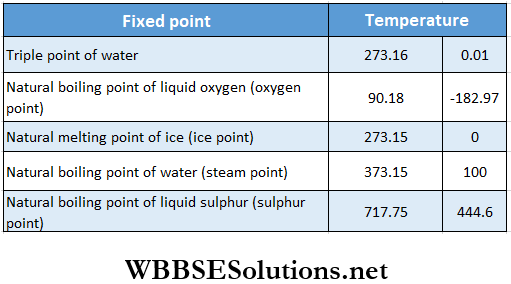
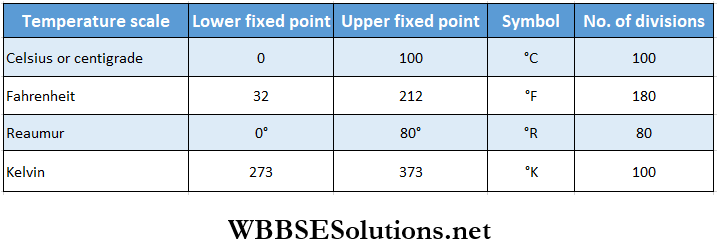
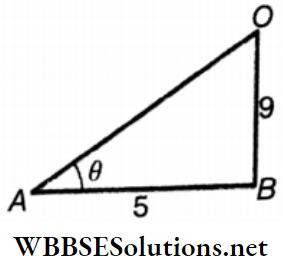
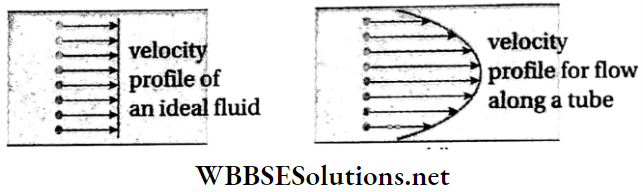
Viscosity And Surface Tension Rate Of Flow Of A Liquid And Continuity
Rate of flow Of liquid: For streamline flow of a perfectly incompressible liquid, the amount of liquid flowing through any cross section of a tube in a given time interval remains constant.
- The rate of flow of a liquid through a tube means the volume of liquid flowing through any cross-section of the tube per second.
- Suppose a liquid flows through a tube of cross-sectional area a with a uniform velocity ν. The volume of liquid flowing through any cross-section of the tube per second is equal to the volume of a cylinder of length v and cross-sectional area α.
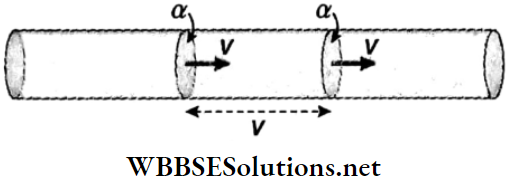
∴ Volume of liquid flowing per second = the rate of flow of the liquid = velocity of flow x area of cross section of the tube = νa
Bernoulli’s Principle Formula
Therefore, the mass of liquid flowing per second = velocity of flow x area of cross-section of the tube x density of the liquid = ναρ [ρ = density of the liquid]
Continuity of flow: For a streamlined flow of a fluid (liquid or gas) through a tube, the mass of the fluid flowing per second through any cross-section of the tube remains constant. This is known as the continuity of flow.

Read and Learn More: Class 11 Physics Notes
Equation Of continuity: Let us consider two sections A and B of a tube having cross-sectional areas a1 and a2 respectively. The velocities of the fluid at sections A and B are ν1 and ν2, and its densities are ρ1 and ρ2 respectively.
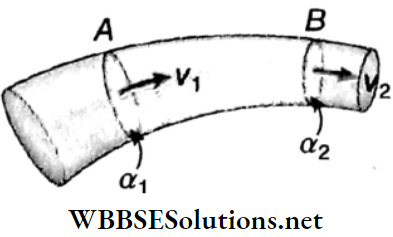
The mass of fluid flowing through section A per second = \(v_1 \alpha_1 \rho_1\) and the mass of fluid flowing through section B per second = \(v_2 \alpha_2 \rho_2\)
For streamline flow, the fluid enters through section A and leaves through section B, and does not remain stored in the region between A and B, hence
⇒ \(v_1 \alpha_1 \rho_1\) = \(v_2 \alpha_2 \rho_2\) …..(1)
The product vαρ is the mass flow rate. If the fluid is incompressible (like a liquid), then its density is constant, and in that case ρ1 = ρ2.
∴ \(v_1 \alpha_1\) = \(v_2 \alpha_2\)…….(2)
or, να = constant …..(3)
Equations (2) and (3) are known as the equations of continuity of liquid flow.
