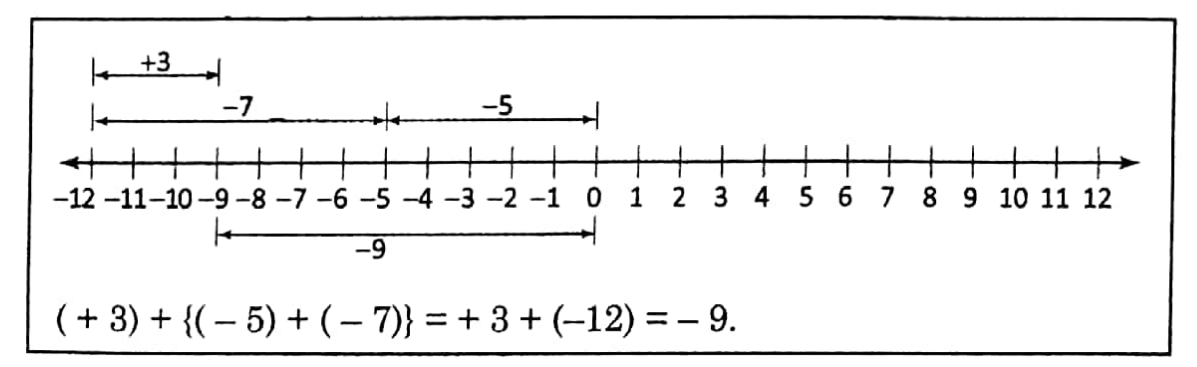
Chapter 5 Energy Synopsis
Energy is the capacity of a body to do work. It is measured by the amount of work that a body can perform.
In CGS system and SI, the absolute units of energy are erg and joule (J) respectively.
W • h and kW • h are also practical units of energy.
1 kW • h = 1000 W • h = 1000 J/s x 3600 s = 3.6 x 106 J
Dimensional formula of energy is ML2T-2.
The energy that a body or a system acquires by virtue of its configuration, position or motion, is known as mechanical energy. Mechanical energy is of two types:
- kinetic energy and
- potential energy.
Kinetic energy is the energy acquired by a body by virtue of its motion. If a body of mass m is moving with a velocity v, then its kinetic energy, Ek = 1/2 mv2.
Read and Learn More WBBSE Solutions for Class 9 Physical Science and Environment
When E is the kinetic energy of a body of mass m, then its linear momentum, p = \(\sqrt{2 m E}\).
The energy acquired by a body by virtue of its position or configuration is called its potential energy.
When a body changes its position against gravity, it gains a certain amount of energy which is called gravitational potential energy.
A body of mass m is raised a height of h against gravity [h \(\ll\) R being the radius of the earth], then its gravitational potential energy, Ep = mgh.
Value of gravitational potential energy on the surface of the earth is taken to be zero. The sum of kinetic energy and potential energy is considered as mechanical energy.
The total mechanical energy of a body or a system remains constant in the absence of resistant forces or other dissipative forces.
Chapter 5 Energy Short And Long Answer Type Questions
Question 1. What is energy? What is its dimensional formula? What are its units in CGS system and SI?
Answer:
Energy, Dimensional Formula And Units In CGS And SI:-
- Energy is the capacity of a body to do work.
- Energy is measured by the amount of work a body can do. So, units and dimensional formulas of energy and work are the same.
- Dimensional formula of energy is ML2T-2.
- Units of energy in CGS system and SI are erg and joule, respectively.
Question 2. Name different forms of energy.
Answer:
Different Forms Of Energy Are:-
Energy is present in nature in different forms. These are
- Mechanical energy,
- Heat energy,
- Chemical energy,
- Sound energy,
- Electric energy,
- Magnetic energy,
- Light energy,
- Atomic energy.
Question 3. What is mechanical energy? How many types of mechanical energy are there? Name them.
Answer:
Mechanical Energy:-
Mechanical energy is the energy that is possessed by an object due to its motion or position or change of configuration or all these three.
Mechanical Energy Is Of Two Types:-
- kinetic energy and
- potential energy.

Question 4. What is kinetic energy? Establish the formula for the kinetic energy of a body of mass m moving with velocity u.
Answer:
Kinetic Energy:-
Kinetic energy is the energy acquired by a body by virtue of its motion.
Formula For The Kinetic Energy Of A Body Of Mass m Moving With Velocity u:-
Suppose, a body of mass m is moving in a straight line with velocity u. A constant force F is applied against the motion of the body to bring it to rest after traversing a distance s.
As the body has traversed a distance s against a force F, it has the capacity to do Fs amount of work. Hence, its kinetic energy = Fs.
If a is the deceleration of the body for applying force F against its motion, then a = \(\frac{F}{m}\).
It stops after traveling a distance s, so its final velocity, v = 0.
From the equation v2 = u2 – 2as, we get
0 = u2 – 2as or, 2as = u2
or, s = \(\frac{u^2}{2 a}\)
∴ kinetic energy of the body,
∴ \(E_k=F s=m a \times \frac{u^2}{2 a}=\frac{1}{2} m u^2\)
Question 5. Can the kinetic energy of a body be negative? Explain.
Answer:
The Kinetic Energy Of A Body Cannot Be Negative:-
The formula for measuring the kinetic energy of a body is 1/2mv2, where m is its mass and v is its velocity.
The value of m can never be negative.
On the other hand, v2 is a squared quantity and hence its value can never be negative. So, the kinetic energy of a body cannot be negative.
Question 6. The kinetic energy of a body of mass m is E, Show that its linear momentum, p = \(\sqrt{2 m E}\)
Answer:
Given
The kinetic energy of a body of mass m is E
Suppose, the body of mass m is moving with uniform velocity v.
∴ kinetic energy, E = \(\frac{1}{2} m v^2\)
or, \(v^2=\frac{2 E}{m} \text { or, } \quad v=\sqrt{\frac{2 E}{m}}\)
Now, the linear momentum of the body is given by
p = \(m v=m \sqrt{\frac{2 E}{m}}=\frac{\sqrt{2 E}}{\sqrt{m}} \times m=\sqrt{2 m E}\)
Question 7. If the kinetic energy of a body moving with velocity v Is E, show its linear momentum; p = 2EV.
Answer:
Given
If the kinetic energy of a body moving with velocity v Is E,
Let m be the mass of the body moving with velocity v.
So, kinetic energy of the body, E = 1/2mv2 momentum p = mv.
∴ \(\frac{P}{E}=\frac{m v}{\frac{1}{2} m v^2} \quad \text { or, } p=2 E v\)
Question 8. The kinetic energies of two bodies (one light and one heavy) are equal. Which one has the higher linear momentum?
