Current Electricity Topic A Coulomb’s Law, Potential Difference, EMF Synopsis
WBBSE Class 10 Current Electricity Overview
1. Electric charge: Electric charge is the physical property of a matter due to which it exerts force on a charged body or on an uncharged body. There are two types of charges—positive charge and negative charge. Similar charges repel each other while opposite charges attract each other.
2. Coulomb’s law: The mutual attractive or repulsive force between two point charges at rest is directly proportional to the product of the charges and inversely proportional.to the square of the distance between them.
This law was given by the French scientist Charles Coulomb. If r is the distance between two point charges
q1 and q2, from Coulomb’s law mutual force,
\(F=k \cdot \frac{q_1 q_2}{r^2}\)
Read and Learn Also WBBSE Solutions for Class 10 Physical Science and Environment
where k is the constant of proportionality. The value of k depends on the surrounding medium and the system of unit used.
3. Units of charge in CGS system and Sl are esu of charge and C (coulomb) respectively, 1C = 3 x 109 esu of charge .
4. 1 esu charge: If two point charges of the same magnitude and of the same nature are placed in vacuum 1 cm apart and exert a repulsive force of 1 dyn on each other, then each charge is called a unit charge or 1 esu of charge or 1 statcouiomb in CGS system.
5. 1 coulomb charge: If two point charges of the same amount and of the same nature are placed in vacuum at a distance of 1 m and exert a repulsive force of 9 x 109 N on each other, then each charge is called a unit charge or 1 C (coulomb) in SI.
6. Electric field: When a charge is kept at any place, it creates a field around it and if any other charge is kept there, it experiences an electric force. This area or field is called electric field.
Understanding Electric Current and Its Measurement
7. Electric potential: Potential at a point in an electric field is defined as the amount of work done in bringing a unit positive charge from infinity to that point.
8. Units of electric potential in CGS system and in SI are respectively esu potential or statvolt and volt.
\(1 \text { volt }=\frac{1}{300} \text { statvolt }\)
9. 1 volt of potential difference: if 1 joule of work has to be done to move 1 coulomb of positive charge from one point to another point in an electric field, then the potential difference between these two points is called 1 volt of potential difference.
\(1 \text { volt }=\frac{1 \text { joule }}{1 \text { coulomb }}=1 \mathrm{~J} / \mathrm{C}\)
10. Electric cell: An electric cell is an arrangement in which other forms of energy like chemical energy, radiant energy can be converted into electric energy without the help of any appliance.
11. Electromotive force of a cell: The amount of work done by an external agency to move a unit positive electric charge from the positive pole of the cell to its negative pole is the electromotive force. V
12. If W work is done in taking a test charge q around the complete circuit of the cell, then emf of the cell is E = W/q.
13. SI unit of emf is volt (V).

Current Electricity Topic A Coulomb’s Law, Potential Difference, EMF Short And Long Answer Type Questions
Question 1. Write the mathematical form of Coulomb’s law.
Answer:
The mathematical form of Coulomb’s law:
Let two point charges q1 and q2 be placed at the points A and B respectively. The distance between these two charges is r. Now if F is the mutual force between the two charges, then according to Coulomb’s law, F ∝ q1, q2
\(F \propto \frac{1}{r^2}\) [when q1, q2 are constants]
Combining the above two conditions, we get \(F \propto \frac{q_1 q_2}{r^2}\) [when q1, q2, and r are variables]
or, \(F=k \cdot \frac{q_1 q_2}{r^2}\)
where k is a constant whose value depends on the surrounding medium and the system of units used.
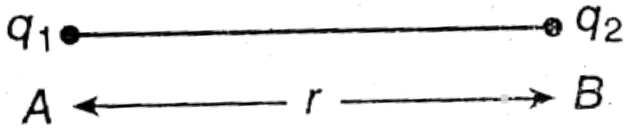
Question 2. Give the definition of unit charge in CGS system with the help of Coulomb’s law.
Answer:
The definition of unit charge in CGS system with the help of Coulomb’s law:
Two point charges q1 and q2 are placed at a distance of r. Now if the mutual force between them is F, then according to Coulomb’s law
⇒ \(F=k \cdot \frac{q_1 q_2}{r^2}\) where k is a constant.
When the two charges are placed in vacuum, k = 1 in CGS system.
∴ \(F=\frac{q_1 q_2}{r^2}\) …….(1)
Here, q1 = q2 = q, r = 1 cm and F = 1 dyn.
So, we get from equation (1), q2 = 1 or, q = ± 1. Therefore, if two point charges of the same magnitude and of the same nature are placed in vacuum 1 cm apart and exert a repulsive force of 1 dyn on each other, then each charge is called a unit charge or 1 esu of charge or 1 statcoulomb in CGS system.
Wbbse Class 10 Physical Science Solutions
Question 3. Give the definition of unit charge in Si with the help of Coulomb’s law.
Answer:
The definition of unit charge in Si with the help of Coulomb’s law:
Two point charges q1 and q2 are placed at a distance of r. Now if the mutual force between them is F, then according to coulomb’s law,
⇒ \(F=k \cdot \frac{q_1 q_2}{r^2}\), where k is a constant.
When the two charges are placed in vacuum, k =9x 109 in SI.
∴ \(F=9 \times 10^9 \cdot \frac{q_1 q_2}{r^2}\) ……(1)
Here, q1 = q2 = q, r = 1 m and F = 9 X 109 N
So from equation (1), we get 9 x 109 = 9 x 109 • q2 or, q2 = 1 or, q = ±1
Therefore, if two point charges of the same amount and of the same nature are placed in vacuum at a distance of 1 m and exert a repulsive force of 9 x 109 N on each other, then each charge is called a unit charge or 1 C (coulomb) in SI.
Question 4. Establish a relation between the units of charge in CGS system and SI.
Answer:
A relation between the units of charge in CGS system and SI:
The unit of charge in SI is C and the unit of charge in CGS system is esu of charge.
Suppose, 1 C = x esu of charge.
Now if two point charges of q1 = q2 = 1 C = x esu are kept at a distance of r = 1 m = 100 cm, then the mutual repulsive force is given by F= 9 x 109 N = 9 x 1014 dyn .
Now according to the coulomb’s law,
⇒ \(F=\frac{q_1 q_2}{r^2}\)
or, x2 = 9 x 1018 or, x = 3 x 109
∴ 1 C = 3 x 109 esu of charge
Question 5. Can we term Coulomb’s constant of the mutual force between two point charges as universal?
Answer:
The mathematical form of the Coulomb’s law regarding electrical force between two point charges q1 and q2 at a distance of r from each other in a medium is \(F=k \cdot \frac{q_1 q_2}{r^2}\) where k is called Coulomb’s constant.
The constant k depends on the nature of the medium. Therefore, mutual electrical force between two point charges depends on the nature of the medium and the value of this force is different in different media. Hence, Coulomb’s constant cannot be termed as universal.
Question 6. How can a charge Q be divded into two parts so that at a particular distance, mutual repulsisve force between them is maximum?
Answer:
Suppose the charge Q is divided into two portions q and (Q – q) and are kept at a distance of r from each other.
∴ mutual repulsive force, \(=k \cdot \frac{q(Q-q)}{r^2}\) [where, k is a constant]
or, \(F=\frac{k}{r^2}\left(Q q-q^2\right) \quad \text { or, } F=\frac{-k}{r^2}\left(q^2-Q q\right)\)
or, \(F=\frac{-k}{r^2}\left\{q^2-2 q \cdot \frac{Q}{2}+\left(\frac{Q}{2}\right)^2-\frac{Q^2}{4}\right\}\)
or, \(F=\frac{-k}{r^2}\left\{\left(q-\frac{Q}{2}\right)^2-\frac{Q^2}{4}\right\}=\frac{k}{r^2}\left\{\frac{Q^2}{4}-\left(q-\frac{Q}{2}\right)^2\right\}\)
For maximum value of F, q-Q/2 = 0 or, q = Q/2
∴ mutual repulsive force is maximum if the charge Q is divided into two equal parts, i.e., Q/2 and Q/2.
Question 7. Two bodies A and B have the same mass. A and B are charged respectively with equal amount of positive and negative charges. Find the change of their masses after charging.
Answer:
Given
Two bodies A and B have the same mass. A and B are charged respectively with equal amount of positive and negative charges.
Let us assume that charge of body A is q and that of body B is -q. Now if the charge of an electron is e, then q/e number of electrons are transferred from A and B. So when the mass of one electron is m, the mass of body A reduces by an amount qm/e and the mass of body B increases by an amount qm/e.
Question 8. Give the idea of potential difference in terms of work done to move one unit of positive charge Give the definition of the units of potential difference in CGS and Si from this.
Answer:
1. Potential difference between two points is the amount of work that has to be done to bring one unit positive charge from the point of lower potential to the point of higher potential.
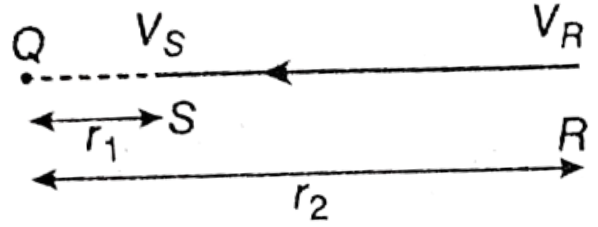
Let, VR and Vs be the electric potentials of the two points R and S kept in an electric field. If VS > VR, then the amount of work done to bring one unit of positive charge from point R to the point S = VS – VR, which is the potential difference between the two points S and R.
2. The units of potential difference in CGS system and Si are esu of potential and volt (V) respectively.
1 esu of potential difference: If 1 erg of work has to be done to move 1 esu of positive charge from one point to another point in an electric field, then the potential difference between those two points is called 1 esu of potential difference.
1 volt of potential difference: If 1 joule of work has to be done to move 1 coulomb of positive charge from one point to another point in an electric field, then the potential difference between these two points is called 1 volt of potential difference.
Question 9. The potentials of two points S and R at a distance of r1 and r2 from a charge Q are VS and VR, respectively. If r2>r1, which point is at higher potential and which one is at lower potential? What is the potential difference between them?
Answer:
Given
The potentials of two points S and R at a distance of r1 and r2 from a charge Q are VS and VR, respectively. If r2>r1,
More the amount of work one to bring one unit positive charge from infinity to any point near another positive charge, more is its potential. For this reason, as r2 > r1, VS > VR. Hence, point S will be at higher potential and point R will be at lower potential.
3. The potential difference between two points is the amount of work that is to be done to bring one unit positive charge from a lower potential point to a higher potential point. Therefore the potential difference between the two given points, V=VS-VR
 Question 10. What is electric charge? How many types of charges are there? Name them.
Question 10. What is electric charge? How many types of charges are there? Name them.
Answer:
Electric charge:
1. Electric charge is that physical property of a matter due to which it exerts force on an electrified body or on an uncharged body.
2. It is of two types, namely positive charge and negative charge.
Question 11. When a material is electrically charged by rubbing, then what is the charge on the two materials?
Answer:
When a material is rubbed with another material, then some electrons get transferred from one material to the other. The material which has deficit of electrons is positively charged and the material which has excess of electrons is negatively charged.
Wbbse Class 10 Physical Science Solutions
Question 12. When does an electrically charged material attract another material? When does it repel another material?
Answer:
1. An electrically charged material attracts an uncharged material and also a material having a charge of opposite nature.
2. It repels a material having charge of similar nature.
Question 13. Write Coulomb’s law.
Answer:
Coulomb’s law:
The mutual attractive or repulsive force between two point charges at rest is directly proportional to the product of the charges and inversely proportional to the square of the distance between them.
Question 14. When the distance between two point charges increases, how does the value of the mutual attractive or repulsive force between the change?
Answer: When the distance between two point charges increases, the value of the mutual attractive or repulsive force gets reduced in proportion to the square of the distance between the two.
Question 15. Define electric field.
Answer:
Electric field:
When a charge is kept at any place, it creates a field around it and if any other charge is placed in that area or field,it experiences an electric force. This area or field is known as an electric field.
Question 16. Define electric potential.
Answer:
Electric potential:
Electric potential at any point in an electric field is the total work done in moving a unit positive charge from infinity to that point under the influence of an external electric field.
In other words, we may say the total amount of work that has to be done in bringing one unit positive charge from infinity to a point near another positive charge is called the electric potential of that point with reference to the second charge.
Question 17. What are the units of electric potential in CGS system and SI? Define them.
Answer:
The units of electric potential in CGS system and SI:
The units of electric potential in CGS system and SI are esu of potential or statvolt and volt (V) respectively.
1. Esu of potential or statvolt: The potential at a point is said to be one esu of potential or one statvolt if 1 erg of work is done in bringing a positive charge of 1 esu from infinity to that point in an electric field.
2. Volt (V): The potential at a point is said to be one volt if 1 J (joule) of work is done in bringing a positive charge of 1 coulomb from infinity to that point in an electric field.
Wbbse Class 10 Physical Science Solutions
Question 18. Establish a relationship between esu of potential and volt (V).
Answer:
A relationship between esu of potential and volt (V)
When the potential difference at any point in an electric field is V, then the work done in bringing a charge of q from infinity to that point is given by
W = qV or, V = W/q
So, \(1 \mathrm{~V}=\frac{1 \mathrm{~J}}{1 \mathrm{C}}=\frac{10^7 \mathrm{erg}}{3 \times 10^9 \mathrm{esu} \text { of charge }}\) = 1/300 esu of potential
∴ 1 esu of potential = 300 V
Wbbse Class 10 Physical Science Solutions
Question 19. What is electromotive force? Or, In reality electromotive force (emf) is not any force—eKpiain the fact.
Answer:
Electromotive force:
1. The electromotive force of an electrical source is defined as the amount of electrical energy that is produced in the source due to the transfer of a positive charge from a lower potential to a higher potential.
2. In reality, electromotive force is not any force at all. It is the energy that is required to move per unit charge from lower potential to higher potential.
Question 20. Define the unit of emf in SI.
Answer:
Unit of emf in SI is J/C or V (volt).
1 volt of electromotive force: If 1J of work has to be done to move a positive charge of 1 coulomb from a point of low potential to a point of high potential in an electrical cell, then the magnitude of the electromotive force of that electrical cell is 1 J/C or 1 V.
Question 21. The emf of an electrical celt is 10 V’ — what do you, mean by this statement?
Answer: ‘The emf of an electrical cell is 10 V’ means that 10 J of work has to be done in order to move an electric charge of 1 C from a lower potential point to a higher potential point in the electrical cell.
Wbbse Class 10 Physical Science Solutions
Question 22. Define electromotive force for an open circuit.
Answer:
Electromotive force for an open circuit:
The electromotive force of a ceil for an open circuit is defined as the amount of work done by an external agency to move a unit positive electric charge from the positive pole of the cell to its negative pole.
Current Electricity Topic A Coulomb’s Law, Potential Difference, EMF Very Short Answer Type Questions Choose The Correct Answer
Question 1. Coulomb force is
- Purely attractive force
- Purely repulsive force
- Either attractive or repulsive
- Neither attractive nor repulsive
Answer: 3. Either attractive or repulsive
Question 2. Electric charges are of
- A single type
- Two types
- Three types
- Four types
Answer: 2. Two types
Question 3. The charge of an electron is
- -3.2 × 10-19 C
- -1.6 × 10-19 C
- 1.6 × 10-19 C
- 3.2 × 10-19 C
Answer: 3. 1.6 X 10-19 C
Question 4. 450 V = how many statvolt?
- 0.5
- 1
- 2
- 1.5
Answer: 4. 1.5
Question 5. 0.1 C = how many esu of charge?
- 3 × 1010
- 3 × 109
- 3 x 108
- 3 x 107
Answer: 4. 3 x 107
Question 6. 1 J/ C is equal to
- 1w
- 1Ω
- 1V
- 1A
Answer: 3. 1V
Wbbse Class 10 Physical Science Solutions
Question 7. 2C charge is equivalent to the charge of how many electrons?
- 6.25 × 1018
- 6.25 × 1019
- 1.25 × 1019
- 12.5 × 1019
Answer: 3. 1.25 × 1019
Question 8. If the charge of an electron be 1.6 x 10-19 C, which one of the following cannot be the charge of a body?
- 1.6 x 10-19 C
- 0.8 x 10-19 C
- 3.2 x 10-19 C
- 4.8 x 10-19 C
Answer: 2. 0.8 x 10-19 C
Question 9. Unit of electromotive force is
- Volt
- Coulomb
- Ohm
- Ampere
Answer: 1. Volt
Question 10. Electromotive force is measured by
- Ammeter
- Voltmeter
- Galvanometer
- Potentiometer
Answer: 2. Voltmeter
Question 11. 1 V = how many esu of potential?
- 3 x 104
- 1/3 x 104
- 1/300
- 300
Answer: 3. 1/300
Current Electricity Topic A Coulomb’s Law, Potential Difference, EMF Answer In Brief
Question 1. What is the unit of electric charge?
Answer: The unit of electric charge in CGS system is esu of charge or stat coulomb.
Question 2. What is the unit of electric charge in SI?
Answer: The unit of electric charge in SI is coulomb (C).
Question 3. On which factors, does the value of the constant k of Coulomb’s law depend?
Answer: The value of the constant k depends on the nature of the surrounding medium and the system of unit.
Question 4. What is the value of k in CGS unit in the equation for Coulomb’s law, \(F=k \cdot \frac{q_1 q_2}{r^2}\) in vacuum?
Answer: The value of k is 1 in CGS unit in vacuum.
Question 5. What is the value of k in SI in the equation for Coulomb’s law, \(F=k \cdot \frac{q_1 q_2}{r^2}\) in vacuum?
Answer: The value of k is 9 x 109 N • m2 • C2 in vacuum.
Question 6. What type of quantity is electric potential?
Answer: Electric potential is a scalar quantity.
Question 7. What is the relationship between electric charge (q) and work (l/V) with electric potential (V)?
Answer: Electric potential, V = W/q
Wbbse Class 10 Physical Science Solutions
Question 8. What is the unit of electric potential in CGS unit?
Answer: The unit of electric potential in CGS unit is esu of potential or statvolt.
Question 9. What is the unit of electric potential in SI?
Answer: The unit of electric potential in SI is volt (V).
Question 10. V • C is the unit of which physical quantity?
Answer: V-C is equal to J (joule) which is the unit of work and energy.
Question 11. What is the unit of electromotive force (emf) in SI?
Answer: The unit of electromotive force (emf) in SI is volt (V).
Question 12. Is Coulomb’s law a universal law?
Answer: No, Coulomb’s law is not a universal law.
Question 13. Does the mass of an uncharged body increase or decrease when it is charged with a positive charge?
Answer: When an uncharged body is charged with a positive charge, its mass decreases.
Question 14. Does the mass of an uncharged body increase or decrease when it is charged with a negative charge?
Answer: When an uncharged body is charged with a negative charge, its mass increases.
Question 15. What is the relationship between quantity of charge and amount of current flown through a conductor?
Answer: Amount of current flown through a conductor = \(\frac{\text { charge }}{\text { time }}\)
Question 16. Unit of which physical quantity is obtained when the unit of electric charge is divided by the unit of electric current?
Answer: Unit of time is obtained when the unit of electric charge is divided by the unit of electric current.
Question 17. Unit of which physical quantity is obtained when the unit of potential difference is multiplied by the unit of charge?
Answer: Unit of work is obtained when the unit of potential difference is multiplied by the unit of electric charge.
Question 18. What is the carrier of electricity in a metal conductor?
Answer: Free electron is the carrier of electricity in a metal conductor.
Current Electricity Topic A Coulomb’s Law, Potential Difference, EMF Fill In The Blanks
Question 1. A body is positively charged if there is ________ of electrons.
Answer: Deficit
Question 2. A body is negatively charged if there is ________ of electrons in it due to friction.
Answer: Excess
Question 3. The active mutual force between two point charges _____ on the nature of the medium.
Answer: Depends
Question 4. The potential at a particular point is _____ volt if 1/300 erg of work is done in bringing one esu of positive charge from infinity to that point in an electric field.
Answer: One
Question 5. Some other form of energy is transformed into _______ in an electric cell.
Answer: Electrical energy
Question 6. Force and electromotive force (emf) are ______ quantities.
Answer: Different
Question 7. Scientist _____ named two opposite type of charges as positive and negative.
Answer: Benjamin Franklin
Question 8. Electric charge is a _____ physical quantity.
Answer: Scalar
Question 9. In equation \(\mathrm{F}=k \cdot \frac{q_1 q_2}{r^2}\) SI unit of k is ______
Answer: n.m2.C-2
Current Electricity Topic A Coulomb’s Law, Potential Difference, EMF State Whether True Or False
Question 1. Charge of an electron is -1.6 x 10-19C.
Answer: True
Question 2. Like charges attract each other and unlike charges repels each other.
Answer: False
Question 3. 1C = 3×1010 esu of charge.
Answer: False
Question 4. According to the Coulomb’s law, mutual force of attraction or repulsion is directly proportional to the square of the distance between the two given point charges.
Answer: False
Question 5. 1 V = 1/300 esu of potential.
Answer: True
Question 6. When a body is earthed, electrons from the earth flow into the body. This means that the body gets positively charged.
Answer: False
Question 7. According to Coulomb’s law if q1 q2 < 0, the force will be repulsive.
Answer: False
Question 8. In electric cell heat energy is converted into electric energy.
Answer: False
Current Electricity Topic A Coulomb’s Law, Potential Difference, EMF Numerical Examples Useful Relations
1. Charge of a body is q = ±ne, where e = charge of an electron and n is an integer.
2. Electrostatic force between two point charges and q2 kept in air or vacuum,
(1) \(F=\frac{q_1 q_2}{r^2}\) (in CGS system)
(2)F = 9 x 109 (in SI)
2. The amount of work done to bring a point charge q from a point of lower potential to a point of higher potential W = qV, V = potential difference between the two points.
1 V = 1/300 esu potential
Question 1. Two-point charges 25 esu and 12 esu are 5 cm apart in air. Calculate the force between the two charges.
Answer:
Given
Two-point charges 25 esu and 12 esu are 5 cm apart in air.
First charge (qx) = 25 esu
Second charge (q2) = 12 esu
Distance between the two charges (r) = 5cm
∴ mutual force between the two charges,
⇒ \(F=\frac{q_1 q_2}{r^2}=\frac{25 \times 12}{5^2}=12 \mathrm{dyn}\)
Question 2. There are two point charges in air at a distance of 2 m from each other. The magnitude of one charge is double than that of the other. If the force of repulsion between them is 7.2 x 1010 N, what is the value of each charge?
Answer:
Given
There are two point charges in air at a distance of 2 m from each other. The magnitude of one charge is double than that of the other. If the force of repulsion between them is 7.2 x 1010 N
Let us assume that first charge, q1 = qC,
Second charge, q2 = 2q C and the distance between the two charges, r = 2 m.
Mutual repulsive force, F= 7.2 x 1010 N
∴ from the equation, \(F=9 \times 10^9 \cdot \frac{q_1 q_2}{r^2}\), we get
⇒ \(7.2 \times 10^{10}=9 \times 10^9 \cdot \frac{q \times 2 q}{2^2}\)
or, \(q^2=\frac{4 \times 7.2 \times 10^{10}}{2 \times 9 \times 10^9}\)
or, q2 = 16 or, \(q=\pm 4\)
So, when \(q=\pm 4\), we get q1 = 4C and q2 = 2 x 4 = 8 C and when q = -4, we get q1 = -4 C and q2 = 2 × (-4) = -8 C
Question 3. Two point charges are kept at a distance of r and the mutual electrostatic force is F . What should be the distance between them for the value of force to become F/2?
Answer:
Given
Two point charges are kept at a distance of r and the mutual electrostatic force is F .
If the charges are q1 and q2, then \(F=k \cdot \frac{q_1 q_2}{r^2}\) ……(1)
where k is a constant.
Let us assume that the distance between the two charges is x so that the value of the mutual force, \(F_1=\frac{F}{2}\).
⇒ \(F_1=k \cdot \frac{q_1 q}{x^2}\) …..(2)
Dividing equation (1) by (2), we get
⇒ \(\frac{F}{F_1}=\frac{x^2}{\dot{r}^2}\)
or, \(2=\frac{x^2}{r^2}\left[because F_1=\frac{F}{2}\right]2=\frac{x^2}{r^2}\left[because F_1=\frac{F}{2}\right]\)
⇒ \(x^2=2 r^2 \quad therefore x=\sqrt{2} r\)
Question 4. A body M is rubbed with another body N and as a result, charge of M is 4.8 μC How many electrons have been exchanged during the process of rubbing?
Answer:
Given
A body M is rubbed with another body N and as a result, charge of M is 4.8 μC
Value of charge of one electron (e) = 1.6 x 10-19 C
Charge of body M, q = 4.8 μC = 4.8 x 10-6C
Let us assume that x number of electrons have been exchanged in this process.
or, \(x=\frac{q}{e} \quad \text { or, } x=\frac{4.8 \times 10^{-6}}{1.6 \times 10^{-19}}=3 \times 10^{13}\)
Hence, 3 x 1013 numbers of electrons have been transferred from body M to body N during the process of rubbing.
Question 5. Charge of body A becomes 6.4 x 10-8 C when rubbed with another body B. What is the change of the mass of body A?
Answer:
Given
Charge of body A becomes 6.4 x 10-8 C when rubbed with another body B.
Value of charge of one electron (e) = 1.6 x 10-19 C
Charge of material A(q) = 6.4 x 10-8C
If x number of electrons are transferred from A to B during the process of rubbing, then
⇒ \(x=\frac{q}{e}=\frac{6.4 \times 10^{-8}}{1.6 \times 10^{-19}}=4 \times 10^{11}\)
Again, the mass (m) = 9.1 x 10-31kg
∴ Decrease of the mass of body A = mx= 9.1 x 10-31 x 4 x 1011 = 3.64 x 10-19 kg
Current Electricity Topic B Ohm’s Law Synopsis
Ohm’s Law Explained with Examples
1. Electric current: The flow of electrically charged particles through any conductor is called electric current. The direction of electric current is given by the direction of motion of positive charges.
2. The amount of electrically charged particles flowing per second through any cross section of a conductor is known as the amount of electric current (l).
\(I=\frac{\text { total charge }(Q)}{\text { total time }(t)}\)
3. Unit of electric current in SI is ampere (A).
4. 1 A: A current of 1 A Is said to flow when a charge of 1C passes through any cross section of a conductor in 1 second.
5. The conventional direction of current is taken to be the direction of flow of positive charge i.e., opposite to the direction of flow of negative charge.
6. Current is a scalar quantity.
7. Ohm’s law: When temperature and other physical conditions of a conductor remain constant, the amount of current flowing through a conductor is directly proportional to the potential difference across the two ends of the conductor.
8. Mathematical expression: If the current flowing through a conductor is l when the potential difference across its two ends is V, then according to the Ohm’s law,
l ∝ V or, l = KV [where K is a proportional constant] or, V = 1/K • l or, V = R; [where 1/K = R(constant)] [where 1/K = R (constant)]
R is called resistance of the conductor.
9. The resistance of a conductor is defined as the property of the conductor due to which the flow of current through it is opposed.
10. Definition of resistance from Ohm’s law: When a potential difference is created across the two ends of a conductor, a current flows through the conductor. The ratio of the potential difference across the conductor to the amount of current flowing through the conductor is called the resistance of the conductor.
11. Unit of resistance in SI is ohm (Ω). \(\text { ohm }=\frac{\text { volt }}{\text { ampere }}\)
12. Those materials which easily conduct electricity are known as conductors.
Example: Any metallic conductor like iron, silver.
Those materials which cannot conduct electricity in general are known as insulators.
Example: Glass, plastic, rubber.
Those materials whose resistivity is more than that of a good conductor but less than that of an insulator are known as semiconductors.
Example: Germanium, Silicon.
13. There is a type of metal or substance whose resistivity becomes zero at a temperature less than a particular temperature. That particular temperature is called critical temperature and this type of substance is called superconductor.
Example: Mercury becomes a superconductor at 4.2K temperature.
14. When an electric current flows through an electric cell, then the constituents of the electric cell provides resistance to the current. This resistance inside the cell is known as the internal resistance.
15. Relation between emf and internal resistance of a cell: Let, an circuit is formed by connecting an electric cell of emf E, internal resistance r and an external resistance R , In closed circuit a current / is passing through the external circuit.
Now from Ohm’s law, we can write \(I=\frac{E}{R+r} \text { or, } E=I R+I r \text { or, } r=\frac{E-I R}{I}\)
15. Resistivity: The resistance of a conductor between the two opposite faces of a unit cube of the substance, when current passes normally through them is called resistivity.
16. If the length and area of cross section of a conductor at a particular temperature are l and A respectively, then its resistance,
\(R=\rho \frac{l}{A}\)
where ρ is resistivity of the material.
16. Unit of resistivity in SI is ohm • m (Ω • m)
17. Conductance: The property of a conductor due to which electricity can be conducted through it is called the conductance of the conductor.
18. If the resistance of a conductor is R, then its conductance, K= 1/R.
19. The unit of conductance is mho (℧). In SI, mho is called siemens (S).
20. The conductance of unit length of any conductor having unit cross sectional area is called the conductivity of the material of that conductor.
21. If the resistivity of the material of a conductor is ρ, its conductivity is \(\sigma=\frac{1}{\rho}\)
22. The unit of conductivity in SI is mho • m-1(℧ • m-1), but another unit S • m-1 is also used.
23. Combination of Resistances: When a number of resistances are connected together in electrical circuit, then the combination is called combination of resistances.
24. Series combination: When several resistances are connected in such a way that the extreme end of one resistance is connected to the beginning of the next resistance and so on, then this combination is called a series combination.
So, if three resistances R1, R2 and R3 are connected in a series combination and if the equivalent resistance is Rs, then Rs = R1 + R2 + R3
25. Parallel combination: When several resistances are connected in such a way that the one of ends of all the resistances are connected to a particular point, while the other ends are also connected to another particular point, then this combination is called a parallel combination.
So, if three resistances R1, R2 and R3 are connected in a parallel combination and if the equivalent resistance is Rp, then \(\frac{1}{R_p}=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}\)
Current Electricity Topic B Ohm’s Law Short And Long Answer Type Questions
Question 1. What are the directions of electric current and the flow of electrons in a metal conductor present in an electric circuit?
Answer:
The directions of electric current and the flow of electrons in a metal conductor present in an electric circuit are
In a metallic conductor, free electrons are the carriers of electricity. If a potential difference is applied across the two sides of a conductor, free electrons move from lower potential to higher potential. As a result, it is said that a current is passing through the conductor.
In a metallic conductor, the direction of electric current is opposite to the motion of the electrons. In other words, it has been assumed that the electrons flow from higher potential to lower potential.
Question 2. Write down Ohm’s law. Or, Explain the formula by which the relationship between the potential difference across the two ends of a conductor and the value of current through it can be known.
Answer:
Ohm’s law
According to Ohm’s law, if the temperature and other physical conditions remain constant, the amount of current flowing through a conductor is directly proportional to the potential difference across the ends of the conductor.
Let us assume the potential across the two ends of a conductor are VA and VB respectively. If VA > VB, then the current flows from A to B.
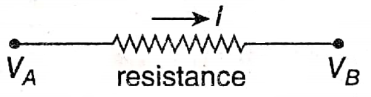
Now, if l is the current, then according to ohm’s law, l ∝ (VA – VB) or, \(\frac{V_A-V_B}{I}=k\)
or, VA – VB = kl, where k is a constant.
Question 3. What do you mean by the resistance of a conductor? Define resistance from Ohm’s law.
Answer:
Resistance of a conductor
1. Resistance is that property of a conductor due to which it obstructs the flow of current through it.
2. If a current l flows through a conductor when V is the potential difference across its two ends, then according to Ohm’s law, V ∝ l or, V/l = R (constant)
This constant is called the resistance of the conductor. In other words, the ratio between the potential difference across the conductor and the amount of current flowing through the conductor is called the resistance.
Question 4. What are ohmic conductor and non-ohmic conductor? Give examples.
Answer:
Ohmic conductor: Those conductors which follow Ohm’s law or those conductors for which l-V graph is a straight line passing through the origin, are called ohmic conductors.
Example: any metallic conductor
Non-ohmic conductor: Those conductors which do not follow Ohm’s law or those conductors for which l-V graph is not a straight line passing through the origin, are called non-ohmic conductors.
Example: electrolytic solution
Question 5. What are the factors on which the resistance of a conductor depends?
Answer:
Factors on which the resistance of a conductor depends are:
- Length of the conductor,
- Cross sectional area of the conductor,
- Nature of the material of the conductor,
- Temperature.
In addition, for some conductors, resistance depends on:
- Intensity of light,
- Strength of magnetic field,
- Pressure.
Question 6. What are the factors on which the resistance of a conductor made of a particular material depends at a definite temperature? How?
Answer:
The factors on which the resistance of a conductor made of a particular material depends at a definite temperature
1. The resistance of a conductor (R) at a particular temperature depends on the length (l) and cross sectional area (A) of the conductor.
2. If the cross sectional area of the conductor remains unchanged, the resistance of the conductor is directly proportional to its length, i.e., R ∝ l [when A is constant].
Again, if the length of the conductor remains unchanged, the resistance of the conductor is inversely proportional to its cross sectional area, i.e., R ∝ 1/A [when l is constant].
Question 7. Define electrical conductance and electrical conductivity. Write down their expressions and their units in SI.
Answer:
Electrical conductance and electrical conductivity
1. The property of a conductor due to which electricity can be conducted through it is called the conductance of the conductor. If the resistance of a conductor is R, its conductance, K = 1/R.
The unit of conductance is mho (℧). In SI, mho is called siemens (S).
2. The conductance of unit length of any conductor having unit cross sectional area is called the conductivity of the material of that conductor. If the resistivity of the material of a conductor is p, its conductivity is σ = 1/ρ.
The unit of conductivity in SI is mho.m-1(℧.m-1), but another unit S.m-1is also used.
Question 8. Write down how the resistance of a metallic wire changes in the following cases:
- Length -of the wire is doubled, diameter and temperature remain unchanged.
- Diameter of cross section of the wire is doubled, length and temperature remain unchanged.
- Keeping length and diameter unchanged, temperature of the wire is increased.
Answer:
1. The resistance of a wire is directly proportional to its length if the diameter and temperature of the conducting wire remain unchanged. Therefore, if length of the wire is doubled, its resistance will also be doubled.
2. If the length of the conducting wire and its temperature remain unchanged, then the resistance of the wire is inversely proportional to its cross section. Suppose, diameter was d initially. Therefore, cross sectional area,
\(A_1=\frac{\pi d^2}{4}\)
Now, if the diameter is doubled, cross sectional area becomes \(A_2=\frac{\pi(2 d)^2}{4}=\pi d^2=4 A_1\)
∴ cross-sectional area will be four times the previous one.
Therefore, present resistance of the wire is 1/4th of the previous resistance.
3. If the length of the conducting wire and its diameter remain unchanged, the resistance of the wire increases with increase of temperature.
Question 9. Two terminals of an electrical cell are connected by a wire. After that, the wire is folded into two and connected again with the same two terminals. In this condition, does the amount of current increase or decrease as compared to the first case?
Answer: As the wire is folded into two and is connected with the cell, the resistance of the folded wire will be half of the resistance of the original wire due to reduction of its length into half. Again, due to doubling of the area of cross section, resistance of the folded wire will be half of the original wire.
Therefore, due to change of length and cross section of the wire, the resistance of the folded wire will be (1/2 Χ 1/2) or 1/4th of its original resistance. Since the potential difference of the cell is constant, current through the folded wire is 4 times the current through the original wire as resistance and current are inversely proportional to each other.
Question 10. A thin and a thick Wire of the same length are connected with the same battery. In which wire is the value of current more and why? Explain.
Answer:
1. When a thin and a thick wire of the same length are connected with the same battery, value of current in the thick wire is more.
2. The resistance of a conductor is inversely proportional to the cross section, so the resistance of a thick wire will be less. Again, if the potential difference remains the same, value of current is inversely proportional to the resistance. Therefore, as a result of decrease of resistance of the thick wire, value of current in the conductor will be more.
Similarly, if the wire is thin, i.e., the cross section is reduced, the resistance of the wire will increase. Hence, as a result of increase of resistance, the value of current through the thin wire will be less.
Question 11. A copper wire is heated keeping its length and radius constant. Find the change in its resistance. If we take a carbon thread instead of a copper wire, will there be any change in the resistance?
Answer:
1. If a copper wire is heated by keeping its length and resistance constant, its resistance increases.
2. If a carbon thread is used instead of a copper wire and heated, the resistance of the thread will decrease. This is because the resistance of all pure metals and most metallic alloys increase with increase of temperature. But the resistance of carbon, electrolytes, rubber, silicon etc. decreases with increase of temperature.
Question 12. 1. How can you connect several resistance of lower value to get a resistance of higher value?
2. How can you connect several resistances of higher values to get a resistance of low value?
Answer:
1. When several resistances of lower values are connected in a series combination, a resistance of higher value can be obtained.
Example: Three wires of resistance 2Ω, 3Ω and 6Ω are connected in a series combination. The equivalent resistance of the combination R = (2 + 4 + 6) Ω =12 Ω
2. When several resistances of higher values are connected in a parallel combination, a resistance of lower value can be obtained.
Example: Three wires of resistances 2Ω, 3Ω and 6Ω are connected in-a parallel combination. The equivalent resistance is given by
⇒ \(\frac{1}{R}=\frac{1}{2}+\frac{1}{4}+\frac{1}{6}=\frac{11}{12}\)
∴ \(R=\frac{12}{11}=1.09 \Omega \text { (approx.) }\)
Types of Current: AC vs. DC
Question 13. Define conductor, insulator and semiconductor at electricity. What is the value of resistivity of each?
Answer:
Conductor: A conductor is a substance which easily conducts electricity.
Example: Any metallic substance.
Insulator: The value of resistivity of a good conductor lies between 10-8 Ω • m and 10-6 Ω • m(approx.). An insulator is a substance which in general, cannot conduct electricity.
Example: Glass, plastic, rubber etc.
Semiconductor: The value of resistivity of an insulator lies between 1010 Ω • m and 1017 Ω • m(approx.).
A semiconductor is a substance whose electrical conductance is less than that of a conductor but more than that of an insulator.
Example: Germanium, silicon.
The value of resistivity of a semiconductor lies between 10-5 Ω • m and 10-2 Ω • m (approx.).
Question 14. Write down the use of insulators in daily life.
Answer:
Use of insulators in daily life
If electric current passes through a conductor and if we touch it during that time, an electric shock can be felt. This is due to the fact that our body is a good conductor of electricity. Insulators protect us from this.
That is why an insulating material like coating of polyethylene is always applied on a conducting wire. For the same reason, a ceramic insulator is used in the overhead wire of trains. Also, handles of electrical instruments like pliers, tester etc. are made up of insulating materials.