∴ \(\nu \propto \frac{1}{\alpha},\) which means that the velocity of liquid flow through any cross-section of a tube is inversely proportional to its cross-sectional area.
The equations of continuity essentially express the law of conservation of mass.
Bernoulli’s Principle Formula
Energy of Liquid in Streamline Flow: At any point inside a flowing liquid, there are three forms of energy
- Kinetic energy,
- Potential energy and
- Energy due to pressure.
1. Kinetic energy: If mass m of a liquid flows with a velocity v, then the kinetic energy of that liquid = = \(\frac{1}{2} m v^2\).
Kinetic energy per unit mass = \(\frac{1}{2}v^2\)
Kinetic energy per unit volume = \(\frac{1}{2} \frac{m}{V} v^2\) [volume of the liquid]
= \(\frac{1}{2} \rho v^2\left[\rho=\frac{m}{V}=\text { density of the liquid }\right]\)
2. Potential energy: If mass m of a liquid is at a height h above the surface of the earth, then the potential energy of that liquid = mgh.
Potential energy per unit mass = gh
Potential energy per unit volume = \(\frac{m g h}{V}\) = ρgh.
Bernoulli’s Principle Formula
3. Energy due to pressure: if a liquid is under the action of some applied pressure, then it acquires some energy and this energy is known as energy due to pressure. The liquid can perform work by expending this energy. Let some liquid of density ρ whose free surface is PQ be kept in a container.
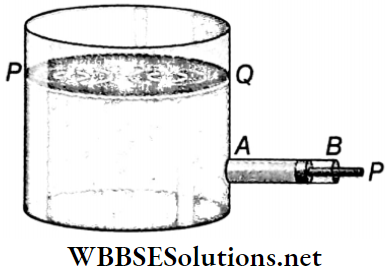
A narrow side tube AB of cross sectional area α is attached near the bottom of the container. This tube is fitted with a piston P, which can move freely along the tube. If the pressure of the liquid at rest along the axis of the narrow tube is p, then the force acting on the piston = pα.
If the piston is slowly pushed inside the tube through a distance x, then work done = pαx. As a result, liquid of volume αx or mass αxρ enters the container. Since the piston is moved slowly, the liquid acquires negligible velocity and hence it will possess no kinetic energy.
So the work done pax remains stored as potential energy in mass αxρ of the liquid that has entered the container. This energy is called the energy due to pressure for the liquid.
The energy due to pressure per unit mass of the liquid = = \(\frac{p a x}{a x \rho}=\frac{p}{\rho} .\)
∴ The energy due to pressure per unit volume of the liquid = \(\frac{p a x}{a x}=p\).
Bernoulli’s Theorem: The Swiss mathematician Daniel Bernoulli established a law for the streamline flow of an ideal fluid (which is incompressible and non-viscous). This law is known as Bernoulli’s theorem. It is an important theorem in Hydrodynamics.
Bernoulli’s Principle Formula
Statement Of the theorem: For a streamline flow of an ideal liquid, the sum of the potential energy, kinetic energy, and energy due to pressure per unit volume of the liquid always remains constant at every point on the I streamline.
If the kinetic energy per unit volume of the liquid = \(\frac{1}{2} \rho v^2\); potential energy = pgh and energy due to pressure = p, then
⇒ \(\frac{1}{2} \rho v^2+\rho g h+p\) = constant
or, \(\frac{1}{2} v^2+g h+\frac{p}{\rho}\) = constant …..(1)
This is the mathematical form of Bernoulli’s theorem. Dividing equation (1) by g, we get,
⇒ \(\frac{v^2}{2 g}+h+\frac{p}{\rho g}\) = constant……..(2)
This also is a form of Bernoulli’s theorem. Here, \(\frac{v^2}{2 g}\) is called the velocity head, h is the elevation head, and \(\frac{p}{\rho g}\) is the pressure head. Each of these heads has the dimension of length.
So, velocity head + elevation head + pressure head
= constant …..(3)
According to relation (3), Bernoulli’s theorem can also be stated as follows.
For a streamline flow of an ideal liquid, the sum of the velocity head, elevation head and pressure head always remains constant at any point in the liquid.
Bernoulli’s theorem is based on the law of conservation of energy for the streamline motion of an ideal fluid. The theorem states that energy remains conserved along any streamline.