Answer:
Given
The kinetic energies of two bodies (one light and one heavy) are equal.
Suppose the masses of the light body and the heavy body are m1 and m2, respectively.
Further, they have the same kinetic energy, E.
∴ linear momentum of the light body, \(p_1=\sqrt{2 m_1 E}\)
and linear momentum of the heavy body, \(p_2=\sqrt{2 m_2 E}\)
∴ \(\frac{p_1}{p_2}=\frac{\sqrt{2 m_1 E}}{\sqrt{2 m_2 E}}=\sqrt{\frac{m_1}{m_2}}<1\)
or, \(p_1<p_2 \text { or } p_2>p_1\)
So, linear momentum of the heavy body is higher if the kinetic energies of the light body and the heavy body are equal.
Question 9. The linear momentum of two bodies (one light and one heavy) are equal. Which one has the higher kinetic energy?
Answer:
Given
The linear momentum of two bodies (one light and one heavy) are equal.
Suppose the masses of the light body and the heavy body are m1 and m2 respectively. Further they have the some momentum, p.
So the kinetic energy of the light body \(E_1=\frac{p^2}{2 m_1}\)
and kinetic energy of the heavy body, \(E_2=\frac{p^2}{2 m_2}\)
So, \(\frac{E_1}{E_2}=\frac{p^2}{2 m_1} \times \frac{2 m_2}{p^2}=\frac{m_2}{m_1}>1\) [because \(m_2>m_1\)]
∴ \(E_1>E_2\)
Hence, if a light body and a heavy body have the same linear momentum, the lighter body has the higher kinetic energy.
Question 10. A body of mass m is moving with constant acceleration from rest. Its velocity is v after t seconds. What is its kinetic energy at time T?
Answer:
Given
A body of mass m is moving with constant acceleration from rest. Its velocity is v after t seconds.
If the acceleration of the body is a, then v = at or, a = \(\frac{v}{t}\)
After time T, its velocity V = aT or, V = \(\frac{vT}{t}\)
∴ kinetic energy of the body after time T,
∴ \(E_k=\frac{1}{2} m V^2=\frac{m}{2} \cdot \frac{v^2 T^2}{t^2}\)
Question 11. Two masses are attached to the two ends of a spring, which Is lying on a smooth horizontal table. The two bodies are pulled in opposite directions and then released. Show that at any moment, the kinetic energy of each body is inversely proportional to its mass.
Answer:
Given
Two masses are attached to the two ends of a spring, which Is lying on a smooth horizontal table. The two bodies are pulled in opposite directions and then released.
Suppose the masses of the two bodies are m1 and m2 respectively and at any moment, their velocities are u1 and u2, respectively.
∴ according to the law of conversation of linear momentum,
⇒ \(m_1 u_1+m_2 u_2=0 \text { or, } m_1 u_1=-m_2 u_2\)
or, \(m_1^2 u_1^2=m_2^2 u_2^2\)……(1)
At that moment, kinetic energy of the first body, \(E_1=\frac{1}{2} m_1 u_1^2\) and the kinetic energy of the second body, \(E_2=\frac{1}{2} m_2 u_2^2\)

∴ \(\frac{E_1}{E_2}=\frac{\frac{1}{2} m_1 u_1^2}{\frac{1}{2} m_2 u_2^2}=\frac{m_1^2 u_1^2}{m_2^2 u_2^2} \times \frac{m_2}{m_1}=\frac{m_2}{m_1}\) [from equation(1)]
or, m1E1 = m2E1
So, at any moment, kinetic energy of each body is inversely proportional to its mass.
Question 12. A body with kinetic energy of 10 J is thrown upward. If air resistance is ignored, what is the potential energy of the body at the highest point?
Answer:
At the highest point, the kinetic energy of the body becomes zero. Hence, according to the law of conservation of energy, potential energy becomes 10J.
Question 13. Explain elastic potential energy and gravitational potential energy with examples.
Answer:
Elastic potential energy
A stone is thrown from a rubber catapult with a high velocity. Here, when the rubber band is stretched to increase its length, it develops a capacity to work which enables it to throw the stone with high velocity. This energy of the stretched string is potential energy in nature and is called elastic potential energy.
Again, work is done if a body is raised to some height against gravity. Then, it also develops some capacity to work.
For example, if a hammer is placed on a nail standing erect on the ground, it does not pierce the ground but if the hammer is raised to some height and then hit straight on the top of the nail, the nail enters a bit into the ground.
Here, displacement of the nail is due to application of force by the hammer. In this case, when the hammer is raised upward, it develops the capacity of doing work. We may say that some amount of gravitational potential energy is stored in the hammer in this way.
Question 14. What is gravitational potential energy? Develop a formula for gravitational potential energy.
Answer:
Gravitational potential energy
- Gravitational potential energy is defined as the potential or capacity to work gained by a body due to the change of its position in a gravitational field.
- Suppose a body of mass m is raised to a height h against gravity (here, h \(\ll\) R, R being the radius of the earth). Force acting on the body m is the weight mg of the body. So, work done against gravity = mg x h = mgh This work is stored in the body as potential energy. Hence, the gravitational potential energy of the body, Ep = mgh.

Question 15. Can gravitational potential energy be negative?
Answer:
Yes, Gravitational Potential Energy Can Be Negative:-
If a body of mass m is raised through a height h from the surface of the earth, gravitational potential energy of the body, Ep = mgh. Again, if it is taken to a depth of h from the earth’s surface, then Ep = -mgh .
So, it may be said that gravitational potential energy depends on the reference plane. Here, surface of the earth is the reference plane. If a body is above the reference plane, its potential energy is positive and if the body is below the reference plane, its potential energy is negative.
Question 16. What is the gravitational potential energy of a body on the surface of the earth if the earth’s surface is considered as the reference plane? At a height of h from the earth’s surface, what is the change of gravitational potential energy of the same body?
Answer:
If earth’s surface is taken as the reference plane, then gravitational potential energy on the surface is zero. It may also be said that if h = 0, then gravitational potential energy = mg x 0 = 0.
If a body of mass is raised to a height of h from the earth’s surface, then gravitational potential energy of the body, Ep = mgh.
Again, if the body is taken at a depth of h below the earth’s surface, then gravitational potential energy, Ep‘ = -mgh.

Question 17. Give an example of the potential energy of a body due to change of its position.
Answer:
Hydroelectricity is produced by confining water in a raised place with a barrier. In this case, potential energy is stored in water due to change of its position.