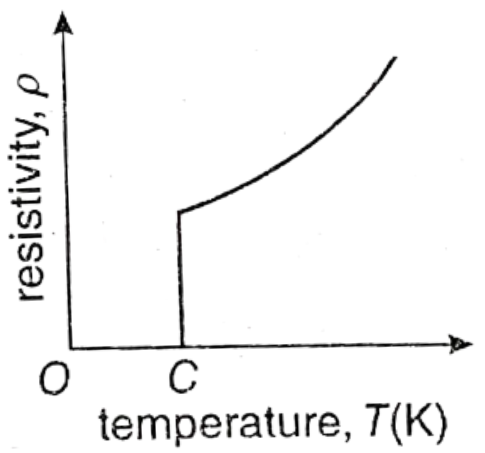
Question 15. What do you mean by superconductivity? Draw a resistivity vs temperature graph to explain this.
Answer:
Superconductivity:
1. In general, if the temperature of a metal is reduced, its resistivity decreases. There is a type of metal or compound whose resistivity becomes zero if their temperature becomes less than a particular temperature. This particular temperature is called critical temperature. This type of phenomenon is called superconductivity and those materials are called superconductors.
2. In the given figure change of resistivity with temperature of this type of material is shown. In the graph, point C is the critical temperature.
For example, mercury at 4.2 K temperature, lead at 7 K temperature and Niobium Nitride (NbN) at 16 K temperature become superconductors.
Question 16. Explain series combination and parallel combination of resistance.
Answer:
Series combination and parallel combination of resistance:
Series combination: When some resistances are connected such that the extreme end of one is connected with the beginning of the next resistance and so on, then the resistance are said to be connected in series. When this combination is joined in an electrical circuit, the same current flows through each resistance after applying a potential difference across the combination.
Parallel combination: When some resistances are connected such that one of the ends of all the resistances is connected at a fixed point while the other end is connected to another point, then the resistances are said to be in parallel combination. When this combination is joined in an electrical circuit, potential difference across each of the resistance remains the same.
Question 17. Determine the equivalent resistance of three resistances connected in series combination.
Answer: The three resistances R1,R2,R3 are connected in series at points A and D in the original circuit. Electric current (I) in the circuit is flowing in the direction from A to D which remains the same for each resistance.
Let us assume that the electric potential at the points A, B, C and D are VA, VB, VC and VD respectively.
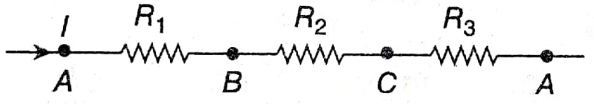
∴ according to Ohm’s law,
VA-VB = IR1 …(1)
VB -VC = IR2 …(2)
VC -VD = IR3 …(3)
Adding the above three equations, we get VA-VD = l(R1 + R2 + R3) …(4)
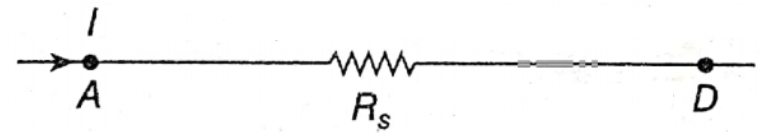
Now, instead of these three resistances, a single resistance Rs is used in between the points A and D. If the same current l flows when the same potential difference is applied between A and D, then according to Ohm’s law, VA-VD = IRS …(5)
By comparing the equations (4) and (5), we get Rs =R1 + R2 + R3 ….(6)
So, RS is the equivalent resistance of the series combination of three resistances.
Question 18. Write the characteristics of the series combination of resistances.
Answer:
The characteristics of the series combination of resistances are:
1. Same amount of electric current passes through each resistance.
2. Equivalent resistance of the combination is the algebraic sum of each individual resistance.
3. When a potential difference is applied between the two sides of the combination, potential difference between the two sides of each resistance is directly proportional to the respective resistance.
4. Equivalent resistance of the combination is more than the value of each resistance.
Question 19. Determine the equivalent resistance of three resistances connected in parallel combination.
Answer: The three resistances R1,R2, R3 are connected in parallel at points A and B in the original circuit.
The potential difference between the points A and B is VA – VB = V. Now, suppose the value of original current in the circuit is l and values of current through R1, R2 and R3 are I1, I2 and l3 respectively. As the resistances are connected in parallel combination, the potential difference across the two sides of each resistance is the same.


