Unit 3 Laws Of Motion Chapter 1 Newton Law Of Motion Long Answer Type Questions
WBBSE Class 11 Long Answer Questions on Newton’s Laws
Question 1. A metal ball is suspended from the roof of a train by a string. When the train is in uniform motion, will the string remain vertical? What happens to the string if the train accelerates?
Answer:
Given
A metal ball is suspended from the roof of a train by a string. When the train is in uniform motion
When the train moves in a straight line with a uniform speed, the speeds of the string and the ball are the same as that of the train. Hence the string remains vertical.
- When the train accelerates, the speed of the attached end of the string increases but the metal ball, because of its inertia of rest, tends to resist the change.
- As a result, the string shifts obliquely backward with respect to the vertical, i.e., shifts opposite to the direction of acceleration.

Question 2. When a bullet is fired at a glass window, it creates a round hole on the glass pane without shattering the whole pane. But when we throw a stone, the glass pane shatters. Explain.
Answer:
Given
When a bullet is fired at a glass window, it creates a round hole on the glass pane without shattering the whole pane. But when we throw a stone, the glass pane shatters.
The interatomic forces between the molecules of a substance are much higher in solids compared to that in liquids and gases. If a part of a solid is set in motion, the neighboring parts also try to be in motion due to this interatomic force. If the speeds of different parts are different, the body breaks.
- In this case, the speed of the bullet is much higher than the speed of the piece of stone thrown. The bullet, therefore, remains in contact with the glass for a very short time.
- In this short period of time, only the part of the glass that comes in direct contact with the bullet gets into a state of motion while the other parts remain static due to inertia.
- Hence, only a small bullet-sized round hole is formed in the glass pane. But the stone, because of its lower velocity, remains in contact with the glass for a longer period and can impact its momentum to a large area of the glass. Thus, within this time, other parts also acquire different speeds and so, the glass shatters.
Question 3. A body is kept on the floor of a train. When the train accelerates forward, the body gains a backward acceleration. Which force is responsible for the backward acceleration?
Answer:
Given
A body is kept on the floor of a train. When the train accelerates forward, the body gains a backward acceleration.
A train moving with an acceleration forms a noninertial frame of reference. A pseudo force develops in such a frame. The pseudo force in this instance acts opposite to the direction of acceleration, and it causes a backward acceleration of the body kept on the floor.
Detailed Explanation of Newton’s First Law
Question 4. Two bodies of equal weight W are suspended exactly from the midpoints of two stretched, horizontal strings. The two strings make angles α1 and α2 (α1 > α2), with the horizontal. In which of the strings, would the tension be higher? Explain.
Answer:
Given
Two bodies of equal weight W are suspended exactly from the midpoints of two stretched, horizontal strings. The two strings make angles α1 and α2 (α1 > α2), with the horizontal. In which of the strings, would the tension be higher
As the weights are suspended from the midpoints, the tensions in the two sides are the same. If it is T, then the resultant of upward components of T = T sinα + T sinα = 2 T sinα. At equilibrium, the resultant has to balance the weight.
∴ W = \(2 T \sin \alpha \quad \text { or, } T=\frac{W}{2 \sin \alpha} .\)
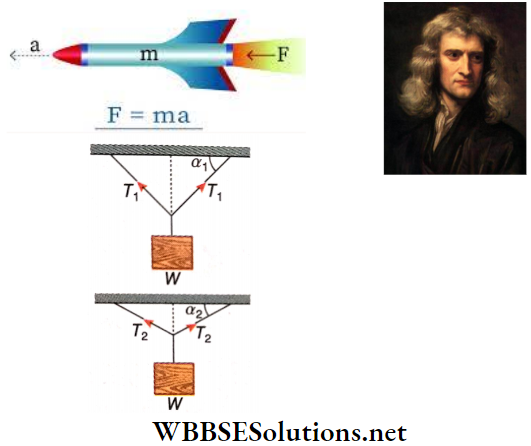
Thus, when the value of a is small, the value of T is large.
In this case, \(T_1=\frac{W}{2 \sin \alpha_1} \text { and } T_2=\frac{W}{2 \sin \alpha_2}\)
As \(\alpha_1>\alpha_2, T_2>T_1\), i.e., the second string has a higher tension.
Question 5. A force is applied on a particle in a lift moving upward with an acceleration aα. Can Newton’s second law of motion be applied to describe the motion of this particle?
Answer:
Given
A force is applied on a particle in a lift moving upward with an acceleration aα.