Question 18. Give an example of the potential energy of a body due to change of its shape.
Answer:
The energy used by an archer to pull the string back in his bow is stored in the elastic material of the bow. This is an example of potential energy due to change of shape.
Question 19. Calculate the reduction in potential energy of a body of mass m at tth second, falling freely from rest.
Answer:
Suppose the body falling freely falls through distances h1 and h2 from rest during time (t-1)s and ts respectively, then
\(h_1=\frac{1}{2} g(t-1)^2 \text { and } h_2=\frac{1}{2} g t^2\)∴ distance covered at the tth second,
h = \(h_2-h_1=\frac{1}{2} g t^2-\frac{1}{2} g(t-1)^2\)
= \(\frac{1}{2} g\left(t^2-t^2+2 t-1\right)=\frac{1}{2} g(2 t-1)\)
Now, as the body is falling down in a perpendicular direction, its potential energy is reduced.
∴ reduction in potential energy during the tth second
= mgh = mg x 1/2 g(2t- 1) = 1/2 mg2(2t- 1)
Question 20. A body has energy but no momentum is this situation possible?
Answer:
Yes, it is possible. A body when thrown upward, it stops for a moment at the highest point. At that moment, it has no momentum but has gravitational potential energy.
Question 21. A ball is kept on the top of a staircase. Is it possible to know without disturbing the ball, if there is any mechanical energy in it? How would one know if there is any potential energy stored in it?
Answer:
The ball is at rest on the top of the staircase. If it is not disturbed, it will forever remain in this state. This is due to the law of inertia. This means that the ball is an inert object and does not move itself.
Thus, if it is not disturbed, it cannot do any work. As energy is defined as a measure of the ability of a body to do work, unless the ball is shaken (disturbed), the external manifestation of energy in it is not possible. So, it is not known whether there is any mechanical energy in the ball or not.
As the ball is pushed from the top of the staircase, it starts rolling down the staircase. As a result, it may be said that the ball possesses kinetic energy. As energy cannot be created, so it may be reasoned that the potential energy stored in the ball changes into this kinetic energy.
Hence, in this way we come to know that gravitational potential energy is stored in this ball while it is on the top of the staircase.
Question 22. Write and explain the law of conservation of mechanical energy.
Answer:
Law Of Conservation Of Mechanical Energy:-
According to the law of conservation of mechanical energy, in the absence of any dissipative force (e.g. friction, air resistance, etc.), the total mechanical energy of a body or an agency always remains constant.
A stone is thrown up in a perpendicular direction. The more it goes up, more is the decrease of its velocity and finally, it comes to rest for a moment. Then it starts falling downwards and as it comes down, its velocity increases.
The stone was thrown upwards with a definite velocity or definite kinetic energy. Now as it goes up, its velocity decreases and kinetic energy also decreases. But its potential energy increases equivalently.
If air resistance is ignored, it can be proved that the decrease in kinetic energy of the stone is equal to the increase in its potential energy. That is, at any moment in its state of motion, the sum of kinetic energy and potential energy of the stone or total mechanical energy remains constant.
There is only a transformation from kinetic energy to potential energy or potential energy to kinetic energy. For example, at the highest point, the stone stops for a moment, and at that time, there is no kinetic energy of the stone.
Entire kinetic energy has been converted to potential energy. Next, when the stone drops downwards, then the potential energy decreases, and the kinetic energy increases. As the stone continues to drop down, its velocity continues to increase.
Question 23. A man pushes a chair on the ground to some distance and pulls it back to the same position, by push. Explain whether the theory of conservation of energy is valid in this case or not.
Answer:
Given
A man pushes a chair on the ground to some distance and pulls it back to the same position, by push.
If a chair is pushed on the ground to some distance, then there is no change in its potential energy. In a moving condition, there is a change of kinetic energy because the velocity of chair slowly diminishes to zero in this case.
Also, some amount of sound energy and heat energy due to friction are develop which are dissipative energies. In the same way, if the chair is brought back to the original position, the transformation of energy and resultant dissipative energy develop again.
Now, according to the theory of conservation of mechanical energy, mechanical energy of anybody is always constant, if there is no loss of energy due to friction or any other opposing force. But in the above case, the theory of conservation of mechanical energy does not hold good as there is loss of energy.
Question 24. Establish the law of conservation of mechanical energy for a freely falling body.
Answer:
Law Of Conservation Of Mechanical Energy For A Freely Falling Body:-
Suppose, a body of mass m is in a static position at point A which is at a height h from the earth’s surface. If the earth’s surface is taken as the reference plane, its kinetic energy at point A, E’k= 0, and potential energy, E’p = mgh.
∴ total mechanical energy of the body at point A is given by,
E’ = E’k + Ep‘ = 0 + mgh = mgh
Now as the body is allowed to fall, it falls under gravity with an acceleration g (acceleration due to gravity). After coming down a distance h1 to B, its velocity v1 at B is governed by the equation \(v_1^2=2 g h_1\).
∴ kinetic energy of the body at point B,
⇒ \(E_k^{\prime \prime}=\frac{1}{2} m v_1^2=\frac{1}{2} m \times 2 g h_1=m g h_1\)
Distance of point B from the surface of the earth is (h – h1).
∴ Potential energy of the body at point B, \(E_p^{\prime \prime}=m g\left(h-h_1\right)=m g h-m g h_1\)

∴ Total mechanical energy at point B, \(E^{\prime \prime}=E_k^{\prime \prime}+E_p^{\prime \prime}\)
= \(m g h_1+m g h-m g h_1=m g h\)
Suppose, just before striking the earth’s surface, the body is at point C.
Its potential energy, E”‘ = mg x 0 = 0
If the velocity of the body at C is v, then v2 = 2gh.
∴ Kinetic energy of the body at point C, \(E_k^{\prime \prime \prime}=\frac{1}{2} m v^2=\frac{1}{2} m \times 2 g h=m g h\)
∴ Total mechanical energy at point C,
⇒ \(E^{\prime \prime \prime}=E_k^{\prime \prime \prime}+E_p^{\prime \prime \prime}=m g h+0=m g h\)
∴ \(E^{\prime}=E^{\prime \prime}=E^{\prime \prime \prime}\)
Hence, the mechanical energy is conserved for a freely falling body.
Question 25. As a balloon filled with hydrogen gas moves upwards, both the kinetic energy and the potential energy of the balloon increase. Is the law of conservation of energy violated in this case?
Answer:
Hydrogen gas is lighter than an equal volume of air. When a balloon is filled with hydrogen gas, it floats as the upward thrust of air on the balloon is greater than the weight of the balloon.