Applying Ohm’s law, we get:
For the first resistance, V = I1 R1 ….(1)
For the second resistance, V = I2 R2 …..(2)
and for the third resistance, V = l3R3 ….(3)
Again, l =I1+I2 +l3
or, \(I=\frac{V}{R_1}+\frac{V}{R_2}+\frac{V}{R_3}\)
or, \(I=V\left(\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}\right)\) ….(4)
Now, if a single resistance RP is used in place of the three resistances in between the points A and B and if the same potential difference is applied to get the same value of current , then according to Ohm’s law,
⇒ \(V=I R_p \quad \text { or, } \quad I=\frac{V}{R_p}\) …..(5)
By comparing equations(4) and (5), we get
⇒ \(\frac{1}{R_p}=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}\) …..(6)
or, \(R_p=\frac{R_1 R_2 R_3}{R_2 R_3+R_1 R_3+R_1 R_2}\)
So, RP is called the equivalentr resistance of the three parallel resistance.
Question 20. Write the characteristics of the parallel combination of resistances.
Answer:
The characteristics of the parallel combination of resistance are:
1. The potential difference across the two terminals of each resistance is the same.
2. The reciprocal of the equivalent resistance of the combination is equal to the sum of the reciprocals of all the resistances.
3. The equivalent resistance of the combination is smaller than the smallest resistance of the combination.
4. As the terminal potential difference is constant, hence the value of current passing through a particular resistance is inversely proportional to that resistance.
Question 21. Calculate the equivalent resistance when n number of resistances, each of value R, are connected in series combination as well as in parallel combination. What is the ratio of thses two values?
Answer:
1. There are n number of resistances with values R1, R2, R3 ••• Rn If they are connected in series combination, then the equivalent resistance
Rs = R1+ R2 + R3 + …+ Rn
Now, as R1 = R2 = R3 = ••• = Rn = R, so, RS= nR Again, if the resistances are connected in parallel combination and their equivalent resistance is RP, then
⇒ \(\frac{1}{R_p}=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}+\cdots+\frac{1}{R_n}\)
As R1 = R2 = R3 = • • • = Rn = R, then \(\frac{1}{R_p}=\frac{n}{R} \text { or, } R_p=\frac{R}{n}\)
2. The ratio of equivalent resistances in the two cases, \(\frac{R_S}{R_p}=\frac{n R}{\frac{R}{n}}=n^2\)
Question 22. Show that the equivalent resistance of three resistances in parallel combination is smaller than the smallest resistance.
Answer: Let us assume that three resistances of values R1, R2 and R3 are connected in parallel combination. R1 is smallest among the three.
If RP is the equivalent resistance of the combination, then
⇒ \(\frac{1}{R_p}=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3} \quad \text { or, } \frac{1}{R_p}=\frac{1}{R_1}+x\)
[Here, \(\frac{1}{R_2}+\frac{1}{R_3}\) is taken as x, where x is a positive quantity.]
∴ \(\frac{1}{R_p}>\frac{1}{R_1} \quad \text { or, } R_p<R_1\)
∴ The equivalent resistance of three resistances.
In parallel combination is smaller than the smallest resistance.
Question 23. What are the constituents of a simple electrical circuit? What Is the direction of electric current in the entire circuit?
Answer:
Constituents of a simple electrical circuit
1. There are two parts of a simple electrical circuit-
(1) External circuit (entire portion except the cell) and
(2) Internal circuit (internal portion of the cell). At least one resistance must be present in the external circuit.
2. Once the key of the circuit is closed, an electric current starts flowing through the circuit and also through the resistance from the end of higher potential to the end of lower potential. In the internal circuit, this flow is from the negative terminal of the cell to the positive terminal.
Question 24. An electrical circuit has been formed by connecting a resistor, a voltmeter, an ammeter, an electric cell and a few pieces of
conducting wires.
1. Out of these, which electrical parts have, their own resistances and which parts have no resistance?
2. If we consider the parts to be ideal, then which ones will be without any resistance?
Answer:
1. According to the question, all the parts, namely, resistor, voltmeter, ammeter, electric cell, conducting wires possess some resistance. Out of these, the resistance of the conducting wire is considered to be negligible compared to the other parts.
2. If all the constituents had been ideal, then except the resistor and voltmeter, then all the other constituents namely ammeter, electric cell and conducting wires would have been without resistance.
Question 25. What is internal resistance of an electric cell? What Is the reason for the existence of this resistance?
Answer:
1. Resistance of the electric cell connected in an electrical circuit is the internal resistance of cell.
2. When an electric cell sends current through a closed circuit, then current flows also through the cell. The active liquid or electrolytic material placed between the two electrodes of this cell offers resistance to this current. This resistance inside the cell is the internal resistance.
Question 26. What is lost volt? What is the cause of its origin?
Answer:
Lost volt:
1. During the flow of electric current through the resistances of the external circuit and the internal circuit, some amount of work has to be done by each electric charge to overcome those resistances.
2. Therefore, some amount of electrical energy is converted to some other form of energy while passing through each resistance. The energy converted in the case of internal circuit is heat energy which cannot be used for any practical purpose. This wasted energy inside an electricai cell is known as lost volt.
3. The presence of the internal resistance of the electrical cell is the cause of origin of lost volt.
Question 27. Write down and explain the mathematical form of Ohm’s law for the entire circuit.
Answer:
According to Ohm’s law, electromotive force (E) for the entire circuit is given by: E=V+V’
where V = potential difference between the two terminals across the resistance R in the external circuit and V’ = amount of work that has to be done by an electric charge to overcome the resistance of the internal circuit, i.e., internal resistance (r) of the cell or lost volt.

Applications of Current Electricity in Daily Life
Question 28. The emf of an electrical cell and its internal resistance are E and r respectively. The cell is connected with an external resistance. What is the value of this external resistance If potential difference is half of the emf of the cell?
Answer:
Suppose the value of external resistance = R.
Current flow in the circuit, \(I=\frac{E}{R+r}\)
The potential difference across the two ends of the resistance (R)= \(R I=\frac{R E}{R+r}\)
According to the question,\(\frac{R E}{R+r}=\frac{E}{2}\) or, R + r = 2R or, r = R or, R = r
∴ The external resistance (R) has the same value as that of the internal resistance (r).
Question 29. Two resistances R1 and R2 are connected in parallel, if an electrical cell is connected with the combination, a current I flows through the circuit. Calculate the values of currents through R1 and R2.
Answer: Suppose the values of currents through R1 and R2 are I1 and I2 respectively.
I = I1+I2 or, I2 = I – I1

Now as the two resistances R1 and R2 are connected in parallel, so the potential difference across the two ends is equal.
⇒ \(\begin{aligned}
& therefore \quad I_1 R_1=I_2 R_2 \\
& \text { or, } I_1 R_1=\left(I-I_1\right) R_2 \text { or, } I_1\left(R_1+R_2\right)=R_2 I \\
& therefore \quad I_1=\frac{R_2}{R_1+R_2} \cdot I \\
& \text { So, } I_2=I-I_1=I-\frac{R_2}{R_1+R_2} \cdot I \\
& \quad=\frac{R_1 I+R_2 I-R_2 I}{R_1+R_2}=\frac{R_1}{R_2+R_2} \cdot I
\end{aligned}\)
Alternative method: As the two resistances are connected in parallel, potential difference across the two ends of each resistance is equal. according to Ohm’s law, V = I1R1 = I2R2
Again, equivalent resistance, \(R=\frac{R_1 R_2}{R_1+R_2}\)
Let the main current = I
⇒ \(So, V=I R=I \frac{R_1 R_2}{R_1+R_2}\)
⇒ \(therefore \quad I_1 R_1=I \cdot \frac{R_1 R_2}{R_1+R_2} and I_2 R_2=I \cdot \frac{R_1 R_2}{R_1+R_2}\)
So, \( I_1=\frac{R_2}{R_1+R_2} \cdot I and I_2=\frac{R_1}{R_1+R_2} \cdot I\)
Question 30. If R = r in the circuit, calculate the value of l.
Answer: Let R1 is the resistance of the external circuit.
So, \(\frac{1}{R_1}=\frac{1}{R}+\frac{1}{R} \quad \text { or, } \frac{1}{R_1}=\frac{2}{R} \quad \text { or, } R_1=\frac{R}{2}=\frac{r}{2}\)
Now, current flow in the circuit, \(I=\frac{E}{R_1+r}=\frac{E}{\frac{r}{2}+r}=\frac{E}{\frac{3 r}{2}}=\frac{2 E}{3 r}\)
Question 31. Ordinary rubber is a bad conductor of electricity but the tyre of an aeroplane is made by a conductor of electricity to some extent. Why?
Answer: An aeroplane runs with high velocity at the time of take-off and landing. As a result, tyres are electrically charged due to friction of the runway and the tyres. There is a possibility of an accident due to this reason. As the tyres are made up of low conductors of electricity, charge produced by this friction goes to the earth and the possibility of an accident is averted.
Question 32. What is the function of an electrical source or electrical cell in an electrical circuit?
Answer: The function of an electrical cell is to create a permanent potential difference between the two ends of a conductor in an electrical circuit. The movement of charge takes place through the conductor as long as there is potential difference between the two ends, that is, the electrical cell is effective.

Question 33. What do you mean by short circuit?
Answer:
Short circuit:
1. If the two poles of any electrical source is connected by a conducting wire of negligible resistance, then that circuit is known as a short circuit.
2. As the resistance is low, flow of current becomes high causing the wire to get extremely heated. The electrical circuit may get damaged due to this.
Question 34. What is electric current?
Answer:
Electric current:
The flow of electrically charged particles through any conductor is called electric current. The magnitude of current is the amount of electrically charged particles flowing per second through any cross section of the conductor.
Question 35. What is the relationship between electric current and charge?
Answer: If q amount of charge passes through any cross section of a conductor in time t, then flow of charge per second = q/t.
∴ Electric current, I = q/t.
Question 36. Define the unit of electric current in SI.
Answer:
Unit of electric current in SI is A (ampere).
1 A: A current of 1 A is said to flow when a charge of 1 C passes through any cross section of a conductor in 1 second.
Question 37. Though electric current has a direction, why is it called a scalar quantity?
Answer: Electric current has both magnitude and direction but it is called a scalar quantity. This is because electric current does not follow the vector addition rule.
Question 38. Define the unit of resistance in SI.
Answer: The unit of resistance in SI is ohm (Ω) and it is given by \(1 \Omega=1 \frac{\mathrm{V}}{\mathrm{A}}\)
1Ω: If a current of 1 A flows through a conductor after the application of a potential difference of 1 V across its two ends, then the resistance of the conductor is called 1Ω.
Question 39. with the help of a graph, express Ohm’s law. Or, With the help of Ohm’s law, draw V-I graph.
Answer: According to Ohm’s law, V = IR.
Taking potential difference V as abscissa and current l through the conductor as ordinate, a graph is drawn. It will be a straight line passing through the origin.

Question 40. Define resistivity.
Answer:
Resistivity
The resistivity of a material is the resistance per unit length of a conductor having unit cross sectional area of the material at a definite temperature. In other words, it is defined as the resistance between the two opposite surfaces of a cube of unit length.
Question 41. What do you mean by the statement— ‘The resistivity of copper is 1.68 x 10-6. cm (at 20°C temperature)’?
Answer:
‘The resistivity of copper is 1.68 x 10-6. cm (at 20°C temperature)’
The statement, ‘The resistivity of copper is 1.68x 10-6 Ω• cm (at 20°C temperature)’ means that the resistance between the two opposite surfaces of a cube of 1 cm side made of copper is 1.68 X 10-6 Ωat 20°C.
Question 42. Determine the unit of resistivity In SI.
Answer:
Unit of resistivity In SI
If ρ is the resistivity of the material of a conductor of length l and cross sectional area A, then
\(R=\rho \cdot \frac{l}{A} \text { or, } \rho=\frac{R A}{l}\)
∴ The unit of resistivity in SI = \(=\frac{\text { unit of resistance in } \mathrm{SI} \times \text { unit of area in } \mathrm{SI}}{\text { unit of length is } \mathrm{SI}}\)
∴ \(=\frac{\Omega \times m^2}{m}=\Omega \cdot m\)
Question 43. How does pressure affect the resistance of a conductor?
Answer: In general, resistance of most of the metals decreases with the application of pressure. For example, resistance of carbon decreases with increase of pressure. But for Li, Ca and Bi, application of pressure increases the resistance instead of decreasing it.
Question 44. How does light affect the resistance of a conductor?
Answer: The resistances of some conductors depend on the intensity of incident light. The resistance of the metal selenium decreases when light is projected on it. With further increase of intensity, resistance keeps on decreasing.
Question 45. Current is passed through a wire With the help of an electrical cell. After sometime, it is observed that the wire gets heated and the amount of current decreases slightly. Explain this phenomenon.
Answer: When an electric current passes through a wire, it gets heated due to the production of heat in the wire. As the temperature of the wire increases, its resistance also increases. As a result, amount of current through it gets reduced. So, after sometime the amount of current flowing through the wire decreases.
Question 46. One silver wire and one iron wire of the same length and the same cross sectional area are taken. They are connected across the same electrical cell individually. The flow of electric current will be more in which case?
Answer: The resistance of a silver wire is less than that of a iron wire having the same length and the same cross section. Now if they are connected across the same electrical cell individually, then current through the silver wire will be more as its resistance is less.
Question 47. Draw- a graph which shows the change of resistivity of a semiconductor with the change of temperature.
Answer: The graph is shown below:

Question 48. There are two wires, one is thin and the other is thick. Both are made up of the same material and have the same length. Which wire will have more resistance and why?
Answer: The resistance of a conductor is inversely proportional to its cross sectional area when its length and material remain constant. Now as cross sectional area of a thin wire is less than that of a thick wire, so the resistance of a thin wire is more than that of a thick wire.
Question 49. What is equivalent resistance?
Answer:
Equivalent resistance
If a single resistance can be used in place of a combination of more than one resistance between the two points of an electrical circuit and when the same current flows through the circuit with the application of the same potential difference, then that single resistance is called an equivalent resistance of those resistances.
Question 50. How do you describe the emf in terms of work done inside a cell?
Answer: The magnitude of emf can be determined by the amount of work done to move a unit positive charge from the negative pole to the positive pole of a cell in an open circuit.
Current Electricity Topic B Ohm’s Law Very Short Answer Type Questions Choose The Correct Answer
Question 1. Which metal is the best conductor of electricity?
- Gold
- Silver
- Aluminium
- Copper
Answer: 2. Silver
Question 2. What is the equivalent resistance of a parallel combination of resistances 3Ω and 6Ω?
- 2Ω
- 4Ω
- 9Ω
- 3Ω
Answer: 1. 2Ω
Question 3. When the potential difference is 10 V and the amount of current is 5 A, then the resistance is
- 5Ω
- 4Ω
- 3Ω
- 2Ω
Answer: 4. 2Ω
Question 4. If R is the equivalent resistance of a parallel combination of three resistances R1, R2 and Rs (R1> R2 > R3), then
- R > R1
- R > R2
- R > R3
- R < R3 < R2 < R1
Answer: 4. R < R3 < R2 < R1
Question 5. The resistance of a superconductor at critical temperature is
- Infinite
- Zero
- 106 Ω
- 109 Ω
Answer: 2. Zero
Question 6. When temperature increases, the resistance of a semiconductor
- Decreases
- Increases
- First increases then decreases
- First decreases then increases
Answer: 1. Decreases
Question 7. What is the value of R at the time of a short circuit?
- ∞
- 0
- 106 Ω
- 1010 Ω
Answer: 2. 0
Question 8. What is the value of R in an open circuit?
- ∞
- 0
- 10 Ω
- 1000 Ω
Answer: 1. ∞
Question 9. 1 A is equal to
- 1 C2/s
- 1 C/s2
- 1 C/s
- 1 s/C
Answer: 3. 1 C/s
Question 10. The carrier of electricity in a metal conductor is
- Orbital electrons
- Electrons and ions
- Free electrons
- Ions
Answer: 3. Free electrons
Question 11. When a charge of 120 C passes through a conductor for 2 minutes, what is the amount of electric current?
- 1 A
- 2 A
- 0.5 A
- 0.25 A
Answer: 2. 2 A
Question 12. 1 A = how many mA?
- 10
- 100
- 1000
- 106
Answer: 3. 1000
Question 13. When 1019 electrons flow through any cross section of a conductor in 2 seconds, the amount of electric current
- 0.8 A
- 1.6 A
- 2.4 A
- 3.2 A
Answer: 1. 0.8 A
Question 14. 1 V/A is equal to
- 1J
- 1C
- 1Ω
- 1W
Answer: 3. 1Ω
Question 15. If temperature increases, resistance of a metal
- Increases at first, then decreases
- Decreases at first, then increases
- Increases
- Decreases
Answer: 3. Increases
Question 16. The unit of resistivity is
- Ω-m2
- Ω-m
- Ω/m
- Ω/m2
Answer: 2. Ω-m
Question 17. When the resistance of a conductor is 0.1 Ω, its conductance is
- 0.1 S
- 1 S
- 10 S
- 100 S
Answer: 3. 10 S
Question 18. When the resistivity of a conductor 2 x 10 8 n • m, then its conductivity is
- 2 x 107 S • m-1
- 2 x 10-8 S • m-1
- 5 x 107 S • m-1
- 5 x 108 S • m-1
Answer: 3. 5 x 107 S • m-1
Question 20. The value of resistivity is lowest in the case of
- Metal
- Insulator
- Semiconductor
- Superconductor
Answer: 4. Superconductor
Question 21. When n number of resistances, each of value R, are connected in series combination, its equivalent resistance is
- (n + 1)R
- (n – 1)R
- nR
- R/n
Answer: 3. nR
Question 22. When n number of resistances, each of value R are connected in parallel combination, its equivalent resistance is
- (n + l)R
- nR
- R/(n+1)
- R/n
Answer: 4. R/n
Question 23. What is the ratio of equivalent resistances of a combination of n number of resistances in series and in parallel, each of value R?
- n2
- n
- 1/n
- 1/n2
Answer: 1. n2
Question 24. In a series combination of several resistances, which of the following remains unchanged for each resistance?
- Potential difference
- Consumed power
- Current
- None of these
Answer: 3. Current
Question 25. In a parallel combination of several resistances, which of the following remains unchanged for each resistance?
- Potential difference
- Consumed power
- Current
- None of these
Answer: 1. Potential difference
Question 26. What is the equivalent resistance of two resistances R1 and R2 in Parallel combination?
- \(\frac{R_1 R_2}{R_1-R_2}\)
- \(\frac{R_1-R_2}{R_1 R_2}\)
- \(\frac{R_1+R_2}{R_1 R_2}\)
- \(\frac{R_1 R_2}{R_1+R_2}\)
Answer: 4. \(\frac{R_1 R_2}{R_1+R_2}\)
Question 27. Three identical resistances are connected in series. The equivalent resistance of the combination is 9 Ω. What is the value of each resistance?
- 3 Ω
- 4.5 Ω
- 18 Ω
- 27 Ω
Answer: 1. 3 Ω
Question 28. Four identical resistances are connected in parallel. The equivalent resistance of the combination is 2 Ω. What is the value of each resistance?
- 4Ω
- 16Ω
- 8Ω
- 32Ω
Answer: 3. 8Ω
Question 29. Two resistances are connected in parallel. If the potential difference is doubled, rate of heat production at a particular resistance becomes
- Two times
- Four times
- Half
- Eight times
Answer: 2. Four times
Question 30. Two resistances are connected in parallel. The ratio of these resistances is 1: 2. What is the ratio of the power consumed by the two resistances?
- 1:2
- 2:1
- 4:1
- 1:4
Answer: 2. 2:1
Question 31. Nichrome is
- An alloy of Ni, Cr, Al
- An alloy of Ni, Cr, Fe
- An alloy of Al, Cr, Fe
- An alloy of Ni, Cr, Fe
Answer: 2. An alloy of Ni, Cr, Fe
Question 32. Which physical quantity is compatible with electromotive force?
- Force
- Momentum
- Acceleration
- Power
Answer: 4. Power
Question 33. If the resistivity of a conductor with length l and cross sectional are a A is p, what is its conductance?
- \(\rho \frac{l}{A}\)
- \(\frac{\rho A}{l}\)
- \(\frac{l}{\rho A}\)
- \(\frac{A}{\rho l}\)
Answer: 4. \(\frac{A}{\rho l}\)
Question 34. If the resistance of a conductor with length l and cross sectional area A is R, its conductivity is
- l/R
- RA/l
- l/RA
- A/Rl
Answer: 3. l/RA
Question 35. The l-V curve of an ohmic conductor makes an angle θ with the positive side of the potential difference axis. What is the value of the resistance (R)?
- R = sinθ
- R = cosθ
- R = tanθ
- R = cotθ
Answer: 4. R = cotθ
Question 36. What is the equivalent resistance between A and B Shown In Fig?
- 1Ω
- 6Ω
- 5Ω
- 4Ω

Answer: 4. 4Ω
Question 37. What is the equivalent resistance between A and B shown in Fig. 16?
- 2Ω
- 3Ω
- 1Ω
- 4Ω

Answer: 1. 2Ω
Question 38. emf of the electric cell is 5 V and its internal resistance is 1Ω. What is the amount of current with a resistance of 4Ω?
- 0.5 A
- 0.75 A
- 1 A
- 1.25 A

Answer: 3. 1 A
Question 39. Amount of current is 0.5 A with a resistance of 9Ω. If the emf of the electric cell is 5V, what is the internal resistance?
- 0.5A
- 0.75Ω
- 1Ω
- 1.5Ω