A lift moving with an acceleration forms a non-inertial frame of reference. Newton’s second law of motion is not applicable in a non-inertial frame of reference directly. In such frames, a pseudo force acts on the particle in addition to the real force applied. Newton’s second law of motion can be applied if the expressions for both the real and the pseudo forces are known.
Question 6. Masses 1 kg and 3 kg move towards each other, due to their mutual attraction; no other external force acts on them. When their velocity of approach is 2 m · s-1, the velocity of the center of mass is 0.5 m · s-1. What will be the velocity of the center of mass of this system when the velocity of approach is 3 m · s-1?
Answer:
Given
Masses 1 kg and 3 kg move towards each other, due to their mutual attraction; no other external force acts on them. When their velocity of approach is 2 m · s-1, the velocity of the center of mass is 0.5 m · s-1.
The two masses may be considered to be constituents of a single system, and there is no external force acting on the system. As per the first law of motion, the velocity of the system remains constant. The velocity of a system is the velocity of its center of mass. So the velocity of the center of mass will continue to be 0.5 m · s-1.
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
Understanding Newton’s Third Law: Long Answer Format
Question 7. Is it possible, for a person sitting inside a car at rest, to make the car move by pushing it from inside?
Answer:
The described push is an internal force, acting only inside the car. For causing the motion of the car on the road, no external force has been applied. By Newton’s first law of motion, in the absence of an external force, the car remains at rest.
Question 8. A meteorite conserved burns in air. Is the momentum of the meteorite conserved?
Answer:
A meteorite conserved burns in air.
Momentum is conserved even when the meteorite bums in the air. The meteorite loses some momentum which gets transferred to the surrounding air particles and to the gases that the meteorite is transformed into. The total momentum is conserved because there is no external force.
Question 9. A man of mass m is standing on a rope ladder attached to and kept vertically by a balloon of mass M at rest in the air. The man starts climbing up the rope ladder at a constant speed v with respect to the ladder. What will be the velocity of the balloon at that time?
Answer:
Given
A man of mass m is standing on a rope ladder attached to and kept vertically by a balloon of mass M at rest in the air. The man starts climbing up the rope ladder at a constant speed v with respect to the ladder.
Since there is no external force, the momentum of the system will be conserved.
Initially, the momentum of the system was zero. When the man starts climbing up, let the velocity with which the balloon moves downwards be V with respect to the ground.
The Upward velocity of the man with respect to the rope ladder = v.
Hence, upward velocity with respect to the ground = v- V.
Hence, from the conservation law of momentum, 0 = m(v-V)-MV or, m(v-V) = Mv
∴ V = \(\frac{m v}{m+M}\).
Question 10. When a body is thrown upwards, the magnitude of its momentum decreases at first and then increases. Does this defy the law of conservation of momentum?
Answer:
Given
When a body is thrown upwards, the magnitude of its momentum decreases at first and then increases.
The momentum of a body is conserved only when there is no external force acting on the body. In the case under discussion, the force of gravity is acting on the body as an external force. So the change in momentum of the body does not signify any violation of the law of conservation of momentum.
Question 11. In a game of tug of war, if both the parties exert a force of Tdyn from either side, what will be the tension in the rope?
Answer:
Given
In a game of tug of war, if both the parties exert a force of Tdyn from either side
Tension in the rope will be T dyn
Long Answer Questions on Force and Acceleration
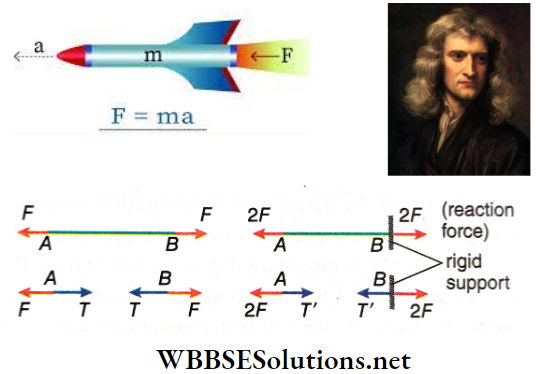
Question 12. Two persons pull a rope by its two ends, exerting equal but opposite forces F. In another situation, one end of the rope is tied to a rigid support and a person pulls it with a force of 2F. In which case will the tension in the rope be greater?
Answer:
Given
Two persons pull a rope by its two ends, exerting equal but opposite forces F. In another situation, one end of the rope is tied to a rigid support and a person pulls it with a force of 2F
Tension in the rope in the second case will be greater i.e., T’ = 2F, whereas the tension, in the first case is T = F.
