
Density of nucleus = \(\frac{M}{V}=\frac{1.67 \times 10^{-27} \times A}{\frac{4}{3} \times 3.14 \times\left(1.2 \times 10^{-15}\right)^3 \cdot A}\)
= 2.3 × 1017 kg.m-3
∴ \(\frac{\text { density of nucleus }}{\text { density of water }}=\frac{2.3 \times 10^{17}}{1000}\)
= 2.3 × 1014
∴ The density of the nucleus is more than 1014 times the density of water.
Practice Problems on Atomic Mass and Number
Atomic Nucleus Discovery Of Radioactivity
Uranium and thorium
Henry Becquerel first observed in 1896 that photographic plates preserved in opaque black paper get affected, when kept close to a compound, uraniumpotassium sulphate, and also found that no external energy source was required to initiate the chemical reaction. Becquerel also observed similar properties in other compounds of uranium and named it radioactivity. Scientist Madam Marie Curie of Poland, discovered radioactivity in the element thorium too.
Polonium and radium:
Marie Curie and Pierre Curie extracted radioactive elements polonium and radium from the uranium ore ‘pitchblend’. Polonium and radium exhibit radioactivity 103 and 10® times more than that exhibited by uranium.
Characteristics of radioactivity:
- Elements of mass number 210 or more, generally exhibit radioactivity.
- All radioactive substances emit highly penetrating radiations (rays) that can easily penetrate thin metal sheets and similar substances.
- Radioactivity is a continuous and spontaneous activity.
- Radioactive rays affect photographic plates.
- Radioactivity is not affected by physical changes brought about by light, heat, electric or magnetic fields.
- Chemical changes of radioactive elements cannot influence the amount of radioactivity. Hence, the radioactivity of an ele¬ ment and that of its compound is the same.
- A chemical change cannot influence the radioactivity of an element and that chemical change involves electrons outside the nucleus. It led to the conclusion that radioactivity is entirely a nuclear phenomenon that happens due to internal changes in the nucleus.
- Radioactivity brings about the transmutation of elements 1000 where one element changes into another.
Some useful definitions
1. Radioactivity or radioactive decay or radioactive disintegration:
The phenomenon of spontaneous emission of rays from an unstable nucleus or due to a nuclear reaction is called radioactivity radioactive decay or radioactive disintegration. Radioactive rays are highly penetrating and originate due to changes in nuclear structure.
Radioactive elements: Elements that exhibit radioactivity spontaneously are called radioactive elements. Generally, the nucleus of a radioactive element is unstable. The nature of stability varies from element to element.
Examples: Uranium, Radium, Thorium.
Parent and daughter atom:
A radioactive element or atom that exhibits radioactivity is called a parent atom. The atom that is left behind after the emission of radioactive radiation is called a daughter atom, which may or may not be radioactive. If it is radioactive then it will be the parent atom for the next decay.
Radioactive sample:
A radioactive sample is a specimen of the material that emits radioactive radiation. From medicine making to paper manufacture, radioactive samples are in wide use. The entire mass of any naturally occurring radioactive substance is not radioactive. Because radioactive decay starts from its beginning, some part of the radioactive substance transforms into stable non-radioactive parts For example, any radioactive sample of uranium also contains some non-radioactive lead.
Radioactive isotopes or radioisotopes:
Radioactivity is not a characteristic of an element One isotope of an element may be radioactive like C14 whereas the other isotope C12 is non-radioactive. Hence, radioisotopes are radioactive isotopes of elements.
Example: The radioactive nature of two uranium isotopes U238 and U235 are different. Again, Pb206, and Pb208 are non-radioactive but Pb210 called RaD is radioactive.
Class 12 physics nucleus chapter notes
Atomic Nucleus Classification Of Radioactive Emissions
Rutherford’s experiment
The radioactive emissions from radioisotopes when subjected to a strong magnetic field at right angles to the plane of the radiations, show different deflections. The experimental arrangement.
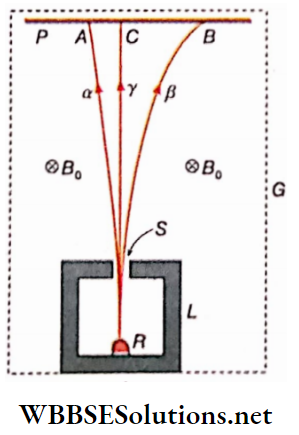
Experimental arrangement
G – A container, nearly evacuated and placed In a dark room.
L – A small, deep, and thick-walled lead container.
R – A mixture of different radioisotopes.
S – Slit on the lid of the lead container that allows radiation to come out upwards.
P – A photographic plate.
B0 – A strong magnetic field perpendicular to the plane of the paper and directed downwards.
1. Observation:
When the photographic plate is examined after a considerable length of time,
Three distinct lines are seen on the plate:
- Line A: It shows a small deviation of some emissions to the left.
- Line B: This shows a significant deviation of some emissions to the right.
- Line C: It shows a part of the radiation not affected by the magnetic field.
2. Inference:
The inferences from the observations are that a mixture of radioactive samples can emit three types of radiation.
