Chapter 3 Chemical Calculations Synopsis
WBBSE Class 10 Chemical Calculations Overview
1. The chemical substances participating in a chemical reaction are known as reactants and the substances produced in the reaction are called products.
2. A chemical equation is the representation of a chemical reaction with the help of symbols and formulas. The reactants are written on the left side while the products are written on the right side of the equation.
3. A chemical equation gives us qualitative as well as quantitative information about the reaction. We can calculate the mass, number of moles and volumes of the involved reactants and products from the chemical equation.
4. Law of conservation of mass: According to the law of conservation of mass proposed by Lavoisier, in any physical or chemical change total mass of all the substances before the change is equal to the total mass of all the substances formed after the change.
5. Law of conservation of mass and energy: The law states that the total amount of mass and energy remains constant before and after any chemical or physical transformation.
Read and Learn Also WBBSE Solutions for Class 10 Physical Science and Environment
6. Equivalence of mass and energy: According to Einstein’s theory of relativity, mass and energy are different aspects of the same entity and are interconvertible.
If a substance ‘A’ of mass ‘m’ is converted into another substance ‘B’ to produce an equivalent amount of energy T, then according to Einstein’s equation of mass-energy equivalence, E=mc2, where c = velocity of light in vacuum.

7. In normal chemical reactions very minute change of mass takes place, which is too small to be measured. Hence, the law of conservation of mass and energy is not applicable to a general chemical reaction. However, the law is applicable to nuclear reactions where a change in energy is detectable.
8. The mass (measured in gram units) of 1 L of a gas at STP is known as the standard density of the gas.
9. The vapour density or relative density of a gas is defined as the ratio of the mass of a given volume of the gas at a given temperature and pressure to the mass of an equal volume of hydrogen gas at the same temperature and pressure.
10. Vapour density is a ratio and hence, it has no unit.
11. Vapour density is independent of temperature and pressure. So, its value is constant at any temperature or pressure.
12. Molecular mass of a gas is twice its vapour density.
13. At STP, the volume of 1 mol of any gas is 22.4 L.
14. At STP, the mass of 1 L of any gas = vapour density of the gas x 0.089
15. Calculation of relative quantities of reactants and products in a chemical reaction is known as stoichiometry.
Chapter 3 Chemical Calculations Short And Long Answer Type Questions
Question 1. Discuss the significance of a balanced chemical equation in chemical calculation.
Answer:
The significance of a balanced chemical equation in chemical calculation
A chemical equation provides us with qualitative as well as quantitative information about a chemical reaction.
Qualitative information: From the chemical equation we can easily identify the reactants and products in a chemical reaction.
- Quantitative information:
It gives us an idea about the number of atoms and molecules of reactants and products involved in the reaction.
- It gives us an idea about the mass of the reactants that reacts with each other and the mass of the products formed.
- If the reactants or products are gases, then at a given temperature and pressure, their volume can be determined.
Question 2. What information can be obtained from the equation \(2 \mathrm{H}_2+\mathrm{O}_2 \rightarrow 2 \mathrm{H}_2 \mathrm{O}\)?
Answer:
Information can be obtained from the equation \(2 \mathrm{H}_2+\mathrm{O}_2 \rightarrow 2 \mathrm{H}_2 \mathrm{O}\)
The equation provides us with the following information:
Qualitative information: Hydrogen and oxygen react with each other to form water.
Quantitative information:
1. 2 mol of hydrogen reacts with 1 mol of oxygen to produce 2 mol of water.
2. Here, 2 X (2 x l)g = 4 g hydrogen reacts with (2 X 16)g = 32g oxygen to produce 2 x (2 x 1 + 16) = 36g water.
3. At the same temperature and pressure, 2 volumes of hydrogen combine with 1 volume of oxygen to produce 2 volumes of water vapour.
4. At STP, 2 x 22.4L = 44.8L hydrogen combines with 22.4 L oxygen to produce 2 x 22.4 L = 44.8 L steam.
Question 3. State the limitations of a chemical equation.
Answer:
A chemical equation cannot give us information about:
1. Concentration of reactants and products.
2. The rate of the reaction or the time required for the completion of the reaction.
3. Whether the reaction has completed or not.
Question 4. In a chemical reaction, mass is conserved explain.
Answer:
In a chemical reaction, mass is conserved as
In a chemical reaction, reactants are transformed into products. The total mass of the products is found to be equal to the total mass of the reactants.
Let us consider, A and B react with each other to produce C and D in a reaction. Hence, the mass of A + mass of B = mass of C + mass of D Thus, the total mass of A and B = total mass of C and D.
Understanding Molar Mass in Chemical Calculations
Question 5. Show with the help of an experiment that mass is conserved in a chemical reaction.
