Radiation And Properties
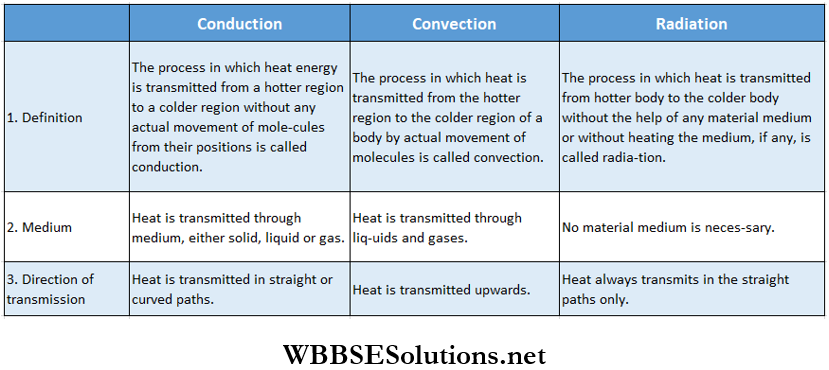
Heat can be transmitted from one place to another by radiation, even without the presence of any material medium. There is no material medium above the atmosphere, between the sun and the earth, but still heat from the sun reaches the earth.
- When we sit near an oven or a fireplace, we feel warm. In this case heat does not reach us by conduction. There is air between the oven and us and we know that air is an insulator.
- Also the air above the oven gets heated and rises upwards while cold air around the oven moves towards it. So heat does not reach us by convection either. Clearly, there is another process by which the heat reaches us. This process is called radiation.
Radiation: The process by which heat is transmitted from one region to another in the form of electromagnetic waves even in the absence of a material medium, or without heating the material medium (if present) is called radiation.
Radiant heat: In the process of radiation, heat energy is transmitted in all directions, from the source, in the form of waves. This heat wave is called thermal radiation or radiant heat.
Generally, all heated bodies spread heat in the form of radiant heat in all directions. During transmission, when the radiated heat gets absorbed by a body, the kinetic energy of its molecules increases. This means that the temperature of the body increases.
Read and Learn More: Class 11 Physics Notes
Similarities between radiant heat and light:
- Radiant heat is an electromagnetic wave like light and can travel in vaccum.
- Radiant heat, like light, travels in a straight line. That is why an umbrella saves our body from the heat of the sun.
- Radiant heat and light have the same speed. In a vacuum, this speed is 3 x 1010 cm • s-1.
- Radiant heat exhibits optical properties like reflection, refraction, interference, diffraction, and polarisation and affects photographic plates.
- Heat radiation obeys the inverse square law, i.e., the intensity at a point at a distance d from the source is inversely proportional to d², which is similar to light. From a source, both radiant heat and light propagate as waves.
- Radiant heat does not heat a medium while passing through it, but it heats bodies that obstruct its flow.

Differences between radiant heat and light:

Nature of Thermal Radiation: We have already seen that radiant heat and light have a lot of similarities. They are of the same nature and they belong to the same family of waves. These waves are known as electromagnetic waves.
- Only the wavelength of radiant heat is greater than that of visible light. In the spectrum of light, red light has the highest wavelength. As the wavelength of radiant heat more than that of red light, radiant heat is known as infrared waves. Radio waves, ultraviolet waves.
- X-rays, and gamma rays to are different kinds of electromagnetic waves. All of them have the same nature; the difference lies only in wavelengths.
- The table provided below lists the wavelength bands of electromagnetic waves with their names and ranges, in order of decreasing wavelength.
- All the electromagnetic waves mentioned above are called radiation although we have seen earlier that radiation generally refers to a specific process. In this context, it should be mentioned that the term radiation is used to mean emission too.

As infrared waves give us the sensation of heat, when they fall on our bodies., they are known as thermal radiation.