Answer: 3. 1Ω
Question 40. Which of the following has the lowest resistivity?
- Gold
- Silver
- Copper
- Aluminium
Answer: 2. Silver
Question 41. A wire of resistance R is cut into two equal parts and these two parts are connected in parallel. What is the equivalent resistance?
- R
- R/2
- R/8
- R/4
Answer: 4. R/4
Current Electricity Topic B Ohm’s Law Answer In Brief
Question 1. What is the unit of resistance in SI?
Answer: Unit of resistance in SI is ohm (Ω).
Question 2. Give example of an ohmic conductor.
Answer: All metal conductors are ohmic conductors.
Question 3. Give examples of three non-ohmic devices.
Answer: Three non-ohmic devices are transistor, LED and filament lamp.
Question 4. What is the term used to describe the fall of potential inside an electric cell?
Answer: The fall of potential inside on electric cell is called lost volts.
Question 5. When an electric cell is connected with an external resistance, what is the direction of current flow in the external resistance?
Answer: The direction of current flow in the external resistance is from the positive pole towards the negative pole.
Question 6. When an electrical cell is connected with an external resistance, what is the direction of current flow inside the cell?
Answer: The direction of current flow inside the cell is from the negative pole towards the positive pole.
Question 7. What is the unit of resistivity in SI?
Answer: The unit of resistivity in SI is Ω• m.
Question 8. What is the unit of conductivity in SI?
Answer: The unit of conductivity in SI is S • m-1.
Question 9. What is the unit of conductance in SI?
Answer: The unit of conductance in SI is siemens (S).
Question 10. When the resistance of the conductor is 100 Ω, what is the value of the conductance?
Answer: When the resistance of a conductor is 100 ft, the conductance of the conductor = 1/100 = 0.01 S 100.
Question 11. When the resistivity of a conductor is 4×10-8 Ω • m, what is the value of its conductivity?
Answer: When the resistivity of a conductor is 4 x 10-8 Ω • m, the value of its conductivity
= \(=\frac{1}{4 \times 10^{-8}}\) = 2.5 x 107 S • m-1
Question 12. What is the value of resistivity of a conductor at a temperature less than the critical temperature?
Answer: The value of resistivity of a conductor at a temperature less than the critical temperature is zero.
Question 13. How does the resistivity of a metal change if its temperature is increased?
Answer: The resistivity. of a metal increases if its temperature is increased.
Common Mistakes in Current Electricity Calculations
Question 14. How does the resistivity of a semiconductor change if its temperature is increased?
Answer: The resistivity of a semiconductor decreases if its temperature is increased.
Question 15. How would you connect some resistances, in series combination or in parallel combination, to get a resistance higher than the resistance of the individual constituents?
Answer: To get a resistance higher than the resistance of the individual constituents, the resistances have to be connected in a series combination.
Question 16. How would you connect some resistances, in series combination or in parallel combination, to get a resistance lower than the resistance of the individual constituents?
Answer: To get a resistance lower than the resistance of the individual constituents, the resistances have to be connected in a parallel combination.
Question 17. The household electrical instruments are connected in which combination?
The household electrical instruments are always connected in parallel combination.
Question 18. Which physical quantity, electric current or potential difference, remains constant for all the resistances connected in a series combination?
Answer: Electric current remains the same through each resistance connected in a series combination of resistances.
Question 19. Which physical quantity, electric current or potential difference, remains constant for all the resistances connected in a parallel combination?
Answer: Potential difference across the two ends of each resistance remains constant in a parallel combination of resistances.
Question 20. Two resistances R1 and R2(R1> R2) are connected in a parallel combination. Which resistance carries more current?
Answer: Between the two resistance R1 and R2, R2 carries more current.
Question 21. Two resistances, each of value R, are connected in a parallel combination. What is the equivalent resistance?
Answer: If two resistances, each of value R, are connected in a parallel combination, its equivalent resistance becomes R/2.
Question 22. What are the values of two resistances such that the amount of current passing through them remains the same, when connected in series and in parallel combination?
Answer: The values of the two resistances should be equal.
Question 23. What is the value of the equivalent resistance, when three resistances 2Ω, 4Ω and 6Ω are connected in a series combination?
Answer: The equivalent resistance of the series combination = 2 + 4 + 6 = 12Ω.
Question 24. Which instrument is used to measure electric current?
Answer: Ammeter is used to measure electric current.
Question 25. Which instrument is used to measure potential difference?
Answer: Voltmeter is used to measure potential difference.
Queswtion 26. What is the condition for steady electric current through a conductor?
Answer: Steady electric current is obtained when the potential difference across the two ends of the conductor remains constant.
Question 27. How does the resistance of the metal selenium change when light rays fall on it?
Answer: When light rays fall on the metal selenium, its resistance decreases.
Question 28. How does the resistance of bismuth change when it is kept in a megnetic field?
Answer: When bismuth is kept in a magnetic field, its resistance increases.
Question 29. How does the resistance of carbon change when pressure on it is increased?
Answer: When pressure on carbon is increased, its resistance decreases.
Question 30. Write one use of superconductivity.
Answer: Superconductive magnet is a very powerful electromagnet. This type of magnet is used in a particle accelerator.
Current Electricity Topic B Ohm’s Law Fill In The Blanks
Question 1. Electric current is a _____ quantity.
Answer: Scalar
Question 2. .cm is the practical unit of ______
Answer: Resistivity
Question 3. The resistivity of a metal is _____ than that of a semiconductor.
Answer: Less
Question 4. If temperature increases, the resistance of germanium (Ge) ________
Answer: Decreases
Question 5. When a suitable impurity is doped inside a semiconductor, its resistivity ________
Answer: Decreases
Question 6. Electric current does not follow the law of _______ addition.
Answer: Vector
Question 7. When the potential difference across the two ends of a conductor is changed, the _______ passing through the conductor also changes.
Answer: Amount of current
Question 8. The nature of the l-V curve of a metallic conductor is a ______
Answer: Straight line
Question 9. If r is the internal resistance of a cell and l is the value of current, then rl is the _______
Answer: Lost volt
Question 10. The ______ of a conductor depends on its length, cross sectional area and its constituent material.
Answer: Resistance
Question 11. When temperature decrease, the resistivity of a metal also _______
Answer: Decreases
Question 12. The handle of an electrical instrument is made of an _______ material in order to avoid electric shock.
Answer: Insulating
Current Electricity Topic B Ohm’s Law State Whether True Or False
Question 1. The SI unit of electric potential is volt.
Answer: True
Question 2. Conductivity of a conductor is the reciprocal of the resistivity of the same conductor.
Answer: True
Question 3. Resistivity of a metal decreases as temperature increases.
Answer: False
Question 4. For a good conductor, resistivity is very low and conductivity is very high.
Answer: True
Question 5. If three resistances R1,R2 ,R3 are connected in series, then the equivalent resistance RS is given by \(R_S=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}\)
Answer: False
Question 6. In series combination of resistances, voltage remains the same through each resistance.
Answer: False
Question 7. In parallel combination of resistances, current remains the same through each resistance.
Answer: False
Question 8. If three resistances R1,R2 ,R3 are connected in parallel, then the equivalent resistance RP is given by \(R_p=\frac{R_1 R_2 R_3}{R_1 R_3+R_1 R_3+R_1 R_2}\)
Answer: True
Question 9. Out of a thin and a thick wire, both made up of same material and having same length, the thin wire has more resistance.
Answer: True
Current Electricity Topic B Ohm’s Law Numerical Examples
1. Electric current intensity I = q/t, where q = amount of charge flows through any cross section of the conductor in the time t
2. q = It
3. Mathematical expression of Ohm’s law is V=IR, where V – potential difference between two ends of the conductor I = electric current passing through the conductor, R = resistance of the conductor.
4. \(R=\rho \frac{l}{A}\), where l = length of the conductor, A = area of cross section of the conductor, ρ = specific resistance
5. Formula for the determination of equivalent resistance in series combination, RS = R1 + R2 + R3 + …… + Rn , if R1 = R2 = … = Rn= R then RS = nR.
6. Formula for the determination of equivalent resistance in parallel combination, \(\frac{1}{R_p}=\frac{1}{R_1}+\frac{1}{R_2}+\cdots+\frac{1}{R_n}\)
For two resistances R1 and ,R2 \(\frac{1}{R_p}=\frac{1}{R_1}+\frac{1}{R_2}=\frac{R_1+R_2}{R_1 R_2}\) or \(R_p=\frac{R_1 R_2}{R_1+R_2}\)
7. E = Ir+V, where E = emf of a cell, r = internal resistance of the cell and V = potential difference
8. Current through external circuit, \(I=\frac{E}{R+r}\)
9. Lost volt = Ir
10. Terminal potential difference of the cell, V = E – lr
Question 1. An electric current of 2 A is passing through a conductor. How much charge will flow in 5 seconds? When the carrier of charge in the conductor are free electrons, how many electrons flow through any cross section of the conductor?
Answer:
Electric current (l) = 2 A
Time (t) = 5 s
If q amount of charge flows, then l = q/t or, q= lt = 2×5 = 10C
Value of the charge of (e) = 1.6 x10-19. C
Number of electrons flowing, through any cross section of the conductor \(=\frac{q}{e}=\frac{10}{1.6 \times 10^{-19}}=6.25 \times 10^{19}\).
Question 2. A current of 0.1 A flows when a potential difference of 10 V is applied across the two ends of a conductor. What is the resistance of the conductor?
Answer: Potential difference across the two ends of the conductor (V) = 10 V
Amount of current flowing (I) = 0.1 A
Now, if the resistance of the conductor = R, then according to Ohm’s law, V= IR or, \(R=\frac{V}{l}=\frac{10 \mathrm{~V}}{0.1 \mathrm{~A}}=100 \Omega\).
Question 3. There are two Conductors, the resistance of the first being three times that of the second. The amount of current flowing through the first is twice that of the second. What is the ratio of the potential difference of the two conductors?
Answer: Let us assume that the resistance and the amount of current passing through the second conductor are R and l respectively.
resistance of the first conductor =3R and current passing through the first conductor = 2I. Potential difference across the two ends of the first conductor,
V1 = 3Rx2I =6RI….(1)
and potential difference across the two ends of the second conductor, V2 = RI
Now dividing equation (1) by (2), we get \(\)or, V1 : V2 = 6 :1
Question 4. An electrical cell with internal resistance of 5 Ω and emf of 2 V is attached to a resistance of 15Ω. What is the potential difference across the two terminals of the cell?
Answer: Emf (E) of the electrical cell = 2 V
Internal resistance (r) = 5 Ω
Resistance of the external circuit (R) = 15 Ω
Let current in the circuit = I

So, \(I=\frac{E}{R+r}=\frac{2}{15+5}\)
∴ I + 0.1A
Hence, potential difference across the two terminals, V=IR = 0.1×15 = 1.5 V
Question 5. An electrical cell has an emf of 5 V. How much work has to be done in an open circuit by an external agency in moving a charge of 1C from cathode to anode inside the cell?
Answer: Emf (E) of the electrical cell = 5 V.
In an open circuit, charge inside the cell, q = 1C Now, work done by an external agency for moving the charge from cathod to anode is given by
W = qE= 1CX5V = 5J
Question 6. The emf and the internal resistance of an electrical cell are 10 V and 1 Ω respectively. The cell is connected to a resistance R = 9 Ω. How much work has to be done by the cell to move a positive charge of 1 C through the resistance R and also through the cell?
Answer: Emf of the cell (E) = 10V and internal resistance (r) = 1 Ω
Resistance in the external circuit (R) = 9 Ω
∴ Current in the circuit, \(I=\frac{E}{R+r}=\frac{10}{9+1}=1 \mathrm{~A}\)
Potential difference across the two ends of the resistance R, V = IR = 1A x 9 Ω = 9 V and lost voltage =Ir= 1 A x 1Ω = 1 V
∴ If the work done to move a positive charge of 1 C through the resistance R and the cell are W1 and W2 respectively, then
W1 = 9V x lC = 9J and W2 =1V x 1C = 1J
Question 7. The resistivity of a material is 9 x 10-6 Ω • cm. What is the length of a wire made of the same material with cross sectional area 0.5 cm2 to have a resistance of 0.5 Ω?
Answer: The cross sectional area of the wire (A) = 0.5cm2
Resistivity of the material (ρ) = 9 x 10-6 Cl • cm
Resistance (ft) = 0.5 Ω
Let the length of the wire = l
So,\(R=\rho \frac{l}{A}\) or, \(l=\frac{R A}{\rho}\)
∴ \(\begin{aligned}
l & =\frac{0.5 \times 0.5}{9 \times 10^{-6}} \\
& =27777.7 \mathrm{~cm}
\end{aligned}\) = 27777.7cm(approx).
Question 8. The ratio of the lengths, radii and resistivities of two metallic wires is 1:2 each. What is the ratio of the resistances of these two wires?
Answer: Suppose the length, the radius and the resistivity, of the first wire are , l1,r1 and ρ respectively. For the second wire, these are I2, r2 and p respectively.
\(\frac{l_1}{l_2}=\frac{r_1}{r_2}=\frac{\rho_1}{\rho_2}=\frac{1}{2}\)
Now, if the areas of cross section and the resistances of the two wires are A1, R1 and A2, R2 respectively, then
\(\frac{R_1}{R_2}=\frac{\rho_1 \frac{l_1}{A_1}}{\rho_2 \frac{l_2}{A_2}}=\frac{\rho_1}{\rho_2} \times \frac{l_1}{l_2} \times \frac{A_2}{A_1}=\frac{1}{2} \times \frac{1}{2} \times \frac{\pi r_2^2}{\pi r_1^2}\)
\(=\frac{1}{4} \times\left(\frac{r_2}{r_1}\right)^2=\frac{1}{4} \times 2^2=1\)
∴ Ratio of the resistances of the wires is 1:1.
Question 9. When an electric cell with emf E and internal resistance r is connected to a resistance 12Ω, a current of 0.5 A flows in the circuit. When it is connected to a resistance of 25 Ω, the current is 0.25 A. Calculate the values of E and r.
Answer: In the first case, current l1 = 0.5 A
∴ \(\frac{E}{R+r}=\frac{E}{12+r}=0.5\) or, E = 0.5(12 + r) •••(1)
In the second case, current I2 = 0.25 A
\(\frac{E}{25+r}=0.25\) = 0.25 or, E= 0.25(25 + r) •••(2)
By comparing equations (1) and (2), we get 0.5(12 + r) = 0.25(25 + r) or, 2(12 + r) = 25 + r
r = 1 Cl
Now from equation (1), we get E = 0.5(12 + 1) = 6.5 V
WBBSE Class 10 Revision Notes on Current Electricity
Question 10. A wire of resistance R is melted and then a new wire is made from that material Whose length is half of the first one. What is the resistance of the new wire?
Answer: Let the length of the original wire =1 and its cross sectional area =A. If the resistivity of the material of the wire = ρ, the resistance of the wire, \(R=\rho \frac{l}{A}\).
Now the length of the wire that is made by melting the original wire, l1 =1/2
Let the cross sectional area of the new wire =AX As the volume of the wire remains unchanged,
l1 A1= lA or,1/2. A1= l.A or, A1 = 2A
∴ Resistance of the new wire, \(R_1=\rho \frac{l_1}{A_1}=\rho \frac{\frac{1}{2}}{2 A}=\frac{1}{4} \times \frac{\rho l}{A}\) or, R1 = R/4
Question 11. A copper wire of diameter 5 mm is made from a piece of copper. Another wire of diameter 1 cm is made from another piece of copper wire of the same mass. What is the ratio of the resistances of the two wires?
Answer: Diameter of the first wire (d1) = 5 mm = 0.5 cm
∴ Cross sectional area of the first wire, \(A_1=\frac{\pi d_1^2}{4}\)
Diameter of the second wire (d2) = 1 cm
∴ Cross sectional area of the second wire, \(A_2=\frac{\pi d_2^2}{4}\)
Let the lengths of the wires be l1 and l2 respectively. As the masses of the wires are the same, their volumes are also the same.
∴ A1l1 = Al2
or, \(\frac{\pi d_1^2}{4} \cdot l_1=\frac{\pi d_2^2}{4} \cdot l_2\)
or, \(\frac{1}{l_2}=\left(\frac{a_2}{d_1}\right)^2=\left(\frac{1}{0.5}\right)^2=4\)
Now let the resistivity of copper =ρ and the resistances of the two wires are R1 and R2 respectively.
Then,\(R_1=\rho \frac{l_1}{A_1}\) and \(R_2=\rho \frac{l_2}{A_2}\)
\(\frac{R_1}{R_2}=\frac{\rho \frac{l_1}{A_1}}{\rho \frac{l_2}{A_2}}=\frac{l_1}{l_2} \times \frac{A_2}{A_1}=4 \times\left(\frac{d_2}{d_1}\right)^2\left[because \frac{l_1}{l_2}=4\right]\) = \(\)
∴ Ratio of the resistances of the two wires is 16: 1.
Question 12. Three resistances of values 1Ω, 2Ω and 3 Ω are connected in parallel. Calculate the equivalent resistance.
Answer: Let the equivalent resistance be R.
So, \(\frac{1}{R}=1+\frac{1}{2}+\frac{1}{3}=\frac{6+3+2}{6}=\frac{11}{6}\)
R = 6/11Ω.
Question 13. The equivalent resistances of two resistances in series and in parallel are 9Ω and 2Ω respectively. Calculate the values of the two.
Answer: Suppose the values of the two resistances are R1 and R2. The equivalent resistance of the resistances connected in series combination is 9Ω.
∴ R1 + R2 = 9 or, R2 = (9-R1) ••(1)
Again the equivalent resistance of the resistances connected in parallel combination is 2Ω.
∴ \(\frac{R_1 R_2}{R_1+R_2}=2 \quad \text { or, } \frac{R_1\left(9-R_1\right)}{9}=2\) [from equation (1)]
or, \(9 R_1-R_1^2\) = 18 or, \(R_1^2-9 R_1\) + 18 = 0 or, (R1-3)(R1-6) = 0
Now when (R1 -3) = 0, then R1 = 3 Ω and R2 = 9-3 = 6Ω
Again, when (R1-6) = 0, then R1 = 6Ω and R2 = 9-6 = 3Ω
∴ Value of these two resistances are 3Ωand 6Ω.
Question 14. A wire of resistance 10 Ω is divided into two equal parts and these two parts are put in a parallel combination. What is the resistance of the combination?
Answer: Resistance of the wire (R) = 10 Ω. As it is divided into two equal parts, resistance of each part, R1= 10/2 = 5 Ω.
Now two resistances, each of 5Ω, are connected in a parallel combination. So the equivalent resistance RP is given by
\(\frac{1}{R_p}=\frac{1}{5}+\frac{1}{5}=\frac{2}{5}\)
∴ Rp = 5/2 =2.5
Question 15. Three resistances; each of value R, are connected in series and parallel combination one after another. If the values of the equivalent resistances in the two cases are RS and respectively, what is the value of \(\frac{R_S}{R_P}\)?
Answer: Equivalent resistance of the series combination of the three resistances, RS = R + R + R = 3R and in case of parallel combination,
\(\frac{1}{R_p}=\frac{1}{R}+\frac{1}{R}+\frac{1}{R}=\frac{3}{R} \quad \text { or, } R_p=\frac{R}{3}\)
So,the required ratio, \(\frac{R_S}{R_P}=\frac{3 R}{\frac{R}{3}}=9\)
Question 16. Calculate the equivalent resistance between the points A and B in the circuit.

Answer: Equivalent resistance of the parallel combination of two resistances 6Ω and 4Ω,
\(R_1=\frac{6 \times 4}{6+4}=\frac{24}{10}=2.4 \Omega\)
Again, equivalent resistance of the parallel combination of two 8Ω and 3Ω,
\(R_2=\frac{8 \times 2}{8+2}=\frac{16}{10}=1.6 \Omega\)
Now, resistances R1 and R2 have formed a series combination.
∴ Equivalent resistance of these two resistance R1 and R2 in series id given by
R = R1+ R2 = 2.4 Ω + 1.6 Ω= 4Ω
∴ Equivalent resistance between the points A and B is 4 Ω.
Question 17. Calculate the value of I in the circuit.