Answer:
With The help of an experiment that mass is conserved in a chemical reaction:
The law of conservation of mass can be proved with the help of rusting of iron.
Materials required: A hard glass test tube, some new iron nails, water, rubber cork, and common balance.
Experiment: A small amount of water is taken in a hard glass test tube and some glossy iron nails are dipped into the water. The mouth of the test tube is covered with the help of a rubber cork. The mass of the test tube is measured with a common balance and is left undisturbed for some days.
Observation: After some days it will be observed that brown-coloured rust is formed on the iron nails. In this condition, the mass of the test tube is measured again with a common balance and it is observed that the mass of the test tube before and after the experiment is equal.
Conclusion: Iron nails react with oxygen and water present in the test tube to form rust.
∴ \(\mathrm{Fe}+\mathrm{O}_2 \text { + water vapour } \rightarrow \mathrm{Fe}_2 \mathrm{O}_3 \cdot x \mathrm{H}_2 \mathrm{O}\)
x = a number of water molecules.
In this case, the total mass of the iron piece along with unreacted oxygen and water vapour is equal to the mass of the rusted iron. Thus, we can say that mass is conserved during chemical reactions.
Question 6. A piece of iron gets heavier when it is kept in moist air for a long time state whether mass is conserved in this case?
Answer:
When iron is kept in moist air for a long time rust is formed over the surface of the iron piece. Oxygen and water vapour present in the air react with iron to form hydrated ferric oxide (Fe2O3 . xH2O).
As a result, the mass increases. So, the total mass of the iron piece along with oxygen and water vapour is equal to the mass of rusted iron. Hence, mass is conserved in this process.
Question 7. On burning, the mass of a candle decreases state whether mass is conserved in this process.
Answer: On burning, the wax of the candle reacts with aerial oxygen to form carbon dioxide (CO2), other gases and water vapour. These products escape into atmosphere. If all the products are collected together, then it would be observed that the total mass of the products is equal to the total mass of wax and used oxygen.
Thus, it can be said mass of wax + mass of used oxygen before burning = mass of remaining candle + mass of produced CO2 + mass of other gases produced + mass of water vapour.
Hence, mass is conserved in this process.
Question 8. Explain with reasons whether law of conservation of mass is followed during electrolysis of acidified water.
Answer:
Reasons whether law of conservation of mass is followed during electrolysis of acidified water
During electrolysis of acidified water, water decomposes to form hydrogen and oxygen.
If electrolysis is carried out in a closed vessel it will be observed that the total mass remains same before and after the electrolysis, i.e., total mass of acidified water = mass of remaining acidified water + mass of produced hydrogen + mass of produced oxygen.
So, it can be said that law of conservation of mass is obeyed during the electrolysis of acidified water.
Question 9. In the normal chemical reactions, mass does not undergo measurable change-explain.
Answer:
In the normal chemical reactions, mass does not undergo measurable change
In the normal chemical reactions, heat is either evolved or absorbed. According to mass-energy equivalence, if heat is evolved in a reaction, an equivalent amount of mass of the reactant will decrease. On the other hand, if heat is absorbed, an equivalent amount of mass of the reactant will increase.
But, in normal chemical reactions, the change in mass observed due to the absorption or evolution of heat is too small to be measured with a balance. Hence, we can say that no measurable change of mass takes place in a chemical reaction.
Question 10. What is the significance of the equation E=mc2? In normal chemical reactions heat is either evolved or absorbed, yet, the change of mass is not observed. Why?
Answer:
Significance of the equation E=mc2
When a substance of mass ‘m‘ is converted into another substance, an equivalent amount of energy E is produced. The equation, E=mc2, represents the conversion of mass into energy and vice versa, where c = velocity of light in vacuum.
The change of mass involved in the normal chemical reactions is too small to be measured. Thus, a change of mass is not generally observed in normal chemical reactions.
Question 11. The law of conservation of mass is not applicable in high energy transformations— explain.
Answer:
The law of conservation of mass is not applicable in high energy transformations
During high-energy transformations such as in nuclear reactions, the total mass is converted into an equivalent amount of energy.
As a result, the large amount of energy produced can be determined from Einstein’s equation, E=mc2. Hence, it can be said that in high-energy transformations law of conservation of mass is not valid. Instead, in such cases, law of conservation of mass and energy is applicable.
Question 12. 1g of a substance is completely converted into energy. Using the equation E=mc2 find the amount of energy obtained due to the conversion.
Answer:
Given
1g of a substance is completely converted into energy. Using the equation E=mc2
The eqaution, E=mc2, represents mass-energy equivalence where,
E = energy, m = mass and c = speed of light in vacuum = 3 x 1010 cm . s-1
∴ E = 1 x (3 x 1010)2erg
= 9 x 1020 erg = 2.14 x 1013 calorie. [∴1 cal = 4.2 x 107 erg]
Question 13. What do you mean by standard density of a gas? What is vapour density or relative density of a gas?