As a result, an upward resultant force acts on the balloon and the balloon goes up with acceleration. In this case, work done by this resultant force is equal to the sum of increase of kinetic energy and increase of potential energy of the balloon. In this case, law of conservation of energy is not violated.
Chapter 5 Energy Very Short Answer type Questions Choose The Correct Answer
Question 1. When the speed of a body is doubled, its kinetic energy becomes
- Doubled
- Quadrupled
- Halved
- Quartered
Answer: 2. Quadrupled
Question 2. When both the mass and the speed of a body is doubled, its kinetic energy becomes
- Four times
- Eight times
- Ten times
- Half
Answer: 2. Eight times
Question 3. Energy is
- The rate of doing work
- The capacity of doing work
- The power of doing work
- All of the above
Answer: 2. The capacity of doing work
Question 4. Amount of energy accumulated by a piece of stone falling freely just before touching the ground is
- Total kinetic energy
- Total potential energy
- Equal amounts of kinetic energy and potential energy
- Heat energy and sound energy
Answer: 1. Total kinetic energy
Question 5. The mechanical energy of a body thrown upward is
- Maximum at the highest point
- Minimum at the highest point
- Highest at the moment of just touching the ground
- Aame at all points
Answer: 4. Aame at all points
Question 6. Total energy of an oscillating simple pendulum for which the theory of conservation of mechanical energy holds good is
- Maximum at the central position
- Maximum at the terminal position
- Minimum at the central position
- Always constant
Answer: 4. Always constant
Question 7. Kinetic energy of the pendulum is
- Maximum at the central position
- Maximum at the terminal position
- Minimum at the central position
- Always constant
Answer: 1. Maximum at the central position
Question 8. potential energy of the pendulum is
- Maximum at the central position
- Maximum at the terminal position
- Minimum at the central position
- Always constant
Answer: 2. Maximum at the terminal position
Question 9. Dimensional formula of energy is
- ML2T-2
- MLT-2
- ML2T-1
- ML2T-3
Answer: 1. ML2T-2
Question 10. Linear momentum of a body of mass m and kinetic energy E is
- 2mE
- √2mE
- \(\frac{2E}{m}\)
- \(\sqrt{\frac{2 E}{m}}\)
Answer: 2. 2mE
Question 11. Kinetic energy of a particle of mass m and momentum p is
- \(\frac{p}{2 m}\)
- \(\frac{p^2}{2 m}\)
- \(\frac{2p^2}{2 m}\)
- \(\frac{p^2}{4 m}\)
Answer: 2. \(\frac{p^2}{2 m}\)
Question 12. Two objects have the same kinetic energy but their masses are in the ratio of 4:9. Ratio of linear momentum of the objects is
- 4:9
- 9:4
- 2:3
- 3:2
Answer: 3. 2:3
Question 13. Ratio of masses of two objects is 1: 2 and ratio of their kinetic energies is 2 :1. What is the ratio of their linear momentum?
- 1:1
- 2:1
- 1:2
- 1:4
Answer: 1. 1:1
Question 14. Linear momentum of two objects is same but the ratio of their masses is 2 : 3. Ratio of kinetic energies of these two objects is
- 3:2
- 2:3
- 4:9
- 9:4
Answer: 1. 3:2
Question 15. What is the kinetic energy of a bullet of mass 10 g moving with a velocity of 400 m/s?
- 100J
- 200J
- 400J
- 800J
Answer: 4. 800J
Question 16. Kinetic energy of a moving object of mass 2 kg is 16 J. Its momentum is
- 2 kg • m • s-1
- 4 kg • m • s-1
- 8 kg • m • s-1
- 12 kg • m • s-1
Answer: 3. 8 kg • m • s-1
Question 17. A mass of 0.03 kg is raised to a height of 20 m above the earth’s surface. The amount of potential energy stored in the mass is(g = 10 m/s2)
- 6J
- 6.28J
- 588J
- 0.588J
Answer: 1. 6J
Question 18. Unit of energy in SI is
- Newton
- erg
- Watt
- Joule
Answer: 4. Joule
Question 19. With increasing height of a body from the surface of the earth, the value of its gravitational potential energy
- Increases
- Decreases
- Remains same
- None of the above
Answer: 1. Increases
Question 20. If X = \(\frac{work}{energy}\), then the dimensional formula energy of X is
- ML-2T-2
- M2L2T-2
- MLT
- M°L°T°
Answer: 4. M°L°T°
Question 21. One empty lorry and a similar lorry loaded with goods are moving with the same speed. Which one of them has greater kinetic energy?
- Loaded
- Empty
- Both have the same energy
- None of the above
Answer: 1. Loaded
Question 22. Which of the following statements is not false?
- A moving body has no kinetic energy
- Potential energy is stored in a compressed spring
- Unit of power is W/m2
- Dimensional formula of work is ML-2T2
Answer: 2. Potential energy is stored in a compressed spring
Question 23. Mechanical energy is the sum of which of the following energies?
- Kinetic energy and potential energy
- Light energy and chemical energy
- Sound energy and light energy
- Heat energy and light energy
Answer: 1. Kinetic energy and potential energy
Question 24. Which of the following statements is not correct?
- SI unit of work is joule
- Potential energy and work have the same dimension
- 1hp = 746W
- Kinetic energy may be negative
Answer: 4. Kinetic energy may be negative
Question 25. Which of the following statements is correct?
- Dimensional formula of kinetic energy is ML2T-3
- Work = power x time
- Sum of mass and energy in this universe is constant
- Mathematical expression of potential energy is 1/2 mv2
Answer: 3. Sum of mass and energy in this universe is constant
Question 26. A body of mass m is falling freely from rest. Decrease of its potential energy in the first second is
- 1/2 mg2
- mg2
- 3/2 mg2
- 2mg2
Answer: 1. 1/2 mg2
Question 27. A body of 2 kg is allowed to fall from the top of a 50 m high tower. After one second of its journey, its kinetic energy is [g = 10 m/s2]
- 50J
- 75J
- 100J
- 200J
Answer: 3. 100J
Question 28. If the kinetic energy of a moving object increase by 20%, its linear momentum increases by
- 10%
- 11%
- 20%
- 21%
Answer: 1. 10%
Question 29. A student travels a distance of 200 m with his school bag of mass 5 Kg in his back his work done against the gravitational force is
- 0
- 0.6 J
- 6 J
- 6 x 102 J
Answer: 1. 0
Question 30. If a body moves with retardation its kinetic energy
- Decreases
- Increases
- Remain unchanged
- Increases first and then decreases
Answer: 1. Decreases
Question 31. A body of mass m is allow to fall freely from a height h. Kinetic energy of the body just before touching the ground is
- 0
- mgh
- \(\frac{mgh}{4}\)
- \(\frac{mgh}{2}\)
Answer: 2. mgh
Question 32. If kinetic energy of a body is 4 times that of the initial, then the ratio of initial momentum to that of the final is
- 1:2
- 4:1
- 1:4
- 2:1
Answer: 1. 1:2
Chapter 5 Energy Answer In Brief
Question 1. What is the relationship between kinetic energy and the momentum of a body of mass m?