Bernoulli’s Theorem Derivation
When the flow of liquid is horizontal, the height of each point in the liquid is assumed to be the same, i.e., h = constant. We can rewrite equation (2) as,
⇒ \(\frac{v^2}{2 g}+\frac{p}{\rho g}=\) constant
or, \(p+\frac{1}{\mathrm{a}} \rho v^2\) = constant
Hence, in the horizontal flow of a liquid, the sum of pressure and kinetic energy per unit volume of the liquid at any point is constant. This implies that where the velocity of flow is high, the pressure is low and vice-versa.
Applications of Bernoulli’s theorem
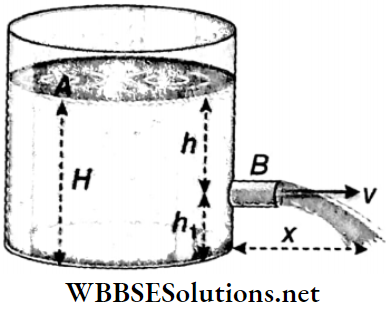
1. Velocity of efflux of a liquid and Torricelli’s theorem: If a small hole is present on the wall of a deep container containing liquid, then the velocity with which the liquid comes out through that small hole is called the velocity of efflux of the liquid.
In Fig, a liquid kept in a large container is emerging with velocity ν through the small hole on the wall of the container. The height of the free surface of the liquid above the hole is h and the depth of the liquid below the hole is h1.
The total depth of the liquid H = h+ h1. Let us consider a point B just outside the hole and another point A on the surface of the liquid. Atmospheric pressure p acts on A and B.
If the container is large and the hole is very small, then the free surface of the liquid will come down so slowly that the velocity of the free surface of the liquid would seem to be almost zero.
Bernoulli’s Theorem Derivation
If we imagine a tube of flow starting from the free surface of the liquid and ending at the point B and apply Bernoulli’s theorem in that tube of flow, then
⇒ \(0+H+\frac{p}{\rho g}=\frac{v^2}{2 g}+h_1+\frac{p}{\rho g}\)
or,\(\frac{v^2}{2 g}=H-h_1=h\)
or, \(v^2=2 g h\)
or, \(v=\sqrt{2 g h}\)
- This is the velocity of efflux of a liquid through a small hole and it is known as Torricelli’s formula. According to this formula, the velocity of efflux of a liquid is the same as that of a body falling freely under gravity through a height h. So, Torricelli’s theorem can be stated as follows:
- The velocity of efflux of a1 liquid through any small hole or orifice is equal to that acquired by a body falling freely from rest under gravity from the free surface of the liquid to the level of the small hole.
- It should be mentioned that this ideal velocity cannot be attained by any liquid in reality because no liquid is non- viscous. It should be remembered that in Bernoulli’s theorem the effect of viscosity of the liquid has been neglected.
Horizontal range: Let, the first drop of liquid emerged from the orifice touches the ground at a distance x after time t. That means, the vertical displacement of the liquid drop is h1.
Now, considering the motion of the liquid along the vertical direction,
initial velocity = 0, acceleration = g
Bernoulli’s Theorem Derivation
From the equation h = ut+1/2gt² we get, h = 0 + 1/2gt²
∴ t = \(\sqrt{\frac{2 h_1}{g}}\)
Again, considering the motion of the liquid aong the horizontal direction, the initial velocity, ν = √2gh, acceleration = 0, time = t.
∴ Horizontal range, \(x=v t=\sqrt{2 g h} \times \sqrt{\frac{2 h_1}{g}}=2 \sqrt{h h_1}\)
2. Venturimeter: A venturimeter is used to measure the rate of flow of liquid through a tube. Its working principle is based on Bernoulli’s theorem.
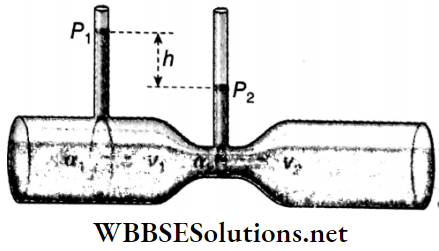
- Fig shows the action of a venturimeter. The two ends of this tube are equally wide and the middle portion is narrow. Liquid flows through this tube in streamlines. The tube is kept horizontal.
- When a liquid flows through a venturimeter, the velocity of the liquid increases at the narrow part of the tube with consequent decrease in pressure. This decrease in pressure is measured with the help of two vertical tubes attached at the wide and the narrow parts of the venturimeter.
Let the velocity of the liquid at the wider part of the tube be ν1 and the pressure be p1 At the narrower part of the tube, the velocity of the liquid is ν1 and the pressure is p2.