Answer:
Density of a gas
1. The mass (measured in gram units) of 1 litre of a gas at STP is known as standard density of the gas.
Vapour densit
2. The vapour density or relative density of a gas is defined as the ratio of mass of a given volume of the gas at a given temperature and pressure to the mass of an equal volume of hydrogen gas at the same temperature and pressure.
Question 14. Deduce the relationship between the standard density and vapour density of a gas.
Answer:
The relationship between the standard density and vapour density of a gas
Vapour density of a gas = mass of a given volume of the gas at a given temperature and pressure/ mass of an equal volume of hydrogen gas at the same temperature and pressure
or, Vapour density of a gas = \(\frac{\text { mass of } V \text { volume of the gas at STP }}{\text { mass of } V \text { volume of hydrogen gas at STP }}\) = \(\frac{\text { mass of } 1 \mathrm{~L} \text { of the gas at STP }}{\text { mass of } 1 \mathrm{~L} \text { of hydrogen gas at STP }}\)
or, Vapour density of the gas =\(\frac{\text { standard density of the gas }}{\text { standard density of hydrogen gas }}\)
or, Vapour density of the gas =\(=\frac{\text { standard density of the gas }}{0.089 \mathrm{~g} \cdot \mathrm{L}^{-1}}\)
Examples of Chemical Calculation Problems
Question 15. Deduce the relationship between molecular mass (M) and vapour density (D) of a gas.
Answer:
The relationship between molecular mass (M) and vapour density (D) of a gas;
Vapour density of a gas = mass of a given volume of the gas at a given temperature and pressure/ mass of equal volume of hydrogen gas at the same temperature and pressure
According to Avogadro’s law, if ‘n’ number of molecules are present in 1/ L of a gas at a given temperature and pressure, ‘n’ number of H2 molecules will also be present in V L of hydrogen gas at the same temperature and pressure.
Hence, vapour density of the gas (D) = \(\frac{\text { mass of } n \text { molecules of the gas }}{\text { mass of } n \text { molecules of hydrogen }}\)
= \(\frac{\text { mass of } 1 \text { molecule of the gas }}{\text { mass of } 1 \text { molecule of hydrogen }}\)
= \(\frac{\text { mass of } 1 \text { molecule of the gas }}{\text { mass of } 2 \text { atoms of hydrogen }}\) (as hydrogen molecule is diatomic)
= \(\frac{1}{2} \times \frac{\text { mass of } 1 \text { molecule of the gas }}{\text { mass of } 1 \text { atom of hydrogen }}\)
= \(\frac{1}{2} \times \text { molecular mass of the gas }(M)\)
∴ \(D=\frac{M}{2}\) ∴ M = 2D
Thus, the molecular mass of gas = 2 Χ vapour density of the gas.
Question 16. How will you understand whether a gas is heavier than air or not? The vapour density of chlorine is 35.5. How will it displace air from a gas jar?
Answer:
1. lf the vapour density of a gas is found to be greater than that of air (14.4), it can be assumed that the gas is heavier than air.
2. The vapour density of chlorine is greater than air. Hence, it is heavier than air. So, there will be an upward displacement of air when chlorine gas is collected into a gas jar filled with air.
Question 17. Vapour density of a gas does not change with temperature justified.
Answer:
Vapour density of a gas does not change with temperature
The vapour density or relative density of a gas is defined as the ratio of the mass of a given volume of the gas at a given temperature and pressure to the mass of an equal volume of hydrogen gas at the same temperature and pressure.
With an increase or decrease in temperature the volumes of hydrogen as well as the concerned gas increase or decrease in the same proportion. Hence, the vapour density of a gas does not change, with temperature.
Question 18. Air contains 74% nitrogen, 24% oxygen and 2% CO2 by volume. Which of the following gases are heavier and which ones are lighter than air CO2, NH3, CH4, and Cl2?
Answer:
Given
Air contains 74% nitrogen, 24% oxygen and 2% CO2 by volume
Average molecular mass of air = \(\frac{28 \times 74+32 \times 24+2 \times 44}{100}\) = 29.28.
So, Vapour density of air = \(\frac{29.28}{2}\) = 14.64.
Now, the vapour density of CO2 = \(\frac{44}{2}\) = 22
Hence, it is heavier than air.
Vapour density of NH3 = \(\frac{17}{2}\) = 8.5.
Hence, it is lighter than air.
Vapour density of CH4 =\( \frac{16}{2}\) = 8.
Hence, it is lighter than air.