- α -rays (alpha rays): Applying Fleming’s left-hand rule it is seen that line A Is produced by the comparatively heavy, positively charged stream of high-speed particles called α – rays or α -particles
- β – (beta rays): By similar analysis, the Une made by light, negatively charged stream of high-speed particles called β -rays or β – particles.
- γ -rays (gamma rays): The emission, that is not deflected by the magnetic field and produces line C, consists of y rays or γ -radiations. γ -radiation is not a stream of charged particles.
3. Discussions:
- Identical results will be obtained when instead of a netic field an electrical field is applied from the right to the left along the plane of the paper.
- No radioactive Isotope can emit all three regulations α, β, and γ simultaneously. Hence, a mixture of different types of radioisotopes needs to be kept in R to obtain the results described. Generally, any radioactive sample contains parent Isotopes us well as a daughter isotope.
- If this daughter Isotope is also radioactive then we can get three types of rays.
- For example: This event may happen parent isotope emits α -rays and the daughter isotope emits β – rays
Alpha (α) rays
1. Nature of α -rays:
Measurements of charge q and specific q/m charge establish that α -rays are high-speed, streams of particles.
- α -particles are positively charged and their charge q – + 2e, that is it contains two units of elementary” charge (e = +1.6 × 10 -12 C ) as that of a proton or electron.
- The mass of an a -particle is four times the mass of a proton.
Experimentally it is found that an a -particle is comparable to a helium-4 nucleus. As the helium-4 nucleus consists of two protons and two neutrons, so alpha particle is denoted by the symbol 2He4.
Properties of α – rays:
Α -rays are not rays, they are a stream of high-speed particles. Each of the particles is known as α -particle.
- Each α -particle is positively charged and it contains two units of elementary charge as that of the electron.
- The mass of each α -particle is equal to the mass of 4 protons.
- From its mass and charge, it is concluded that α -particles are structurally identical to a helium nucleus.
- As α -particles are positively charged they can be deflected by the electric or magnetic field
- The initial velocity and kinetic energy of α -particles depend on radioisotopes from which a -particles are emitted. In most cases, the initial velocity is nearly 109cm s -1 and initial kinetic energy is within the limit of 5 MeV to 10 MeV.
- 1C1 From its high initial kinetic energy it can be concluded that α-particles are emitted from the nucleus of an atom. CD α -particles have low penetrating power in comparison to β and γ -rays and can be completely absorbed in mm thick aluminum plate.
- As penetrating power is less, or -particles have high ionization power in comparison to β and γ -rays. In a gaseous medium a -particles dislodge orbiting electrons and ionize the gas.
- Affects the photographic plate. When it strikes fluorescent material (like zinc sulfide) it produces scintillation (flashes of light).
- In a gaseous medium α -rays cannot travel beyond a certain range. This range is determined by the nature ofthe the emission source. α -rays are used in nuclear reactions and in artificial transmutation of one element into another:
Beta(β)rays
1. Nature Of β -rays:
From the experiment, we know that like α -rays, β -rays are also a stream of fast-moving particles.
From the measurement of charge q and the specific q/m charge of β-rays, it is proved that each β-particle is an electron, i.e.,
- Charge of β -particle e = -1.6 × 10-19 C and
- Mass of β -particle =9.1 × 10– 31 1 kg
Since the mass of an electron is negligible compared to the mass of the proton, therefore the mass number of β -particle is taken as zero. Due to its unit negative charge fi -particle is sometimes expressed as -1β2 or -1e2
Class 12 physics nucleus chapter notes Properties of β -rays:
β -rays are not rays rather they are a stream of high-speed particles known as β – particles.
Each β -particle is an electron
- As β -particles are light and negatively charged they are significantly deflected by the electric or magnetic field.
- Initial velocity, as well as the kinetic energy of each particle, depends on the radioisotopes from which the particle is emitted. Initial velocity may take any value from zero to the velocity of light.
- Similarly, kinetic energy ranges from zero to a certain upper limit Generally this value ranges from 5 MeV to 10 MeV.
- From its high initial kinetic energy, it can be concluded that they are emitted from the nucleus of an atom.
- Inside a nucleus when a neutron transforms into a proton, an electron is generated. As nuclear force does not influence electrons, it cannot confine the electron in the nucleus and so it comes out. This electron is β – particle and not orbital.
- The penetration power of β -rays is 100 times greater than that of β -rays but \(\frac{1}{100}\) part of that of γ -rays. It is completely absorbed by a 1 cm thick aluminum plate.
- β-rays can ionize gas but its ionizing power is \(\frac{1}{100}\) of that of a -rays.
- It affects photographic plates and produces weak scintillation on falling on a fluorescent screen. -β rays are used in the nuclear reaction and artificial trans¬ mutation.
- Whenever a β -particle is emitted from the nucleus of a radioactive element a massless, chargeless particle called neutrino is formed, the existence of which was originally suggested by Wolfgang Pauli in the year 1930. The name was given by Fermi (in 1934) while giving his neutrino theory of β -decay. It was detected in 1956 by Reines and Cowan.
Comparison between cathode rays and β – rays:
1. Similarities:
- Both are streams of moving electrons.
- Both possess penetrating and ionizing properties.
- Both affect photographic plates and exhibit fluorescence when falling on compounds like zinc sulfide etc.