Answer: Equivalent resistance R1 of the parallel combination of two resistances 6 Ω and 4 Ω,
\(\frac{1}{R_1}=\frac{1}{6}+\frac{1}{4}=\frac{2+3}{12}=\frac{5}{12}\)
∴ R1 = 12/5 = 2.4
Now equivalent resistance of two resistances R1 and 1.6 Ω connected in series combination is R = (2.4 + 1.6)Ω = 4Ω
Emf of the cell (E) = 5 V
Internal resistance (r) = 1Ω
∴ \(I=\frac{E}{R+r}=\frac{5}{4+1}=\frac{5}{5}=1 \mathrm{~A}\)
Current Electricity Topic C Heating Effect Of Electric Current, Electrical Power Synopsis
1. Joule’s law:
(1)First law: The amount of heat (H) produced in a conductor is directly proportional to the square of the current (l) passed, when the resistance (R) of the conductor and the time (t) of flow of current remain constant, i.e., H ∝ I2 [when R and t remain constant].
(2)SecondLaw: The amount of heat(H) produced in a conductor is directly proportional to the resistance (R) of the conductor, when the amount of current (I) and the time (t) of flow of current remain constant, i.e.; H ∝ R [when I and t are constants].
(3)Third Law: The amount of heat (H) produced in a conductor is directly proportional to the time (t) for which the current passes, when the amount of current (I) and resistance (R) of the conductor remain constant, i.e., H ∝ t [when R and l are constants].
2. If a current l is sent through a resistance R for a time t, an amount of heat H is produced. Then according to Joule’s law, \(H=\frac{I^2 R t}{4.2} \mathrm{cal}\)
3. Electric Fuse: Electric fuse is a safety device which is used to limit the current in an electric circuit Fuse protects the circuit and the appliances connected in that circuit from being damaged. Generally an alloy of lead and tin (Pb 75%, Sn 25%) is used to make a fuse wire.
4. Electric energy: The capacity to do work by an electrical instrument is called its electric energy.
5. SI unit of electric energy is joule (J).
6. Electric power: Electrical power (P) of any electrically driven machine is defined as its rate of consumption of electrical energy with respect to time.
\(P=V I=I^2 R=\frac{V^2}{R}\)
7. Unit of electric power in SI is watt (W). 1W is defined as the rate of consumption of electrical energy in an electrical appliance when a current of 1A passes through the appliance at a potential difference of 1V.
1 kW = 103 watt, 1 MW = 106 watt
8. kW • h: The amount of electrical energy consumed by an electrical appliance of 1 kW power in Ih is called lkW • h or BOT unit.
1 kilowatt • hour -1000 x 3600 j = 3.6 X 106 J
9. The rating of an electric bulb is 220 V -100 W. This means, that if the potential difference across the two ends of a iarnp is 220 V, it has the brightest glow and the lamp spends 100 j of electrical energy per second.
10. LED (Light Emitting Diode) lamp saves more electricity than CFL- (Compact Fluorescent Lamp). CFL produces around 7% to 9% of expended energy into visible light whereas LED produces nearly 4% to 18% of expended energy into visible light.
Current Electricity Topic C Heating Effect Of Electric Current, Electrical Power Short And Long Answer Type Questions
Question 1. Write down Joule’s laws of heating effect of electric current.
Answer:
Joule’s laws of heating effect of electric current:
In the year 1841, British physicist James Prescott Joule published three laws with respect to heating effect of electric current. These are:
1. Law of electric current: The amount of heat produced in a conductor (H) is directly proportional to the square of the current passed (I), when the resistance (R) of the conductor and the time of flow of current (t) remain constant, i.e., H ∝ I2 [when R and t remain constant].
2. Law of resistance: The amount of heat produced in a conductor (H) is directly proportional to the resistance of the conductor (R), when the amount of current (l) and the time of flow of current (t) remain constant, i.e., H oc R [when / and t are constants].
3. Law of time: The amount of heat produced in a conductor (H) is directly proportional to the time for which the current passes (t), when the amount of current (I) and resistance of the conductor (R) remain constant, i.e. H ∝t [when R and I are constants].
Question 2. A steady current l is flowing through a resistance 8 connected in a circuit for time (t). Potential difference across its two ends is V. Answer the following questions.
1. If e is the charge of a single electron, then how many electrons do flow across any cross section of the resistor during that interval of time?
2. What is the work done in moving +Q charge potential difference across the potential difference V and what is the power supplied to circuit by the source?
3. What happens to the energy expended by the source? If the work done is totally converted into heat, then how much heat is produced In the resistor?
Answer:
1. Let n numbers of electron flow across any cross section of the resistor in time t.
So net charge flow, q=ne
Hence from the equation, l = q/t We get, q = It or, ne = It or, n = lt/e
2. Work done in moving +Q charge through a potential difference V is W = VQ ….(1)
Hence the power input to the circuit by the source is P = W/t = V .Q/t
3. Energy expended by the source gets dissipated in the resistor as heat.
Now, if the amount of heat produced is H, then from the law of equivalence of heat and work, we may write
W = JH or, H = W/J •••(2)
Hence from equations (1) and (2), we get
\(H=\frac{V Q}{J}=\frac{V I t}{J}=\frac{I^2 R t}{J}\)
where J = 4.2 J/cal is the mechanical equivalent of heat.
∴ \(H=\frac{I^2 R t}{4.2} \mathrm{cal}\)
Question 3. Electric current is passing through a metallic wire. How does the amount of heat that is produced according to Joule’s law, change for the following cases?
1. Value of electric current is tripled, keeping resistance and time of flow of current unchanged.
2. Potential difference across the two ends of the wire is doubled, keeping resistance and time of flow of current unchanged.
Answer: 1. According to Joule’s law, the amount of heat produced in a metallic wire is directly proportional to the square of the current passed, when the resistance of the conductor and time of flow of current remain constant. In this case, as the value of current is tripled, heat produced becomes 32 or 9 times.
2. Amount of heat produced is given by \(H=I^2 R t=\left(\frac{V}{R}\right)^2 R t=\frac{V^2 t}{R} \quad\left[because I=\frac{V}{R}\right]\)
So, amount of heat produced in a metallic wire is directly proportional to the square of the potential difference across the two ends of the wire, when the resistance of the conductor and time of flow of current remain constant. In this case, as the value of potential difference is doubled, heat produced becomes 22 or 4 times.
Question 4. What is the change in the lieat produced jp a conductor due to flow of current in the flowing two cases?
1. Current is allowed to flow in the opposite direction
2. ac is applied instead of dc.
Answer:
1. Heat produced in a conductor due to flow of current does not depend on.the direction of the current. If the magnitude of current and time of its flow through the conductor remain constant for a fixed resistance, then amount of heat produced does not change even if the direction of current is changed.
2. When ac is sent through a conductor instead of dc, then also heat is produced. If the magnitude of current and time of its flow through the conductor remain unchanged, then amount of heat produced in the conductor remains unchanged even if ac is applied instead of dc.
Question 5. Two resistances R1 and R2 are connected In parallel. This combination is connected to a circuit having a definite potential difference. and H2 are the heat produced in them in-a time t. Prove that H1 : H2= R2 : R1.
Answer: Let us assume, thar l1 and l2 are the respective currents passing resistances R1 and R2 combination.

∴ ratio of heat produced in these two resistances in time t,
\(H_1: H_2=I_1^2 R_1 t: I_2^2 R_2 t=I_1^2 R_1: I_2^2 R_2\)
\(\begin{aligned}
& =\frac{V^2}{R_1^2} \cdot R_1: \frac{V^2}{R_2^2} \cdot R_2 \cdot\left[because I=\frac{V}{R}\right] \\
& =\frac{1}{R_1}: \frac{1}{R_2}=R_2: R_1 \text { (proved) }
\end{aligned}\)
Question 6. What is electrical power? How is it measured?
Answer:
Electrical power:
1. Electrical power of any electrically driven machine is defined as the rate of consumption of electrical energy with respect to time.
2. When an electric current l is passed through an electrically driven machine for a time period t, then electrical energy consumed, W = VIt
where V is the potential difference between the two ends of the resistance of the instrument. Therefore, electrical power is given by
\(P=\frac{W}{t}=\frac{V I t}{t}=V I\) ….(1)
Again, if R is the resistance of the instrument, then according to the Ohm’s law, we get V=IR
So from equation (1), P =V1 = IR.I = I2R …(2)
Further, l =V/R
∴ \(P=\left(\frac{V}{R}\right)^2 R=\frac{V^2}{R^2} \cdot R=\frac{V^2}{R}\) ….(3)
Therefore from equations (1), (2) and (3), the electrical power is given by P = Vl + I2R +V2/R
Question 7. How is the rating of an electrical lamp done? Or, What do you mean by voltage rating and power rating of an electric lamp?
Answer: Any electrical appliance is manufactured in such a way that at a particular potential difference, it would be most effective without getting damaged. This value of potential difference is mentioned on the body of the appliance. This is called voltage rating.
If 220 V is written on the body of an electrical lamp, it indicates that the lamp, if connected to a 220 V supply line, would be most effective without getting damaged. If the potential difference is less than 220 V, it glows with less brightness and if the potential difference is more than 220 V, this lamp may get damaged or burnt.
In addition to voltage rating, there is another rating which is called power rating. This is also known as watt rating.
Power rating or watt rating is the electrical energy that is consumed per unit time by the electric lamp when a potential difference mentioned in the voltage rating is applied to it.
Question 8. Caiculate the resistance and the maximum current passing through a 220 V- 100 W lamp.
Answer: Rating of the lamp is 220 V-100 W. This means that when a potential difference V= 220V is applied across the two ends of the lamp, power consumed, P = 100 W.
If R is the resistance of the lamp, then \(P=\frac{V^2}{R}\) or \(R=\frac{V^2}{P}=\frac{220^2}{100}=484 \Omega\)
Again if l is the maximum current passing through the lamp, then
P=VI = or, I = P/V = 100/220 = 0.454A
Question 9. Two electric lamps of 240V-60W and 240 V-100 W are connected in series combination. Which one glows brighter?
Answer: If p electrical energy is consumed when a potential difference V is applied across the two ends of a lamp of resistance R, then
\(P=\frac{V^2}{R} \quad \text { or, } R=\frac{V^2}{P}\) ….(1)
So, resistance of the first lamp, \(R_1=\frac{240^2}{60}=960 \Omega\) and resistance of the second \(R_2=\frac{240^2}{100}=576 \Omega\)
Now, if the two lamps are connected in a series combination, the same current (I) flows through each lamp.
From Joule’s law, H ∝ I2Rt
Now as the resistance of first lamp is greater than that of the second lamp, more heat is produced in this case and so the first lamp glows brighter.
Question 10. Two filament bulbs of ratings 3 V-1 W and 3 V-2 W are connected in series and in parallel combination. In each case, which bulb does glow brighter?
Answer:
Resistance of 3V-1W bulb is given by \(R_1=\frac{3^2}{1}=9 \Omega\)
Resistance of 3V-2W bulb is given by \(R_2=\frac{3^2}{2}=\frac{9}{2}=4.5 \Omega\)
Case 1: When the bulbs are connected in series, current passing through them is the same. Here, the bulb which has greater resistance produces more heat and thus glows brighter. So, 3V-1W bulb glows brighter.
Case 2: When the bulbs are connected in parallel, potential difference across the two ends is the same. Here, the bulb which has less resistance produces more heat and thus glows brighter. So, the 3 V-2 W bulb glows brighter.
Question 11. There are two lamps of ratings 240 V- 500 W and 240 V -1000 W. Which one has got a thicker filament?
Answre: If P is the energy consumed when a potential difference V is applied across the two ends of a lamp of resistance R, then \(P=\frac{V^2}{R} \quad \text { or, } R=\frac{V^2}{P}\) ……(1)
So, resistance of the first lamp, \(R_1=\frac{240^2}{500}\) = 115.2Ω and resistance of the second lamp, \(R_2=\frac{240^2}{1000}=57.6 \Omega\) = 57.6Ω
Now, if the length (l) of conductor remains unchanged, A ∝ 1/R, where A is the cross sectional area of the conductor.
As the resistance of the second lamp is less, the cross sectional area of the filament of the second lamp is more, i.e., this filament is thicker than the filament of the first one.
Question 12. What happens if a 220 V-100 W lamp is connected to a 440V line? Further, what happens if it is connected to a 160 V line?
Answer:
1. When a 220 V-100 W lamp is connected to a 440V line, current greater than the highest admissible amount of current passes through it and as a result, huge amount of heat is produced and the coil of the lamp is burnt or damaged. So, the lamp glows brightly for a very short interval of time and then gets fused.
2. If the lamp is connected to a 160 V line, current lower than the highest admissible amount of current passes through it and as a result, the lamp glows less brightly.
Question 13. Describe an electric lamp and its working principle.
Answer:
An electric lamp
The practical application of the heating effect of electric current is best demonstrated in the case of an electric lamp. Two thick conducting wires are inserted in a sealed glass bulb. A long filament made of tungsten is attached to the two ends of the wires.

Now if current is passed through it, the filament gets extremely heated and emits light due to incandescence. As the melting point of tungsten is high (3380°C), it does not melt at high temperature. Again, to prevent it from corrosion, the bulb is filled with some inert gas like argon and in some cases, also with nitrogen gas. Nowadays, the filament is made up of wolframite (an alloy of iron, manganese and tungsten) instead of tungsten.
Question 14. Why Is an electric bulb filled with an inert gas or nitrogen instead of making It free from air or filling it up with air?
Answer: When an electric bulb is made free from air, its filament vaporises at high temperature. After a few days, the filament decays, disintegrates and then finally vaporises to form a coating inside the bulb.
As a result, the transparency of the glass is reduced and brightness of the bulb is decreased. On the other hand, if the bulb is filled with air, the filament will be corroded by oxygen of air to produce a metallic oxide at high temperature and the working power of the bulb is slowly reduced.
For all these reasons, an electric bulb is filled with an inert gas or nitrogen. Only then there is no possibility of corrosion and as vaporisation is very low, the bulb works for a longer period of time.
Question 15. Describe an electric lamp and its working principle.
Answer:
An electric lamp and its working principle:
In an electric heater, there is a notched roundshaped disc made of a bad conductor of electricity, like mica or fire clay, inside a metallic container. A long wire made of nichrome (an alloy of Ni, Cr and Fe) is set inside by twisting it like a spring. If the two ends of the nichrome wire are connected to an electric line with the help of a plug, the coil becomes hot and red.

As the resistivity of nichrome is high, it can be heated more and it is not corroded easily. Further, melting point of nichrome is high. For these reasons a nichrome wire is used in a heater. There is a base of porcelain on the mica or fire clay disc on which the cooking utensils are kept so that there is no possibility of connection between the cooking utensils and electricity.
Question 16. Though heat is generated continuously in an electric heater, its temperature becomes stable after sometime. What is the reason behind this?
Answer: When electrical current is passed through a heater, heat is produced in the coil of the heater according to Joule’s law and the temperature of the coil increases. As a result, difference of temperature between the coil and the surrounding goes on increasing.
More the difference of temperature, more is the rate of radiation of heat by the coil. In this way, the temperature of the coil increases and at a particular temperature, rate of production of heat in the coil is equal to the rate of radiation of heat by the coil, in this state, the temperature of the coil stops increasing further and becomes stable.
Question 17. What happens when a small length” of the coll of the nichrorne wire of a heater is cutoff?
Answer: When the length of the nichrorne wire is reduced, resistance of the coil also gets reduced. We know that different electrical appliances in our houses remain in parallel combination and due to this parallel combination, potential difference at the two ends of different electrical appliances always remain constant.
In this case, heat produced is inversely proportional to the resistance \(\).
Now as the resistance gets reduced in this case, so more heat is produced. Therefore, when the length of the coil made of nichrorne wire is reduced, more heat is produced.
Question 18. Describe an electric Iron and its working principle.
Answer:
Electric Iron
A nichrorne wire coil is fitted within two trianguler shaped mica sheets in an electric iron. This is kept securely in an iron covering. When current is passed through the coil, it gets heated and as a result, the iron covering also gets heated.

Question 19. What is a fuse? Write down the working principle of an electric fuse. Why is It used?
Answer:
Fuse:
1. Electric fuse is an arrangement that protects a circuit from excess flow of electric current. This is a type of wire with low melting point and is made of an alloy of tin and lead (tin 75%, lead 25%). The wire is joined with a holder made of porcelain and then connected to an electric line.
2. Electric fuse is connected in a series combination with the live wire of an electric line. The melting point of a fuse wire is low and it cannot carry current of a value more than that of a fixed value. Due to any reason, when value of current in that electric line increases suddenly such that its crosses that fixed value, the wire melts to disconnect the circuit. Thus, electric fuse saves electrical appliances from getting burnt or damaged.
3. Due to joining of a fuse wire in a line, costly electrical appliances in the house (for example, refrigerator, washing machine, TV etc.) are protected against damage in case of excess flow of electric current. Above all, possibility of a fire due to burning of a line by short circuit in the house is also considerably reduced.
Question 20. Make a comparative discussion on incandescent lamp, CFL and LED with respect to saving of energy.
Answer: In an incandescent lamp, nearly 98% of consumed electrical energy is converted to heat energy and only 2% produces visible light.

CFL (Compact Fluorescent Lamp) converts 7% to 9% of consumed energy into visible light whereas LED (Light Emilting Diode) converts nearly 4% to 18% of consumed energy into visible light. So, incandescent lamp does not save energy. CFL and LED save more energy than the incandescent lamp. Though CFL and LED are costlier than incandescent lamps, these reduce electric bill by saving a good deal of electrical energy in the long run.
Question 21. What are the main advantages of LED bulb in comparison to CFL?
Answer:
LED bulb is an energy-saver compared to CFL in many ways. Some of the main advantages of LED bulb are as follows:
1. LED bulb produces very low green house gas.
2. Consumption of energy is comparatively lower in LED bulb.
[For example, whereas a CFL of 12-15 W consumes 55 kW-h of energy in a particular time interval, an LED bulb consumes only 28 kW-h of energy in the same time interval.]
3. The light of an LED bulb goes out immediately when it is switched off but the same does not happen in case of a CFL.
4. The life of an LED bulb is many times more than that of a CFL. [For example, whereas one CFL can glow easily for 10000 hours, an LED bulb gives light for nearly 50000 hours.]
5. Light energy received from an LED bulb is much more than the same from one CFL of the same wattage.
Question 22. The coil of a heater does not radiate light whereas the filament of an electric lamp radiates light. Why?
Answre: The filament of an electric lamp is made of tungsten and is very thin. Again, the coil of a heater is made of nichrome whose area of cross section is more than the filament. As a result, the resistance of the filament of lamp is comparatively greater than the coil.
Due to its high resistance, the filament is heated more and after becoming white hot, it starts radiating light. But as the resistance of the coil of the heater is less, it is not heated to that extent and does not become white hot to radiate light.
Question 23. How is the energy rating mark of an electrical machine done? There are two washing machines manufactured by two different companies. Four stars are marked on one machine and five stars are marked on another. Which one is more economical in terms of consumption of electrical energy?
Answer:
1. The same machine of different models consume different amounts of electrical energy. To show this difference in consumption of electrical energy, some stars are marked on the machines. Amount of energy consumed by the machine can be compared by the number of stars.
More the number of these stars on the body of a machine, less is the amount of energy spent by that machine as compared to other models. Therefore more stars signify more savings. This is how energy rating mark of an electrical machine is done.