According to Bernoulli’s theorem, \(\frac{v_1^2}{2 g}+\frac{p_1}{\rho g}=\frac{v_2^2}{2 g}+\frac{p_2}{\rho g}\)
the elevation head, h1=h2 since the tube is horizontal.
∴ \(\frac{p_1-p_2}{\rho g}=\frac{1}{2 g}\left(v_2^2-v_1^2\right) \text { or, } p_1-p_2=\frac{\rho}{2}\left(v_2^2-v_1^2\right)\)
or, \(h \rho g=\frac{\rho}{2}\left(v_2^2-v_1^2\right)\)
[h = difference in liquid levels in the vertical tubes attached to the venturimeter]
∴ h = \(\frac{1}{2 g}\left(v_2^2-v_1^2\right)\)
If the cross-sectional areas of the wide and the narrow parts of the venturimeter are α1 and α2 respectively, then according to the equation of continuity, we get,
⇒ \(\alpha_1 v_1=\alpha_2 v_2 \text { or, } \frac{v_1}{v_2}=\frac{\alpha_2}{\alpha_1}\)
∴ h = \(\frac{v_2^2}{2 g}\left(1-\frac{v_1^2}{v_2^2}\right)=\frac{v_2^2}{2 g}\left(1-\frac{\alpha_2^2}{\alpha_1^2}\right)\)
or, \(v_2^2=2 g h \cdot \frac{\alpha_1^2}{\alpha_1^2-\alpha_2^2}\)
or, \(v_2=\frac{\alpha_1}{\sqrt{\alpha_1^2-\alpha_2^2}} \cdot \sqrt{2 g h}\) …..(1)
Therefore, the volume of liquid flowing out per second,
Venturimeter Derivation Class 11
V = \(\alpha_2 v_2=\frac{\alpha_1 \alpha_2 \sqrt{2 g h}}{\sqrt{\alpha_1^2-\alpha_2^2}}=\alpha_1 \alpha_2 \sqrt{\frac{2 g h}{\alpha_1^2-\alpha_2^2}}\)…….(2)
So, when α1 and α2 are known, by measuring h we can determine the rate of flow of the liquid through the tube with the help of equation (2).
3. Pitot tube: A pitot tube is also used to measure the rate of flow of liquids. Its working principle is similar to that of a venturimeter. Its action also depends on Bernoulli’s theorem.
- In this instrument, two tubes AB and CED, open at both ends are introduced vertically and side by side inside the liquid. The open end B of the tube AB remains parallel to the flow of the liquid. The DE part of the tube CED is so bent that the opening D faces the flowing liquid normally.
- The height of the liquid column in the tube AB expresses the pressure of the liquid at the point B. Since the liquid flow is obstructed at the portion DE of the tube CED, the velocity of flow at point D is zero.
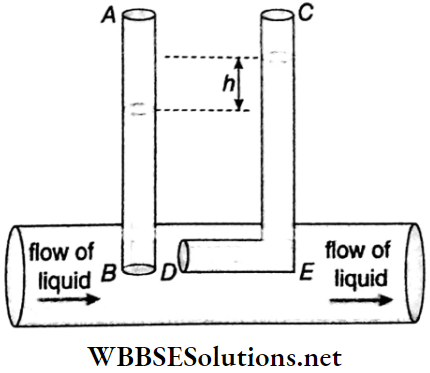
The difference in the liquid levels in the two tubes = h.
Let the velocity of liquid flow be ν.
The points B and D lie on the same horizontal plane; therefore, according to Bernoulli’s theorem,
⇒ \(\frac{1}{2 g} v^2+\frac{p_B}{\rho g}=0+\frac{p_D}{\rho g}\) [ρ= density of the liquid]
or, \(\frac{1}{2 g} v^2=\frac{p_D-p_B}{\rho g}\)
or, \(\frac{1}{2} v^2=\frac{h \rho g}{\rho} v^2=2 g h or, v=\sqrt{2 g h}\)
Pressure and Velocity Relationship in Bernoulli’s Principle
Venturimeter Derivation Class 11
If the cross-section of the pipe where the two tubes are placed is a, then the volume of liquid flowing per second through that section, V = \(\alpha v=\alpha \sqrt{2 g h} .\)
When an aeroplane is in motion, the velocity of air currents can be determined with the help of a pitot tube.
4. Sprayer or atomizer: A sprayer or atomizer is used for spraying water, insecticides, etc. Its action also depends on Bernoulli’s theorem.