- Both are deflected by an electric or a magnetic field.
2. Dissimilarities:

Gamma rays
Nature of γ-rays:
- γ-rays are electromagnetic rays like light rays and with the same speed as that of light in a vacuum.
- According to Planck’s quantum theory, γ-ray is constituted of a stream of photons. As frequency is high, so energy of each photon is also high. Its wavelength is shorter than that of X-rays, ranging from 0.005 Å to 0.5 Å.
Example: The energy of γ -ray photon of wavelength 0.01Å is 1.24 MeV.
Properties of γ -rays:
Like light, γ-rays are electromagnetic waves and travel at the same speed as that of light in any medium.
The wavelength of γ -rays is in the range from 0.005Å to 0.5Å γ -rays are neutral and therefore they are not deflected by an electric or a magnetic field.
According to quantum theory, γ -rays comprise high-energy photons. The energy of each photon is considerably high and can measure up to a few MeV
The high energy of γ-ray photons implies that γ-rays are emitted from the nucleus. When a and β -particles are emitted from a nucleus the nucleus acquires an excited energy state. To return to the ground state γ -rays are emitted
The penetrating power of γ -rays is high in comparison with a or ft -rays. It can penetrate a few centimeters of lead plate. The ratio of the penetrating power of α, β, and γ- rays is 1 : 102: 104.
The ionizing power of 7 -rays is comparatively less than that of α and β -rays.
γ -ray, like X-rays, undergoes diffraction.
γ -rays, can affect photographic plates and adversely affect the cells of the human body. Therefore, for the treatment of cancer and tumor γ -rays are used. Powerful γ -ray bursts are used to probe star formation.
γ -rays are used in nuclear reactions and artificial trans¬ mutation operations.
γ -ray photon of energy of a few MeV or more, when pass¬ ing close to a heavy nucleus changes into an electron and a positron (particle identical to an electron but with positive charge). This is called “pair production’’ which is an example of energy changing to mass hence the energy associated with a γ -ray photon can be taken as E = 2me × c² J.
Comparison between X-rays and γ -rays:
1. Similarities:
- Both X-rays and γ-rays are electromagnetic waves. ElSl Both can create fluorescence and affect photographic plates.
- Both have ionizing and penetrating power. Crystals can diffract both X -rays and γ -rays. ‘Both X -rays and γ -rays remain unaffected by an electric or a magnetic field.
- x -rays and 7 -rays travel with the speed of light in a vacuum.
2. Dissimilarities:
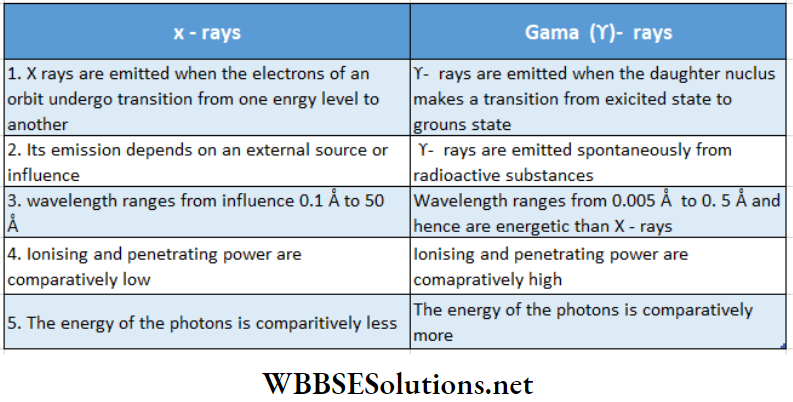
Nuclear Physics Class 12 Notes
Atomic Nucleus Activity
Activity Definition:
The rate of radioactive disintegration with time is called the activity of the sample.
Mathematically, activity (A) \(\frac{d N}{d t}=\lambda N\) = the numerical value
There fore activity A∝ N and \(A \propto \lambda \propto \frac{1}{T} .\)
From this, we can say, a radioactive sample has greater activity if
- The sample contains a large number of radioactive atoms [N)
- Decay constant is high or half-life is low.
Also if A0 and A are the activities initially and after a time t, then
⇒ \(A_0=\lambda N_0, \text { and } A=\lambda N\)
∴ \(\frac{A}{A_0}=\frac{N}{N_0}=e^{-\lambda t}\)
Or, A = \(A_0 e^{-\lambda t}\)
∴ Activity also decreases exponentially with time
Units to measure activity
The activity of a radioactive substance is measured in terms of the number of disintegrations per unit of time. In SI, the unit is becquerel or Bq and 1 Bq = 1 dis¬ integration per second or 1 dps.