Vapour density of Cl2 = \(\frac{71}{2}\) = 35.5.
Hence, it is heavier than air.
Question 19. Dry air is heavier than moist air. Explain with reason.
Answer:
Dry air is heavier than moist air:
The average vapour density of air is 14.4 while that of water vapour is 9. Hence, a given volume of dry air is heavier than an equal volume of water vapour. Moist air contains more amount of water vapour as compared to dry air. That is why dry air is heavier than moist air.
Chapter 3 Chemical Calculations Very Short Answer Type Questions Choose The Correct Answer
Question 1. Law of conservation of mass was proposed
- Dalton
- Lavoisier
- Arrhenius
- Proust
Answer: 2. Lavoisier
Question 2. The difference in mass between reactants and products is
- Observed in low-energy transformations
- Observed in a general chemical reaction
- Observed in high-energy transformations
- Never observed
Answer: 3. Observed in high-energy transformations
Question 3. Two substances, A and B react with each other to produce C and D. Which of the following statements is true?
- Total mass of A and B > total mass of C and D
- Total mass of A and B < total mass of C
- Total mass of A and B = total mass of C and D
- Mass of A = mass of 8 and mass of C = mass of D
Answer: 3. Total mass of A and B = total mass of C and D
Question 4. The mass-energy equivalence is represented by the equation
- E2=mc
- E=mc2
- E= m2c
- E = mc
Answer: 2. E=mc2
Question 5. Which of the following cannot be determined from a chemical equation?
- Number of moles of reactants & products
- Volume of reactants and products at STP
- Concentration of reactants and products
- Mass of reactants and products.
Answer: 3. Concentration of reactants and products
Question 6. The equation E=mc2 is related to
- Newton
- Planck
- Avogadro
- Einstein
Answer: 4. Einstein
Question 7. Measurable change in mass takes place in
- General chemical reactions
- Nuclear reactions
- Exothermic reactions
- Endothermic reactions
Answer: 2. Nuclear reactions
Question 8. In the equation E=mc2, the terms E, m and c respectively represents
- Mass, energy and speed of light in vacuum
- Speed of light in vacuum, mass and energy
- Energy, mass and speed of light in vacuum
- Energy, speed of light in vacuum and mass
Answer: 3. Energy, mass and speed of light in vacuum
Question 9. According to mass-energy equivalence, in an exothermic reaction the mass of products
- Will decrease in an equivalent amount
- Will increase in an equivalent amount
- Will increase or decrease in an equivalent amount
- Will remain unchanged
Answer: 1. Will decrease in an equivalent amount
Question 10. According to mass-energy equivalence, in an endothermic reaction the mass of products
- Will decrease in equivalent amount
- Will increase in equivalent amount
- Will increase or decrease in equivalent amount
- Will remain unchanged
Answer: 2. Will increase in equivalent amount
Question 11. Total amount of mass and energy after any physical or chemical transformation
- Decreases
- Increases
- Remains constant
- Norie of these
Answer: 3. Remains constant
Question 12. The relationship between mass and number of moles is given by
- \(\text { number of moles }=\frac{\text { molar mass }}{\text { given mass }}\)
- \(\text { molar mass }=\frac{\text { number of moles }}{\text { given mass }}\)
- \(\text { number of moles }=\frac{\text { given mass }}{\text { molar mass }}\)
- Number of moles = given mass x molar mass
Answer: 3. \(\text { number of moles }=\frac{\text { given mass }}{\text { molar mass }}\)
Question 13. The amount of N2 produced from 64g NH4NO2 is
- 14g
- 28g
- 32g
- 40g
Answer: 2. 28g
Question 14. The volume of NO2 produced when 2 mol of NO reacts with O2 at STP is
- 11.2 L
- 44.8 L
- 22.4 L
- 5.6 L
Answer: 3. 22.4 L
Question 15. Which of the following relations is correct?
- At STP, mass of 1L of a gas = vapour density of the gas x 0.89
- At STP, mass of 1L of a gas = vapour density of the gas x 0.089
- Vapour density of a gas = 0.089 x mass of 1L of a gas at STP
- Vapour density of a gas = 2.24 x molecular masss of the gas
Answer: 2. At STP, mass of 1L of a gas = vapour density of the gas x 0.089
Question 16. 1 mol of nitrogen combines with 3 mol of hydrogen to produce.