Question 13. A uniform rope of length L rests on a smooth plane. One end of the rope is pulled with a force F. What is the tension in the rope at a distance l from that end?
Answer:
Given
A uniform rope of length L rests on a smooth plane. One end of the rope is pulled with a force F.
Let the mass of the rope be M.
Hence, mass per unit length of the uniform rope = \(\frac{M}{L}\).
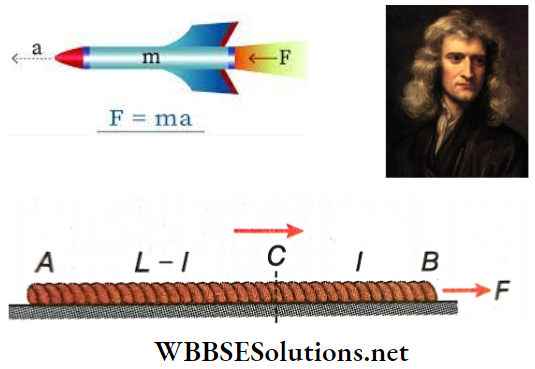
Length of the part BC of the rope =l, length of the part AC = L-l,
and mass of the part AC = \(\frac{M}{L}\)(L- l).
Acceleration of the rope due to the force F is a = \(\frac{F}{M}\).
Let the tension at C be T.
Hence, part AC of the rope gains acceleration due to the tension T.
∴ T = mass of AC x acceleration of the rope
= \(\frac{M}{L}\)(L- l). \(\frac{F}{M}\) = \(\frac{F}{L}\)(L-l)
WBBSE Class 11 Sample Questions on Motion
Question 14. A boat is floating on still water. A man walks from one end of the boat to the other. What will be the displacement of the boat?
Answer:
Given
A boat is floating on still water. A man walks from one end of the boat to the other.
The initial momentum of the man-boat system is zero, and no external force acts on it. So, as the person walks from one end of the boat to the other, the boat moves backward as per the law of conservation of momentum.
Let the length of the boat = L, t = time required by the man to move from one end to the other. In this time, let the distance which the boat moves back by be x.
Hence, the average velocity of the boat = \(\frac{x}{t}\), and the average velocity of the man with respect to the bank = \(\frac{L – x}{t}\).
Let the mass of the boat be M and that of the man be m.
So, \(\frac{m(L-x)}{t}-\frac{M x}{t}=0\), from the conservation law of momentum.
∴ Displacement of the boat, x = \(\frac{m L}{m+M}\)
Question 15. A jet plane usually flies at a considerable height but a propeller plane files at a low altitude, Expalin?
Answer:
Given
A jet plane usually flies at a considerable height but a propeller plane files at a low altitude
A jet plane functions on the principle of conservation of linear momentum. Gas is ejected from the rear end of the plane at a high speed and thus the plane moves forward.
- At high altitudes, though the density of air is low, it is sufficient for providing oxidants to the fuel of the plane. Moreover, as the density of air is low at higher altitudes, the production of heat due to friction is reduced.
- On the other hand, a propeller plane exerts a backward thrust on the air by rotating its blades. The reaction causes the plane to move. This thrust depends on the density of air and therefore this plane flies at lower altitudes where the air density is higher.
Question 16. Which principle of conservation can explain the flight of a rocket?
Answer:
Given
The flight of a rocket can be explained by the principle of conservation of momentum. The initial momentum of the rocket, along with its fuel, is zero.
- The burnt fuel is ejected backward, through a hole, with some momentum. This momentum should be canceled by an equal and opposite momentum so that the net momentum is still zero.
- This requirement for an equal and opposite momentum is responsible for the forward motion of the rocket.
Real-Life Examples of Newton’s Laws: Long Answer Questions
Question 17. Two persons are facing each other on two boats floating on still water. They are holding the two ends of a rope. When either of them or both pull the rope, then the boats meet at the same point whatever the pull on the rope may be. Give reasons for this. Is there any difference in the times taken by the boats to meet, for different forces applied on the rope?
Answer:
Given
Two persons are facing each other on two boats floating on still water. They are holding the two ends of a rope. When either of them or both pull the rope, then the boats meet at the same point whatever the pull on the rope may be
Let the masses of the first and the second men together with their respective boats be m1 and m2.
Let the force applied by one of them on the rope be F.
