Superposition Of Waves Beats
WBBSE Class 11 Superposition of Waves Overview
Beats
When two progressive waves with equal or nearly equal amplitudes, but with a slight difference in then- frequencies, move in the same direction and superpose in a region of space, the amplitude and intensity of the resultant wave increase and decrease periodically. This phenomenon is known as beats.
Principle Of Superposition
Beats Experiment: Let two tuning forks of the same frequency be kept on a hollow box. If the two forks are vibrated simultaneously, nothing special is observed in the emitted sound. Now, one arm of one of the forks is waxed slightly. Thus the frequency of this fork decreases slightly.
The two forks are vibrated again simultaneously keeping them close to each other. It is observed that the loudness of the emitted sound rises and falls periodically. This phenomenon is known as beats.
Characteristics Of Beats:
- Two progressive waves with equal or nearly equal amplitudes, but differing slightly in frequencies are allowed to superpose. The resultant amplitude rises and falls periodically and beats are formed.
- Rise and fall of resultant amplitude result in rise and fall of intensity. This corresponds to periodic increments and decreases of the loudness for sound waves and the brightness for light waves.
- If the resultant intensity goes to maximum (or minimum) n times per second, then n is called the number of beats per second or beat frequency. It is equal to the difference in the frequencies of the component waves, i.e., if n1 and n2 are the component frequencies (n1>n2), the beat frequency is n = n1-n2.
- The persistence of hearing of the human ear is \(\frac{1}{10}\)s. This means that if more than one sound of the same type comes to our ear within \(\frac{1}{10}\)s, we cannot distinguish them. So, if the beat frequency is more than 10 Hz, we cannot feel the effect of rise and fall of the intensity. We may conclude that to feel the effect of beats, the two superposing waves should have a frequency difference of less than 10 Hz.
- For example, if two sound waves of frequencies 200 Hz and 205 Hz superpose, the beat frequency is (205 – 200) = 5; that can be easily perceived.
- For sound waves, two sources having a frequency difference of less than 10 Hz are often realized in practice. But for light waves, individual frequencies are of the order of 1015 Hz in the visible range. So, a frequency difference of less than 10 Hz is practically impossible to observe. For this reason, the formation of beats is a phenomenon useful for sound waves but has no physical significance for light waves.
The above discussions provide the precise definition of beats:
Beats Definition: The periodic rise and fall of the loudness of the resultant sound wave, produced by the superposition of two progressive sound waves of equal amplitude but of slightly different frequencies, is called beats.
Graphical Representation Of Beat Formation: Let two sound waves of nearly equal amplitudes be incident on each other with the initial phase. For example, we take two progressive waves of frequencies 10 Hz and 8 Hz.
- They superpose in a region of space with equal amplitude and with the same initial phase. Shows the displacements y1 and y2 of the two waves A and B, respectively. The resultant displacement is shown by C. From the principle of superposition, the algebraic sum (y1 + y2) denotes the resultant displacement y at any instant.
- When t = 0, \(\frac{1}{2}\)s and 1 s, the two waves are in the same phase. So, the resultant y is the maximum. On the other hand, at t = \(\frac{1}{4}\)s and \(\frac{3}{4}\)s, the two waves are in opposite phase and the resultant y is nearly zero, i.e., minimum.
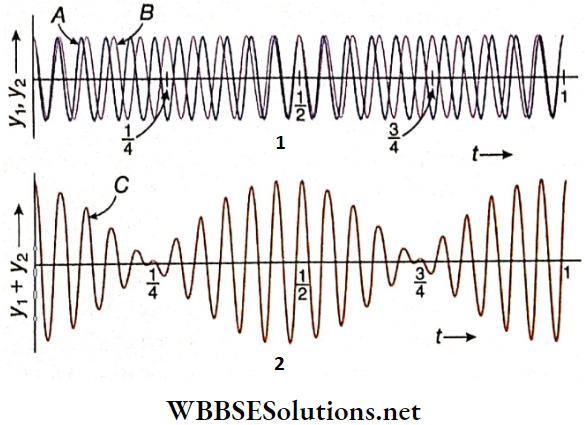
- So in a span of 1 s, rise or fall in the intensity of sound occurs twice, i.e., two beats are formed per second.