Answer: If E is the kinetic energy of the mass m, its linear momentum, P = \(\sqrt{2 m E}\).
Question 2. If a body is raised some distance from the earth’s surface and then released, which energy is transformed to which energy?
Answer: In this case, gravitational potential energy stored in the body is transformed into its kinetic energy.
Question 3. What is the nature of energy conversion in the case of an arrow being shot from a bow?
Answer: An archer stores potential energy in the bow by bending its string which gets converted to kinetic energy when the arrow is shot.
Question 4. What is the condition of applicability of the law of conservation of mechanical energy?
Answer: Condition for applicability of the law of conservation of mechanical energy is that no dissipative force should be present.
Question 5. If air resistance is taken into consideration, is mechanical energy conserved in case of a body falling under gravity?
Answer: No, if air resistance is taken into consideration, mechanical energy is not conserved in case of a body falling under gravity.
Question 6. A body has momentum but no energy—is this situation possible?
Answer: No, it is not possible because if a body has momentum, it must have kinetic energy.
Question 7. What is the dimensional formula of kinetic energy?
Answer: Dimensional formula of energy is ML2T-2.
Question 8. What is the kinetic energy of a body of mass m with momentum p?
Answer: Kinetic energy of the body is \(\frac{p^2}{2 m}\)
Question 9. What is the condition of applicability of gravitational potential energy, Ep = mgh?
Answer: The condition of applicability of gravitational potential energy, Ep = mgh is h\(\ll\)R, where R is radius of the earth.
Question 10. Mechanical energy is the sum of which energies?
Answer: Mechanical energy is the sum of kinetic energy and potential energy.
Question 11. What is the relationship between work and energy?
Answer: Energy is the capacity of a body to do work.
Question 12. What is the relationship between the units of work and energy?
Answer: Energy is measured in the same unit as that of work.
Question 13. What is potential energy?
Answer:
Potential energy
Energy acquired by a body by virtue of its position or configuration is called its potential energy.
Question 14. What will be the kinetic energy of a body when its mass is made four times and the velocity is doubled?
Answer: Initial kinetic energy = 1/2 mv2 where m is mass and v is velocity of the body.
∴ Final kinetic energy of the body
= 1/2 x 4m x (2v)2 = (1/2 mv2) x 16
So, the kinetic energy becomes 16 times its initial value.
Question 15. Name the type of energy possessed by a raised hammer.
Answer: Potential energy.
Question 16. Two bodies of masses m1 and m2 have equal kinetic energies. What is the ratio of their linear momenta?
Answer:
Given
Two bodies of masses m1 and m2 have equal kinetic energies.
Momentum, P = \(\sqrt{2 E m}\) where E is kinetic energy and m is mass.
∴ \(\frac{P_1}{P_2}=\frac{\sqrt{2 E m_1}}{\sqrt{2 E m_2}}=\sqrt{\frac{m_1}{m_2}}\)
where P1 is momentum of the first body and P2 is momentum of the second body.
Chapter 5 Energy Fill In The Blanks
Question 1. When the velocity of a body is doubled, then its kinetic energy is ______
Answer: Quadrupled
Question 2. When the kinetic energy of a body is increased nine times, linear momentum becomes ______
Answer: Three times
Question 3. If two bodies have different masses but same kinetic energy, then the ______ body has more momentum.
Answer: Heavier
Question 4. If two bodies with different masses but same momentum, then the _______ body will have less kinetic energy.
Answer: Heavier
Question 5. If resistance due to air is neglected, its kinetic energy at a distance of \(\frac{h}{2}\) from the ground is _______
Answer: \(\frac{mgh}{2}\)
Question 6. According to the law of conservation of energy, _______ can neither be created nor destroyed.
Answer: Energy
Question 7. Total mechanical energy of a body is ________ when it falls under the influence of gravity.
Answer: Conserved
Question 8. Kinetic energy ______ be negative.
Answer: Cannot
Question 9. Gravitational potential energy _______ be negative.
Answer: Can
Question 10. If a bullet is fired from a gun, kinetic energy of the bullet is _________ than the kinetic energy of the gun.
Answer: Greater
Question 11. If the kinetic energies of two bodies of mass m and Am are in the ratio 2:1, then ratio of the momentum of the bodies is _________
Answer: 1:√2
Question 12. Gravitational potential energy is present due to existence of ________
Answer: Gravitational
Question 13. ________ is the energy acquired by a body by virtue of its motion.
Answer: Kinetic energy
Question 14. Energy is expressed in the unit of ________
Answer: Work
Question 15. Total ________ energy of a gas-balloon increases when it goes up with acceleration.
Answer: Mechanical
Question 16. Kinetic energy of a mass of m moving with a velocity v is \(\frac{1}{2}\) x m x ________
Answer: v2
Question 17. ________ energy of a freely falling body gradully increases.
Answer: Kinetic
Question 18. ________ energy of a freely falling body gradully decreases.
Answer: Potential
Question 19. ________ of a body of mass m with a kinetic energy E is \(\sqrt{2 m E}\)
Answer: Linear momentum
Question 20. Dimensional formula of kinetic energy is _________
Answer: ML2T-2
Question 21. __________ potential energy depends on the plane of reference.
Answer: Gravitational
Question 22. Total sum of ________ and sum of energy of this universe is a constant.
Answer: Mass
Question 23. Energy can neither be _______ nor _______
Answer: Created, destroyed
Question 24. Presence of ________ potential energy is not felt if there is no change in the height of the body.
Answer: Gravitational
Question 25. Two individuals A and B reach the peak of a mountain following two different paths. The value of their stored gravitational potential energy is _______
Answer: Same
Question 26. Potential energy of a body of mass m at a height h from the earth’s surface depends on the _______ of the earth.
Answer: Gravitational acceleration
Chapter 5 Energy State Whether True Or False
Question 1. In SI, the absolute unit of energy is joule (J).
Answer: True
Question 2. Energy acquired by a body by virtue of its position or configuration is called its kinetic energy.