Other practical units are:
Curie 1 Ci= 3.70 × 1010 dps
Rutherford = 1 Rd = 106 dps
Atomic Nucleus Activity Numerical Examples
Example 1. Po210 has a half-life of 140 d. In lg Po210 how many disintegrations will take place every second? (Avogadro’s number = 6.023 × 1023 )
Solution:
Disintegration constant,
λ = \(\frac{0.693}{T}=\frac{0.693}{140 \times 24 \times 60 \times 60} \mathrm{~s}^{-1}\)
Number of atoms in 210 g Po210
= Avogadro’s number = 6.023 × 1023
Number of atoms in 1 g Po210, N = \(\frac{6.023 \times 10^{23}}{210}\)
Disintegration per second
= Activity = λN
= \(\frac{0.693}{140 \times 24 \times 60 \times 60} \times \frac{6.023 \times 10^{23}}{210}\)
= 1.64 × 1014 dps
Example 2. A radioactive sample of half-life 30 d contains 1012 particles at an instant of time. Find the activity of the 1 sample
Solution:
Decay constant
λ = \(\frac{0.693}{T}\)
= \(\frac{0.693}{30 \times 24 \times 60 \times 60} \mathrm{~s}^{-1}\) and N = 1012
∴ Activity , λN = \(\lambda N=\frac{0.693 \times 10^{12}}{30 \times 24 \times 60 \times 60}\)
= 2.67 × 105 dps
Examples of Nuclear Reactions
Example 3. How much 84Po210 of a half-life of 138 days is required to produce a source of α -radiation of intensity 5 (millicurie)?
Solution:
Decay constant, \(\frac{0.693}{T}=\frac{0.693}{138 \times 24 \times 60 \times 60} \mathrm{~s}^{-1}\)
Activity, A =5 mCi = 5 × 3.70 × 107 dps
Now, A = λN
N = \(\frac{A}{\lambda}=\frac{5 \times 3.7 \times 10^7 \times 138 \times 24 \times 60 \times 60}{0.693}\)
Again, the number of atoms contained in 210 g Po210
= Avogadro’s number = 6.023 × 1023
Hence, the mass of N such particles
= \(\frac{210 \times 5 \times 3.7 \times 10^7}{6.023 \times 10^{23}} \times \frac{138 \times 24 \times 60 \times 60}{0.693}\)
1.11 × 10-6 (approx)
Nuclear physics class 12 notes
Example 4. A 280-day-old radioactive substance shows an activity of 6000 dps, 140 days later its activity becomes 3000 dps. What was its initial activity?
Solution:
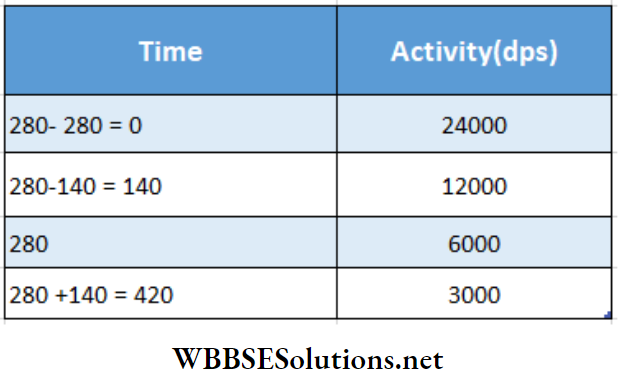
In the table, the last two values of activity are given. These are used to calculate the first two values.
Hence, initial activity =24000 dps
Nuclear physics class 12 notes
Atomic Nucleus Nuclear Fission
Nuclear Fission Definition:
Breaking up of a heavy nucleus into two nuclei of almost equal masses is called Nuclear Fission
Peripheral reactions: In most nuclear reactions, the emitted particle is not heavier than a projectile particle. This indicates that there is a small change in the atomic number and mass number ofthe target nucleus. These are termed peripheral reactions because the core of the nucleus is practically unaffected. For example,
⇒ \({ }_7 \mathrm{~N}^{14}+{ }_2 \mathrm{He}^4 \rightarrow{ }_8 \mathrm{O}^{17}+{ }_1 \mathrm{H}^1 ;{ }_7 \mathrm{~N}^{14}+{ }_0 \mathrm{n}^1 \rightarrow{ }_6 \mathrm{C}^{14}+{ }_1 \mathrm{H}^1\)
Collision of thermal neutron with U-235: This results in the formation of almost two equally heavy nuclei, due to the disintegration of the heavier U-235 nucleus. For example
⇒ \({ }_0 \mathrm{n}^1+{ }_{92} \mathrm{U}^{235} \rightarrow{ }_{35} \mathrm{Br}^{85}+{ }_{57} \mathrm{La}^{148}+3{ }_0 \mathrm{n}^1\) …………………….. (1)
⇒ \({ }_0 \mathrm{n}^1+{ }_{92} \mathrm{U}^{235} \rightarrow{ }_{36} \mathrm{Kr}^{92}+{ }_{56} \mathrm{Ba}^{141}+3{ }_0 \mathrm{n}^1\) …………………….. (2)
Such splitting up of the nucleus cannot be termed a peripheral reaction. Generally, it is called nuclear fission. This was invented in 1939 by Otto Han and Strassman
The energy released In nuclear fission:
Mass lost during nuclear fission changes to energy as per mass-energy equivalence. In the equation (1),
Initial mass = total mass of U-235 and neutron
= 235.1 + 1.009 = 236.1 u (approx.) J
Final mass = total mass of Br-85 , La-148 and 3 neutrons
= 84.9 + 148.0 + 3 × 1.009 = 235.9 u (approx.)