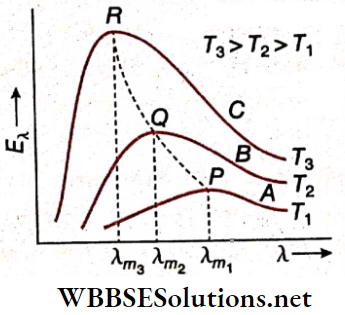
- Everybody, at any temperature above absolute zero (T = 0K), emits and absorbs radiation. It has been observed experimentally that the lower the temperature of a body, the higher is the wavelength of the radiation emitted by it and vice versa.
- This is why when a body is heated at first it emits infrared radiation. As the temperature rises, the emitted radiation falls in the visible light region. The body goes from being ‘red-hot’ to ‘white-hot’ as its temperature increases.
WBBSE Class 11 Radiation and Properties Notes
Prevost’s Theory of Heat Exchanges: Any hot substance radiates heat to as well as absorbs radiant heat from its surroundings at all times. Hence, there always exists a radiant heat exchange between a body and its surroundings.
Therefore, the rise or fall of temperature of a body depends on the exchange of heat between the body and its surroundings. This is called Prevost’s theory of heat exchange. According to this theory,
- when the rate of absorption of heat from the surroundings is greater than the rate of radiation of the body, the temperature of the body rises up.
- when the rate of absorption of heat from the surroundings is lower than the rate of radiation of the body, the temperature of the body decreases.
- when the rate of radiation equals the rate of absorption, the temperature of the body remains constant and the body remains at thermal equilibrium with its surroundings.
For example, when we stand near a fireplace, we feel hot. The radiant heat absorbed by our body from the fireplace is more than the heat that our body radiates hence the feeling.
Again, when we stand near a big chunk of ice we feel because the amount of radiant heat absorbed by our botljr from the ice is less than the heat that our body radiates.
Absorption of Radiation: When radiation is incident on a surface, it is, in general, divided into three parts:
1. Reflection from the surface: A part of the incident radiation is reflected. The reflective power or reflectance (r) is defined as,
r = \(\frac{\text { amount of reflected radiation }}{\text { amount of incident radiation }}\)
Normally, r ≤ 1. The value r = 1 refers to a perfectly white surface, commonly called a perfectly white body.
2. Transmission across the surface: A part of the incident radiation is transmitted across the surface. The transmitting power or transmittance (t) is defined as,
t = \(\frac{\text { amount of transmitted radiation }}{\text { amount of incident radiation }} \text {. }\)
Here also, t ≤ 1. For t = 1, the material of the surface is called a perfectly transparent medium, or a diather- manous substance. On the other hand, t = 0 corresponds to a perfectly opaque, or athermanous substance.
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
3. Absorption by the surface: A part of the incident radiation is absorbed by the surface. The absorptive power (a) of a surface is defined as,
a = \(\frac{\text { amount of radiation absorbed }}{\text { amount of incident radiation }}\)
Here again, a ≤ 1. A surface with a = 1 is called a perfectly black surface, or simply a perfectly black body. The value of a for lamp black and platinum black are 0.96 and 0.98, respectively.
All of the three quantities r, t, and a are ratios of two similar quantities (amounts of radiation). So, each of them is dimensionless and has no unit.
In fact, all of the three quantities r, t and a depend on the wavelength (λ) of radiation. So, rλ, tλ, and aλ denote the reflective power, transmitted power, and absorptive power of a surface at a certain temperature T and for a certain wavelength λ. For example,
1. A blue surface is a good reflector of blue radiation, a negligible part being transmitted or absorbed. So for blue radiation incident of such a surface, r is high, but t or a is low.
2. Glass is almost transparent for radiations of short wavelengths (temperature of source ~ 1200°C), like X-rays
or visible light So, t ≈ 1. But for longer wavelengths (temperature of source ~ 100°C), relevant to thermal radiations, glass behaves as an almost opaque or other- manous medium, so that t ≈ 0. This principle is utilized in greenhouses.
The temperature of a body increases due to radiation only when some part of the incident radiation is absorbed by its surface. Therefore, the absorptive power a turns out to be a very important property in case of thermal radiations.