Answer: False
Question 3. If a body changes its position against gravity, it gains certain amount of energy which is called gravitational potential energy.
Answer: True
Question 4. Kinetic energy of a body can be negative.
Answer: False
Question 5. Mechanical energy of a freely falling body is conserved.
Answer: True
Question 6. Linear momentum of a body of mass m and kinetic energy E is 2mE
Answer: False
Chapter 5 Energy Numerical Examples
Useful Information
- Kinetic energy of a body of mass m moving with velocity v is E = 1/2 mv2
- linear momentum of a body of mass m moving with kinetic energy E is p \(=\sqrt{2 m E} \quad \text { or, } E=\frac{p^2}{2 m}\)
- Gravitational potential of a body of mass m at a height h from the earth’s surface (h\(\ll\)R, R = radius of the earth) is Ep = mgh [g is acceleration due to gravity] and that of the body at a depth h from the earth’s surface is Ep = -mgh
- For a free falling body, total mechanical energy at any instant = kinetic energy + potential energy.
Question 1. What is the kinetic energy of a bullet of mass 20 g which is moving with a velocity of 500 m/s?
Answer:
Mass of the bullet, m = 20g = 0.02 kg
Velocity, v = 500 m/s
∴ kinetic energy of the bullet,
⇒ \(E_k=\frac{1}{2} m v^2=\frac{1}{2} \times 0.02 \times(500)^2=2500 \mathrm{~J}\)
Question 2. A body of mass 1 kg is allowed to fall from the top of a tower of height 100 m. What is the kinetic energy of the body
- after 1s of fall?
- at the moment of just reaching the base of the tower?
Answer:
Given
A body of mass 1 kg is allowed to fall from the top of a tower of height 100 m.
Mass of the body, m = 1 kg, acceleration of the body, a = g = 9.8 m/s2.
1. Velocity of the body after t = Is, v = at
or, v = gt = 9.8 x 1 = 9.8 m/s
∴ kinetic energy of the body after 1 s
= 1/2 mv2 = 1 x 1 x 9.82 = 48.02 J
2. Height of the tower, h – 100 m
If the velocity of the body at the base of the tower = v1, then
⇒ \(v_1^2=2 a h \quad \text { or, } v_1^2=2 g h\)
∴ kinetic energy at the base of the tower
= \(\frac{1}{2} m v_1^2=\frac{1}{2} \times m \times 2 g h=m g h\) = 1 x 9.8 x 100 = 980J
Question 3. The kinetic energy of a moving body of mass10 g is 2J. What is the linear momentum of the body?
Answer:
Given
The kinetic energy of a moving body of mass10 g is 2J.
Mass of the body, m = 10 g = 0.01 kg
Kinetic energy, E = 2 J
Velocity of the body = v
∴ linear momentum, p = mv
or, p2 = m2v2 = 2m x 1/2 mv2 = 2mE
∴ p = \(\sqrt{2 m E}\)
= \(\sqrt{2 \times 0.01 \times 2}=0.2 \mathrm{~kg} \cdot \mathrm{m} \cdot \mathrm{s}^{-1}\)
Question 4. The linear momentum of a moving body of mass 50 kg is 600 g • cm • s-1. What is its kinetic energy?
Answer:
Given
The linear momentum of a moving body of mass 50 kg is 600 g • cm • s-1.
Mass of the body, m = 50g
Linear momentum, p = 600g • cm • s-1
If velocity of the body = v, mv = p
or, \(v=\frac{p}{m}=\frac{600}{50}=12 \mathrm{~cm} \cdot \mathrm{s}^{-1}\)
∴ kinetic energy of the body,
⇒ \(E_k=\frac{1}{2} m v^2=\frac{1}{2} \times 50 \times 12^2=3600 \mathrm{erg}\)
Alternative method:
Mass of the body, m =50 g
∴ linear momentum, p = 600g • cm • s-1
∴ kinetic energy,
⇒ \(E_k=\frac{p^2}{2 m}=\frac{600^2}{2 \times 50}=3600 \mathrm{erg}\)
Question 5. If the kinetic energy of a body is increased by 44%, what is the percentage change in its linear momentum?
Answer:
Suppose, mass of the body =m and kinetic energy = E.
∴ linear momentum, p = \(\sqrt{2 m E}\) •••(1)
If the kinetic energy increases by 44%, then the new kinetic energy becomes
⇒ \(E_1=E+\frac{44}{100} E=1.44 E\)
and the new linear momentum becomes
⇒ \(p_1=\sqrt{2 m E_1}=\sqrt{2 m \times 1.44 E}\)
= \(1.2 \times \sqrt{2 m E}\)
or, p1 = 1.2p [from equation (1)]
∴ \(\frac{p_1-p}{p} \times 100 \%=\frac{1.2 p-p}{p} \times 100 \%\)
= 0.2×100% = 20%
So, if the kinetic energy of a body is increased 44%, its linear momentum increases by 20%.
Question 6. If the linear momentum of a moving body is increased by 30%, what is the change of its kinetic energy?
Answer:
Suppose the mass of the body is m and its linear momentum is p.
∴ kinetic energy, E = \(\frac{p^2}{2 m}\)……(1)
If the linear momentum is increased by 30%, the new linear momentum becomes
⇒ \(p_1=p+\frac{30}{100} p=1.3 p\)
and the new kinetic energy becomes
⇒ \(E_1=\frac{p_1^2}{2 m}=\frac{(1.3 p)^2}{2 m}=1.69 \frac{p^2}{2 m}=1.69 E\)
∴ \(\frac{E_1-E}{E} \times 100 \%=\frac{1.69 E-E}{E} \times 100 \%\)
= 0.69 x 100% = 69%
So, if the linear momentum of the body is increased by 30 %, its kinetic energy increases by 69%.
Question 7. A body of mass 200 g is thrown downwards with a velocity of 4.9 m/s from the top of a tower of height 15m. What is its kinetic energy just a moment before it strikes the ground? What is the total time of its fall?
Answer:
Giiven
A body of mass 200 g is thrown downwards with a velocity of 4.9 m/s from the top of a tower of height 15m
Mass of the body, m = 200 g = 0.2 kg
Initial velocity of the body which is going downwards, u = 4.9 m/s
Height of the tower, h = 15 m
If v is the velocity just a moment before it strikes the ground, then
v2 = u2 + 2gh = 4.92 + 2 x 9.8 x 15 = 318.01
or, v = 17.83 m/s
∴ kinetic energy of the body at that time,
⇒ \(E_k=\frac{1}{2} m v^2\)
= \(\frac{1}{2}\) X 0.2 x 318.01 = 31.801J
If total time of its fall = t, then v = u + gt
or, 17.83 = 4.9 + 9.81 or, 9.81 = 12.93 12.93
∴ t = \(\frac{12.93}{9.8}\) = 1.32 s
Question 8. A bomb of mass 4 m is disintegrated into two pieces of masses m and 3 m after an explosion. If the velocity of the first piece is u, how much energy is emitted in this explosion?