∴ Mass loss =236.1- 235.9 = 0.2 u
∴ Energy released =0.2 × 931MeV = 186 MeV (approx.) (as- 1 u ≈ 931 MeV ). This energy is available from only one nucleusÿ of U-235
Considering the number of atoms of U-235 in 1 g of.U-235, the energy released during nuclear fission is of the order of 7.6 x 1010 J per g. This energy is equivalent to the energy that can be obtained by burning 3000 tons of coal.
Moderator
The three neutrons released in nuclear fission practically absorb the released energy (approximately 186 . MeV) and change to high-speed neutrons as kinetic energy increases. For further use of these neutrons for fission reaction, these are to be slowed down as thermal neutrons. Substances like heavy water (D2O), and graphite can slow down the high-speed neutrons when neutrons pass through them. These are called moderators
Chain reaction
Nuclear reactions sustained by the product of the initial reaction leading from one reaction to the other consecutively is called chain reactions. In equation (1), one neutron is bombarded on the U-235 target and 3. neutrons are released. They are slowed down to thermal neutrons.
Now they are used to set up further fission of 3 U-235 nuclei, releasing 9 new neutrons and so on. The number of fissions, like 1, 3, 9, 27, 81, is increasing in the form of multiple progression. Hence, in a short time, a large number of fissions take place, t releasing a huge amount of thermal energy. This is the principle’ i of an atomic bomb
Critical Size
Neutrons formed during fission tend to escape without hitting the target nucleus. This decreases the number of available neutrons to sustain the chain reaction, ultimately resulting in the cessation of the chain tion.
To prevent this, the following two methods are applied:
- The radioactive sample Uiat is taken in the shape of a sphere, which has less surface area compared to its volume.
- Mass of the sample taken is a little more than the calculated value. To continue nuclear fission sustaining its chain reaction the minimum size of the sample required is called critical size.
Controlled fission: Nuclear reactor
The energy released during nuclear fission should not be misused. Rather it should be used for useful and necessary purposes like the tion of electricity.
But for that, the following precautions should be taken:
- The huge energy produced should not go out of control causing immense destruction and
- Energy supply should continue almost at the same rate for a long time. Fission brought about conforming to the above two conditions is called controlled chain reaction or controlled fission.
The device where controlled fission and subsequent generation of electricity is conducted is called a nuclear reactor.
The effective number of fission neutrons produced per absorp¬ tion in the fuel in each successive step of a chain reaction is the balled neutron reproduction factor. In case of an uncontrolled chain reaction in an atomic bomb, the ratio is 2.5 or above.
In a nuclear reactor, the factor is kept close to 1 or slightly more to attain the condition stated above. This Is the guiding principle of an atomic reactor.
Out of different types of reactors, a Pressurised Water Reactor or PWR is most widely used.
A schematic diagram of a PWR is shown in Fig. 2.7. The reactor consists of:
Core:
Inside the core the nuclear reaction takes place. Core contains
- Fuel rod,
- Control rod
- Moderator and
- Coolant.
1. U-235 ’is used as a fuel rod. Heat is generated when U-235 is bombarded with neutrons.
2. A steel rod with a coating of boron is used as a control rod. Boron absorbs surplus thermal neutrons
3. Heavy water is usually used as a moderator. Moderator slows down the high energy neutrons produced1 =, due to the nuclear reaction in the core to thermal neutrons to sustain the chain reaction.
4. Generally water is used as a coolant. The heat generated due to fission is absorbed by coolant water maintained at high 1 pressure to avoid boiling
WBCHSE physics class 12 nucleus notes Heat exchanger:
In this part, heat from coolant which is radioactive is transferred to non-radioactive water for further use. Radioactive water from coolant is kept confined within the core area by protective concrete shielding.
Turbine:
Non-radioactive water, at high temperature, is piped out of the shielding and converted to steam to rotate the turbine to produce electricity in the same manner as in a Thermal Power Station.
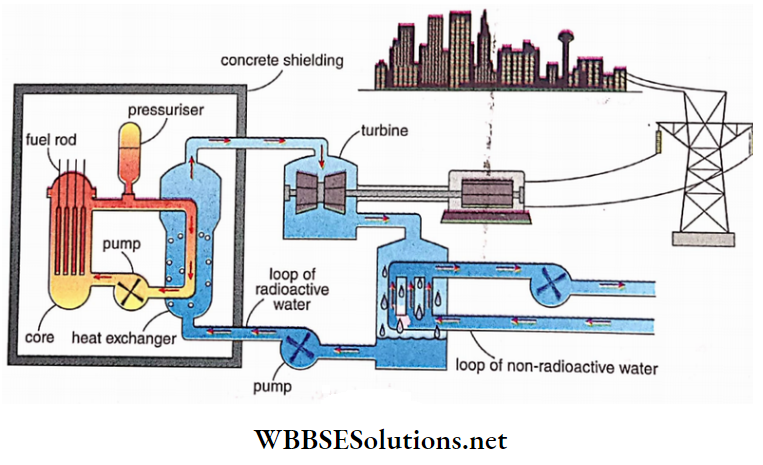
WBCHSE physics class 12 nucleus notes
Atomic Nucleus Nuclear Fission Numerical Examples
Example 1. The kinetic energy of a slow-moving neutron is 0.04 eV. What fraction of the speed of light is the speed of this neutron? At what temperature will the average kinetic energy of a gas molecule be equal to the energy of this neutron? [mass of neutron : 1.675 × 10-27 kg, Boltzmann constant, kB = 1.38 × 10-23 J. K-1
Solution:
The kinetic energy of the slow neutron
= 0.04 eV = 0.04 × (1.6 × 10-19)J
Kinetic Energy, \(E_k=\frac{1}{2} m v^2\)
v = \(\sqrt{\frac{2 E_k}{m}}=\sqrt{\frac{2 \times 0.04 \times 1.6 \times 10^{-19}}{1.675 \times 10^{-27}}}\)
= 2764 m.s–1
⇒ \(\frac{v}{c}=\frac{2764}{3 \times 10^8} \times 100 \%\)
= 0.00092%
Average kinetic energy at temperature T
= \(\frac{3}{2} k_B T=0.04 \times\left(1.6 \times 10^{-19}\right) \mathrm{J}\)
T = \(\frac{2 \times 0.04 \times\left(1.6 \times 10^{-19}\right)}{3 \times\left(1.38 \times 10^{-23}\right)}\)
= 309 K
= 36° C
Conceptual Questions on Radioactive Decay
2. Example In a typical nuclear fission reaction, it was found that there was a loss of mass of 0.2150 u. How much energy in MeV will be released from this reaction? (c = 3× 108ms-1).