Emission Of radiation: The rate of emission of radiation from a unit surface area of a body depends only on two properties of the body:
- Nature of the surface: The rate of emission depends on the colour of the surface, on whether the surface is rough or shiny, and on whether is it smooth or porous, etc. This dependence is described by Kirchhoff’s law.
- Temperature of the body: Higher the temperature of a body, more is the rate of emission. The rate falls to zero only when a body is at the temperature of absolute zero, i.e., at T = 0. This dependence is described by Stefan’s law.
Emissive power (e): The amount of radiation emitted from a unit surface area of a body in unit time is called the emissive power of that particular surface kept at a particular temperature.
In general, this emissive power is different for different colours of radiation, i.e., for different wavelengths (λ).
If eλ denotes the emissive power of a surface for a wavelength λ, then the amount of radiation emitted per unit area of the surface per second between the wavelengths λ and λ + dλ will be eλdλ.
Total emissive power of the surface for all possible wavelengths is, e = \(\int_0^{\infty} e_\lambda d \lambda\)
The units of emissive power are cal • cm-2 • s-1 in CGS system and J • M-2 • s-1 in SI.
1 \(\mathrm{~J} \cdot \mathrm{m}^{-2} \cdot \mathrm{s}^{-1} =\frac{1 \mathrm{~J}}{1 \mathrm{~m}^2 \times 1 \mathrm{~s}}=\frac{\frac{1}{4.2} \mathrm{cal}}{10^4 \mathrm{~cm}^2 \times 1 \mathrm{~s}}\)
= \(\frac{1}{42000} \mathrm{cal} \cdot \mathrm{cm}^{-2} \cdot \mathrm{s}^{-1}\)
A perfectly black surface is the best emitter of radiation at a particular temperature. In this context, the emission from any surface is sometimes compared with that from a perfectly black surface.
A new quantity called relative emittance or emissivity or coefficient of emission (ε) of a surface is then defined as,
ε = \(\frac{\text { heat radiated from the surface }}{\begin{array}{c}
\text { heat radiated from the same area of a perfectly } \\
\text { black surface at the same temperature } \\
\text { in an equal interval of time }
\end{array}}\)
= \(\frac{e}{E}\), where E = emissive power of a perfectly black
Being a ratio between two amounts of heat, ε is dimensionless and has no unit. Of course, ε ≤ 1, the equality sign stands for a perfectly black surface.
Understanding Heat Radiation in Physics
Black body: A body that absorbs radiations of all wavelengths falling on it, without reflecting or transmitting any of it, is called an ideal black body. As it is an ideal absorber it is an ideal emitter as well.
- At a particular temperature, energy radiated by a black body is greater than that radiated by any other substance. Hence, radiation emitted by an ideal black body is called total radiation or black body radiation.
- In reality there is no ideal black body, though lamp black and platinum black is considered to be nearly ideal. Lamp black absorbs about 96% and platinum black absorbs about 98% of the radiation falling on them.
Ferry’s black body: A hollow double-walled metal sphere kept at a steady temperature with a fine hole behaves like an ideal black body. It is blackened inside with lamp black and nickel-polished on the outside.
- The space between the two walls does not have any air. So heat is not lost due to conduction and convection. Heat radiation entering the sphere through the small opening O gets completely absorbed inside the sphere after a few successive reflections.
- The projection P, opposite to the opening O prevents normal reflection of the radiation. So no radiation entering the sphere through the opening can be out of the sphere. This means that the total radiation gets absorbed. Therefore the sphere acts as an ideal black body This is known as Ferry’s blackbody.
- Thermal radiation comes out of the opening when the body is heated. In fact the opening O acts as a black body and the intensity and nature of radiation depends only on the temperature of the body. Hence, this radiation is also called temperature radiation.
- In fact, any hollow enclosure with a small opening acts as a nearly ideal black body. Radiant heat entering it has a negligible chance of coming out through the small opening.

Good absorbers are good emitters so these hollow enclosures emit intense black body radiations at high temperatures. In a coal furnace, the narrow gaps between the burning pieces of coal appear to be brighter than any piece of burning coal itself.