- 1 mol NH3
- 2 mol NH3
- 3 mol N3
- 4 mol NH3
Answer: 2. 2 mol NH3
Question 17. The relationship between molecular mass and vapour density is given by
- 2M = D
- M = D2
- M = 3D
- M = 2D
Answer: 4. M = 2D
Question 18. The vapour density of SO2 is
- 16
- 32
- 64
- 48
Answer: 2. 32
Question 19. The molecular mass of a gas is 46. Its vapour density is
- 23u
- 23 amu
- 23 g
- 23
Answer: 4. 23
Question 20. The vapour density of a gas is 22. The volume of 44g of the gas at STP is
- H.2L
- 5.6L
- 22.4L
- 44.8L
Answer: 3. 22.4L
Question 21. The average vapour density of air is
- 14.4
- 16.5
- 17.2
- 20.1
Answer: 1. 14.4
Question 22. The molecular mass of chlorine is 71. Which of the following statements is correct?
- Chlorine gas is lighter than air
- Chlorine gas is heavier than air
- Chlorine gas has the almost same mass as that of air
- Unpredictable
Answer: 2. Chlorine gas is heavier than air
Question 23. Mass of 112 mL O2 at NTP is
- 0.64 g
- 0.96 g
- 0.32 g
- 0.16g
Answer: 4. 0.16g
Question 24. Mass of 6.022 x1022 number of molecules of water is
- 18 g
- 1.8 g
- 9 g
- 0.18 g
Answer: 2. 1.8 g
Question 25. Volume of CO2 gas at STP, obtained by heating 1 mol of CaCO3 is
- 22.4 L
- 11.2 L
- 5.6 L
- 44.8L
Answer: 1. 22.4 L
Question 26. The percentage increase in mass of an Mg wire after its combustion in pure oxygen is
- 100%
- 66.7%
- 24%
- 80%
Answer: 2. 66.7%
Question 27. The volume of hydrogen at STP obtained from the reaction of 4 mol Na with water is
- 22.4 L
- 44.8 L
- 11.2 L
- 5.6L
Answer: 2. 44.8 L
Question 28. The number of moles of NH3 gas formed by the reaction of 2 mol N2 with 6 mol H2 is
- 6 mol
- 2 mol
- 8 mol
- 4 mol
Answer: 4. 4 mol
Question 29. Vapour density of sulphur trioxide is
- 40
- 32
- 36
- 80
Answer: 1. 40
Question 30. Vapour density of a gas is 32. Which of the following is the molecular mass of the gas?
- 8
- 16
- 32
- 64
Answer: 4. 64
Question 31. The volume of CO2 at STP, obtained by the complete combustion of 1.2 g of C is
- 1.2 L
- 2.24 L
- 4.8 L
- 4.4 L
Answer: 2. 2.24 L
Question 32. Molecular mass of methane is 16. Which of the following is its vapour density?
- 22.4
- 8
- 16
- 32
Answer: 2. 8
Question 33. The volume of 2 mol CO2 at STP is
- 11.2 L
- 22.4 L
- 44.8 L
- 44.8 mL
Answer: 3. 44.8 L
Question 34. The mass of 1 L of H2 gas at STP is
- 1g
- 2g
- 0.089 g
- 22.4 g
Answer: 3. 0.089 g
Question 35. Amount of nitrogen required to produce 4 mol NH3 is
- 14 g
- 28 g
- 42 g
- 56 g
Answer: 4. 56 g
Question 36. The volume of 1.7 g of ammonia gas, at STP is
- 22.4 L
- 2.24 L
- 11.2 L
- 1.12 L
Answer: 2. 2.24 L
Question 37. The volume of 2.8 g ethylene, at STP is
- 224 L
- 22.4 L
- 2.24 L
- 0.224 L
Answer: 3. 2.24 L
Question 38. Vapour density of a gas is 16. The mass of 11.2 L of the gas at STP is
- 22.4 g
- 8 g
- 16 g
- 11.2 g
Answer: 3. 16 g
Question 39. Ratio of the standard density of a gas at NTP to its vapour density is
- 0.89
- 0.98
- 0.089
- 8.9
Answer: 3. 0.089
Question 40. The volume of 17 g of a gas at STP is 22.4 L. Vapour density of the gas will be
- 17
- 0.85
- 8.5
- 1.7
Answer: 3. 8.5
Question 41. The ratio of molecular mass to the vapour density of a gas with respect to hydrogen is
- 2:1
- 1:2
- 1:1
- 1:3
Answer: 1. 2:1
Question 42. Number of molecules formed by the complete reaction of 1 mol of C with 1 mol of O2 is
- 6.022 × 1023
- 1.806 × 1024
- 6.022 × 1022
- 6.022 × 1024
Answer: 1. 6.022 X 1023
Question 43. Value of c in the equation, E=mc2 is
- 3 x 1010 m/s
- 3 x 109 cm/s
- 3 x 109 m/s
- 3 x 1010 cm/s
Answer: 4. 3 x 1010 cm/s
Question 45. Mass of 11.2 L CO2 at STP is
- 88 g
- 44 g
- 32 g
- 22 g
Answer: 4. 22 g
Question 46. Molar mass of water (in g . mol-1 unit) is
- 16
- 18
- 20
- 22
Answer: 2. 18
Question 47. Mass of 12.046 × 1023 number of molecules of N2 is
- 28 g
- 56 g
- 14 g
- 7g
Answer: 2. 56 g
Question 48. If the volume of 44 g of CO2 at STP be 22.4L, the mass of 5.6 L of CO2 will be
- 11g
- 22g
- 33 g
- 44 g
Answer: 1. 11g
Question 49. According to E=mc2 equation, the unit of E will be
- meV
- MeV
- mev
- mV
Answer: 2. MeV
Question 50. What is the amount of chalk to be reacted with HCI to produce 5.6 L of CO2 at STP?