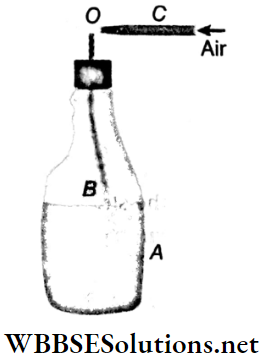
- The liquid to be sprayed is kept in a container A and its mouth is closed with the help of a cork or cap. A narrow tube B passes through the cap of the container. C is another tube through which air is blown. The tube C has a narrow tip.
- When air comes out from this narrow tip O with a high velocity, pressure at O decreases. Since O lies just above the open end of the tube B, the liquid rises through the tube B due to this low pressure, and as it meets the high-velocity air coming out of the tube C, it sprays out in the form of fine droplets.
Explanation of Some Phenomena with Bernoulli’s Theorem
1. It is not safe to stand near a fast-moving train: Due to the very high speed of the train, the air near the train also flows at a very high speed. As a result, pressure in that region decreases compared to the air pressure of the surrounding region. This excess surrounding pressure behind a person pushes him towards the train and may cause a serious accident.
2. The tin roof of a house is sometimes blown off during a storm: Since the velocity of the wind above the roof is very high, pressure becomes very low. The air inside the room is still and so the higher pressure from inside pushes the roof upwards and hence the roof may be lifted and blown off with the wind.
3. Two boats or ships moving side by side have a tendency to come closer: The speed of water in the narrow gap between boats or ships is greater than the speed of water on the other sides of the vessels. So, the pressure in that narrow region decreases. As a result, due to higher water pressure on the other sides of the boats or ships, they experience a lateral force and, hence, come closer.
Venturimeter Derivation Class 11
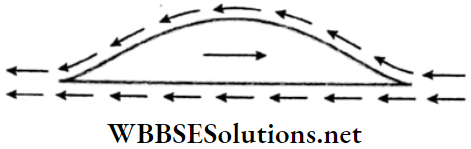
4. Flying in air of typical-shaped objects: Let us take an object moving through air towards right.
- Its lower surface is flat, but the upper surface is oval-shaped. The relative motions of the streamlines of air moving above and below it are shown by arrows.
- Clearly, the upper streamline traverses a greater distance in any fixed interval of time; so its velocity is higher. Then, according to Bernoulli’s theorem, the air pressure above the object is less than that below it.
- As a result, a net upward pressure acts on the object. This helps the object to fly through air, provided its weight is sufficiently low. This is one of the principles utilised to fly an aeroplane.
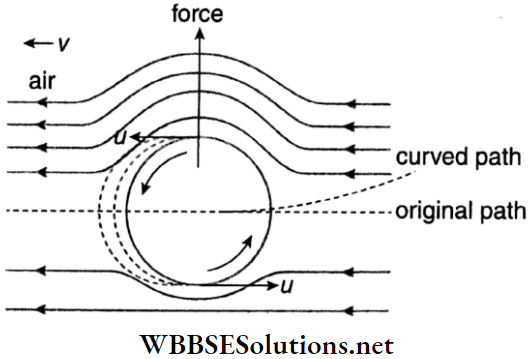
5. Magnus effect: When a spinning ball is thrown horizontally with a large velocity, it deviates from its usual parabolic path of spin free motion. This deviation can be explained on the basis of Bernoulli’s principle.
- When a ball moves forward, the air ahead the ball, moving with velocity v (say), rushes to fill up the vacant space behind the ball left evacuated by the motion of the ball.
- As the ball spins, the layer of air surrounding the ball also moves with the ball at a velocity u (say). From the fig, it can be stated that the resultant velocity of air above the ball becomes (v+ u) while that below that ball is (v- u).
- This difference in the velocities of air results in the pressure difference between the lower and upper faces of the ball. This pressure difference exerts a net upward force on the ball due to which it moves along a curved path as shown in Fig.
- If the spin of the ball is opposite to that shown in the Fig. a net downward force will act on it, deviating it from its original path. This effect is known as Magnus effect. If the surface of the ball is rough, more air is dragged and the path of the ball becomes more curved.
6. Blood flow and heart attack: An artery may get constricted due to the accumulation of plaque on its inner walls. In order to drive blood through this constriction the speed of the flow of blood is increased.
- This increased velocity lowers the blood pressure in the constricted region and the artery may collapse due to the external pressure.
- As a result, the heart exerts more pressure to open this artery and forces the blood through. As the blood rushes through the opening, the internal pressure once again drops due to some reasons leading to a repeat collapse which results in heart attack.