Kirchhoff’s Law: The relation between emissive power and absorptive power of a substance is expressed by Kirchhoff’s law. It states that the ratio between the emissive power and the absorptive power of any substance at a fixed temperature is equal to the emissive power of an ideal black body at that temperature which is constant.
Let at a fixed temperature T, the emissive power of a substance be e, its absorptive power be a, and the emissive power of an ideal black body at that temperature be E. Then, by Kirchhoff’s law, \(\frac{e}{a}=E\)…(1)
Conclusions drawn from Kirchhoff’s law:
- The value of the ratio \(\frac{e}{a}\) increases with the increase in temperature as E increases with the rise in temperature for a fixed wavelength.
- When T is kept constant, E is also a constant for a particular wavelength of radiation. This means, if e is large, a is also large. So a good emitter is also a good absorber and vice versa for the same wavelength.
- Relative emittance or coefficient of emission, \(\varepsilon=\frac{e}{E}\) from Kirchhoff’s law, ε is equal to a, the absorptive power.
Conclusions drawn from Kirchhoff’s law Applications:
- A white china bowl is partially smeared with lamp black. Now, the bowl is heated to 1000°C. If we take this bowl to a dark room then the black coloured parts appear brighter than the rest. The black part is a better absorber than the white part. So, from Kirchhoff’s law, it is a good emitter as well. Thus, it appears brighter as emits more heat.
- In a solar spectrum, we see many black lines. These are called Fraunhofer lines. The origin of these lines can be explained with the help of Kirchhoff’s law.
Examples of Emission and Absorption of Heat
- Cups and saucers, mugs, etc. which are used to serve hot beverages like tea and coffee are usually white and shiny. This helps to keep the beverage hot for a long period of time as heat loss due to radiation is very small.
- Dark or black-colored wet clothes dry up faster in sun than white wet clothes do. This happens because black clothes absorb the heat from the sun at a higher rate.
- It is comfortable to wear dark clothes in winter and white clothes in summer. Dark clothes keep our bodies warm by absorbing more heat emitted by the sun. During the summer, the white clothes absorb a very small fraction of the heat and reflect most of it. So our bodies remain cool.
- The cloth of an umbrella is usually made black. Being a good absorber, it collects the sun rays and also radiates out the heat fast. The radiated heat does not heat up the air below the umbrella and hence keeps the body cool.
- Let us take two thermometers and blacken the bulb of one of them. If we keep these thermometers in the sun, after a while the temperature of the blackened thermometer will read more than the other one. This hap¬pens because the blackened bulb absorbs more heat from the sun.
- Humid air is a better absorber of heat than dry air. On a cloudy and humid day, air absorbs more heat from the sun and becomes warmer. During night, the surface of the earth cools by radiating heat. But humid air is rather- manous to heat and it prevents the passage of heat to outer space. Thus, humid and cloudy days and nights are warmer than clear ones.
- It is difficult to stay in houses with tin roofs during summer afternoons because tiniabsorbs heat. But at night, tin radiates the heat rapidly and cools. So the room becomes cold.
- Outer surface of a spacecraft is made smooth and shiny to reflect off radiated heat from the sun so that the spacecraft cannot get much heated.
- In coal ovens, spaces between the glowing pieces of coal appear brighter than the glowing coal pieces themselves. Any hole behaves like an ideal black body. Hence, absorptive power, as well as emissive power of the spaces, is very high. So the spaces appear brighter.
- Bottom surface of a cooking container is made black and rough so as to absorb more heat. Hence food is cooked faster in these containers than those with white and polished bottom.
- Highly polished shoes are more comfortable to wear since they reflect most of the incident heat and absorb very little of it.
- In deserts, days are unbearably hot and nights are cold. Air in those regions being very dry, is diathermanous. During the daytime, this dry air allows heat to easily pass through the atmosphere and heat the surface. At night, the earth’s surface radiates heat which easily escapes the atmosphere leaving the surface and the air around it cold. This is why extreme temperature changes are observed in deserts.
Greenhouse: In cold countries glass-houses and sometimes garden-houses with glass roofs are constructed. These are used to preserve plants and vegetables from withering away due to the low temperatures and so are called greenhouses. Glass is diathermanous to short waves.