Answer:
Given
A bomb of mass 4 m is disintegrated into two pieces of masses m and 3 m after an explosion. If the velocity of the first piece is u
Suppose, the velocity of the piece with mass 3 m is v.
From the law of conservation of linear momentum, we get mu + 3mv = 0
or, 3 mv = -mu or, v = –\(\frac{u}{3}\)
∴ the energy emitted in the explosion,
E = \(\frac{1}{2} m u^2+\frac{1}{2} \cdot 3 m v^2=\frac{1}{2} m u^2+\frac{3}{2} m\left(-\frac{u}{3}\right)^2\)
= \(\frac{1}{2} m u^2+\frac{m u^2}{6}=\frac{2}{3} m u^2\)
Question 9. A tennis ball coming with a velocity of 15 m/s is sent back in the opposite direction with a velocity of 20 m/s. If the change of kinetic energy of the ball is 8.75 J, what is the change of momentum of the ball?
Answer:
Given
A tennis ball coming with a velocity of 15 m/s is sent back in the opposite direction with a velocity of 20 m/s. If the change of kinetic energy of the ball is 8.75 J,
Suppose the mass of the ball is m.
If the direction of the return path of the ball is taken as positive, then
initial velocity, u = -15m/s,
final velocity, v = 20 m/s,
change of kinetic energy of the ball is 8.75 J.
∴ \(\frac{1}{2} m v^2-\frac{1}{2} m \dot{u}^2=8.75 \text { or, } \frac{m}{2}\left(v^2-u^2\right)=8.75\)
or, \(m(v-u)(v+u)=2 \times 8.75\)
or, \(m(v-u)=\frac{2 \times 8.75}{(v+u)}=\frac{2 \times 8.75}{20-15}\)
= \(\frac{2 \times 8.75}{5}=3.5 \mathrm{~kg} \cdot \mathrm{m} \cdot \mathrm{s}^{-1}\)
So, change of momentum of the tennis ball is 3.5 kg • m • s-1.
Question 10. From a height of 8 m above the earth’s surface, a body of mass 1kg is thrown upwards in a perpendicular direction with a velocity of 250cm/s. What is its kinetic energy just before touching the ground?
Answer:
Given
From a height of 8 m above the earth’s surface, a body of mass 1kg is thrown upwards in a perpendicular direction with a velocity of 250cm/s
Mass of the body, m = 1 kg.
The body is thrown upwards in a perpendicular direction with a velocity of 250 cm/s.
Initial velocity of the body in the downward direction,
u = -250 cm/s = -2.5 m/s
If the velocity of the body is v just before touching the ground,
v2 = u2 + 2gh = (-2.5)2 + 2 X 9.8 x 8
or, v2 = 163.05
∴ kinetic energy of the body at that time,
⇒ \(E_k=\frac{1}{2} m v^2=\frac{1}{2} \times 1 \times 163.05\)
= 81.525 J
Question 11. The ratio of the kinetic energies of two bodies of masses m and 2m is 1:2. What is the ratio of their linear momentum?
Answer:
If the kinetic energy of the first body is E1, its linear momentum, \(p_1=\sqrt{2 m E_1}\)
and if the kinetic energy of the second body is E2, its linear momentum,
⇒ \(p_2=\sqrt{2 \cdot 2 m \cdot E_2}\)1
0∴ \(\frac{p_1}{p_2}=\frac{\sqrt{2 \cdot m \cdot E_1}}{\sqrt{2 \cdot 2 m \cdot E_2}}=\sqrt{\frac{E_1}{2 E_2}}\)
= \(\sqrt{\frac{1}{2} \times \frac{1}{2}}\left[because \frac{E_1}{E_2}=\frac{1}{2}\right]\)
or, \(\frac{p_1}{p_2}=\frac{1}{2}\)
∴ \(p_1: p_2=1: 2 \)
Question 12. What is the potential energy of a body of of mass 2 kg which is kept at a height of 15 m from the surface of the earth?
Answer:
Mass of the body, m = 2 kg.
Height of the body from the earth’s surface, h = 15 m
Acceleration due to gravity, g = 9.8 m/s2
∴ the potential energy of the body,
Ep = mgh = 2 kg x 9.8 m/s2 x 15 m = 294 J
Question 13. A ball of mass. 100g is thrown 5 m upwards in a vertical direction. What form of energy is present in the ball at the highest point? What is the value of this energy? What is its total energy just before striking the ground?
Answer:
Given
A ball of mass. 100g is thrown 5 m upwards in a vertical direction.
At the highest point, the ball does not have any velocity.
So, at a distance of 5 m from the ground, it has only potential energy stored in it.
∴ value of this energy = mgh = 100 g x 9.8 m/s2 x 5 m
= \(\frac{100}{1000}\)kg x 9.8 m/s2 x 5 m = 4.9 J
Just before it strikes the ground, its distance (h) from the ground is zero.
So, its kinetic energy = total mechanical energy.
According to the law of conservation of mechanical energy, this kinetic energy is equal to the potential energy stored in the ball when it is at the highest distance = 4.9J.
Question 14. Velocity-time graph of a body is shown

- What is the change of kinetic energy from t = 5 s to t = 10 s?
- What is the acceleration of the body?
- What is the value of force applied on the body?
- Calculate the amount of work done by the applied force during time t = 5 s to t = 10 s. Show that the amount of work done during this time is equal to the Increase of kinetic energy of this body.
Answer:
1. From the graph, at time t1 = 5s, velocity v1 of the body is 15 m/s and at time t2 = 10 s, velocity v2 of the body is 20 m/s.
∴ change of kinetic energy during 5 s to 10 s,
⇒ \(E_k=\frac{1}{2} m v_2^2-\frac{1}{2} m v_1^2=\frac{m}{2}\left(v_2^2-v_1^2\right)\)
= \(\frac{2}{2}\left(20^2-15^2\right)=175 \mathrm{~J}\)
2. Acceleration of the body,
⇒ \(a=\frac{v_2-v_1}{t_2-t_1}=\frac{20-15}{10-5}=1 \mathrm{~m} / \mathrm{s}^2\)
3. Force applied on the body, F = ma = 2 x 1 = 2 N
Distance covered by the body during the time from 5 s to 10 s,
s = area of the trapezium EBCF
= \(\frac{1}{2}\)(BE+ CF) x EF = \(\frac{1}{2}\)(15+ 20)(10-5) = 87.5 m
∴ work done by the applied force during this time, W = F.S = 2 x 87.5 = 175 J
∴ W = E
Question 15. A body of mass 50 kg is falling freely from a height of 10 m. What is its total mechanical energy at any point of time?