Acceleration of the 1st boat = \(\frac{F}{m_1}=a_1\), and that of the 2nd boat = \(\frac{F}{m_2}=a_2\).
Let the displacements of the two boats before meeting be Sj and s2 respectively. They meet after a time t.
∴ \(s_1 =\frac{1}{2} a_1 t^2=\frac{1}{2} \frac{F}{m_1} t^2 ;\)
∴ \(s_2=\frac{1}{2} a_2 t^2=\frac{1}{2} \frac{F}{m_2} t^2\)
Hence, \(\frac{s_1}{s_2}=\frac{m_2}{m_1}\).
Thus for any value of the force F, the meeting point of the two boats divides the initial distance in the ratio of m2: m1. Hence, the contact point is a fixed point.
Now, \(t^2=\frac{2 s_1 m_1}{F}=\frac{2 s_2 m_2}{F}\)
As s1m1= s2m2, time remains unchanged for a fixed F. But the time taken would be different for different values of F.
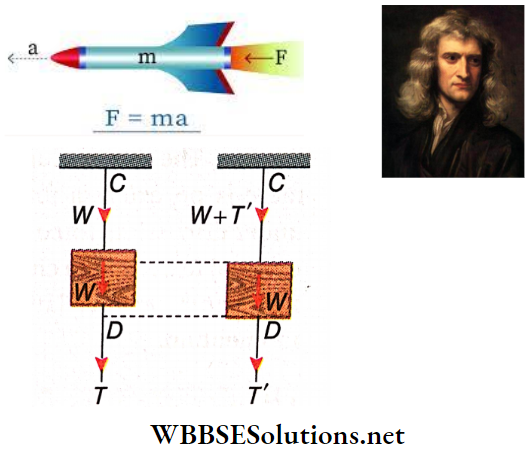
Question 18. A block of mass m is suspended from the ceiling using a string C. Another piece of string D is fitted to the other end of the block. When the string D is pulled suddenly, it snaps. But when D is pulled slowly, the string C snaps. Explain.
Answer:
Given
A block of mass m is suspended from the ceiling using a string C. Another piece of string D is fitted to the other end of the block. When the string D is pulled suddenly, it snaps. But when D is pulled slowly, the string C snaps.
The application of a sudden pull (T) on string D does not disturb the block due to its inertia.
- The extension of the string C is negligible as the block practically remains at the same place. In this case, only the weight W acts on C and the string does not snap. On the other hand, the force T may be sufficient for D to snap.
- But when string D is slowly pulled, the applied force acts on the whole system. So, the tension T’ on the string D becomes less than the tension T’ + W on C as the block also starts coming down. The tension, being higher, causes the string C to snap.
Question 19. Rocket is the only means of travel in space—explain.
Answer:
Rocket is the only means of travel in space
Usually, a vehicle applies some force on its surrounding objects; the reaction force exerted by those objects on the vehicle is actually responsible for its motion.
- On the other hand, let a system of two comparable masses be initially at rest so that the total momentum is zero. Let one of the masses be somehow detached, and allowed to move with a certain momentum in a definite direction.
- Then the other mass would acquire an equal momentum in the opposite direction.
- Hence, the total momentum will still be zero, i.e., be conserved. In this way, the masses can acquire motion without any external force, and without the aid of the surrounding objects.
- A rocket in space utilizes this principle. The spaceship and the fuel constitute the two different masses. As the fuel is ejected backward, the spaceship successfully moves forward. It should be noted that, in space,
- No external force can be applied on a vehicle, and
- The vacuum in outer space can provide no surrounding object. A rocket is, therefore, the only means of travel in outer space.
Question 20. Two balls of different masses have the same volume. If the air resistance on both the balls is the same, prove that, when dropped from the same height, the heavier ball reaches the ground earlier.
Answer:
Given
Two balls of different masses have the same volume. If the air resistance on both the balls is the same
Let the mass of one of the balls be m, and the air resistance acting upward, be f.
Hence, total downward force on the ball, F = mg – f
∴ Acceleration, = \(\frac{F}{m}=\frac{m g-f}{m}\)
= \(g-\frac{f}{m}\)
Thus, from the equation, the ball with more mass has a smaller value of \(\frac{f}{m}\) and therefore a greater acceleration. So, it reaches the ground earlier.
Applications of Newton’s Second Law: Long Answers
Example 21. A balloon of mass M, carrying some sand is descending with an acceleration a. When 1/4th of the sand is emptied out of the balloon, the balloon descends with a uniform velocity. Find the initial mass of sand in the balloon.