- Sun rays, because of its high temperature, are rich in shorter waves and can enter through the roof. Materials in the shade absorb the radiation and warm up. The radiations from these preserved materials mostly consist of long waves like heat waves as they are at quite low temperatures.
- Glass being athermanous to long waves, does not allow the radiation to escape and the interior of the house remains warm throughout the year.
Greenhouse effect: Due to the presence of certain gasses in the earth’s atmosphere, the earth acts as a huge greenhouse. These gases are present in trace amounts and are known as greenhouse gases.
- Water-vapour, carbon dioxide- methane, nitrous oxide, dhloroflurocarbons) etc. are some important greenhouse gases. These gases form a layer in the atmosphere which is diathermanous to short heat waves, but athermanous to long waves, So, the sun rays, being of short wavelength can enter the earth’s atmosphere, However, the heat radiated from the earth’s surface.
- Is of longer wavelength and cannot pass through the gaseous layer, Ihls warms up the air and the earth’s surface. This Is called the greenhouse effect. In the absence of this effect the average temperature of the earth’s surface would have been lower by 30 °C-35 °C approximately. The atmosphere would then be adverse for life.
Enhanced greenhouse effect: Due to human activities, the amount of greenhouse gases In air Is Increasing alarmingly. This results in the increase In air and earth’s temperature regularly. This is called enhanced greenhouse effect. If this enhanced greenhouse effect is not controlled, the existence of life on earth will be in danger In the future.
Harmful effects of global warming:
- There will be an acute shortage of water for consumption and for use In agricultural and electricity production. There will also be degradation in the quality of drinking water,
- Due to the melting of ice on mountains and polar regions sea level will rise causing large areas on the earth’s surface to submerse under water.
- Death rate of plants and animals will increase.
- Forest resources will show signs of destruction.
Stefan-Boltzmann Law Explained
Stefan’s Law: After careful analysis of the results of a few experiments by scientists Dulong, Petit and Tyndall, famous physicist Joseph Stefan formulated the relation connecting the amount of radiation emitted by a body and the temperature of the body.
This law is known as Stefan’s law. The law states that, the total heat radiated (E) per second from per unit surface area of a body at an absolute temperature T is directly proportional to the fourth power of the absolute temperature of the body.
i.e., \(E \propto T^4\) or \(E=\sigma \cdot T^4\)
Here σ is called Stefan’s constant, whose experimental value is
σ = \(5.672 \times 10^{-5} \mathrm{erg} \cdot \mathrm{cm}^{-2} \cdot \mathrm{s}^{-1} \cdot \mathrm{K}^{-4}\)
= \(5.672 \times 10^{-8} \mathrm{~W} \cdot \mathrm{m}^{-2} \cdot \mathrm{K}^{-4} .\)
Clearly, for T= 0, E = 0 which means that a body at T = 0 K does not emit thermal radiation.
Later in 1884 Boltzmann theoretically proved the law from thermodynamical considerations and established that the law is valid only for black body radiation.
The law is, thus, also known as Stefan-Boltzmann law. It states that the total radiant energy emitted per second per unit surface area of a perfect black body is directly proportional to the fourth power of its absolute temperature.
ie., E ∝ T4 or, E = σT4
If for any surface, the relative emittance = £ and the surface are = A, then die heat radiated in time t is, Q = σεAtT4
The law is sometimes called Stefan’s fourth power law.
- It should be noted diat die amount of heat radiated by a body due to its temperature can be obtained from Stefan’s law. But the law does not refer to the net loss or gain of heat by radiation due to exchange with its surroundings. It should be modified in that case.
- As the black body at absolute temperature T radiates heat, it absorbs heat as well from its surroundings at absolute temperature T0. Hence, by Prevost’s tiieory of heat exchange, the net rate of heat radiation by a unit surface area of a black body is equal to the difference in die rates of emission and absorption.
i.e., \(E=\sigma\left(T^4-T_0^4\right)\)
If the body is not a black body, then radiation per second per unit area, E = ∈σ = \(\epsilon \sigma\left(T^4-T_0^4\right)\), where ∈ is the relative emittance of the die surface.