Solution:
Loss of mass
Δm = 0.2150 u = 0.2150 × (1.66 × 10-27) kg
Associated release of energy,
ΔE = Δm c²
= \(0.2150 \times\left(1.66 \times 10^{-27}\right) \times\left(3 \times 10^8\right)^2\)
= \(3.2121 \times 10^{-11} \mathrm{~J}=\frac{3.2121 \times 10^{-11}}{1.6 \times 10^{-19}}\)
200 × 106 eV
= 200 MeV
WBCHSE Physics Class 12 Nucleus Notes
Atomic Nucleus Nuclear Fusion
Nuclear fusion Definition:
The phenomenon in which two or more light nuclei combine to form a comparatively heavy nucleus is called nuclear fusion
Fusion is the reverse phenomenon of fission.
Example:
The probability of fusion of two hydrogen nuclei is very low. A good example of nuclear fusion is the fusion between two deuterons i,e.f two heavy hydrogen nuclei (jH2)
⇒ \({ }_1 \mathrm{H}^2+{ }_1 \mathrm{H}^2 \rightarrow{ }_2 \mathrm{He}^3+{ }_0 \mathrm{n}^1\) …………… (1)
The probability of fusion of another hydrogen isotope, tritium (jH3) with deuteron is also high
⇒ \({ }_1 \mathrm{H}^3+{ }_1 \mathrm{H}^2 \rightarrow{ }_2 \mathrm{He}^4+{ }_0 \mathrm{n}^1\) …………… (1)
The energy released In nuclear fusion: Mass lost during nuclear fusion changes to energy as per mass-energy equivalence. In the equation (1) :
Initial mass = total mass of 2 deuterons = 2 × 2.015 = 4.030 u
Final mass = total mass of He3 and neutron
= 3.017 + 1.009 = 4.026 u
Mass loss = 2 × 2.015 -(3.017 + 1.009)
= 0.004 u
The energy released =0.004 × 931 MeV
= 3.7 MeV (approx.)
Hence, the energy released from 1 g of deuterium will be about
“For fusion of tritium with deuteron, the released per gram will be more and is about 30 × 1010 J.
Thus, the energy released from comparatively easily available deuterium or tritium fusion is greater than tÿat obtained from the fission of U-235
In addition, in a fusion reaction, a greater percentage of the nucleus takes part than the participant in fission nuclei in a fission reaction. A hydrogen bomb is made, based on this fusion reaction.
Conditions of Nuclear Fusion:
1. light dement:
For bringing about (Vision of two post* tively charged nuclei. the electrostatic force of repulsion needs to be overcome. Hydrogen-like lighter elements arc convenient because of die low positive charge contained in them and thereby there is less force of repulsion.
2. High temperature:
To bring about nuclear (Vision, hydrogen isotopes are to be raised to a few awe degrees Celsius temperature. That is why fusion reaction Is a thermonuclear reaction. To reach a high temperature, the most effective way is to set up an uncontrolled fission reaction. Therefore, to get nuclear energy from fusion, nuclear fission has to take place first.
The energy of the sun and the stars:
In the sun and other stars, the energy at the center is produced by the thermonuclear reaction. The core of stars being at a very high temperature, favours the process. According to the presently accepted theory, the thermonuclear reaction cycle in the sun is completed in steps. In ever)’ Cycle, primarily due to nudear fusion of four protons, one helium nudes, and two positrons are formed.
1H1 + 1H1 + 1H1 + 1H1 → 2He4 + +1e0 + +1e0
The mass defect = mass of 4 protons- combined mass of 2He4 and 2 positrons = 4 × 1.008 – (4.003 + 2 0×.00055) = 0.0279 u Corresponding energy =0.0279 × 931 = 26MeV (approx.)