Answer:
Given
A balloon of mass M, carrying some sand is descending with an acceleration a. When 1/4th of the sand is emptied out of the balloon, the balloon descends with a uniform velocity.
Let the initial mass of sand in the balloon be m and the upward thrust of air on the balloon or air resistance be F.
Hence, the equation of motion initially is, (M + m)g-F = (M+ m)a…(1)
After 1/4th of the sand is emptied out, the equation changes to \(\left[M+\left(m-\frac{m}{4}\right)\right] g-F=0\)…(2)
From (1) and (2), \((M+m) g-\left[M+\left(m-\frac{m}{4}\right)\right] g=(M+m) a\)
or, \(\frac{m}{4} g=(M+m) a\)
or, \(m=\frac{M a}{\left(\frac{1}{4} g-a\right)}=\frac{4 M a}{g-4 a}\)
Question 22. Two forces, F1 and F2 are applied at the two ends of a rope of length l. F2 > F1 and they are oppositely directed. What will be the force that will act at a point at a distance x from one of the extreme ends of the rope?
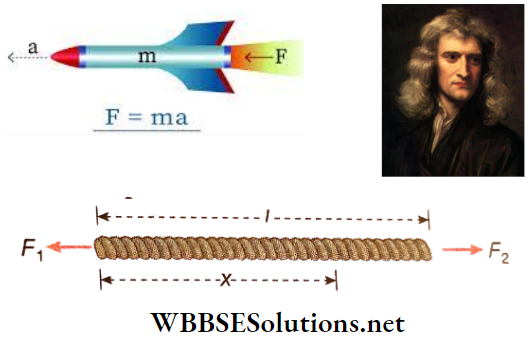
Answer:
Given
Two forces, F1 and F2 are applied at the two ends of a rope of length l. F2 > F1 and they are oppositely directed.
Let the mass of the rope be M.
∴ Mass per unit length of the rope = \(\frac{M}{l}\).
∴ Mass of length x of the rope = \(\frac{Mx}{l}\)
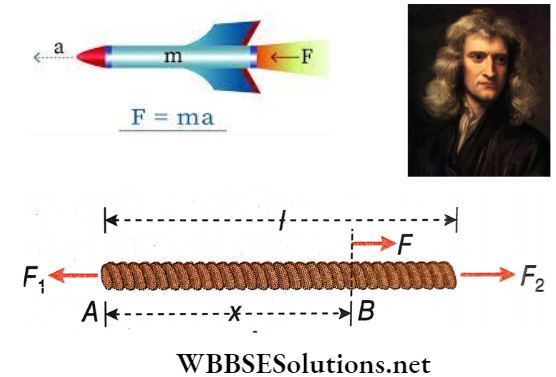
Now if the acceleration of the entire rope is a, F2 – F1 = Ma …(1)
Suppose, the force acting at a point, say B, at a distance x from the left end is F. Therefore, the equation of motion of the length AB of the rope is, \(F-F_1=\frac{M x}{l} \cdot a\)…(2)
From (1) and (2) we get, \(F-F_1=\frac{x}{l}\left(F_2-F_1\right) \quad \text { or, } F=\frac{l-x}{l} F_1+\frac{x}{l} F_2 \text {. }\)
Question 23. If you stand on the floor it exerts an upward reaction on you. Then why don’t you go up?
Answer:
Here, two forces act on the body:
- The downward force of weight due to earth’s attraction and
- Reaction of the ground. They are equal and opposite.
So, resultant force = 0, and there is no motion of the body
Question 24. A body of weight W1 is suspended from the ceiling of a room by a rope of weight W2. What is the force exerted by the ceiling on the body?
Answer:
Given
A body of weight W1 is suspended from the ceiling of a room by a rope of weight W2.
In this case, a total weight ( W1 + W2) is suspended from the ceiling. It is the force exerted on the ceiling. Hence, the ceiling exerts a reaction force of (W1+ W2) on the body.
Question 25. A ball of mass m is suspended by a light thread attached to the hook of a car At the precise moment when the car began to descend a smooth inclined plane under gravity, the thread was perpendicular to the plane. When the car begins to descend, what angle will the thread make with the plane? The angle of inclination = a.