2. Five stars marked washing machines is more economical in terms of consumption of electrical energy.
Question 24. When we received the electric bill, we found that 24 units were consumed last month. What is the meaning of this unit? What is its name?
Answer:
1. The meaning of consumption of 24 units in the last month is that 24 BOT units of electrical energy were spent in that house last month. That is the unit of consumed electrical power.
2. The name of the unit is Board of Trade Unit (BOT unit) or kilowatt-hour (kW-h).
Question 25. What do you mean by a short circuit?
Answer:
Short circuit
When two electrical lines of opposite nature or two opposite poles of an electrical cell are connected through very low resistance, excessive amount of current flows in the circuit. This is called short circuit. As excessive amount of heat is produced in a short circuit, sometimes leading to a fire in the circuit or a damage to the circuit components.
Question 26. Define the unit of electrical power in SI.
Answer: The unit of electrical power in SI is watt (W).
1 W power: 1W is defined as the rate of consumption °f electrical energy in an electrical appliance When a current of 1 A passes through the appliance at a potential difference of 1V.
Question 27. What is W • h and kW • h or BOT unit? These are the units of which physical quantity?
Answer:
(1)1 W-h: The amount of electrical energy consumed by an electrical appliance of 1W power in 1 h is called 1W • h.
1 kW-h or BOT unit: The amount of electrical energy consumed by an electrical appliance of lkW power in lh is called lkW• h or BOT unit.
(2)W•h and kW• h or BOT unit are the unit of electrical energy.
Question 28. Express 1W • h and1 kW • h in J unit.
Answer: 1 W•h = \(1 \frac{\mathrm{J}}{\mathrm{s}}\) = 3600 J and 1 kW • h = 1000 W.h = \(1000 \frac{\mathrm{J}}{\mathrm{s}} \times 3600 \mathrm{~s}=3.6 \times 10^6 \mathrm{~J}\)
Question 29. MW and kW are the units of which physical quantity? Express MW in terms of kW.
Answer:
1. MW and kW are the practical units of electrical power.
2. 1 MW = 106 W = 103 • 103 W = 1000 kW
Question 30. 220 V-100 W is written on the body of an electric lamp. What information does it convey?
Answer: If 200 V – 100 W is written on the body of an electric lamp, we can infer that if a potential difference of 220 V is applied across the two ends of the lamp, it will glow with maximum brightness and 100 J of electrical energy is consumed by the lamp every second.
Question 31. Why is the filament of an electric lamp made of tungsten?
Answer: Tungsten does not melt even at high temperatures as its melting point is 3380°C. In addition to this, as the resistivity of tungsten is high, the resistance of the wire is also high and thus it becomes very hot due to current flow to radiate light. Also, thin and long wire can be drawn from tungsten. For all these reasons, the filament of an electric lamp is made of tungsten.
Question 32. There are two thick conducting wires in an electric bulb at the ends of which the filament is attached. Why as light not emitted from them, even though the same amount of current passes through these wires?
Answer: As these two wires are short and their cross sectional area is large, the resistance is Sow. As the resistance is low, the heat produced is less in spite of passing the same amount of current. That is why no light is emitted.
Question 33. Why is a nichrome wire used in an electric heater?
Answer: Nichrome is an alloy of nickel, chromium and iron. As the resistivity of nichrome is high, so it can be heated more and as its melting point is also high (1400°C approx.), it will not melt so easily. Further, it does not corrode easily at high temperature. For all these reasons, a nichrome wire is used in a heater.
Question 34. What do you mean by a 10A fuse?
Answer:
10A fuse
A 10A fuse means that if an electric current of more than 10A passes through that fuse wire, the wire will become extremely hot and will melt immediately to disconnect the circuit. As a result, electric current through the circuit will stop flowing.
Word Problems on Current Electricity with Solutions
Question 35. The same amount of current is passed through one thin and another thick wire of the same material and of the same length for the same period of time. Heating of which wire will be more?
Answer: The resistance of a thin wire is more than that of a thick wire of the same material and of the same length. Now we know that, H = Therefore, the thin wire will become hotter than the thick wire when the same current is passed through it for the same period of time.
Question 36. Two wired of the same material and of the same cross sectional area are taken. The length of the first one is greater than that of the second. When the same amount of current is passed through both the wires for the same period of time, heating of which wire will be more?
Answer: Out of the two wires made of the same material and having the same cross sectional area, the one with greater length has higher resistance. Therefore the resistance of the first wire is more than that of the second wire. So, when the same amount of current is passed through both the wires for the same period of time, the first wire gets more heated.
Question 37. If same amount of electric current is passed for the same time duration through two copper wires-one thick and one thinheating of which wire will be more? Explain with reasons.
Answer: Between the two copper wire, the resistance of the thin wire is more. We know that if electric current and time of flow of current remain unchanged, then heat produced in the conductor is directly proportional to the resistance of the wire. Therefore, heat produced will be more in the case of the thin copper wife.
Question 38. Through two copper wires, one long and the other short, the same amount of current passed for the same duration of time. why the longer wire gets heated more? Explain with reasons.
Answer: The resistance of wires of the same cross section and made of the same materia! is directly proportional to the length of the wires. So, the resistance of the longer wire is more than the resistance of the shorter wire.
Now we know that the amount of heat produced in a conductor is directly proportional to the resistance of the conductor, when the amount of current and time of flow remain constant. Therefore, as the same amount of current is sent through both the wires for the same time duration, more heat is produced in the longer wire as its resistance is more. As a result, longer wire gets heated more.
Question 39. The same amount of current is passed through two wires of the same type for 1 minute and 5 minutes respectively. The heat produced in the second wire is how many times the heat produced in the first wire?
Answer: We know that the amount of heat produced in a conductor is directly proportional to the time of flow of current through the conductor when the amount of current and the resistance of the conductor remain constant.
As the same amount of current passes through these two wires of the same type and the time of flow in the second wire is 5 times the time of flow in the first wire, heat produced in the second wire is 5 times the heat produced in the first wire.
Question 40. Two wires of the same length but of different cross section and made of the same material are connected separately with a battery for a certain time. In which case is more heat produced?
Answer: We know that heat produced in a conductor,
\(H=\frac{V^\tau t}{4.2 \times R}\)
As V and t are constant, so, H ∝ 1/R
As the resistance of the thick wire is less than that of the thin wire, so more heat is produced when the thick wire is connected for the same time with the battery.
Question 41. Electricity is passing through a wire from east to west and it Is passing through another wire of the same resistance from west to east, is the amount of heat produced in these two wires during the same time duration equal?
Answer: The amount of heat produced in these two wires will be equal. This is because the heat produced in a conductor due to passing of electricity does not depend on the direction of current. Since both the wires have the same resistance and current flows through them for the same time duration, the amount of heat produced is also the same.
Current Electricity Topic C Heating Effect Of Electric Current, Electrical Power Very Short Answer Type Questions Choose The Correct Answer
Question 1. Keeping the resistance and time constant, if the amount of current is doubled, heat produced becomes
- Two times
- Four times
- Six times
- Eight times
Answer: 2. Four times
Question 2. When a number of resistances are kept in a parallel combination and the amount of current (l) and time of flow of current (t) remain constant, then the relation between the produced heat (H) and resistance (R) is
- H ∝ R
- H ∝ 1/R
- H ∝ R2
- H ∝ 1/R2
Answer: 2. H ∝ 1/R
Question 3. When the resistance (R) of a conductor and the time of flow of current (t) remain constant, then the relation between the heat produced (H) and the amount of current (l) is
- H ∝ R
- H ∝ 1/R
- H ∝ R2
- H ∝ 1/R2
Answer: 3. H ∝ R2
Wbbse Class 10 Physical Science Solutions
Question 4. When the resistance (R) of the conductor and the amount of current (I) remain constant, then the relation between the heat produced (H) and time (t) is
- H ∝ t2
- H ∝ t
- H ∝ 1/t
- H ∝ 1/t2
Answer: 2. H ∝ t
Question 5. When some resistances are kept in a series combination and the amount of current (I) and the time of flow of current (t) remain constant, the relation between the heat produced (H) and the resistance (R) is
- H ∝ R
- H ∝ R2
- H ∝1/t
- H ∝ 1/t2
Answer: 1. H ∝ R
Question 6. A fuse wire is made of
- Tin
- Lead
- An alloy of tin and lead
- An alloy of aluminium and copper
Answer: 3. An alloy of tin and lead
Question 7. Which of the following does not indicate 1 W (watt)?
- 1 V x 1A
- 1 A2 x 1Ω
- 1 V2/1Ω
- 10V x 1Ω
Answer: 4. 10V x 1 Ω
Question 8. The resistance of a 240 V – 60 W lamp is
- 480 Ω
- 960 Ω
- 240 Ω
- 720 Ω
Answer: 2. 960 Ω
Question 9. What is the emf of the cell, if 10 J of work is done in moving 2 C of charge once around an electric circuit?
- 10 V
- 5 V
- 2.5 V
- 1 V
Answer: 2. 5 V
Question 10. Star sign on an electric machine indicates
- Voltage rating
- Watt rating
- Energy rating
- Ampere rating
Answer: 3. Energy rating
Question 11. Power consumed to send a current of 2 A through a potential difference of 5 V is
- 20 W
- 5 W
- 10W
- 15 W
Answer: 3. 10W
Question 12. Power consumed to send a current of 4 A through a resistance of 2 ft is
- 64 W
- 16 W
- 24 W
- 32 W
Answer: 4. 32 W
Question 13. Power consumed to maintain a potential difference of 10 V at the two ends of a resistance of 10 ft is
- 10 W
- 100 W
- 1 W
- 1000 w
Answer: 1. 10 W
Question 14. 0.2 W • h = how many joules?
- 360
- 720
- 1080
- 1440
Answer: 2. 720
Question 15. Which of the following bulbs has the highest resistance?
- 220 V-25 W
- 220 W-60 W
- 220 V-100 W
- 220 V-40 W
Answer: 3. 220 V-100 W
Question 16. Which of the following bulbs has the lowest resistance?
- 220 V-25 W
- 220 V-60 W
- 220 V-100 W
- 220 V-40 W
Answer: 3. 220 V-100 W
Question 17. When the following four bulbs are connected in series, which one will glow brightest?
- 220 V-25 W
- 220 V-60 W
- 220 V-100 W
- 220 V-40 W
Answer: 1. 220 V-25 W
Question 18. Among the following light emitters of equal watts, which one saves maximum power?
- CFL
- LED bulb
- Incandescent bulb
- Tube light
Answer: 2. LED bulb
Question 19. Electric iron works on the principle of
- Action of magnet on electric current
- Action of electric current on magnet
- Action of electromagnetic induction
- Production of heat due to flow of current
Answer: 4. Production of heat due to flow of current
Wbbse Class 10 Physical Science Solutions
Question 20. A constant potential difference is applied at the two sides of a uniform wire. Heat produced is doubled if
- Radius of the wire is doubled
- Both length and radius are doubled
- Both length and radius are halved
- Length is doubled but radius is halved
Answer: 3. Both length and radius are halved
Question 21. The resistance of a 220 V-100 W lamp is
- 968 Ω
- 1936 Ω
- 484 Ω
- 242 Ω
Answer: 3. 484 Ω
Question 22. A current of 1 A is passed through a resistance of 10 ft for 4.2 minutes. What is the amount of heat produced?
- 500 cal
- 600 cal
- 700 cal
- 800 cal
Answer: 2. 600 cal
Question 23. 1 kW = how many W?
- 100
- 1000
- 500
- 5000
Answer:
Question 24. 1 MW = how many W?
- 102
- 106
- 103
- 104
Answer: 2. 106
Question 25. 1W • h = how many J?
- 36
- 360
- 3600
- 36000
Answer: 3. 3600
Question 26. Which one is the unit of power?
- A • s
- W • h
- A2/ ohm
- A2 • ohm
Answer: 4. A2 • ohm
Question 27. The amount of heat produced in a resistor when a current is passed through it can be found using
- Faraday’s law
- Ampere’s law
- Joule’s law
- Ohm’s law
Answer: 3. Joule’s law
Current Electricity Topic C Heating Effect Of Electric Current, Electrical Power Answer In Brief
Question 1. If H calories of heat is produced when a current of I A passes through a resistance of R Ω for time t, then write the expression for H.
Answer: \(H=\frac{I^2 R t}{4.2} \mathrm{cal}\)
Question 2. If H joules of heat is produced when a current of I A passes through a resistance of R Ω for time t, then write the expression for H.
Answer: H = I2Rt J
Question 3. What is the step taken to prevent the corrosion of the filament of an electric bulb?
Answre: To protect the filament from oxygen in the air, the air in the bulb is either removed or replaced with an inert gas like neon or argon.
Question 4. What is a gas-filled bulb?
Answer: A bulb filled with an inert gas or nitrogen (N2) is called a gas-filled bulb.
Question 5. Between the filament and the fuse wire of an electric bulb, which one has greater cross section?
Answer: The cross section of a fuse wire is more than that of a filament of an electric bulb.
Question 6. What is the material used for making the heating coil of a heater?
Answer: The heating coil of a heater is made of an t alloy of Ni, Cr and Fe known as nichrome.
Question 7. What happens when there is a hole in the mica sheet of an electric iron?
Answer: There is possibility of getting an electric shock if there is a hole in the mica sheet of an electric iron.
Question 8. Which material is used for the manufacturing of an electric fuse?
Answer: An alloy of tin and lead (tin 25%, lead 75%) is used for the manufacturing of an electric fuse.
Question 9. What is the nature of the resistivity and the melting point of a fuse wire?
Answer: Both the resistivity and the melting point of a fuse wire are very low.
Question 10. What is the unit of electric power in SI?
Answer: The unit of electric power in SI is watt (W).
Question 11. What is the voltage at which a 220 V-100 W lamp glows with maximum brightness?
Answer: A lamp of 220 V-100 W glows with maximum brightness when the potential difference across it is 220 V.
Question 12. While purchasing an electrical appliance, which of the following factors should be given maximum importance? Voltage rating, watt rating, energy rating.
Answer: While purchasing an electrical appliance, the utmost importance should be given to energy rating.
Question 13. What is the amount of current passing through a 220 V- 100 W bulb connected across a potential difference of 200 V?
Answer: Amount of current passing through the lamp = 100/220 = 0.45A
Question 14. What is the commercial unit of electrical energy?
Answer: BOT unit is the commercial unit of electrical energy.
Wbbse Class 10 Physical Science Chapter 6 Question and Answers
Question 15. There are two bulbs of 220 V-100 W and 220 V-50 W respectively. Which one has more resistance?
Answer: The 220V-50W bulb has more resistance than the 220 V-100 W bulb.
Question 16. Write full form of CFL.
Answer: CFL stands for Compact Fluorescent Lamp.
Question 17. Which harmful element is used in CFL?
Answer: Mercury is used in CFL.
Question 18. What is the full form of LED?
Answer: The full form of LED is Light Emitting Diode.
Question 19. Between CFL and LED which have long lifespan?
Answer: Between CFL and LED, LED have long lifespan.
Current Electricity Topic C Heating Effect Of Electric Current, Electrical Power Fill In The Blanks
Question 1. The unit of electrical energy in SI is ______
Answer: Joule
Question 2. The value of the mechanical equivalent, of heat is _______
Answer: 4.2 J/cal
Question 3. An electric fuse saves us from accidents and short-circuits by ____ when an excess current flows through it.
Answer: Melting
Question 4. The filament of an electric bulb is made of ______
Answer: Tungsten
Question 5. The coil of the nichrome wire in an electric iron is covered with a _____ foil.
Answer: Mica
Question 6. 1 MW = _____ kW
Answer: 1000
Question 7. The unit of electrical power is multiplied by the unit of ____ to get an unit of electrical energy.
Answer: Time
Question 8. W • h is the unit ______
Answer: Electric Energy
Question 9. In a plant producing thermal electricity, plenty of ______ gas is emitted which pollutes the environment.
Answer: CO2
Question 10. 1BOT unit = _______ W • h
Answer: 1000
Question 11. 1 W/A = 1 _____
Answer: V(volt)
Current Electricity Topic C Heating Effect Of Electric Current, Electrical Power State Whether True Or False
Question 1. In an electric cell, electrical energy gets converted into chemical energy.
Answer: False
Question 2. If the potential difference across the two ends of a wire is halved, keeping resistance and time of flow of current same, then the amount of heat produced will become 4 times.
Answer: False
Question 3. The filament of an electric bulb is made of tungsten.
Answer: True
Question 4. Lamps, electric fans and refrigerators used in our house are connected in series combination.
Answer: False
Question 5. CFL, LED lamps are more efficient than incandescent lamps.
Answer: True
Question 6. A fuse is used to avoid short circuit damage.
Answer: True
Question 7. If ac is passed through a conductor istead of dc, heat is not produced.
Answer: False
Question 8. 1 watt = 1 V x 1 A
Answer: True
Question 9. Maximum current that can passes through a 200V-25 W lamp is 1/8 A.
Answer: True
Question 10. Resistance of a 220 V-100 W lamp is 484Ω.
Answer: True
Question 11. Poisonous mercury vapour used in CFL.
Answer: True
Current Electricity Topic C Heating Effect Of Electric Current, Electrical Power Numerical Examples
1. If Q charge flows through a section under potential difference V, then the amount of electrical work done, W = QV
2. Heat evolved in a conductor of resistance R due to current flowing through it for time t, H =I2Rt (in J unit)
=\(\frac{I^2 R t}{4.2} \text { (in cal unit) }\)
3. If I be the current through resistance R and potential difference between its two ends is V then, electrical power
P = VI = I2R = \(=\frac{V^2}{R}\)
3. 1 BOT unit = 1kW • h = 3.6 x 106 J
4. If rating of a electric lamp be xV-yW, then resistance of the filament of the lamp, R = \(\frac{x^2}{y}\) unit and current through the filament, I=y/x unit.
Question 1. How much heat Is produced if a current of 0.8 A passes through a resistance of 10 Ω for 1 minute?
Answer: Resistance of the conductor (R) = 10 Ω
Electric current (I) = 0.8 A
Time of flow of current (t) = 1 minute = 60 seconds
∴ Amount of heat produced in the conductor, H = I2Rt = 0.82 X 10 X 60 = 384 J
∴ \(H=\frac{384}{4.2}\) cal = 91.42 cal 4.2
Question 2. There are two bulbs with ratings 220 V-40 W and 220V-60W.
1. Which one has greater resistance?
2. When the two bulbs are connected in series, which one glows more brightly?
3. When the two bulbs are connected in parallel, which one glows more brightly?
Answer:
1. We know, power (P) = \(\frac{V^2}{R} \text { or, } R=\frac{V^2}{P}\)
∴ Resistance of the 220 V-40 W bulb \(\left(R_1\right)=\frac{220^2}{40} \Omega\)
And resistance of the 220 V-60 W bulb 2202 \(\left(R_2\right)=\frac{22 U^2}{60} \Omega\)
2. When the two bulbs are connected in series, equal amount of current passes through each of them.
Therefore, power spent by the bulb of resistance R1( P1) = I2 R1 and power spent by the bulb of resistance R2(P2) = I2 R2
As R1 >R2, therefore P1 > P2. Again since heat produced, H α P, so the first bulb glows more brightly.
3. When the two bulbs are connected in parallel, the potential difference across the two ends of each of them is equal.
In that case, power consumed by the bulb of resistance \(R_1\left(P_1\right)=\frac{V^2}{R_1}\) and power consumed by the bulb of resistance \(R_2\left(P_2\right)=\frac{V^2}{R_2}\).
As R1 > R2, therefore, P2 > P1 and consequently, H2 > H1 [∴ H ∝ P]. Hence the second bulb glows more brightly.
Wbbse Class 10 Physical Science Chapter 6 Question and Answers
Question 3. The emf of an electric cell is 6 V and its internal resistance is 1Ω. The cell is connected to a resistance of 9Ω. How much heat is generated in the resistance in 21 seconds?
Answer: The emf of an electrical cell (E) = 6 V and its internal resistance (r) = 1Ω
Resistance in the external circuit (R) = 9 Ω
∴ Electric current in the circuit,
\(I=\frac{E}{R+r}=\frac{6}{9+1}=0.6 \mathrm{~A}[latex]
Heat produced in resistance R in t = 21 seconds is given by
[latex]H=\frac{l^2 R t}{4.2}=\frac{0.6^2 \times 9 \times 21}{4.2}=16.2 \mathrm{cal}\)

Question 4.6 bulbs of 40 W each, 4 fans of 80 W each, one TV of 100 W run daily for 5 hours in a house. What is the monthly (30 days) energy consumption of that house in BOT unit?
Answer: 1 kW • h = 1 BOT unit = 1 unit Light, fan and TV run daily for 5 hours.
∴ Power consumed = 40 x 6 + 80 x 4 + 100 = 660 W
∴ Electricity consumed in one day = 660 W x 5 h = 3300 W • h 3300
=\(\frac{3300}{1000}\) kW.h = 3.3 BOT unit.
∴ Electricity consumed in a month containing 30 days = 3.3 X 30 = 99 BOT unit.
Question 5. There are 1 light of 60 W, 2 lights of 100 W, 1 fan of 80 W and 1 heater of 1000 W in a house. If in the month of May, all lights burn for 6 h, fan runs for 12 h and heater burns for 4 h, what is the electric bill for that month? Cost of 1Bot Unit = ₹ 5
Answer: The month of May contains 31 days.
Electricity consumption per day for electric lights, fan and heater = (1 X 60 X 6 + 2 X 100 X 6 + 1 X 80 X 12
+ 1 x 1000 x 4) W – h = 6520 W • h = 6.52 kW • h = 6.52 BOT unit
∴ Electricity consumption in the month of May = 6.52 x 31 = 202.12 BOT unit
Hence electricity bill for the month of May
= 202.12 x 5 = ₹ 1010.60
Question 6. A 220 V-60 W lamp is connected to an electrical line with a potential difference of 220 V. What is the amount of current passing through the lamp? What is the resistance of the filament?
Answer: We know, watt = volt x ampere Suppose, a current l is passing through the lamp. So, 60 = 220 X I or, I = 60/220 = 0.27A
Now if the resistance of the filament of the lamp is R, we get V = IR or, 220 = 60/220 x R
\(R=\frac{220 \times 220}{60}=806.67 \Omega \text { (approx.) }\)
Question 7. What is the change of rate of production of heat, if the length and radius are both doubled by keeping the same potential difference across the two ends of a conducting wire?
Answer: Let the length of the wire = l and radius = r in the first case
∴ Cross sectional area, A = πr2
When the resistivity of the material of the wire is ρ, then resistance of the wire, \(R=\rho \frac{l}{A}\)
Let V be the potential difference across the two ends of the wire, then rate of heat production,
\(H=\frac{V^2}{4.2 R} \mathrm{cal} / \mathrm{s}\) …..(1)
Now in the second case, the length of the wire, (l1) = 2l and radius, (r1) = 2r
∴ Cross sectional area, \(A_1=\pi r_1^2=4 \pi r^2=4 \mathrm{~A}\)
Now, resistance of the wire, \(R_1=\rho \frac{l_1}{A_1}=\rho \cdot \frac{2 l}{4 A}=\frac{1}{2} \times \frac{\rho l}{A}=\frac{R}{2}\)
Hence the change of rate of production of heat,
\(H_1=\frac{V^2}{4.2 R_1}=\frac{2 V^2}{4.2 \times R}=2 \mathrm{H}\)…(2)
∴ Rate of production of heat in the second case is double of that in the first case.
Wbbse Class 10 Physical Science Chapter 6 Question and Answers
Question 8. What Is the total electric bill for 30 days if an electric lamp of 220 V-100 W is lighted everydayfor 6 hours? Cost of lBQT unit = ₹5
Answer: Electrical energy consumed in one day = 100 x 6 = 600 W • h
∴ Electrical energy consumed in 30 days = 600 X 30 = 18000 W- h = 18 kW • h = 18 BOTunit
∴ Amount of electric bill = 5 x 18 = ₹90
Question 9. Two bulbs have ratings 240 V-500 W and 240V-1000W. Filament of which bulb is thicker?
Answer: Rating of the first bulb is 240 V-500 W.
∴ Resistance of the first bulb,
\(R_1=\frac{V_1^2}{P_1}=\frac{240 \times 240}{500}=115.2 \Omega\)
Rating of the second bulb is 240 V-1000 W.
∴ Resistance of the second bulb,
\(R_2=\frac{V_2^2}{P_2}=\frac{240 \times 240}{1000}=57.6 \Omega\)
Since R ∝1/A, when other parameters are kept A constant. A is area of cross section.
Hence resistance of the second bulb is less than that of the first.
∴ Area of cross section of the second bulb is greater than that of the first i.e., the filament of th second bulb is thicker than that of the first.
Question 10. An electric cell of emf 10 V and internal resistance 1 Ω is connected with a 3Ω resistor. What is the rate of production of heat by the cell?
Answer: emf of the cell (E) = 10 V
Internal resistance, r=1Ω
Resistance in the external circuit R = 3Ω
∴ Current through the circuit, \(I=\frac{E}{R+r}=\frac{10}{3+1}=2.5 \mathrm{~A}\)
Rate of production of heat by the cell =I2(R+r) = 2.52(3+1) = 25J/s = 25/4.2 cal/s = 5.95 cal/s
Question 11. A lamp of resistance 440 Ω is used for 10 h in 220 V supply line. Find the amount of energy consume by the lamp in BOT unit.
Answer: Resistance of the lamp (R) = 440
potential difference, V = 220 V
∴ The amount of energy consumed \(=\frac{V^2}{R} \cdot t=\frac{220^2 \times 10}{440 \times 1000} \mathrm{~kW} \cdot \mathrm{h}=1.1 \mathrm{~kW} \cdot \mathrm{h}\) =1.1Bot unit
Current Electricity Topic D Electromagnetism Synopsis
1. Ampere’s swimming rule: When a man swims along a current-carrying wire in the direction of the flow of current with his face turned towards the compass needle, then the north pole of the compass needle gets deflected towards his left hand.
2. Right hand grip rule: If a current carrying wire is held in the right hand such that the thumb points towards the direction of the flow of current, then the other fingers holding the wire indicate the direction of the magnetic lines of force.
3. Fleming’s left hand rule: If the thumb, forefinger and the middle finger of the left hand is held mutually perpendicular to each other in such a way that the forefinger points Electromagnetism to the direction of the magnetic field and the middle finger to the direction of the electric current, then the thumb indicates the direction of motion of the conductor or the direction of operative force on the conductor.
4. Barlow’s wheel: Barlow’s wheel is an arrangement to demonstrate action of magnet on current. Rotation of Barlow’s wheel can be explained using Fleming’s left hand rule.Electrical energy is converted into mechanical energy in Barlow’s wheel. Barlow’s wheel rotates only when direct current (dc) is pass.
5. If the direction of either the magnetic field or the electric current is reversed, the rotation of the wheel also gets reversed.
6. Speed of rotation of the wheel increases with the increase of
(1)Flow of current through the circuit,
(2)Intensity of the magnetic field. induction is the phenomenon in which an emf is induced in a coil if there is a change in the magnetic flux linked with the coil.
7. Faraday’s laws of electromagnetic induction:
(1)First law: When there is a change in the magnetic fiux linked with a dosed coil,fan electromotive force is induced In the coil, thereby producing electric current.
(2)Second law: in case of electromagnetic induction, the magnitude of the induced emf (electromotive force) is directly proportional to the time rate of change of magnetic flux linked with the coil.
(3)Lens’s law: in case of electromagnetic induction, the direction of the induced current (or emf) is such that the current opposes the very cause which produces it.
8. Fleming’s right hand rule: Stretch the thumb, middle finger and forefinger of your right hand In a mutually perpendicular way. If the forefinger of indicates the direction of magnetic field and the thumb indicates the direction of motion of conductor, then the middle finger will indicate the direction of induced current.
9. An electric motor is an electrical instrument where an electric-carrying coil rotates around a fixed axis under the influence of a fixed magnetic field and transforms electrical energy into mechanical energy.
10. Direct current and alternating current: If an electric current is always unidirectional then it Is called dc (direct current). if the direction of an electric current reverses at regular intervals, it is called ac (alternating current).
11. Advantages of ac over dc:
(1)The voltage of ac can be steped up or steped down as required by using step up or step down transformer respectively. But dc voltage can not be steped up or steped down.
(2)Production and distribution cost of ac is lower than that of dc.
12. Dynamo: Dynamo is an instrument by which mechanical energy is converted into electrical energy by utilising the principal of electromagnetic induction.
13. For domestic supply of electricity, two wires are drawn from the overhead or underground cable of the electric supply company. One is live wire and the other is neutral wire. In general, the live wire is covered with red plastic and the neutral wire is covered with black plastic. According to recent international guidelines, live wire is now covered with brown plastic and neutral wire is covered with light blue (sky blue) plastic.
Current Electricity Topic D Electromagnetism Short And Long Answer Type Questions
Question 1. Describe Oersted’s experiment to demonstrate the magnetic effect of electric current. Or, Show the magnetic effect of electric current with the help of a simple experiment.
Answer: Scientist Hans Christian Oersted performed an experiment in 1820 to demonstrate the magnetic effect of electric current. The following experiment is done on the basis of the same.
Required instruments: Two batteries of 1.5 V each, a thick copper wire, connecting wires, battery holder, switch, magnetic needle, resistor. Experiment: Two batteries are put in the battery holder and then connection is made as shown in Fig 30 with thick copper wire, resistor (R) and v switch through connecting wires.
The entire setup is kept by the side of a table. AB is the thick copper wire. Now wire AB is placed outside the table so that wire AB does not fall off or slide. Further, wire AB is kept such that A remains in the south and B in the north.
The magnetic needle is kept below the wire AB with the switch remaining off. In this state, magnetic needle remains along the direction of north-south. Now, when the switch is flipped on, it can be seen that the north pole of the magnetic needle is deflected towards the west.