- This means that \(\frac{1}{2}\)s time is required to produce a single beat. Moreover, shows that the time interval between two maximum amplitudes = \(\frac{1}{2}\) – 0 = \(\frac{1}{2}\)s; the time interval between two minimum amplitudes = \(\frac{3}{4}\) – \(\frac{1}{4}\) = \(\frac{1}{2}\)s.
So, it may be concluded that a beat is formed between two consecutive maxima or between two consecutive minima of the resultant amplitude.
Mathematical Analysis Of Beats: Let us consider two sound waves having frequencies n1 and n2 (n1> n2) traveling through a medium. They have equal amplitude A and equal initial phase. The displacements of the two waves at any point is \(y_1=A \sin 2 \pi n_1 t \text { and } y_2=A \sin 2 \pi n_2 t\)
According to the principle of superposition, the resultant displacement is
y = \(y_1+y_2=A\left(\sin 2 \pi n_1 t+\sin 2 \pi n_2 t\right)\)
= \(2 A \sin \left(2 \pi \frac{n_1+n_2}{2} t\right) \cos \left(2 \pi \frac{n_1-n_2}{2} t\right)\)
or, y = \(A^{\prime} \sin 2 \pi n t\)….(1)
where n = \(\frac{n_1+n_2}{2}\)…(2)
and \(A^{\prime}=2 A \cos 2 \pi \frac{\left(n_1-n_2\right) t}{2}\)…(3)
The factor sin2πnt in equation (1) implies that the resultant wave is a sound wave with frequency n = \(\frac{n_1+n_2}{2}\) = average of the component frequencies.
Besides, equation (3) shows that the amplitude of the resultant wave (A’) is not a constant, rather it varies with time t. As the intensity of sound is proportional to the square of the amplitude, this intensity also varies with time.
The amplitude changes with time between a maximum and a minimum. As a result, the intensity of sound periodically increases or decreases. This is known as beats.
Beat Frequency:
1. If \(\cos 2 \pi \frac{\left(n_1-n_2\right) t}{2}=0\), the intensity of sound becomes zero, i.e., A’ = 0.
In this condition, no sound is heard.
Here, \(\cos 2 \pi \frac{\left(n_1-n_2\right) t}{2}=0\)
This corresponds to \(2 \pi \frac{\left(n_1-n_2\right) t}{2}=\frac{\pi}{2}, \frac{3 \pi}{2}, \frac{5 \pi}{2}, \cdots\)
or, t = \(\frac{1}{2\left(n_1-n_2\right)}, \frac{3}{2\left(n_1-n_2\right)}, \frac{5}{2\left(n_1-n_2\right)}, \cdots\)
So, the time interval between two consecutive minima is
\(t_0 =\frac{3}{2\left(n_1-n_2\right)}-\frac{1}{2\left(n_1-n_2\right)}=\frac{1}{n_1-n_2}\)= \(\frac{5}{2\left(n_1-n_2\right)}-\frac{3}{2\left(n_1-n_2\right)}\)
i.e, the number of minima per second = \(\frac{1}{t_0}=n_1-n_2\).
2. If \(\cos 2 \pi \frac{\left(n_1-n_2\right) t}{2}= \pm 1\), the intensity of sound becomes maximum, i.e., A’ = ±2A (maximum amplitude). In condition, aloud sound is heard.
Here, \(\cos 2 \pi \frac{\left(n_1-n_2\right) t}{2}= \pm 1\)
This corresponds to \(2 \pi \frac{\left(n_1-n_2\right) t}{2}=0, \pi, 2 \pi, \cdots\)
or, t=0, \(\frac{1}{n_1-n_2}, \frac{2}{n_1-n_2}, \frac{3}{n_1-n_2}, \cdots\)
The time interval between two consecutive maxima is \(t_0^{\prime}=\frac{1}{n_1-n_2}-0=\frac{1}{n_1-n_2}=\frac{2}{n_1-n_2}-\frac{1}{n_1-n_2} ;\)
i.e., the number of maxima per second
= \(\frac{1}{t_0^{\prime}}=n_1-n_2\)
So, the number of beats per second or the beat frequency = n1 ∼ n2 = magnitude of difference in frequencies of the two superposed waves.
It is to be noted that if the amplitudes A and B of the two superposed waves are slightly different, then for the resultant wave, maximum amplitude = (A+B) and minimum amplitude = (A-B)≠0
Definition of Superposition of Waves for Class 11
Superposition Of Waves
In this case, the sound intensity never falls to zero, that is, absolute silence is never achieved. Rather, a feeble sound is heard between every two loud maxima.