Solar Constant and Solar Temperature: Surface temperature of the sun can be determined from the knowledge of the solar constant and Stefan’s law.
Solar constant: It is defined as the amount of radiant energy received per minute per unit area by a perfectly black body placed on earth at die mean distance of the earth from the sun in a direction perpendicular to the direction of rays from the sun.
Its value is nearly 1300 J • m-2 • s-1, i.e., 1300 W • m-2
Determination of solar temperature: The central region of the sun is very hot. This region is called the photosphere which is surrounded by a comparatively cooler atmosphere called the chromosphere. For all external purposes, fire temperature of the sun means the temperature of the chromosphere.
Let, r be the radius and TK be the temperature of the sun. To determine the solar temperature, the sun is treated as a perfectly black body. According to Stefan’s Jaw, the energy emitted by the sun in t seconds E = \(4 \pi r^2 \sigma T^4 t\)
Imagine a sphere of radius R drawn around the sun. R is the mean distance of the earth from the sun. The surface area of the sphere = 4πR²,
Therefore, the energy incident normally per unit area of the earth’s surface in time
t = \(\frac{E}{4 \pi R^2}=\frac{4 \pi r^2 \sigma T^4}{4 \pi R^2}=\frac{r^2 \sigma T^4 t}{R^2}\)

If t=60s then this quantity is nothing but the solar constant S.
∴ S = \(\frac{r^2 \sigma T^4 \times 60}{R^2} \text { or, } T^4=\left(\frac{R}{r}\right)^2 \times \frac{S}{\sigma} \times \frac{1}{60}\)
or, T = \(\left[\left(\frac{R}{r}\right)^2 \times \frac{S}{\sigma} \times \frac{1}{60}\right]^{1 / 4}\)….(1)
From the equation (1), the solar temperature can be determined.
Here, S = \(0.032 \mathrm{cal} \cdot \mathrm{cm}^{-2} \cdot \mathrm{s}^{-1} \text { (approx.) }\)
σ = \(1.37 \times 10^{-12} \mathrm{cal} \cdot \mathrm{cm}^{-2} \cdot \mathrm{s}^{-1}\)
r = \(7 \times 10^{10} \mathrm{~cm}\)
R = \(15 \times 10^{12} \mathrm{~cm}\)
Putting these values in equation (1) the solar temperature (T) becomes 5722 K (approx.).
Wien’s Displacement Law Overview
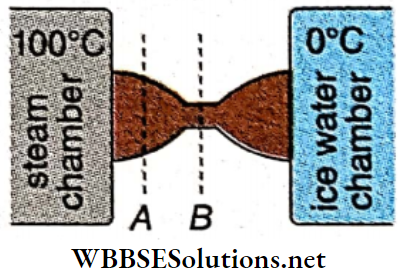
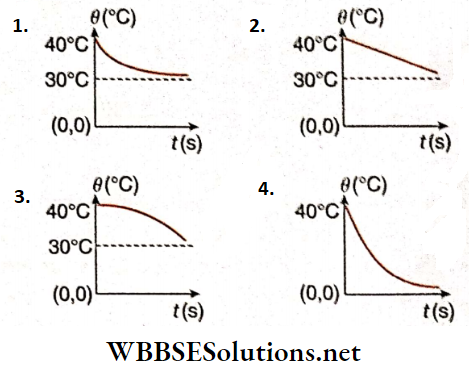
Newton’s Law of Cooling Statement: The rate of loss of heat by a body, due to radiation, is direedy proportional to the temperature difference between the body and its surroundings, provided the temperature difference is small.
Time of cooling: Let a body, of mass m and specific heat s, at a temperature θ be placed in a surrounding of temperature θ0, where θ0 < θ.
According to Newton’s law of cooling, the rate of loss of heat by the body, \(\frac{d Q}{d t} \propto-\left(\theta-\theta_0\right)\)
Also from calorimetry, \(\frac{d Q}{d t}=m s \frac{d \theta^{I R}}{d t}, \text { where } \frac{d \theta}{d t}\) = rate of fall of temperature.