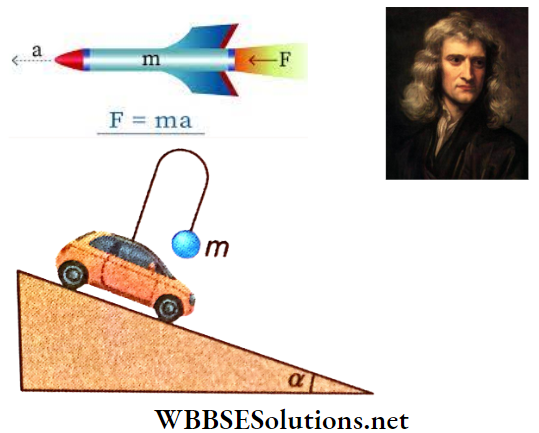
Answer:
Given
A ball of mass m is suspended by a light thread attached to the hook of a car At the precise moment when the car began to descend a smooth inclined plane under gravity, the thread was perpendicular to the plane. When the car begins to descend
As the car descends along the inclined plane under gravity, its acceleration is gsinα.
The acceleration of the ball is also gsinα.
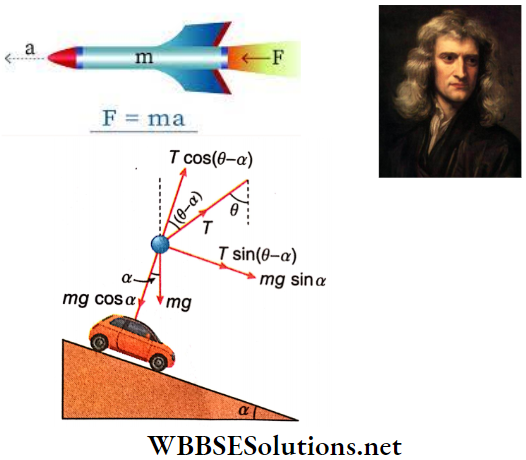
Suppose, while descending, the thread makes an angle θ with the vertical.
So, the equation of motion of the ball is, Tsin(θ-α) + mgsinα = m · gsinα
or, sin(θ-α) = 0 or, θ = α
Therefore, the thread will be inclined at an angle α with the vertical, i.e., it will still be perpendicular to the inclined plane.
Question 26. A body of mass 1 kg initially at rest explodes and breaks into three fragments of masses in the ratio 1:1:3. The two pieces of equal mass fly off perpendicular to each other with a speed of 30 m/s each. What is the velocity of the heavier fragment?
Answer:
Given
A body of mass 1 kg initially at rest explodes and breaks into three fragments of masses in the ratio 1:1:3. The two pieces of equal mass fly off perpendicular to each other with a speed of 30 m/s each.
Let the 1kg mass break into three fragments with masses m1, m2, and m3 and their velocities are v1, v2, and v3 respectively.
Here, \(m_1+m_2+m_3=1 \mathrm{~kg}\)
As \(m_1: m_2: m_3=1: 1: 3\)
∴ \(m_1=m_2=0.2 \mathrm{~kg}, m_3=0.6 \mathrm{~kg}, v_1=v_2=30 \mathrm{~m} / \mathrm{s}\)
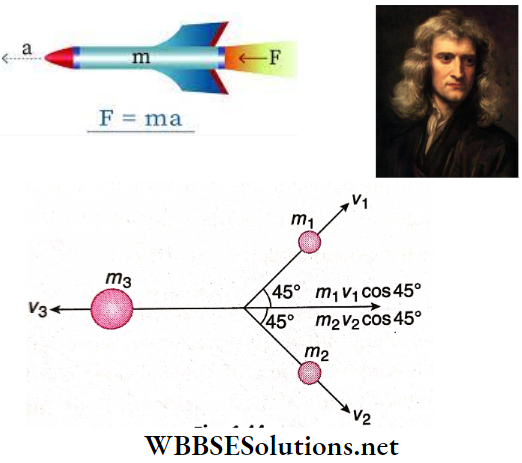
Applying the law of conservation of linear momentum along the horizontal direction, \(m_3 v_3=m_1 v_1 \cos 45^{\circ}+m_2 v_2 \cos 45^{\circ}\)
or, \(0.6 v_3=0.2 \times 30 \times \frac{1}{\sqrt{2}}+0.2 \times 30 \times \frac{1}{\sqrt{2}}\)
or, \(v_3=\frac{2 \times 0.2 \times 30}{0.6} \times \frac{1}{\sqrt{2}}=\frac{20}{\sqrt{2}} \mathrm{~m} / \mathrm{s}=14.14 \mathrm{~m} / \mathrm{s}\)