Conditions For The Recognition Of Beats: The beat frequency must be less than 10 Hz, i.e., more than one beat must not be formed within a time span of \(\frac{1}{10}\)s. Otherwise, beats cannot be separately recognized by the ear and a continuous sound is heard. This is known as a beat note.
The amplitudes and intensities of die two superposed waves must be equal or nearly equal. If they are widely different, the intensity variation between the maxima and the minima of the resultant wave is much less, and it becomes hard to recognize the resultant beats.
Application Of Beats:
Determination Of An Unknown Frequency: To determine the unknown frequency of a source of sound, a few tuning forks of known frequencies are taken. Let it be the unknown frequency of a source of sound and n1, n2, n3, ….. be the known frequencies of the standard tuning forks.
At first, die tuning forks, one by one, are vibrated simultaneously with the source of sound to detect whether recognizable beats are formed or not. Suppose, a particular tuning fork of frequency nf is able to form such beats. If the beat frequency is N, then
Either, n-ni = N i.e., n = ni + N;
or, n1 – n = N i.e., n = ni – N
Examples of Superposition of Waves in Real Life
- For example, if a tuning fork of frequency 200 Hz forms 4 beats per second, the unknown frequency of the source will be either 200 4 = 204 Hz or 200 – 4 = 196 Hz
- Now, the correct value between 204 Hz and 196 Hz is to be ascertained. For this purpose, a small drop of wax is put or a small piece of paper is pasted on one arm of the tuning fork.
- This results in a slight decrease in the frequency of the fork. So the beat frequency will also change slightly, it may be less or more than its previous value.
- If the number of beats is reduced, the unknown frequency will be 196 Hz. On the other band, if the number of beats is increased, the unknown frequency wall he 204 Hz.
Tuning Of A Musical Instrument: A musical instrument can be tuned with die help of beats. Let a particular string of a musical instrument be brought to unison with a standard source of musical sound. The string and the standard source are vibrated simultaneously.
The length and the tension of the string are gradually varied until beats are formed and recognized. It indicates that the frequency of the string is now within 10 Hz of the frequency of the standard source. Then a little more variation of the length and the tension would bring the string tuned exactly with the source. No more formation of beats would confirm this tuning.

Superposition Of Waves Beats Numerical Examples
Mathematical Representation of Superposition of Waves
Example 1. When two tuning forks are vibrated simultaneously, 4 beats are heard per second. The second fork is waxed slightly and then 6 beats are heard per second when both are vibrated again simultaneously. Find out the frequency of the second fork. Given, that the frequency of the first fork is 510 Hz.
Solution:
Given
When two tuning forks are vibrated simultaneously, 4 beats are heard per second. The second fork is waxed slightly and then 6 beats are heard per second when both are vibrated again simultaneously.
Let n2 be the frequency of the 2nd tuning fork.
We know, the number of beats per second = difference in frequencies of the two tuning forks.
If n2 > 510 Hz , then n2 -510 = 4 or, n2 = 514 Hz. When the 2nd fork is waxed, its frequency becomes less than 514 Hz. Then the number of beats per second decreases. So, 6 beats will not be formed per second.
If n2 < 510 Hz , then 510- n2 = 4 or, n2 = 506 Hz . The frequency decreases further for waxing. So, 6 beats can be formed per second. This means that the value n2 = 506 Hz matches with the given problem.
| Class 11 Physics | Class 12 Maths | Class 11 Chemistry |
| NEET Foundation | Class 12 Physics | NEET Physics |
Example 2. 2 of length 78 cm and 80 cm of a sonometer are kept at the same tension. A tuning fork produces 4 beats per second with each of them. Find out the frequency of the tuning fork.
Solution:
Given
2 of length 78 cm and 80 cm of a sonometer are kept at the same tension. A tuning fork produces 4 beats per second with each of them.