Venturimeter Derivation Class 11
Rate Of Flow Of A Liquid Numerical Examples
Example 1. The pressure at an orifice situated at the lower side of a vessel filled with a liquid is greater than the atmospheric pressure by 9.8 x 103 N · m-2. What is the velocity of efflux if the density of the liquid is 2500 kg · m-3? [g = 9.8 m · s-2]
Solution:
Given
The pressure at an orifice situated at the lower side of a vessel filled with a liquid is greater than the atmospheric pressure by 9.8 x 103 N · m-2.
Velocity of efflux of the liquid, v = √2gh
According to the problem, hρg = 9.8 x 103
or, \(g h=\frac{9.8 \times 10^3}{2500}\)
∴ v = \(\sqrt{\frac{2 \times 9.8 \times 10^3}{2500}}=2.8 \mathrm{~m} \cdot \mathrm{s}^{-1} .\)
Step-by-Step Guide to Bernoulli’s Equation
Example 2. Water is flowing through a horizontal tube of unequal cross-section. At a point where the velocity of water is 0.4 m · s-1, the pressure is 0.1 m Hg. What is the pressure at a point where the velocity of water is 0.8 m · s-1? The density of mercury = 13.6 x 103 kg · m-3.
Solution:
Given
Water is flowing through a horizontal tube of unequal cross-section. At a point where the velocity of water is 0.4 m · s-1, the pressure is 0.1 m Hg.
According to Bernoulli’s theorem,
⇒ \(\frac{1}{2} v_1^2+\frac{p_1}{\rho}=\frac{1}{2} v_2^2+\frac{p_2}{\rho}\) (since the tube is horizontal)
or, \(p_2=p_1+\frac{1}{2} \rho\left(v_1^2-v_2^2\right)= 0.1 \times\left(13.6 \times 10^3\right) \times 9.8\) + \(\frac{1}{2} \times 10^3 \times\left\{(0.4)^2-(0.8)^2\right\}\)
= \(13088 \mathrm{~Pa}=\frac{13088}{\left(13.6 \times 10^3\right) \times 9.8} \mathrm{~m} \mathrm{Hg}\)
= 0.0982 m Hg
Example 3. A pitot tube is connected to a main pipeline of diameter 16 cm. The difference in height of the water columns in the two arms of the tube is 10 cm. Determine the rate of flow of water through the main pipe.
Solution:
Given
A pitot tube is connected to a main pipeline of diameter 16 cm. The difference in height of the water columns in the two arms of the tube is 10 cm.
Volume of water flowing through the pipe per second,
V = αv [v = velocity of water]
V = \(\alpha \sqrt{2 g h} \text { (Here, } \alpha=\pi(8)^2=64 \pi \mathrm{cm}^2, h=10 \mathrm{~cm} \text { ) }\)
∴ V = \(64 \pi \sqrt{2 \times 980 \times 10}=64 \pi \times 140\)
= 2.8 x 104 cm3 = 0.028 m3.
The rate of flow of water through the main pipe = 0.028 m3.
Venturimeter Derivation Class 11
Example 4. A venturimeter has been connected between two points of a pipe. The radii of the pipe at these two points are 5 cm and 3 cm respectively. The difference in pressure between these points is equal to that of a water column of height 5 cm. Determine the rate of flow of water through the pipe.
Solution:
Given
A venturimeter has been connected between two points of a pipe. The radii of the pipe at these two points are 5 cm and 3 cm respectively. The difference in pressure between these points is equal to that of a water column of height 5 cm.
The rate of flow of water,
V = \(\alpha_1 \alpha_2 \sqrt{\frac{2 g h}{\alpha_1^2-\alpha_2^2}}\)
Here, \(\alpha_1=\pi(5)^2=25 \pi \mathrm{cm}^2, \quad \alpha_2=\pi(3)^2=9 \pi \mathrm{cm}^2\), h=5 \(\mathrm{~cm}, g=980 \mathrm{~cm} \cdot \mathrm{s}^{-2}\)
= \(25 \pi \cdot 9 \pi \sqrt{\frac{2 \times 980 \times 5}{(25 \pi)^2-(9 \pi)^2}} \approx 3000.6 \mathrm{~cm}^3 \cdot \mathrm{s}^{-1} .\)
Example 5. Water from a tap falls vertically with a velocity of 3 m · s-1. The area of the cross-section of the mouth of the tap is 2.5 cm2. If the flow of water is uniform and steady throughout, then determine the cross-sectional area of the tube of flow of water at a depth of 0.8 m from the mouth of the tap. [g = 10 m · s-2]
Solution:
Given
Water from a tap falls vertically with a velocity of 3 m · s-1. The area of the cross-section of the mouth of the tap is 2.5 cm2. If the flow of water is uniform and steady throughout
The area of cross-section of the mouth of the tap, A1 = 2.5 cm², and velocity of water flow there, v1 = 3 m · s-1.