- 25 g
- 30g
- 15 g
- 20 g
Answer: 1. 25 g
Question 51. If the vapour density of a compound be 13, its molecular formula may be
- CO2
- C2H4
- C2H6
- C2H2
Answer: 4. C2H2
Question 52. The gas whose vapour density is equal to that of CO gas is
- O2
- N2
- H2
- Cl2
Answer: 2. N2
Question 53. Amount of carbon required to produce 2 mol CO2is
- 12 g
- 6g
- 24 g
- 44 g
Answer: 3. 24 g
Question 54. The percentage of Ca in CaCO3 is
- 25
- 40
- 60
- 80
Answer: 2. 40
Question 55. Amount of hydrogen formed by electrolysis of 5 mol of water is
- 4 g
- 5 g
- 20 g
- 10g
Answer: 4. 10g
Question 56. Vapour density of carbon dioxide is
- 22
- 11
- 44
- 88
Answer: 1. 22
Question 57. 22 g CO2 is formed by the reaction of 6 g carbon with oxygen, the required amount of oxygen is
- 6g
- 18g
- 16g
- 28g
Answer: 3. 16g
Chapter 3 Chemical Calculations Answer In Brief
Common Mistakes in Chemical Calculations
Question 1. What do you mean by ‘conservation of mass’ in a chemical reaction?
Answer:
‘Conservation of mass’ in a chemical reaction
In a chemical reaction, total mass of all the reactants is equal to total mass of all the products. This is known as the ‘conservation of mass’ in a chemical reaction.
Question 2. State the law of conservation of mass and energy.
Answer:
Law of conservation of mass and energy
The law of conservation of mass and energy states that the total mass and energy before and after a transformation is always constant.
Wbbse class 10 physical science chapter 3 question answers
Question 3. When is the ‘law of conservation of mass and energy’ applicable instead of ‘law of conservation of mass’?
Answer: In high energy transformations like nuclear reactions, ‘law of conservation of mass and energy’ is applied instead of ‘law of conservation of mass’.
Question 4. Define ‘reactants’ and ‘products’.
Answer:
‘Reactants’ and ‘products’
The chemical substances participating in a chemical reaction are known as ‘reactants’. The substances produced due to chemical reactions among the reactants are known as ‘products’.
Question 5. Is the change in mass detectable in normal chemical reactions?
Answer: No, the change in mass is not detectable in normal chemical reactions.
Question 6. Write the relationship between molecular mass (M) and vapour density (D) of a gas.
Answer: Molecular mass (M)=2x vapour density (D).
Question 7. Vapour density of SO2 gas at 25°C and normal pressure is 32.-What will be its vapour density at 50°C?
Answer: Since, vapour density is independent of temperature and pressure, hence, the vapour density of SO2 at 50°C will remain 32.
Question 8. Define ‘stoichiometry’.
Answer:
‘Stoichiometry’
Stoichiometry is defined as the calculation of relative quantities of reactants and products in a chemical reaction.
Wbbse class 10 physical science chapter 3 question answers
Question 9. What is the unit of vapour density?
Answer:
Unit of vapour density
Since vapour density is a ratio, thus it has no unit.
Question 10. Mass of 5.6 L of a gas at STP is 11g. Find the vapour density of the gas.
Answer: Mass of 5.6 x 4 = 22.4L of the gas at STP is 11 x 4 = 44g. Hence, vapour density of the gas = 44/2 = 22.
Question 11. Name the physical quantity whose unit is ‘mole’.
Answer:’Mole’ is the SI unit of the amount of substance.
Question 12. What is the number of molecules in 22.4 L of a gas at STP?
Answer: 6.022 x 1023.
Question 13. What is the number of moles in m g of a gas having molar mass of M g/mol?
Answer: Number of moles of the said gas = M/N.
Question 14. What is the number of molecules in 1.8 g water?
Answer: Number of molecules present in 1.8 g water = 6.022 X 1023 X 1.8/18 = 6.022 x 1022
Question 15. How much ammonia is equivalent to 4 mol ammonia?