Fundamental frequency of the 78 cm long wire, \(n_1=\frac{1}{2 \times 78} \sqrt{\frac{T}{m}}\)
Fundamental frequency of the 80 cm long wire, \(n_2=\frac{1}{2 \times 80} \sqrt{\frac{T}{m}}\)
∴ \(\frac{n_1}{n_2}=\frac{80}{78} ; \text { clearly, } n_1>n_2\)
If n is the frequency of the tuning fork, then \(n_1-n=4 \text { and } n-n_2=4\)
∴ \(n_1-n_2=8 \text { or, } \frac{n_1}{n_2}-1=\frac{8}{n_2}\)
or, \(\frac{80}{78}-1=\frac{8}{n_2} \text { or, } \frac{2}{78}=\frac{8}{n_2} \text { or, } n_2=312 \mathrm{~Hz}\)
∴ n = \(312+4=316 \mathrm{~Hz}\).
Example 3. A 75 cm long stretched string is tuned with a tuning fork. If the length of the string is reduced by 3 cm, it produces 6 beats with the tuning fork per second. Find out the frequency of the timing fork.
Solution:
Given
A 75 cm long stretched string is tuned with a tuning fork. If the length of the string is reduced by 3 cm, it produces 6 beats with the tuning fork per second.
Frequency of the tuning fork, n = fundamental
Frequency of the 75 cm long string = \(\frac{1}{2 \times 75} \sqrt{\frac{T}{m}}\)
Again, the fundamental frequency of the (75 – 3) cm or 72 cm long string, \(n^{\prime}=\frac{1}{2 \times 72} \sqrt{\frac{T}{m}}\)
∴ \(\frac{n}{n^{\prime}}=\frac{72}{75} ; \text { clearly, } n<n^{\prime}\) .
As 6 beats are produced per second, we have,
n’ – n = 6
or, \(\frac{n^{\prime}}{n}-1=\frac{6}{n}\)
or, \(\frac{75}{72}-1=\frac{6}{n} \text { or, } \frac{3}{72}=\frac{6}{n} \text { or, } n=144 \mathrm{~Hz}\).
Question 4. A tuning fork of unknown frequency produces 5 beats per second with another tuning fork. The second fork can cause a closed organ pipe of length 40 cm to vibrate in its fundamental mode. The beat frequency decreases when a small amount of wax is dropped on the first fork. Find out the frequency of the first tuning fork. Given, the speed of sound in air = 320 m · s-1.
Solution:
Given
A tuning fork of unknown frequency produces 5 beats per second with another tuning fork. The second fork can cause a closed organ pipe of length 40 cm to vibrate in its fundamental mode. The beat frequency decreases when a small amount of wax is dropped on the first fork.
Length of the organ pipe =40 cm = 0.4 m
So, its fundamental frequency = \(\frac{V}{4 l}=\frac{320}{4 \times 0.4}=200 \mathrm{~Hz}\)
∴ The frequency of the 2nd tuning fork, n2 = 200 Hz.
Clearly, the frequency of the 1st tuning fork is n1 = (200+ 5) Hz or, (200-5) Hz;
The wax would decrease the frequency of the 1st fork. This decreased frequency must be closer to n2 = 200 Hz, as the beat frequency also decreases. So, the frequency of the 1st tuning fork is n1 = 205 Hz.
Example 5. Two tuning forks A and B, when vibrated simultaneously, produce 5 beats per second. 40 cm and 40.5 cm lengths of a sonometer wire, kept in the same tension, are tuned with the forks A and B, respectively. Find out the frequencies of the two tuning forks.
Solution:
Given
Two tuning forks A and B, when vibrated simultaneously, produce 5 beats per second. 40 cm and 40.5 cm lengths of a sonometer wire, kept in the same tension, are tuned with the forks A and B, respectively.
According to the question, \(n_A=\frac{1}{2 \times 40} \sqrt{\frac{T}{m}} \text { and } n_B=\frac{1}{2 \times 40.5} \sqrt{\frac{T}{m}}\)
∴ \(\frac{n_A}{n_B}=\frac{40.5}{40}=\frac{81}{80} ; \text { clearly, } n_A>n_B\)
As the number of beats per second is 5, we have, \(n_A-n_B=5 \quad \text { or, } \frac{n_A}{n_B}-1=\frac{5}{n_B} \quad \text { or, } \frac{81}{80}-1=\frac{5}{n_B}\)
or, \(\frac{1}{80}=\frac{5}{n_B} \quad \text { or, } n_B=400 \mathrm{~Hz}\)
∴ \(n_A=n_B+5=400+5=405 \mathrm{~Hz}\)
WBBSE Class 11 Revision Notes on Superposition
Example 6. Two wires are tied on a sonometer. The tensions, lengths, diameters, and densities of the materials of the two wires are in the ratio 8: 1, 36: 35, 4: 1, and 1:2, respectively. Find out the number of beats produced per second when the two wires are vibrated simultaneously. Given, the frequency of the wires are vibrated simultaneously. The frequency of the wire emitting a tone of higher pitch is 360 Hz.