Let the area of cross-section of the tube of flow of water at a depth of 0.8 m below the tap be A2 and the velocity of water flow there be v2
∴ \(v_2^2=v_1^2+2 g hp\)
= (3)² + 2 x 10 x 0.8
= 9 + 16 = 25
or, v2 = 5 m · s-1
We know that, A1 V1 = A2 V2
or, \(A_2=\frac{A_1 v_1}{v_2}=\frac{2.5 \times 3}{5}=1.5 \mathrm{~cm}^2 .\)
Bernoulli’s Principle in Everyday Life
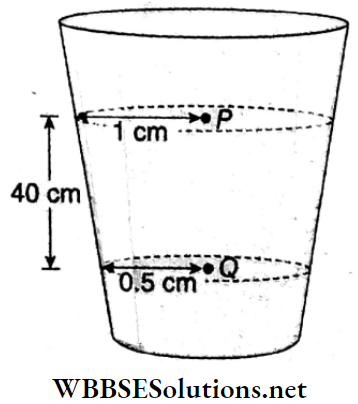
Example 6. Water flows in streamline motion through a vertical tube of non-uniform cross-section. The radius of the tube at point P and at point Q are 1 cm and 0. 5 cm respectively. Point Q is at a distance of 40 cm from point P. The pressure difference between these points is 39.3 cm of water. Determine the rate of flow of water through the tube.
Solution:
Given
Water flows in streamline motion through a vertical tube of non-uniform cross-section. The radius of the tube at point P and at point Q are 1 cm and 0. 5 cm respectively. Point Q is at a distance of 40 cm from point P. The pressure difference between these points is 39.3 cm of water.
Accoring to the equation of continuity,
⇒ \(A_1 v_1=A_2 v_2\)
∴ \(v_2^2=\frac{A_1^2}{A_2^2} v_1^2\)
According to Bernoulli’s theorem,
⇒ \(\frac{v_1^2}{2 g}+h_1+\frac{p_1}{\rho g}=\frac{v_2^2}{2 g}+h_2+\frac{p_2}{\rho g}\)
or, \(\frac{v_2^2-v_1^2}{2 g}=\left(h_1-h_2\right)+\left(\frac{p_1-p_2}{\rho g}\right)\)
or, \(\frac{v_2^2-v_1^2}{2 g}=40+39.3 \frac{\rho g}{\rho g}=79.3 \mathrm{~cm}\)
∴ \(\frac{v_1^2}{2 g}\left[\frac{A_1^2}{A_2^2}-1\right]=79.3\)
or, \(\frac{v_1^2}{2 g}\left[\frac{(\pi)^2}{(0.25 \pi)^2}-1\right]=79.3\)
or, \(v_1=101.79 \mathrm{~cm} / \mathrm{s}\)
The rate of flow of water = \(A_1 v_1=101.79 \times \pi\)
=319.78 cm3
The rate of flow of water through the tube =319.78 cm3
Real-World Applications of Bernoulli’s Principle
Example 7. A liquid of density 1000 kg/m3 is flowing in streamline motion through a tube of the non-uniform cross-section. The tube is inclined with the ground, The area of cross sections at points P and Q of the tube are 5 x 10-3 m2 and 10 x 10-3 m2 respectively. The height of the points P and Q from the ground are 3 m and 6 m respectively. The velocity of the liquid at point P is 1 m/s. Calculate the work done per unit volume due to
- the pressure and
- gravitational force for the flow of liquid from point P to point Q.

Solution:
Given
A liquid of density 1000 kg/m3 is flowing in streamline motion through a tube of the non-uniform cross-section. The tube is inclined with the ground, The area of cross sections at points P and Q of the tube are 5 x 10-3 m2 and 10 x 10-3 m2 respectively. The height of the points P and Q from the ground are 3 m and 6 m respectively. The velocity of the liquid at point P is 1 m/s.