Answer: 4 mol ammonia is equivalent to 17 x 4 = 68 g of ammonia.
Question 16. What is the mass of 12.046 x 1023 number of nitrogen molecules?
Answer: Mass of 6.022 x 1023 number of nitrogen molecules = 28 g
∴ Mass of 12.046 x 1023 number of nitrogen molecules
∴ \(\frac{28 \times 12.046 \times 10^{23}}{6.022 \times 10^{23}} \mathrm{~g}\)
WBBSE Class 10 Revision Notes on Chemical Calculations
Question 17. Calculate the molecular mass of a gas if the volume of 50 g of the gas is 44.8 L at STP.
Answer: Gram-molecular mass of the gas = mass of 22.4L of the gas at STP = \(\frac{50 \times 22.4}{44.8}\) = 25g
∴ Molecular mass of the gas is 25.
Question 18. What is the relationship between standard density and vapour density?
Wbbse class 10 physical science chapter 3 question answers
Answer: Standard density = 0.089 x vapour density.
Question 19. If the vapour density of a bimolecular gas be 14, calculate its atomic mass.
Answer: Molecular mass of the gas = 2 x 14 = 28
Since the gas is bimolecular, hence its atomic mass = 28/2 =14.
Chapter 3 Chemical Calculations Fill In The Blanks:
Question 1. A chemical equation. provides us with _________ and ____________ information.
Answer: Quantitative, qualitative
Question 2. The velocity of light in a vacuum is _________.
Answer: 3×108m.s-1.
Question 3. The change in mass is __________ in normal chemical reactions.
Answer: Undetectable
Question 4. Mass is ____________ in case of rusting of iron.
Answer: Conserved
Question 5. The volume of H2S produced from 88g FeS is __________.
Answer: 22.4L
Question 6. When hydrogen reacts with oxygen to form water, the number of moles ___________.
Answer: Decreases
Question 7. Vapour density is __________ of temperature and pressure.
Answer: Independent
Question 8. CO2 is heavier than air as the__________ of CO2 is greater than air.
Answer: Vapour density
Question 9. If the vapour density of a gas is 20, the number of moles of 80g of the gas is ________.
Answer: 2
Question 10. The mass of 1L H2 at STP is ________.
Answer: 0.089g
Question 11. In a chemical reaction, the total mass of reactants is = the total mass of the _________.
Answer: products
Wbbse class 10 physical science chapter 3 question answers
Question 12. The proposer of E=mc2 equation is ____________
Answer: Albert Einsein
Question 13. At STP/ 22.4 L of H2 reacts with 22.4 L of Cl2 to produce ________ L of HCl.
Answer: 44.8
Question 14. Vapour density is a ______ less quantity.
Answer: Unit
Chapter 3 Chemical Calculations State Whether True Or False
Question 1. The total mass remains constant before and after a chemical reaction.
Answer: True
Question 2. The vapour density of a gas is twice its molecular mass.
Answer: False
Question 3. 35Cl atom is taken as the standard for the determination of atomic mass.
Answer: False
Wbbse class 10 physical science chapter 3 question answers
Question 4. 8g oxygen contains 3.011 Χ 1023 molecules.
Answer: False
Question 5. Vapour density is a unitless quantity.
Answer: True
Question 6. 16g oxygen is equal to 0.5 mole oxygen.
Answer: True
Question 7. 6.022 x 1023 molcules of water weighs 18g.
Answer: True
Question 8. The substances produced in a chemical reaction are called reactants.
Answer: False
Question 9. The volume of lg gas at STP is 22.4L.
Answer: True
Wbbse class 10 chemical calculation
Question 10. Scientist Lavoisier proved the ‘law of conservation of mass’.
Answer: True
Question 11. Interconversion of mass and energy is possible.
Answer: True
Question 12. At STP, the mass of 22.4 L of any substance refers to its molecular mass.
Answer: False
Question 13. The amount of O2 formed by heating 4 mol of KCIO3 is 2 mol.
Answer: False
Question 14. Vapour density of a gas x 0.089 =standard density of the gas.
Answer: True
Question 15. If the molecular mass of a gas is 44, its vapour density will be 24.
Answer: False
Chapter 3 Chemical Calculations Numerical Examples
Question 1. 3 g of carbon reacts completely with 8 g oxygen to produce carbon dioxide. How much carbon dioxide will be produced? Justify your answer.
Answer:
Given
3 g of carbon reacts completely with 8 g oxygen to produce carbon dioxide.
During chemical reactions, law of conservation of mass is obeyed and hence, according to the law, total mass of the reactants must be equal to the total mass of the products.