∴ \(\frac{d \theta}{d t} \propto-\left(\theta-\theta_0\right) \quad \text { or, } \frac{d \theta}{d t}=-C\left(\theta-\theta_0\right)\)
where C is 2 constant and the —vs sign indicates fell in temperature with time.
∴ \(\frac{d \theta}{\theta-\theta_0}=-C d t\)
By integration, \(\int_{\theta_1}^{\theta_2} \frac{d \theta}{\theta-\theta_0}=-C \int_0^t d t\),
At the time of cooling the temperature of the body decreases from θ1 to θ2 in time t, where θ1 > θ2]
or, \(\left[\log _e\left(\theta-\theta_0\right)\right]_{\theta_1}^{\theta_2}=-C t\)
or, \(\log _e\left[\frac{\theta_1-\theta_0}{\theta_2-\theta_0}\right]\) = Ct
or, \(t=\frac{1}{C} \log _{\ell}\left[\frac{\theta_1-\theta_0}{\theta_2-\theta_0}\right]\)……(1)
Newton’s few of cooing from Stefan-Bottzmonn few: When the difference in temperature between the body and its surroundings is low, Stefan-Boltzmann’s few becomes equal to Newton’s law of cooling. Let the absolute temperature of die body and That of the surroundings be T and T0 respectively (T— T0) is very small.
From Stefan’s law, heat radiation per unit area per unit time,
E = \(\sigma\left(T^4-T_0^4\right)=\sigma\left(T-T_0\right)\left(T^3 \div T^2 T_0 \div T T_0^2 \div T_0^3\right)\)
If (T- T0) is very small, ie., if T ∝ T0, we can write the equation as,
E = \(\sigma\left(T-T_0\right) \times 4 T_0^3=C\left(T-T_0\right) \text {, where } C=4 \sigma T_0^3\)
At a constant surrounding temperature, C Is a constant
Hence E ∝ (T – T0), which is Newton’s law of cooling.
The body will cool by radiation following Newton’s laws of moving only when the temperature difference between the body and its surroundings is small.
Newton’s law of cooling is valid for cooling by conduction when k, A, and d are constants. It is also valid for some cases of cooling by convection.
Transmission Of Heat Radiation And Properties Numerical Example
Example 1. The initial temperature of a body is 353 K. It reduces to 337 K in 5 mins and to 325 K In 10 mins. What will be its temperature after 15 mins? What is the temperature of the surroundings?
Solution:
From Newton’s law of cooling we have,
In\(\frac{\theta_1-\theta_0}{\theta_2-\theta_0}=Ct\)
After 5 mins, \(\ln \frac{353-\theta_0}{337-\theta_0}=5 C\)…(1)
After the next 5 mins, \(\ln \frac{337-\theta_0}{325-\theta_0}=5 C\)….(2)
Yet after another \(5 \mathrm{mins}, \ln \frac{325-\theta_0}{\theta_3-\theta_0}=5 \mathrm{C}\)…(3)
[where θ3 is the temperature of the body after 15 mins]
From equations (1) and (2) we have, \(\frac{353-\theta_0}{337-\theta_0}=\frac{337-\theta_0}{\theta_3-\theta_0} \text { or, } \theta_0=289 \mathrm{~K}\)
∴ Temperature of the surroundings =289K.
From equations (2) and (3) we have, \(\frac{337-\theta_0}{325-\theta_0}=\frac{325-\theta_0}{\theta_3-\theta_0} \)
or, \(\frac{337-289}{325-289}=\frac{325-289}{\theta_3-289} \quad \text { or, } \theta_3=316 \mathrm{~K}\)
Applications of Thermal Radiation
Example 2. The temperature of a black body is increased from 27°C to 927°C. What is the ratio of the heat radiated at these two temperatures?