From equation of continuity, \(A_1 v_1=A_2 v_2\)
or, \(v_2=\left(\frac{A_1}{A_2}\right) v_1=\left(\frac{5 \times 10^{-3}}{10 \times 10^{-3}}\right) \cdot(1)=\frac{1}{2} \mathrm{~m} / \mathrm{s}\)
Applying Bernoulli’s theorem we get,
⇒ \(p_1+\frac{1}{2} \rho v_1^2+\rho g h_1=\rho_2+\frac{1}{2} \rho v_2^2+\rho g h_2\)
or, \(\rho_1-\rho_2=\rho g\left(h_2-h_1\right)+\frac{1}{2} \rho\left(v_2^2-v_1^2\right)\)……….(1)
1. Work done per unit volume of the liquid due to the pressure of the streamline flow from P to Q is,
⇒ \(W_p =p_1-p_2\)
⇒ \(W_p =\rho g\left(h_2-h_1\right)+\frac{1}{2} \rho\left(\nu_2^2-v_1^2\right)[\text { from equation (1)] }\)
= \(\left[(1000)(9.8)(6-3)+\frac{1}{2}(1000)\left(\frac{1}{4}-1\right)\right]\)
= \(\left[3 \times 9.8-\frac{3}{8}\right] \times 10^3=29025 \mathrm{~J} / \mathrm{m}^3\)
2. Work done due to gravitational force for the streamline motion from P to Q is,
Wg = ρg(h1 – h2) = 1000 x 9.8 x (3 – 6)
= -29400 J/m3
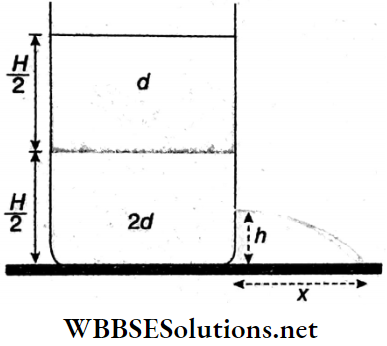
Example 8. A big container is kept on a horizontal surface of uniform area of cross-section, A. Two non-viscous liquids of densities d and 2d which are not mixed with each other and not compressed, are kept in the container. The height of both liquid column is H/2 and the atmospheric pressure on the open surface of the liquid is p0. A small orifice is created at a height h from the bottom of the container,
- Calculate the initial velocity of efflux of the liquid through the orifice,
- Calculate the horizontal distance x at which the first liquid drop emerged from the orifice will reach,
- What will be the value of h if the value of the horizontal distance x to be maximum xm? Also, calculate the value of xm neglecting the air resistance.
Solution:
Given
A big container is kept on a horizontal surface of uniform area of cross-section, A. Two non-viscous liquids of densities d and 2d which are not mixed with each other and not compressed, are kept in the container. The height of both liquid column is H/2 and the atmospheric pressure on the open surface of the liquid is p0. A small orifice is created at a height h from the bottom of the container,
1. Let, the initial velocity of efflux of the liquid = v.
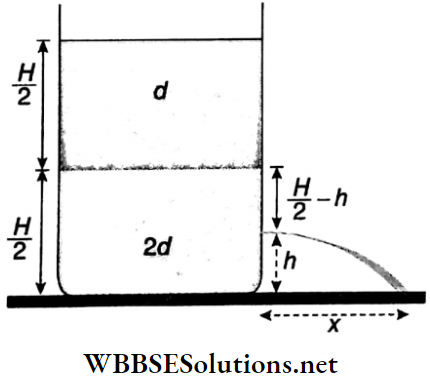
According to Bernoulli’s theorem,
⇒ \(p_0+d g\left(\frac{H}{2}\right)+2 d g\left(\frac{H}{2}-h\right)=p_0+\frac{1}{2}(2 d) v^2\)
or, \(v^2=\left(\frac{H}{2}+\frac{2 H}{2}-2 h\right) g or, v=\sqrt{(3 H-4 h)_2^g}\)
2. The time required to reach the ground of the first liquid drop emerged from the orifice is,
t = \(\sqrt{\frac{2 h}{g}}\)
∴ The horizontal distance traversed by the liquid is,
x = vt = \(\sqrt{(3 H-4 h)_2^g} \sqrt{\frac{2 h}{g}}\)
= \(\sqrt{h(3 H-4 h)}\)
3. The condition for x to be maximum (xm):
x = \(\sqrt{h(3 H-4 h)}=\sqrt{-\left(4 h^2-3 h H\right)}\)
= \(\sqrt{\left\{(2 h)^2-2 \cdot 2 h \cdot \frac{3}{4} H+\left(\frac{3}{4} H\right)^2-\left(\frac{3}{4} H\right)^2\right\}}\)
= \(\sqrt{\frac{9}{8} H^2-\left(2 h-\frac{3}{4} H\right)^2}\)
∴ x = \(x_m \text { when } 2 h-\frac{3}{4} H=0\)
Hence, h= \(\frac{3}{8}H\)
∴ \(x_m=\sqrt{\frac{9}{8} H^2}=\frac{3}{2 \sqrt{2}} H\)