Sun has a huge hydrogen stored, but per year only 1 part in 1011 of the hydrogen stored in the sun is used. Also, the energy released due to thermonuclear reaction is about 4 × 1026 W. It is estimated that the sun will continue producing energy for another 5 billion years before the total store of energy fuels is exhausted
Atomic Nucleus Uses Of Radioactive Isotopes
Medical science:
- Studying blood circulation patterns and investigating of
ailments, radioactive sodium (Na-24) and radioactive
phosphorus (P-32) are used.
- Radioactive radium or strontium are used to destroy
cancer cells. Presently radioactive cobalt (Co-GO) is extensively used for this purpose.
- Radioactive phosphorus (P-32) is very effective in treating blood cancer and brain tumors. S3 Radioactive iodine (1-131) is used in the treatment of the thyroid gland.
Radioactive tracer or indicator:
For various investigation purposes, P-32 and Na-24 are used as tracers or indicators. Examples: Different chemical reactions in plants and animals, the reaction of phosphorus-containing manure for agriculture, and detecting cracks in dams and reservoirs.
Radioactive pigments:
A paint in which traces of radium and a fluorescent ZnS are mixed glows even in the darkness. This pigment is used in watch dials, electrical switches, roads, etc.
Radiocarbon dating:
Cosmic rays bring about a nuclear reaction with atmospheric nitrogen producing some C-14 of half-life about 5600 years. C-14, in the atmosphere, changes to CO2 (carbon dioxide) and during the process of photosynthesis enters into the plant body. In living plant and animal bodies, a definite ratio is maintained between radioactive C-14 and normal C-12.
Assume this ratio is 1: x. The quantity of radiocarbon C-14 decreases exponentially after the death of a carbon-enriched sample, but the quantity of C-12 remains constant. So, at the time T, 2T, 3T, ……….., the ratio of C-14 and C-12 will be 1/2 :x, 1/4 :x, 1/8 :x, respectively. Hence, by estimating this ratio in an archaeological sample, the age of the sample can be estimated. Thus, radiocarbon C-14 acts as a radioactive clock.
Geological time determination:
The half-life of c-14 is only 5600 years while Earth and other geological specimens are more ancient. Therefore C-14 clock cannot be used for determine their age. Here uranium clock is used by noting the ratio of lead and uranium (half-life = 450 crores of years) in the sample. Q Production of energy: Radioactive uranium or plutonium is used as fuel in nuclear power stations.
WBCHSE physics class 12 nucleus notes
Atomic Nucleus Uses Of Radioactive Isotopes Numerical Examples
1. In a piece of ancient wood, C and C-12 are present. The ratio of C-14 and C-12 in this wood at present is part of their ratio in the ancient wood. The half-life of C14 is 5570 y. What is the age of the wood?
Solution:
Half-life C14 , T = 5570 y
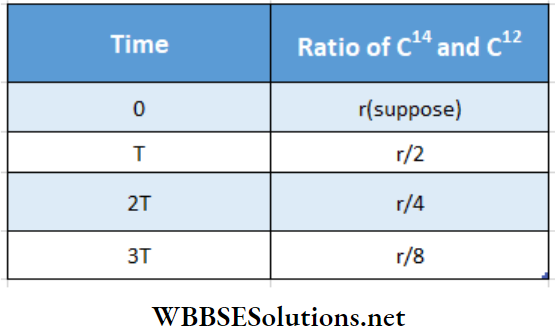
From the table, in time 3T ratio of C and C is \(\frac{r}{8}\) i.e., \(\frac{1}{8}\) of that ratio when T = 0
∴ The age of the piece of wood
3T = 3 × 5570
= 16710 y
Real-Life Applications of Nuclear Energy
Atomic Nucleus Very Short Questions And Answers
Question 1. What is the relation between a unified atomic mass unit (u) and an electron volt?
Answer: [lu = 931.2 MeV]
Question 2. The mass of a proton or 1.67 × 10-24 g. What is its equivalent energy in MeV?
Answer: [939.4 MeV]
Question 3. What is the order of magnitude of the density of nuclear matter?
Answer: [1017kg. m-3]
Question 4. What is the difference in the structure’s nuclei?
Answer: Cl37 has 2 extra neutrons
Question 5. What is the relation between the atomic number (Z) and the mass number (A) of two isobars?
Answer: [Z is different, but A is the same]
Question 6. What is the difference in the properties of the two carbon isotopes C12 and C14, in the context of radioactivity?
Answer: C14 is radioactive, but C12 is not
Question 7. What is the approximate ratio of the penetrating power of rays α, β and ϒ
Answer: 1: 102:104
Question 8. What is the relation between the half-life and decay constant of a radioactive isotope?
Answer: \(\lambda=\frac{\ln 2}{T}\)
Question 9. When a β -particle is emitted from the radioactive isotope 15P32, it is converted into 16S32. Write down the required transformation equation.
Answer: \({ }_{15} \mathrm{P}^{32} \rightarrow{ }_{16} \mathrm{~S}^{32}+{ }_{-1} \beta^0\)
Question 10. When an α -particle is emitted from a uranium nucleus (atomic no, 92, mass number 238), a new nucleus is formed. From this nucleus β -particle is also emitted What will be the atomic number and mass number of the final nucleus?