Solution:
Given
Two wires are tied on a sonometer. The tensions, lengths, diameters, and densities of the materials of the two wires are in the ratio 8: 1, 36: 35, 4: 1, and 1:2, respectively.
Mass per unit length of a wire, m = \(\frac{\pi d^2}{4} \rho\), where d = diameter of the wire and ρ = density of the material.
∴ Fundamental frequency, n = \(\frac{1}{2 l} \sqrt{\frac{T}{m}}=\frac{1}{2 l} \sqrt{\frac{4 T}{\pi d^2 \rho}}=\frac{1}{l d} \sqrt{\frac{T}{\pi \rho}}\)
So, for the two wires, \(\frac{n_1}{n_2}=\frac{l_2}{l_1} \cdot \frac{d_2}{d_1} \cdot \sqrt{\frac{T_1}{T_2} \cdot \frac{\rho_2}{\rho_1}}=\frac{35}{36} \times \frac{1}{4} \times \sqrt{\frac{8}{1} \times \frac{2}{1}}=\frac{35}{36}\)
Clearly, n2 >n1 so the pitch of the tone emitted by the 2nd wire is higher.
Here, n2 = 360 Hz
∴ \(\frac{n_1}{360}=\frac{35}{36} \quad \text { or, } n_1=350 \mathrm{~Hz}\)
∴ Number of beats per second =360-350 = 10.
Example 7. Two tuning forks A and B, when vibrated simultaneously, produce 5 beats per second. The forks produce resonances respectively with 36 cm and 37 cm long air columns of a tube closed at one end. What are the frequencies of the tuning forks?
Solution:
Given
Two tuning forks A and B, when vibrated simultaneously, produce 5 beats per second. The forks produce resonances respectively with 36 cm and 37 cm long air columns of a tube closed at one end.
If is the velocity of sound air, the fundamental frequency of a tube of length l closed at one end, is
n = \(\frac{V}{4 l}\)
∴ \(n_A=\frac{V}{4 \times 36} ; n_B=\frac{V}{4 \times 37}\)
So, \(\frac{n_A}{n_B}=\frac{37}{36}\) ; clearly, \(n_A>n_B\).
As 5 beats are produced per second, we have \(n_A-n_B=5\)
or, \(\frac{n_A}{n_B}-1=\frac{5}{n_B} \quad \text { or, } \frac{37}{36}-1=\frac{5}{n_B} \quad \text { or, } \frac{1}{36}=\frac{5}{n_B} \\
\text { or, } n_B=180 \mathrm{~Hz}\)
∴ \(n_A=n_B+5=180+5=185 \mathrm{~Hz} .\)
Example 8. A diver sends an audio signal from some depth underwater. This signal produces 5 beats per second with the note emitted by a 20 cm long pipe whose one end is closed. Find out the frequency and the wavelength of the audio signal inside water. Given, the velocity of sound in air = 360 m · s-1 and that in water = 1500 m · s-1.
Solution:
Given
A diver sends an audio signal from some depth underwater. This signal produces 5 beats per second with the note emitted by a 20 cm long pipe whose one end is closed.
The fundamental frequency of the 20 cm long pipe closed at one end, \(n_0=\frac{V}{4 l}=\frac{360}{4 \times(20 \times 0.01)}=450 \mathrm{~Hz}\)
As 5 beats are produced per second, the frequency of the audio signal is either, (450- 5) = 445 Hz or (450 + 5) = 455Hz
The frequency does not change due to refraction from water to air. So the wavelength inside water is
Either \(\frac{1500}{445}=3.37 \mathrm{~m} \quad \text { or, } \frac{1500}{455}=3.30 \mathrm{~m}\)
Example 9. Two waves from 10 beats in 3s in a gas, The wavelength are 1m and 1.01m, respectively. Find out the velocity of sound in the gas.
Solution:
Given
Two waves from 10 beats in 3s in a gas, The wavelength are 1m and 1.01m, respectively.