Now, the poles of the batteries are interchanged. As a result, flow of current is from B to A in the wire AB or from north to south direction. As done previously, if the magnetic needle is kept below the wire AB, it is observed that the deflection of the north pole of the magnetic needle is towards east.
Conclusion: From this experiment of Oersted, we can conclude that a magnetic field is produced around a current carrying wire and if the direction of current is reversed, the direction of magnetic field is also reversed.
Question 2. In which direction does the deflection of the north pole of a magnetic needle take place when it has been kept below a current carrying wire in Oersted’s experiment, when the direction of current flow is from:
1. South to north
2. North to south
Answer:
1. When the direction of current flow through the wire is from south to north, then the north pole of the magnetic needle gets deflected towards west.
2. When the direction of current flow through the wire is from north to south, then the north pole of the magnetic needle gets deflected towards east.

Question 3. Explain the similarity between the magnetic fields created by a bar magnet and by the flow of current in a circular conductor.
Answer: The lines of force obtained in case of a bar magnet and in case of a circular current carrying conductorshow that a circular current carrying conductor is equivalent to a bar magnet since their lines of force are similar.

If the current flows in a clockwise direction in the circular conductor, the front face of the circular conductor acts like the south pole of a bar magnet.

If seen from the opposite side, the direction of flow is anticlockwise and that side acts like the north pole.
Question 4. If a current carrying circular coll is kept in a hanging position, what is the final alignment of the coil? Why?
Answer: When a current carrying circular coil is kept hanging, it settles such that its axis is along north-south direction. If we look at the coil from the south, flow of current is clockwise and if it is seen from the north, flow of current is anticlockwise. This is because a current carrying circular coil acts like a bar magnet.

The side from which the flow of current is found to be clockwise acts as the south pole and the side from which the flow of current is found to be anticlockwise acts as the north pole.
Question 5. Write down Fleming’s left hand rule.
Answer: According to Fleming’s left hand rule, if the thumb, the forefinger and the middle finger of the left hand is held mutually perpendicular to each other in such a way that the forefinger points to the direction of the magnetic field and the middle finger points to the direction of the electric current, then the thumb indicates the direction of motion of the conductor or the direction of the force acting on the conductor.

Question 6. Explain the working principle, construction and working of a Barlow’s wheel with a diagram.
Answer:
Working principle: A force acts on an electrical conductor due to the effect of magnetic field. In Barlow’s wheel, a star-shaped metal wheel with several spokes rotates continuously due to this force. Here, electrical energy is converted into mechanical energy.
Construction: A star-shaped copper wheel with several sharp teeth is mounted on a metal rod (B) in such a way that the wheel rotates along a horizontal axis in a perpendicular plane. The metal rod is attached with the wooden base by the help of a stand (T).
Just below the wheel and above the wooden base, a container filled with mercury (M) is placed in such a way that each tip of the tooth just dips into the container while rotating. The container (M) is placed in between the two poles of a horse-shoe magnet (NS). Now the axis of the wheel and mercury are connected to a battery and switch with the help of two binding screws (S1 and S2) and conducting wires.
Working: If the switch is flipped on, electric current passes along the path shown by arrows. Now, as the direction of the magnetic field and the direction of current through the wheel are mutually perpendicular to each other, so, according to Fleming’s left hand rule, a force is applied on the tooth of the wheel which touches mercury.

Due to this force, the wheel rotates towards right. Now as the wheel starts rotating, the tip of the tooth which is in contact with mercury rotates and the circuit gets disconnected. But due to the inertia of motion, the next tooth comes in contact with mercury and again the whole process continues and the wheel of Barlow’s wheel keeps rotating continuously.
Question 7. What happens to the rotation of Barlow’s wheel If:
1. The current flow is in opposite direction?
2. The two poles of the magnet are reversed?
3. The current flow is in opposite direction and the poles of the magnet are reversed simultaneously?
4. ac is applied instead of dc?
Answer:
1. If the current flows in the opposite direction- in a Barlow’s wheel, then by keeping the direction of magnetic field the same, the wheel starts rotating in the opposite direction.
2. If the current flows in the same direction but the two poles of the magnet are reversed, the Barlow’s wheel starts rotating in the opposite direction.
3. If the current flows in the Barlow’s wheel in the opposite direction and the two poles are reversed simultaneously, the Barlow’s wheel keeps rotating in the same direction.
4. If ac is applied instead of dc, the rotation of the Barlow’s wheel stops.
Question 8. Describe the construction and working of an electric motor.
Answer:
Construction: The main parts of an electric motor are:
1. Field magnet,
2. Armature,
3. Commutator and
4. Brush.
Answer:
1. Field magnet: This is a powerful horseshoe electromagnet. The strength of the magnet may be increased as required by increasing the amount of current or the number of wounds per unit length.

2. Armature: This is a rectangular coil ABCD made of insulated copper wire. The coil is wound on a soft iron bar. The coil is kept in such a way that the two arms of the coil; AB and CD are at right angles to the magnetic field.
3. Commutator: The two ends of the armature are joined with two half-rings E and F. (This is also called split-ring device.) This is called a commutator. When the armature rotates, these two half-rings also rotate.
4. Brush: The outer surfaces of the two half-rings touch two carbon brushes B1 and B2. A battery and a switch are connected to these two brushes through conducting wires.
Working: When current is passed, it goes from A to B in the arm AB and from C to D in the arm CD. According to Fleming’s left hand rule, it is observed that the force acting on AB acts downwards and the force acting on CD acts upwards. Hence, the coil rotates anticlockwise. When the coil ABCD is vertical, then two brushes B1 and B2 come in the gap between the two halfrings E and F and connection is broken.
But due to inertia of motion, half-ring F touches brush B1 and half-ring E touches brush B2. As a result, direction of current in the arm AB is from B to A and in the arm CD, it is from D to C. In this state, according to Fleming’s left hand rule, the arm AB is deflected upwards and the arm CD is deflected downwards. In this way, current continues to flow in the circuit and the armature rotates in the same direction.
Question 9. What is the direction of magnetic field? How would you determine the direction of a magnetic field?
Answer:
1. The direction towards which a magnetic field exerts force on a small isolated north pole placed in it gives the direction of the magnetic field.
2. As it is not possible to get an isolated magnetic pole, so the deflection of the north pole of a magnetic needle is used to determine the direction of a magnetic field.
Question 10. When an electric current is passed through a conducting wire, does the wire get magnetised?
Answer: No, the wire does not get magnetised in this case. If we keep some iron filings on a piece of paper which is in contact with a conducting wire, we would find that the iron filings are not attracted by the wire.
Visual Representation of Electric Circuits
Question 11. Write down Ampere’s swimming rule.
Answer: According to Ampere’s swimming rule, if a man swims along the current carrying wire in the direction of the flow of current with his face always towards the compass needle, then the north pole of the compass needle gets deflected towards his left hand.

Question 12. Write down the right hand grip rule.
Answer: According to right hand grip rule, if a current carrying wire is held in right hand such that the thumb points towards the direction of flow of current, then the other fingers holding the wire indicate the direction of magnetic field lines.

Question 13. Can Barlow’s wheel he called a motor?
Answer: In an electric motor, electrical energy is transformed into kinetic energy. In Barlow’s wheel, electrical energy is transformed into rotational kinetic energy. Hence, Barlow’s wheel can be called an electric motor.
Question 14. How would you increase the speed of rotation of a Barlow’s wheel?
Answer: When the pole strength of the magnet or the amount of electric current increases, then the speed of rotation of Barlow’s wheel also increases.
Wbbse Class 10 Physical Science Chapter 6 Question and Answers
Question 15. When current is passed through a conducting circular coil, which magnetic pole is formed in which face?
Answer: South pole is formed on that face of the coil through which current flows in a clockwise direction, whereas north pole is formed on that surface of the coil through which current flows in an anticlockwise direction.

Question 16. What is an electric motor?
Answer:
Electric motor:
Electric motor is an electrical appliance where a current carrying coil rotates around a fixed axis under the influence of a uniform magnetic field and transforms electrical energy into mechanical energy.
Question 17. A dc motor is rotating clockwise. How is it possible to change this direction?
Answer: When the poie of the magnet is reversed or the direction of current is reversed, then the direction of rotation of a motor is also reversed, i.e., it rotaties in an anticiockwise direction.
Question 18. Write the uses of an electric motor. How would you increase the speed of rotation of the armature of an electric motor?
Answer:
1. Electric motor is used in electric fans, MP3 players, pumps, trains, rolling mills etc.
2. The speed of rotation of the armature can be increased by increasing the number of windings or increasing the current or increasing the strength of the magnetic field.
Question 19. The north Pole and the south pole of a magnetic needle are not indicated. How would you identifythe poles ofa magnetwith the help of a conducting wire and a battery?
Answer: The conducting wire is connected to the battery. Now, the wire is kept in such a way that the electric current in the wire flows from south to north. If the magnetic needle is then kept below the wire, the pole that gets deflected towards the west is the north pole. The remaining one is the south pole.
Question 20. The positive terminal and the negative terminal of a battery are not indicated How would you identify the terminals of the battery with the help of a conducting wire and a magnetic needle?
Answer: Generally the magnetic needle aligns itself in the north-south direction. The conducting wire is connected with the battery and is placed along
the north-south direction. Now, the magnetic needle is kept below the wire. If the north pole is deflected towards west, then the wire at the south end has been connected with the positive terminal and if the north pole is deflected towards east, then the wire at the, south end has been connected with the negative terminal.
Question 21. What is the function of caron in dc motor?
Answer:
The function of caron in dc motor:
Carbon brushes make contact with the commutator to supply current to the armature in on brushes motor.
Current Electricity Topic D Electromagnetism Very Short Answer Type Questions Choose The Correct Answer
Question 1. Electrical energy is produced from mechanical energy in
- Barlow’s wheel
- Electric motor
- Electric generator
- Transformer
Answer: 3. Electric generator
Question 2. Barlow’s wheel rotates when
- dc is passed
- ac is passed
- ac or dc is passed
- No current is passed
Answer: 1. dc is passed
Question 3. Barlow’s wheei is a
- Simple dc motor
- Simple ac motor
- dc generator
- ac generator
Answer: 1. Simple dc motor
Question 4. The liquid used in the arrangement of Barlow’s wheel is
- Glycerin
- Alcohol
- Mercury
- Kerosene
Answer: 3. Mercury
Question 5. Which of the following converts electrical energy into mechanical energy?
- Dynamo
- Transformer
- Electric motor
- Inductor
Answer: 3. Electric motor
Question 6. Rotation speed of the armature of a motor can be increased by
- Increasing current intensity
- Increasing number of turns of the coil
- Increasing the strength of the magnetic field
- All of these
Answer: 4. All of these
Question 7. According to Fleming’s left hand rule, the middle finger indicates the direction of
- Magnetic field
- Current
- Motion
- Force
Answer: 2. Current
Question 8. Rotation of Barlow’s wheel occurs according to
- Fleming’s left hand rule
- Ampere’s swimming rule
- Right hand grip rule
- Fleming’s right hand rule
Answer: 1. Fleming’s left hand rule
Current Electricity Topic D Electromagnetism Answer In Brief
Question 1. What can be determined with the help of Ampere’s swimming rule?
Answer: The direction of the magnetic field around a long wire carrying electric current can be determined with the help of Ampere’s swimming rule.
Question 2. When a man swims towards east, what is the direction of deflection of the north pole of the magnetic needle according to the Ampere’s swimming rule? [Suppose the current flows from west to east.]
Answer: There is no deflection of the north pole of the magnetic needle.
Question 3. According to Ampere’s swimming rule, in which direction a man should swim for the deflection of the north pole of a magnetic needle to be towards west. [Suppose the current flows from south to north.]
Answer: The man should swim from south towards north.
Question 4. In a magnetic field, when a force acts on the south poje of a magnet towards the east direction, then what is the direction of the magnetic field?
Answer: The direction of the magnetic field is towards the west.
Question 5. Would two magnetic lines of force intersect each other?
Answer: No, two magnetic lines of force wouuld never intersect each other.
Question 6. Can ac (alternating current) rotate a Barlow’s wheel?
Answer: No, an ac cannot rotate a Barlow’s wheel.
Question 7. What is magnetic flux?
Answer: The number of magnetic lines of force passing perpendicularly through a plane placed in a magnetic field is called magnetic flux.
Question 8. What type of electric current can rotate a Barlow’s wheel?
Answer: dc (direct current) can rotate a Barlow’s wheel.
Wbbse Class 10 Physical Science Chapter 6 Question and Answers
Question 9. What type of energy transformation does take place in an electric motor?
Answer: Electrical energy is transformed into mechanical energy in an electric motor.
Question 10. What type of energy transformation does take place in a dynamo?
Answer: Mechanical energy is transformed into electrical energy in a dynamo.
Question 11. Between thermal energy and hydroelectric energy which one is renewable?
Answer: Hydroelectric energy is renewable energy
Question 12. How do you determine the direction of rotation of Barlow’s wheel?
Answer: The direction of rotation of Barlow’s wheel is determined by Fleming’s left hand rule.
Question 13. When the south pole of a bar magnet is brought towards a closed coil along its axis, what is the direction of current in the front face of the coil?
Answer: The direction of current in the front face of the coil is clockwise.
Question 14. Name the device which is used to reverse the direction of current in the coil of a motor after every half rotation.
Answer: Commutator.
Question 15. State the function of a split ring in a dc motor.
Answer: Function of split ring in a dc motor is to reverse the direction of current in the coil of a motor after every half rotation.
Current Electricity Topic D Electromagnetism Fill In The Blanks
Question 1. In Fleming’s left hand rule, the forefinger indicates the direction of ______
Answer: Magnetic Field
Question 2. Magnetic lines of force emerge from the ______ pole of a magnet.
Answer: North
Question 3. When an electric current is passed through a conducting wire, a _________ field is produced around it.
Answer: Magnetic
Question 4. When current is flowing through a conductor, the conductor is not ______
Answer: Magnetised
Question 5. For a circular current carrying loop, the face of the coil in which current appears clockwise develops ______ pole.
Answer: North
Question 6. If ac is passed through a dc motor, its armature does not ______
Answer: Rotate
Question 7. Magnetic lines of forces may be _____ curve.
Answer: Closed
Current Electricity Topic D Electromagnetism State Whether True Or False
Question 1. Direaction of rotation of Barlow’s wheel is determined by right hand grip rule.
Answer: False
Question 2. A static charge may produce mgnetic field.
Answer: False
Question 3. The direction of magnetic field at any point on a magnetic line of force is along the tangent drawn on that point.
Answer: True
Question 4. In right hand thumb rule the thumb indicates the direction of flow of urrent.
Answer: True
Question 5. A circular wire carrycing current acts as a bar magnet.
Answer: True
Question 6. Magnetic field intensity inside a spiral current carrying conductor increases when an iron rod is placed inside the coil.
Answer: True
Question 7. Strength of an electromagnet can be increased as much as desired.
Answer: False
Question 8. Galvanometer is an electrical device which is based on the principle of magnetic effect of electric current.
Answer: True
Question 9. The magnitude of force applied on a long straight current carrying wire is maximum when it is placed perpendicular to the direction of magnetic field.
Answer: True
Question 10. Direction of the magnetic field around a long wire carrying current is determined by right hand thumb rule.
Answer: True
Current Electricity Topic E Electromagnetic Induction And Domestic Electrical Circuit Synopsis
1. Electromagnetic induction is the phenomenon in which an emf is induced in a coil if there is a change in the magnetic flux linked with the coil.
2. Faraday’s laws of electromagnetic induction:
3. First law: When there is a change in the magnetic fiux linked with a dosed coil, an electromotive force is induced in the coil, thereby producing electric current
Wbbse Class 10 Physical Science Chapter 6 Question and Answers
Second law: In case of electromagnetic induction, the magnitude of the induced emf (electromotive force) is directly proportional to the time rate of change of magnetic flux linked with the coil.
4. Lenz’s law: In case of electromagnetic induction, the direction of the induced current (or emf) is such that the current opposes the very cause which produces it.
5. Fleming’s right hand rule: Stretch the thumb, middle finger and forefinger of your right hand in a mutually perpendicular way. If the forefinger of indicates the direction of magnetic field and the thumb indicates the direction of motion of conductor, then the middle finger will indicate the direction of induced current.
6. Direct current and alternating current: If an electric current is always unidirectional then it is called dc (direct current). If the direction of an electric current reverses at regular intervals, it is called ac (alternating current).
7. Advantages of ac over dc:
(1)The voltage of ac can be steped up or steped down as required by using step up or step down transformer respectively. But dc voltage can not be steped up or steped down.
(2) Production and distribution cost of ac is lower than that of dc.
8. Dynamo: Dynamo is an instrument by which mechanical energy is converted into electrical energy by utilising the principal of electromagnetic induction.
9. For domestic supply of electricity, two wires are drawn from the overhead or underground cable of the electric supply company. One is live wire and the other is neutrai wire. In general, the live wire is covered with red plastic and the neutral wire is covered with black plastic. According to recent international guidelines, live wire is now covered with brown plastic and the neutral wire is covered with light blue (sky blue) plastic.
Current Electricity Topic E Electromagnetic Induction And Domestic Electrical Circuit Short And Long Answer Type Questions
Question 1. Write down Faraday’s electromagnetic induction. Or, Write down the two laws of Faraday regarding electromagnetic induction.
Answer:
First law: Whenever there is a change in the magnetic flux linked with a closed coil, an electromotive force is induced in the coil. The induced emf lasts as long as the magnetic flux continues to change.
Second law: In case of electromagnetic induction, the magnitude of the induced emf (electromotive force) is directly proportional to the rate of change of magnetic flux linked with the coil.
Question 2. State Lenz’s law. Explain Lenz’s law with tHe help of the law of conservation of energy.
Answer:
1. According to Lenz’s law, the direction of any magnetic induction effect is such as to oppose the cause of the effect.
2. Let us assume that the N pole of a bar magnet is brought near a closed coil along its axis and due to this, the direction of induced current in the coil is clockwise. That means, S pole is formed in the front face of the coil.
This S pole attracts the N pole of the bar magnet. As a result, the bar magnet accelerates towards the coil. In this case, it is found that without the supply of any external energy, kinetic energy of the magnet and also electrical energy are obtained which go against the principles of law of conservation of energy.
Therefore, when the N pole of the bar magnet proceeds along the axis of the coil, then flow of current in the coil is anticlockwise. As a result, N pole is created in the front face of the coil instead of S pole and it repulses the N pole of the bar magnet.
To move the bar magnet towards the coil along its axis, some mechanical work has to be done. This mechanical work is converted into electrical energy. Therefore, the direction of the induced emf is such that it opposes the very cause responsible for its production—this is Lenz’s law.
Question 3. Show that Lenz’s law supports the law of conservation of energy
Answer: It is known from the phenomenon of electromagnetic induction that if there is a relative motion between a magnet and a coil, a current is induced in the coil. It has been found by experiment that if the magnet is brought near or taken away from the coil, some resistance acts always.
As a result, in order to maintain a relative motion between the magnet and the coil, positive work has to be done against this resistance. This work is manifested as electrical energy in the coil. In other words, the law of conservation of energy is valid in this case.
Lenz’s law states that the direction of induced emf is such that it opposes the cause responsible for its production. Hence, Lenz’s law supports the law of conservation of energy.
Question 4. By keeping a copper ring horizontal, a bar magnet is dropped freely from a steep height through the centre of the ring, is the acceleration of the failing ring equal to or more than the acceleration due to gravity?
Answer: When the bar magnet is dropped from a steep height towards the centre of the copper ring, then magnetic flux linked to the copper ring increases, that is, there is a change of magnetic flux linked to the copper ring. As a result, an electromotive force is induced in the ring and electric current starts to flow.
The direction of induced current is such that it opposes the very cause which produces it. In this case, the fall of the bar magnet is opposed. Therefore, a repulsive force acts upward on the bar magnet so that it falls downwards with lesser acceleration than the acceleration due to gravity.
Question 5. What do you mean by direct current and alternating current?
Answer:
Direct current (dc): When the direction of an electric current is always the same, it is called dc.
Alternating current (ac): When the direction of an electric current reverses at regular intervals, it is called ac.
Change of electric current (I) is shown with change of time (t) with the help of a graph.
The direction of current does not change but in the direction of current reverses at regular intervals. So current is dc and it is ac.