Answer: 91, 234
Question 11. What are the atomic number and the mass number of the plutonium isotope produced due to two successive β – decays of the isotope 92PU239 of uranium
Answer: 94, 239
Question 12. Which fundamental particle was first discovered from artificial transmutation?
Answer: Neutron
Question 13. \({ }_1 \mathrm{H}^2+{ }_1 \mathrm{H}^3 \rightarrow{ }_2 \mathrm{He}^4+\) __________
Answer: 0n1
Question 14. Write down the decay scheme of a free neutron.
Answer: n→ p+e
Question 15. \({ }_1 \mathrm{H}^1+{ }_1 \mathrm{H}^1+{ }_1 \mathrm{H}^1+{ }_1 \mathrm{H}^1 \rightarrow{ }_2 \mathrm{He}^4+2\) ______________
Answer: +1β0
Question 16. Four nuclei of an element undergo fusion to form a heavier nucleus, with a release of energy. Which of the two the parent or the daughter nucleus would have higher binding energy per nucleon? The d
Answer:
Daughter nucleus in nuclear fusion would have higher binding energy per nucleon
Atomic Nucleus Assertion Type
Direction: These questions have statement 1 and statement 2. Of the four choices given below, choose the one that best describes the two statements.
- Statement 1 Is true, statement 2 Is true; statement 2 Is a correct explanation for statement 1
- Statement 1 Is true, statement 2 Is true; statement 2 Is not a correct explanation for statement 1
- Statement 1 Is true, and statement 2 Is false
- Statement I is false, statement 2 Is true
Question 1.
Statement 1: Negative charges are never emitted from the nucleus of an atom.
Statement 2: Nucleus of an atom is constituted only of protons and neutrons.
Answer: 4. Statement I is false, statement 2 Is true
Question 2.
Statement 1: The Mass of the O16 nucleus is less than the sum of masses of 8 protons and 8 neutrons.
Statement 2: Some internal energy is needed to keep the protons and neutrons bound in the nucleus.
Answer: 1. Statement 1 Is true, statement 2 Is true; statement 2 Is a correct explanation for statement 1
Question 3.
Statement 1: At any specific instant, if the number of atoms in two radioactive samples of radium-226 and polonium-210 is equal, then the activity of the radium sample will be less because the half-life of radium and that of polonium are 1600 y and 140 d respectively.
Statement 2: The activity of a radioactive sample is proportional to its decay constant.
Answer: 1. Statement 1 Is true, statement 2 Is true; statement 2 Is a correct explanation for statement 1
Question 4.
Statement 1: Some energy is released when a heavy nucleus disintegrates into two nuclei of moderate size
Statement 2: The more the mass number of the nucleus, the more is the binding energy for each proton of the neutron.
Answer: 3. Statement 1 Is true, statement 2 Is false
Question 5.
Statement 1: No natural radioisotope can emit positron.
Statement 2: Some artificially transmuted isotopes show radioactivity some of these may emit positrons.
Answer: 3. Statement 1 Is true, statement 2 Is false
Question 6.
Statement 1: The greater the decay constant of a radioactive element, the smaller its half-life.
Statement 2: An element, although radioactive, can last longer, if its decay with time is slow.
Answer: 2. Statement 1 Is true, and statement 2 Is true; statement 2 Is not a correct explanation for statement 1
WBCHSE physics class 12 nucleus notes
Atomic Nucleus Match The Columns
Question 1. Match column A with column B.
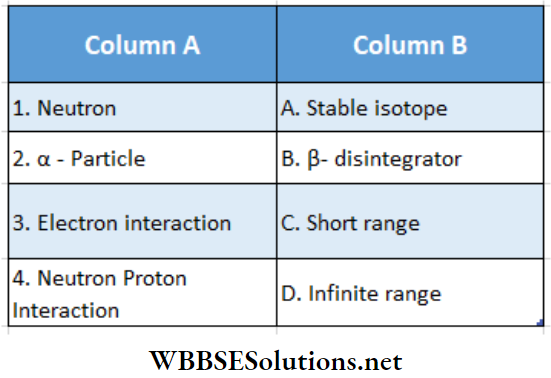
Answer: 1 – B, 2 – A, 3 – D, 4 -C
Question 2. The half-life of radium-226 is about 1600y. Match the columns for a sample rich in radium
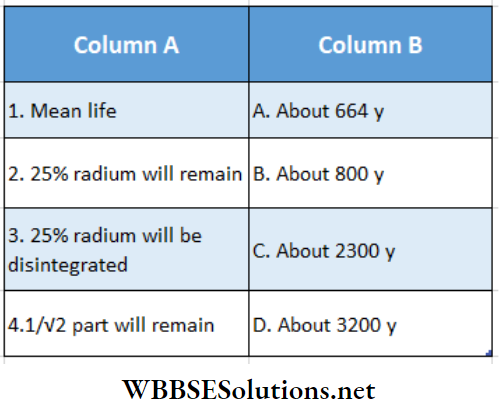
Answer: 1 – C, 2 – D, 3 – A, 4 – B




























