Let V be the velocity of sound in the gas.
As 10 beats are formed in 3 s, the beat frequency = \(\frac{10}{3}\)Hz.
The frequency of the 1st wave, \(n_1=\frac{V}{\lambda}=\frac{V}{1} \mathrm{~Hz} \text {; }\)
The frequency of the 2nd wave, \(n_2=\frac{V}{\lambda_2}=\frac{V}{1.01} \mathrm{~Hz}\)
Here, \(n_1>n_2\)
∴ \(n_1-n_2=\frac{10}{3} \text { or, } \frac{V}{1}-\frac{V}{1.01}=\frac{10}{3}\)
or, V = \(336.7 \mathrm{~m} \cdot \mathrm{s}^{-1}\) .
Example 10. 24 tuning forks lire Otttutged lit the ascending writer of their frequencies. Itaeh fork produces 4 between per second with Ha Immediately preceding fork, The tell fork emits tut octave to Hint emitted by the first one, Find out the frequencies of the first the the last tuning forks.
Solution:
Given
24 tuning forks lire Otttutged lit the ascending writer of their frequencies. Itaeh fork produces 4 between per second with Ha Immediately preceding fork, The tell fork emits tut octave to Hint emitted by the first one,
Let the frequencies In ascending order of the 24 tuning forks be n1, n2,……… n24. frequencies tell that
\(\begin{gathered}n_2-n_1=4 \\
n_3-n_2=4 \\
\ldots \ldots \ldots \ldots \ldots \ldots \\
n_{24}-n_{23}=4 \\
\hline \text { (on addition) } n_{24}-n_1=23 \times 4=92
\end{gathered}\)
As the 24th tuning fork emits an octave to that emitted by the first one, n24 = 2n1
∴ 2n1 – n1 = 92 or, n1 = 92 Hz.
∴ n24 = 2n1 = 2 x 92 = 104 Hz.
Example 11. A wire of length 25 cm and muss 2.5 g Is stretched with a fixed tension. The length of a pipe dosed at one end Is 40 cm. During vibrations, the first overtone of the wire produces 8 beats per second with the fundamental emitted by the pipe. The number of beats reduces with the decrease In tension In the wire. If the velocity of sound In air Is 320 m · s-1, find the tension in the wire.
Solution:
Given
A wire of length 25 cm and muss 2.5 g Is stretched with a fixed tension. The length of a pipe dosed at one end Is 40 cm. During vibrations, the first overtone of the wire produces 8 beats per second with the fundamental emitted by the pipe. The number of beats reduces with the decrease In tension In the wire. If the velocity of sound In air Is 320 m · s-1,
The fundamental frequency of the pipe closed at one end, \(n_1=\frac{V}{4 l}=\frac{(320 \times 100)}{4 \times 40}=200 \mathrm{~Hz}\)
The mass per unit length of the wire,m = \(\frac{2.5}{25}=0.1 \mathrm{~g} \cdot \mathrm{cm}^{-1}\)
The 1st overtone is the 2nd harmonic of the wire; the frequency is \(n_2=2 \times \frac{1}{2 l} \sqrt{\frac{T}{m}}=\frac{1}{25} \sqrt{\frac{T}{0.1}}=\frac{\sqrt{10 T}}{25} \mathrm{~Hz}\)
Clearly, n2 reduces with the decrease in tension T; so (n2 – n1) also decreases. For this reason, the beat frequency reduces. This means, n2 > n1.
As B beats are produced per second, we get n2 – n1 = 8
or, n2 = n1 + 8 = 200 + 8 = 208 Hz.
∴ 208 = \(\frac{\sqrt{107}}{25} \text { or, } 10 T=(208 \times 25)^2\)
or, T = \(\frac{(200 \times 25)^2}{10}=27.04 \times 10^5 \mathrm{dyn}=27,04 \mathrm{~N} .\)
Example 12. A sonometer wire Is stretched by hanging a 10cm high brass cylinder vertically from one of Its ends. The wire resonates with a tuning fork of frequency 250 Hz. Now the cylinder Is partially Immersed In water. If the wire and the tuning fork are vibrated simultaneously, 4 beats are heard per second. Calculate the length of the portion of the cylinder that was immersed In water. Given, the density of brass = 0,5 g · cm-3.