Question 6. What are the advantages of ac over dc?
Answer:
The advantages of ac over dc are as follows:
1. With the help of a transformer, ac may be transformed from low voltage to high voltage (step-up transformer) and from high voitage to low voltage (step-down transformer). This advantage is not available in case of dc. i The cost of generation of ac by a generator is less than that of dc.
2. Loss of energy while transmission comparatively less in ac
Question 7. Discuss the working principle of an ac dynamo in brief.
Answer:
The working principle of an ac dynamo in brief:
The main parts of an ac dynamo are shown. The main parts are field magnet, armature, slip ring and brush. N and S are the two poles of a horse-shoe magnet, which is called a field magnet. But, generally, an electromagnet is used in place of a field magnet as a permanent magnet.
This field magnet creates a nearly constant magnetic field between the two poles, directed from north pole to south pole. ABCD is a rectangular coil made by winding insulated copper wires on a rectangular bar made of soft iron. This is called an armature.
The open ends of the armature coil are connected with two perfectly smooth round-shaped rings made of with two perfectly smooth round-shaped rings made of result, electromotive force is induced in the coil and current flows in the circuit.

When the arm AB of the coil ABCD comes down, then the direction of current in the resistance R becomes opposite to the direction of the current when the arm AB goes up. Hence, during one full rotation of the coil ABCD, the direction of current in the resistance R changes twice.
In this way, as the coil ABCD rotates with a uniform angular velocity, the direction of current in the resistance changes direction periodically at a fixed interval of time and the generated current is ac in nature.
Question 8. Discuss the working principle of a dc dynamo in brief.
Answer:
The working principle of a dc dynamo in brief:
The main parts of a dc dynamo are shown. The main parts are field magnet, armature, commutator and brush. N and S are two poles of a horseshoe magnet, which is called a field magnet. But, in general, an electromagnet is used in place of a field magnet as a permanent magnet. This field magnet creates a nearly constant magnetic field between the two poles, directed from north pole to south pole.
ABCD is a rectangular coil made by winding insulated copper wires on a soft iron cylindrical bar. This is called an armature. The open ends of the armature coil are connected with two semi-circular sheets R1 and R2 made of brass. R1 and R2 together form a commutator. When the coil rotates, the commutator also rotates. B1 and B2 are two carbon brushes.
When the coil AB rotates with the commutator, then the brushes touch the two sheets of the commutator lightly. A resistance R is connected with the brushes B1 and B2 in the external circuit. Now when the coil rotates, magnetic flux linked with the coil changes. As a result, electromotive force is induced in the coil and current flows in the circuit.
When current is passed through the coil in the direction of ABCD, then applying Fleming’s left hand rule we can say that the arm AB experiences a downward force while arm CD experiences an upward force. After crossing the vertical position, the arm AB tries to move upwards and the arm CD tries to come downwards due to inertia of motion. At that time, sheet R1 comes in contact with brush B2 and sheet R2 comes in contact with brush Br.
As a result, the current is unidirectional instead of rotating in the opposite direction. Thus, it flows through the armature. The open ends of the armature coil are connected with two semi-circular sheets R1 and R2 made of brass. R1 and R2 together form a commutator. When the coil rotates, the commutator also rotates. B1 and B2 are two carbon brushes.
When the coil AB rotates with the commutator, then the brushes touch the two sheets of the commutator lightly. A resistance R is connected with the brushes B1 and B2 in the external circuit. Now when the coil rotates, magnetic flux linked with the coil changes. As a result, electromotive force is induced in the coil and current flows in the circuit.
When current is passed through the coil in the direction of ABCD, then applying Fleming’s left hand rule we can say that the arm AB experiences a downward force while arm CD experiences an upward force. After crossing the vertical position, the arm AB tries to move upwards and the arm CD tries to come downwards due to inertia of motion.
At that time, sheet R1 comes in contact with brush B2 and sheet R2 comes in contact with brush Br. As a result, the current is unidirectional instead of rotating in the opposite direction. Thus, it flows through the resistance R in the same direction in the external circuit and dc is generate.

Question 9. Write down the difference between a dynamo and an electric motor.
Answer:
The diffrence between a dynamo and an electric motot as follows:
| Dynamo |
Electric motor |
| 1. Mechanical energy is converted into electrical energy in a dynamo |
1. Electrical energy is converted into mechanical energy in an electric motor. |
| 2. Dynamo is constructed on the basis of the principle of electromagnetic induction. |
2. Electric motor is constructed on the basis of the magnetic action of electric current. |
Question 10. Describe, in brief, the principle generation of thermal electricity.
Answer:
Principle generation of thermal electricity
In a simple turbine, some blades are attached to the end of a rod which is connected to the coil of a dynamo. If the turbine is rotated, the coil also rotates and electrical energy is generated
In a thermal power station, water is boiled by burning coal or any other fuel and transformed into steam. This steam rotates the blades of a turbine and consequently, the armature coil of the dynamo rotates.

As a result, there is a change in the magnetic flux linked with the coil and an electromotive force is induced. Thermal electricity is generated in this way.This steam is again condensed in a condenser and is sent back by Rankine cycle to the place where it is heated.
Question 11. Briefly describe the principle of generation of hydroelectricity
Answer:
The principle of generation of hydroelectricity:
In a hydroelectric power plant, water is stored in reservoirs. Now this water stored at a great height is allowed to fall downwards. As the mass of water falls down, its potential energy decreases and kinetic energy increases.
If a blade of a turbine is kept in this falling water, it rotates and the armature coil of the dynamo also rotates with it. As a result, there is a change in the magnetic flux linked with the coil causing an electromotive force to be induced and thus hydroelectricity is generated.
Huge amount of CO2 is generated in a thermal power plant which pollutes the environment. For this reason, many countries of the world are now giving importance to hydroelectricity. At present, 16% of the total generated electricity is hydroelectricity.
Question 12. Explain the arrangement of electric fine in a house with diagram.
Answer:
The arrangement of electric fine in a house with diagram:
For domestic supply of electricity, two wires are drawn from the overhead or underground cable of the electric supply company. One is live wire and the other is neutral wire. In general, live wire is covered with red plastic and neutral wire is covered with black plastic. According to international guidelines, live wire is now covered with brown plastic and neutral wire is covered with light blue (sky coloured) plastic.
Live wire and neutral wire are first connected with the meter. Now, the live wire is passed through the main fuse and neutral wire is taken directly from the meter and connected with the main switch. The line of the house can be switched ‘on’ and ‘off’ as per requirement with the help of the main switch.
As the potential difference between the earth and the neutral line is not always zero, an iron rod is inserted inside the ground and that rod is connected with the conducting wire. This arrangement is called earthing and the wire used for this is called the earth wire. The earth wire is green or yellow in colour.
The earth wire is taken to the switch board meant for electrical appliance (iron, refrigerator, table fan etc.) through the main switch. Live wire goes to different switch boards from the main switch through several branch lines. Points are made, according to requirement,in the switch board for use of fan, tv, lamp etc.
Live wire is connected to each switch board through, a fuse. Next, the wiring of the house is done from switch board through live wire and neutral wire. Lamp, refrigerator, electric fan etc., in the house remain in a parallel combination.

Question 13. What is earthing? Why is it done? How earthing is done?
Answer:
Earthing:
1. Connecting an electrical circuit or an electrical appliance to the earth through a conducting wire is known as earthing.
2. Due to any defect in the electrical connection, the metal coating of the appliance may get electrified. There is a possibility of danger due to that. If anybody standing on the ground touches that appliance, the person may get an electric shock. Earthing is done to avoid that possibility.
3. For earthing, a conducting rod made of metal is driven into the ground. In the household circuit, there is an extra wire (earthing wire) connected to this rod. In case of a three-pin socket, the comparatively thicker hole is connected to this wire.
The resistance of this wire is very low There is a connection between the metal coating of the electrical appliance and the earthing wire. The potential of the earth is zero. So, if anyhow the coating of the instrument is electrified, that electricity goes to the earth directly. Therefore the possibility of getting a shock is minimised.
Question 14. Explain the functioning of a switch and a main switch.
Answer:
The functioning of a switch and a main switch:
Switch is an arrangement in an electrical circuit by which the flow of electric current can be made ‘off’ and ‘on’. Switch is generally made of ebonite and is fixed on the respective board attached on the surface of the wall. Generally, a 5A switch is used in the circuit of electric fan, bulb, tubelight and a 10A or 15A switch is used in the circuit of pump, refrigerator, heater.

Main switch is an arrangement which may be made ‘off’ and ‘on’ according to necessity to disconnect or connect the electric line of the house from the main electric supply line. As a result, the flow of electric current in homes can be stopped or started according to one’s will.

Question 15. What are electromagnetic induction and induced current?
Answer:
Electromagnetic induction and induced current
If there is change of magnetic flux associated with a closed coil, an electric current originates in the coil. This phenomenon is called electromagnetic induction and the current flowing in the coil is called induced current.
Question 16. What is induced electromotive force?
Answer:
Induced electromotive force
If there is a relative motion between a magnetic field and a conductor, then an electromotive force originates in the conductor which is called induced electromotive force.
Question 17. The north poles of two identical bar magnets are kept at the same height from the centres and at right angles to the planes of two identical closed circular conducting coils. Now both the bar magnets are brought near the coils to the same height, first one rapidly and the second one slowly. In which case is the induced current more
Answer: Induction of current is more in the first coil because the first magnet was brought rapidly and thus, the rate of change of magnetic flux linked with the first coil is more than the second one. As result, the amount of induced electromotive force is also more. Hence, induction of current is more in the first case.
Question 18. Along the axis of a circular coil made of wire, a cylindrical bar magnet Is kept. The fragment rotates about the axis. Is there any induction of current in that coil?
Answer: When the magnet rotates about the common axis of the magnet and the circular coil, there is no change in the flux linked to the coil. So, no electromotive force is induced in the coil. As a result, no current is induced in the coil.

Question 19. A bar magnet is kept along the axis of a circular conducting coil. Now both the coil and the bar magnet are moved in the same direction with the same velocity. Is there any induction of current in the coil?
Answer: When both the coil and the bar magnet are moved in the same direction with the same velocity, there is no relative velocity between them. As a result, there is no change in the magnetic flux linked to the coil. Therefore, no electromotive force is induced and thus, no current is induced in the coil.
Question 20. What is a dynamo? How many types of dynamo are there? Name them.
Answer:
Dynamo
1. Dynamo is a device in which mechanical energy is converted into electrical energy by utilising the principle of electromagnetic induction.
2. It is of two types: (1) ac dynamo and (2) dc dynamo.
Question 21. What is the combination in which electric lamp, electric fan, refrigerator etc. are connected with the electric line of house? Why?
Answer: Electric lamp, electric fan, refrigerator etc. are connected in a parallel combination with the electric line of a house because any one of them can be switched on or off as per requirement and the potential difference across the two ends of every electrical appliance remains constant.
Question 22. Write down the formation of a three-pin plug in brief.
Answer:
Formation of a three-pin plug in brief
In a three-pin plug, there are three metal pins. The top one is longer and thicker than the other two which are of equal size. The two lower pins are connected with the live wire and the neutral wire respectively. Earthing is done with the thicker and longer one.
Question 23. Why do electrical appliances like electric bulb, heater, iron etc. work both with ac and dc?
Answer: The electrical appliances like electric bulb, heater, iron etc. work on the principle that heat is produced when electric current passes through a conductor. Heat produced in a conductor due to the flow of electric current depends on the resistance, amount of current and time of flow of current, which are independent of direction. Hence, produced heat does not depend on ac or dc.
Question 24. Why is the earth pin of a three-pin plug made longer and thicker?
Answer: The earth pin of a three-pin plug is made thicker so that this pin can not be inserted in the other two holes by mistake. Also, the earth pin is made longer so that the end of this pin gets connected with the socket before the other two pins and thereby reducing the chance of the user getting electric shock.
Question 25. Write down the construction of a socket in brief.
Answer:
Construction of a socket in brief
A socket is an important component of an electric circuit in which a plug is inserted. There are three holes in a socket. The lower two holes are connected with live wire and neutral wire whereas the upper, bigger hole is connected with earth wire. This socket is connected with the respective board.
Question 26. Why is the front portion of every pin of a three-pin plug split?
Answer: The front portion of every pin of a three-pin plug is split length-wise so that proper connection can be made with the socket. Without proper connection between the pin and the socket, sparks may fly due to loose connection which can be very dangerous.

Current Electricity Topic E Electromagnetic Induction And Domestic Electrical Circuit Very Short Answer Type Questions Choose The Correct Answer
Question 1. The colour of a live wire is
- Brown
- Black
- Green
- Sky blue
Answer: 1. Brown
Question 2. The colour of a neutral wire is
- Brown
- Black
- Green
- Skyblue
Answer: 2. Black
Question 3. Which of the following helps in the conversion of low voltage ac into high voltage ac?
- Converter
- Rectifier
- Step-up transformer
- Step-down transformer
Answer: 3. Step-up transformer
Question 5. Electromotive force (emf) is induced in a closed coil, when magnetic flux linked with the coil
- Purely increases
- Remains constant
- Purely decreases
- Either increases or decreases
Answer: 4. Either increases or decreases
Question 6. The direction of the induced emf in a circuit is determined by which law?
- Lenz’s law
- Faraday’s first law of electromagnetic induction
- Faraday’s second law
- Ampere’s swimming rule
Answer: 1. Lenz’s law
Question 7. No emf is induced in a closed coil when magnetic flux linked with the coil
- Decreases
- Is constant
- Increases
- Changes
Answer: 2. Is constant
Question 8. In electromagnetic induction, the induced emf in a coil is independent of
- Change in the flux
- Time
- Resistance of the coil
- All of these
Answer: 3. Resistance of the coil
Question 9. Lenz’s law is a consequence of the law of conservation of
- Charge
- Momentum
- Energy
- Mass
Answer: 3. Energy
Question 10. The instrument which converts mechanical energy into electrical energy is
- Dynamo
- Motor
- Galvanometer
- Barlow’s wheel
Answer: 1. Dynamo
Question 11. The basic principle in which dc generator works is
- Magnetic effect of electricity
- Electromagnetic induction
- Chemical effect of electricity
- Heating effect of electricity
Answer: 2. Electromagnetic induction
Question 13. In domestic circuit switch is connected in the
- Live wire
- Neutral wire
- Earth wire
- Fuse wire
Answer: 1. Live wire
Question 14. The thicker and longer pin of the three-pin plug is connected to
- Live wire
- Neutral wire
- Live or neutral wire
- Earth wire
Answer: 4. Earth wire
Question 15. The main fuse is connected in
- Live wire
- Neutral wire
- Both the live and earth wire
- Earth wire
Answer: 1. Live wire
Current Electricity Topic E Electromagnetic Induction And Domestic Electrical Circuit Answer In Brief
Question 1. When the north pole of a bar magnet is brought towards a closed coil along its axis, what is the direction of current in the front face of the coil?
Answer: The direction of current in the front face of the coil is anticlockwise.
Question 2. When the north pole of a bar magnet is taken away from a closed coil along its axis, what is the direction of current in the front face of the coil?
Answer: The direction of current in the front face of the coil is clockwise.
Question 3. When the south pole of a bar magnet is taken away from a closed coil along its axis, what is the direction of current in the front face of the coil?
Answer: The direction of current in the front face of the coil is anticlockwise.
Question 4. With the help of which instrument, alternating current (ac) of high voltage can be transformed into ac of low voltage and vice versa?
Answer: With the help of a transformer, alternating current (ac) of high voltage can be transformed into ac of low voltage and vice versa.
Question 5. In which direction does current pass through a live wire?
Answer: Current through a live wire passes from the direction of electricity supply station to the direction of electrical appliances.
Question 6. In which direction does current pass through a neutral wire?
Answer: Current through a neutral wire passes from the direction of electrical appliances to the direction of electricity supply station.
Question 7. What is the colour of live wire used in electric line in our homes?
Answer: The colour of live wire used in electric line in our homes is brown.
Question 8. What is the colour of neutral wire used in electri c line in our homes?
Answer: The colour of neutral wire used in electric line in our homes is sky blue.
Question 9. By means of which gadget, the electric line in our homes can be switched on and off?
Answer: With the help of the main switch, the electric line in our homes can be switched on and off.
Question 10. What is the colour of earthing wire?
Answer: The colour of earthing wire is either green or green with yellow stripe.
Question 11. Which wire is connected with the top big hole of a three pin plug?
Answer: Earthing wire is connected with the top big hole of a three pin plug.
Question 12. Which type of wire is generally used in house wiring?
Answer: Copper wire is generally used in house wiring.
Question 13. Which metal wire is generally used for the transmission of electricity to a distant place?
Answer: Aluminium wire is generally used for the transmission of electricity to a distant place.
Question 14. What is electromagnetic induction?
Answer: Electromagnetic induction is the phenomenon in which an emf is induced in a coil if there is a change in the magnetic flux linked with the coil.
Current Electricity Topic E Electromagnetic Induction And Domestic Electrical Circuit Fill In The Blanks
Question 1.If there is change of _____ linked with a closed coil, an emf is induced in the coil.
Answer: Magnetic Flux
Question 2. The cost of production generator is _____ than that of ac.
Answer: More
Question 3. When ac with high voltage is sent from a power station, loss of energy due to transmission of electricity is _____
Answer: Less
Question 4. When magnetic flux in a closed coil remains _____ no emf is induced in the coil
Answer: Stationary
Question 5. In ______ mechanical energy converted into electrical energy.
Answer: Generator
Question 6. Frequency of dc current is ______
Answer: Zero
Question 7. ac can be converted into dc by ______
Answer: Rectifier
Question 8. Lenz’s law actually follows the principle of conservation of _______
Answer: Energy
Question 9. In our domestic supply line frequency of ac is ______ Hz.
Answer: 50
Question 10. According to international convention, colour of the insulation of the neutral wire is ______
Answer: Blue
Current Electricity Topic E Electromagnetic Induction And Domestic Electrical Circuit State Whether True Or False
Question 1. Lenz’s law supports the law of conservation of energy.
Answer: True
Question 2. Induced emf is produced in a closed coil when it is placed in a magnetic field.
Answer: False
Question 3. If the north pole of a bar magnet moves towards a closed coil, the direction of induced current is anticlockwise in the front side of the coil.
Answer: False
Question 4. Magnitude of induced emf can be calculated from Faraday’s first law.
Answer: False
Question 5. In electroplating ac is used.
Answer: False
Question 6. Production cost of ac is less than that of dc.
Answer: True
Question 7. dc can be converted to ac by using converter.
Answer: True
Question 8. EMF produced by a generator is directly proportional to its number of turns
Answer: True
Question 9. Switches are connected in neutral wire.
Answer: False
Question 10. Fuse is always connected in the beginning of the circuit in live wire.
Answer: True
Question 11. According to colours of insulation are brown for live, light blue of neutral and green for earth
Answer: False
Current Electricity Miscellaneous Type Questions Match The Column
Question 1.
| Column A |
Column B |
| Equivalent resistance in Ω |
1. 48 |
| Current flowing through the circuit in A |
2. 3 |
| Heat produced in the circuit for 2 seconds in J |
3. 96 |
| Power consumed in W |
4. 4 |
Answer:
Equivalent resistance in Ω: 2. 3
Current flowing through the circuit in A: 4. 4
Heat produced in the circuit for 2 seconds in J: 3. 96
Power consumed in W: 1. 48
Question 2.
| Column A |
Column B |
| 6 x 108 esu of charge is equal to |
1. 0.1 W • h |
| 360 joule is equal to |
2. 4 x 10-8 Ω • m |
| 2.5 x 107 S • m-1 is equal to |
3. 107 W |
| 10 MW is equal to |
4. 0.2 C |
Answer:
6 x 108 esu of charge is equal to: 4. 0.2 C
360 joule is equal to: 1. 0.1 W • h
2.5 x 107 S • m-1 is equal to: 2. 4 x 10-8 Ω • m
10 MW is equal to: 3. 107 W







![]()

![]()
![]() as the functional group is acetone (CH3COCH3)
as the functional group is acetone (CH3COCH3)

![]()
![]()






















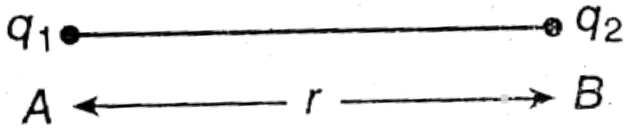
 Question 10. What is electric charge? How many types of charges are there? Name them.
Question 10. What is electric charge? How many types of charges are there? Name them.