Solution:
⇒ \(T_1=27^{\circ} \mathrm{C}=(27+273) \mathrm{K}=300 \mathrm{~K}\)
⇒ \(T_2=927^{\circ} \mathrm{C}=(927+273) \mathrm{K}=1200 \mathrm{~K}\)
According to Stefan’s law, \(E \propto T^4\)
or, \(\frac{E_1}{E_2}=\left(\frac{T_1}{T_2}\right)^4=\left(\frac{300}{1200}\right)^4=\left(\frac{1}{4}\right)^4=\frac{1}{256} .\)
Example 3. A spherical black body of radius 12 cm radiates 450 W heat, energy at 500 K. Now If the radius is reduced to half the temperature doubled, what will be the amount of heat radiated?
Solution:
The surface area of a sphere, \(A=\pi r^2, i.e., A \propto r^2\)
Rate of radiation from the surface of a black body, \(E \propto A T^4 \quad \text { or, } E \propto r^2 T^4 \quad\left[because A \propto r^2\right]\)
∴ \(\frac{E_1}{E_2}=\left(\frac{r_1}{r_2}\right)^2\left(\frac{T_1}{T_2}\right)^4\)
or, \(E_2=E_1\times\left(\frac{r_2}{r_1}\right)^2\left(\frac{T_2}{T_1}\right)^4=450 \times\left(\frac{1}{2}\right)^2 \times\left(\frac{2}{1}\right)^4\)
= 1800 W
Short Answer Questions on Heat Radiation
Example 4. The filament of on electric bulb of 40 W has an average temperature of 2500 °C. If the length and diameter of the filament are 10 cm and 0.1mm respectively, then determine Its coefficient of radiation (ε), provided all the heat Is emitted only through radiation. Given, σ = 5.67 x 10-5 CGS unit
Solution:
We know, \(\frac{Q}{t}=\epsilon A \sigma T^4\)
Here, \(\frac{Q}{t}\) = radiation per second = 40 W = 40 x 107 erg · s-1,
A = πld = π x 10 x 0.01 = 0.1 π cm²
σ = 5.67 x 10-5 CGS unit,
T = 2500 + 273 = 2773 K.
∴ 40 x 107 = ∈ x 0.1 π x 5.67 x 10-5 x (2773)4
or, ∈ = 0.38 cal °C-2 cm-2.
Example 5. With a body temperature of 37° C, a person of a body surface area of 1.40 m² and a coefficient of emission of 0.85, stands In a room kept at a temperature of 20°C. If Stefan’s constant, σ = 5.67 x 10-8 W • m-2 • K-4 find the rate of loss of heat by the man by radiation only.
Solution:
Heat lost by the man per min is given by,
Q = \(\epsilon A \sigma\left(T^4-T_0^4\right) \times t\)
= \(0.85 \times 1.40 \times 5.67 \times 10^{-8}\left[310^4-293^4\right] \times 60\)
= \(7550 \mathrm{~J}=1800 \mathrm{cal} \text { (approx). }\)
Emissivity and Its Importance
Example 6. The earth receives radiation from the sun at the rate of 1400 W • m-2. The distance of the centre of the sun from the surface of the earth is 1.5 x 1011 m and the radius of the sun is 7 x 108 m. Treating the sun as a black body, determine Its surface temperature in Kelvin scale, σ = 5.67 x 10-8 W • m-2 · K-4
Solution:
If T be the surface temperature of the sun, then
T = \(\left[\left(\frac{R}{r}\right)^2 \times \frac{S}{\sigma}\right]^{1 / 4}\)
Here, \(S=1400 \mathrm{~W} \cdot \mathrm{m}^{-2}, R=1.5 \times 10^{11} \mathrm{~m}, r=7 \times 10^8 \mathrm{~m}, \sigma=5.67 \times 10^{-8} \mathrm{~W} \cdot \mathrm{m}^{-2} \cdot \mathrm{K}^{-4}\)
∴ T = \(\left[\frac{\left(1.5 \times 10^{11}\right)^2}{\left(7 \times 10^8\right)^2} \times \frac{1400}{5.67 \times 10^{-8}}\right]^{1 / 4}\)
= \(\left[0.1128 \times 10^{16}\right]^{1 / 4}=5800 \mathrm{~K}\)