Solution:
Given
A sonometer wire Is stretched by hanging a 10cm high brass cylinder vertically from one of Its ends. The wire resonates with a tuning fork of frequency 250 Hz. Now the cylinder Is partially Immersed In water. If the wire and the tuning fork are vibrated simultaneously, 4 beats are heard per second.
The apparent loss of weight of the partially Immersed cylinder reduces the tension m the wire.
So, the frequency of the vibrating wire decreases. When the cylinder is partially immersed In water, the fundamental frequency of the wire is \(n_2=n_1-4=256-4=252 \mathrm{~Hz}\)
Now, \(\frac{n_1}{n_2}=\sqrt{\frac{T_1}{T_2}} or, \frac{256}{252}=\sqrt{\frac{T_1}{T_2}}\)
Here, T1 = weight of the cylinder
= 10αρg = 10α x 0.5 x g
T2 = weight of the partially immersed cylinder
= weight of the cylinder in the air – the weight of the water displaced
= 10α x 8.5 x g – lα x 1 x g
where α = area of the cross-section of the cylinder
l = height of the cylinder immersed in water
∴ \(\frac{256}{252}=\sqrt{\frac{10 \alpha \times 8.5 \times g}{10 \alpha \times 8.5 \times g-l \alpha \times 1 \times g}}=\sqrt{\frac{85}{85-l}}\)
or, \(\frac{85}{85-l}=\left(\frac{256}{252}\right)^2 \text { or, } 85-l=85 \times\left(\frac{252}{256}\right)^2\)
or, \(l=85 \times\left[1-\left(\frac{252}{256}\right)^2\right]=2.64 \mathrm{~cm}\)
Example 13. Two progressive waves y1 = 4 sin 500πt and y2 = 2 sin506π t are supported. Find the number of beats produced in one minute.
Solution:
Given
Two progressive waves y1 = 4 sin 500πt and y2 = 2 sin506π t are supported.
Comparing the given equations with the standard equation y = Asinωt,
We have, \(\omega_1=500 \pi \text { or, } n_1=\frac{\omega_1}{2 \pi}=\frac{500 \pi}{2 \pi}=250 \mathrm{~Hz} ;\)
and \(\omega_2=506 \pi \text { or, } n_2=\frac{\omega_2}{2 \pi}=\frac{506 \pi}{2 \pi}=253 \mathrm{~Hz}\)
∴ Number of beats per second = difference in frequencies =253-250 =3
∴ A Number of beats produced In one minute = 3 x 60 = 100
Example 14. Three transverse progressive waves are x1 = Acos(kx-ωt), x2 = Acos(kx + ωt) , x3 = Acos (ky-ωt). How may these be superposed to generate
- A stationary wave,
- A wave propagating In a direction inclined at an angle of 45° with both the positive x and y-axes? In each case, find out the positions where the resultant intensity would always be zero.
Solution:
Given
Three transverse progressive waves are x1 = Acos(kx-ωt), x2 = Acos(kx + ωt) , x3 = Acos (ky-ωt).
1. The first and the second waves are two identical but oppositely directed waves. So, they would generate a stationary wave.
The equation of the resultant wave would be
z = \(z_1+z_2 =A[\cos (k x-\omega t)+\cos (k x+\omega t)]\)
= \(2 A \cos k x \cdot \cos \omega t\)
The resultant intensity is zero, where 2Acos kx = 0
∴ coskx = 0 or, x = \(\frac{(2 n+1) \pi}{2 k}\{n=0,1,2,3, \ldots\}\)
2. The first wave directed along the positive x-axis and the third wave directed along the positive y-axis are identical waves. So the resultant wave propagates in a direction that Is Inclined at 45″ with both the x and the yaxes. The equation of the resultant wave would be
z = \(z_1+z_3=A[\cos (k x-\omega t)+\cos (k y-\omega t)]\)
= \(2 A \cos \frac{k(x+y)-2 \omega t}{2} \cdot \cos \frac{k(x-y)}{2}\)
Tire resultant intensity is zero, where \(2 A \cos \frac{k(x-y)}{2}=0\)
∴ \(\cos \frac{k(x-y)}{2}=0\)
or, \(\frac{k(x-y)}{2}=\frac{(2 n+1) \pi}{2}[n=0,1,2,3, \ldots]\)
or, \(x-y=\frac{(2 n+1) \pi}{k}\) .