Answer:
Given
A body of mass 50 kg is falling freely from a height of 10 m.
Total potential energy stored in a body of mass 50 kg at a height of 10 m and at rest is converted to total mechanical energy at any point of time during its free fall.
∴ required total mechanical energy = mgh = 50 x 9.8 x 10J = 490 J
Question 16. At what height above the surface of the earth, does the total energy of a freely falling body of mass 15 kg at any moment becomes 1176 J?
Answer:
The total mechanical energy of a body falling freely from rest always remains constant.
Suppose, the body falls from a height h.
So, mgh = 1176
or, h = \(\frac{1176}{m g}=\frac{1176}{15 \times 9.8}\) = 8m
Question 17. What amount of total work can a man do in 10 minutes, working with a power of 6 J/s?
Answer:
Power of the man, P = 6 J/s
Time of doing work, t = 10 min = 10 x 60s
∴ work done by the man,
W = P X t = 6 x 10 x 60J = 3600 J
Question 18. What is the velocity with which a bullet of mass 80 g has to be fired so that the value of its kinetic energy is 400 J?
Answer:
Mass of the bullet, m = 80 g = 80 x \(\frac{1}{1000}\) kg
Kinetic energy of the bullet, Ek = 400 J
Suppose the velocity of the bullet is v at the time of firing.
Now, according to the equation \(E_k=\frac{1}{2}mv^2\), \(v^2=\frac{2 E_k}{m}\)
or, \(v =\sqrt{\frac{2 E_k}{m}}=\sqrt{\frac{2 \times 400}{80 \times \frac{1}{1000}}}=\sqrt{\frac{2 \times 400 \times 1000}{80}}\)
= \(\sqrt{10 \times 1000}=100 \mathrm{~m} / \mathrm{s}\)
Question 19. A body of mass 100g is rolled up to the top of a smooth plane inclined at 30°. The height of the inclined plane from the ground is 1 m. Determine the value of gravitational potential energy stored in the body when it is at the top of the inclined plane.
Answer:
Given
A body of mass 100g is rolled up to the top of a smooth plane inclined at 30°. The height of the inclined plane from the ground is 1 m.
When the body is rolled up to the top of the inclined plane, its perpendicular height from the surface of the earth is 1 m.
Mass of the body, m – 100 g = 1/100 kg
Acceleration due to gravity, g = 9.8 m/s2
∴ the required gravitational potential energy,
Ep = mgh = 10 x 9.8 x 1 = 0.98 J
Question 20. A body of mass 100 g is thrown upwards in a vertical direction with a velocity of 10m/s. Calculate the kinetic energy of the body at a height of 2 m from the earth’s surface with the help of the law of conservation of mechanical energy.
Answer:
Given
A body of mass 100 g is thrown upwards in a vertical direction with a velocity of 10m/s.
Mass of the body, m = 100 g = 0.1 kg
Velocity with which the body is thrown, u = 10 m/s
Kinetic energy at the moment when the body is thrown,
⇒ \(\dot{E}_k=\frac{1}{2} m u^2=\frac{1}{2} \times 0.1 \times 10^2=5 \mathrm{~J}\)
Gravitational potential energy, Ep = 0
At a height of h = 2 m from the surface of the earth, gravitational potential energy,
⇒ \(E_p^{\prime}=m g h=0.1 \times 9.8 \times 2=1.96 \mathrm{~J}\)
If the kinetic energy of the body is Ek in that position, then from the law of conservation of mechanical energy, we get
⇒ \(E_k^{\prime}+E_p^{\prime}=E_k+E_p \quad \text { or, } E_k^{\prime}+1.96=5+0\)
∴ \(E_k^{\prime}=5-1.96=3.04 \mathrm{~J}\)
Question 21. A body of mass 1 kg is allowed to fall freely from a height of 20 m from the surface of the earth. Calculate the
- kinetic energy and potential energy of the body after 1 s of its release.
- kinetic energy and potential energy of the body after 2 s of its release (fall).
- Show that in the above two cases, total mechanical energy remains unchanged.
Answer:
Given
A body of mass 1 kg is allowed to fall freely from a height of 20 m from the surface of the earth.
Mass of the body, m = 1kg
Initial height, h = 20m
Acceleration due to gravity, g = 9.8 m/s2.
1. If v1 is the velocity of the body after t1 = 1s, then, v1 = gt1 = 9.8 x 1 = 9.8 m/s
If the body descends a distance h1 in one second,
⇒ \(h_1=\frac{1}{2} g t_1^2=\frac{1}{2} \times 9.8 \times 1^2=4.9 \mathrm{~m}\)
Kinetic energy of the body after 1 second, \(E_k^{\prime}=\frac{1}{2} m v_1^2=\frac{1}{2} \times 1 \times 9.8^2=48.02 \mathrm{~J}\)
and potential energy, E’p =mg(h – h1)
= 1 x 9.8(20 – 4.9) = 147.98J
2. If v2 is the velocity of the body after t2 = 2s, v2 = gt2 = 9.8 X 2 = 19.6 m/s
If the body descends a distance h2 in two seconds, then
⇒ \(h_2=\frac{1}{2} g t_2^2=\frac{1}{2} \times 9.8 \times 2^2=19.6 \mathrm{~m}\)
So, kinetic energy of the body after 2 seconds,
⇒ \(E_k^{\prime \prime}=\frac{1}{2} m v_2^2=\frac{1}{2} \times 1 \times 19.6^2=192.08 \mathrm{~J}\)
and potential energy, \(E_p^{\prime \prime}=m g\left(h-h_2\right)\)
= 1 x 9.8 X (20 – 19.6)= 3.92 J
3. Total kinetic energy of the body after 1 second,
⇒ \(E^{\prime}=E_k^{\prime}+E_p^{\prime}\) = (48.02 + 147.98) J =196J
and total kinetic energy of the body after 2 seconds,
⇒ \(E^{\prime \prime}=E_k^{\prime \prime}+E_p^{\prime \prime}\)
= (192.08+ 3.92)J = 196 J
∴ E’ = E ”
Hence, total mechanical energy remains unchanged.