Here, mass of carbon = 3g; mass of oxygen = 8g
Hence, mass of produced CO2 = (3 + 8)g = 11 g
Applications of Chemical Calculations in Real Life
Question 2. 15.9 g copper sulphate and 10.6 g sodium carbonate react with each other to produce 14.2 g sodium sulphate and 12.3 g copper carbonate. Show that the results are in accordance with the law of conservation of mass.
Answer:
Given
15.9 g copper sulphate and 10.6 g sodium carbonate react with each other to produce 14.2 g sodium sulphate and 12.3 g copper carbonate.
According to the given data,
Mass of copper sulphate \(=15.9 \mathrm{~g}\)
Mass of sodium carbonate \(=10.6 \mathrm{~g}\)
So, total mass of reactants =26.5 g similarly for the products,
Wbbse class 10 chemical calculation
Mass of sodium sulphate 14.2 g
Mass of copper carbonate =12.3 g
Total mass of products =26.5 g
Hence, in the given reaction, total mass of the reactants = total mass of the products. So, we can say that the given data is in accordance with the law of conservation of mass.
Question 3. When 3.0g ethane (C2H6) is burnt in air 8.8g CO2 and 5.4g H2O are produced. Show that the result is in accordance with law of conservation of mass.
Answer:
Given
When 3.0g ethane (C2H6) is burnt in air 8.8g CO2 and 5.4g H2O are produced.
When ethane is burnt in air, following reaction takes place
2\(\mathrm{C}_2 \mathrm{H}_6+7 \mathrm{O}_2 \rightarrow 4 \mathrm{CO}_2+6 \mathrm{H}_2 \mathrm{O}\)
Gram-molecular mass of ethane = 30g.
Hence, from the equation it can be deduced that (2 x 30) g ethane reacts with (7 x 32)g oxygen.
∴3g ethane reacts with \(\frac{7 \times 32 \times 3}{2 \times 30}\) = 11.2 g oxygen.
Thus, total mass of reactants = (3 + 11.2 g) =14.2g
Total mass of products = (8.8 + 5.4)g = 14.2g.
Thus, the result agrees with the law of conservation of mass.
Question 4. 30g potassium chlorate on heating produces 14.9g potassium chloride and 9.6g oxygen. How much potassium chlorate remains unreacted?
Answer:
Given
30g potassium chlorate on heating produces 14.9g potassium chloride and 9.6g oxygen.
Let us consider, the mass of unreacted potassium chlorate is xg.
In a chemical reaction mass is conserved. Hence,mass of potassium chlorate taken = mass of unreacted potassium chlorate + mass of produced potassium chloride + mass of produced oxygen
∴30 = x+ 14.9 + 9.6
X = 5.5
Hence, 5.5 g potassium chlorate will remain unreacted.
Wbbse class 10 chemical calculation
Question 5. When 12.25g of potassium chlorate is strongly heated, it produces 7.45g potassium chloride and gives out 3.36 L oxygen at standard temperature and pressure. Show that the reaction obeys the law of conservation of mass.
Answer:
Given
When 12.25g of potassium chlorate is strongly heated, it produces 7.45g potassium chloride and gives out 3.36 L oxygen at standard temperature and pressure.
The molecular mass of oxygen = 32.
Hence, at SIP 22.41 oxygen weighs 32g.
∴ At STP 3.36 L oxygen weights \(\frac{32 \times 3.36}{22.4}\) g= 4.8g.
Mass of potassium chlorate = 12.25 g.
Total mass of produced oxygen and KCI = (4.8 + 7.45)g = 12.25g.
Thus, we can say that the reaction obeys the law of conservation of mass.
Question 6. What amount of sodium chloride will react with 3.41g silver nitrate to produce 1.70g sodium nitrate and 2.87g silver chloride? Consider that the mass is conserved completely in the reaction.
Answer:
1. As the mass is conserved in a chemical reaction, we can say, mass of sodium chloride + mass of silver nitrate = mass of sodium nitrate + mass of silver chloride or, mass of sodium chloride + 3.41g
Therefore, mass of sodium chloride = (1.70 + 2.87 -3.41)g = 1.16 g
Question 7. 6.3g NaHCO3 when added to 15g acetic acid solution produces 18g of residue. Find the mass of evolved CO2.
Answer:
Given
6.3g NaHCO3 when added to 15g acetic acid solution produces 18g of residue.
According to the law of conservation of mass in a chemical reaction, total mass of reactants = total mass of products
Here, total mass of reactants = (6.3 + 15) g = 21.3 g
Hence, 21.3g = mass of residue + mass of evolved CO2 or, 21.3 g = 18 g + mass of evolved CO2
Therefore, mass of evolved CO2 = (21.3- 18) g = 33 g.
Wbbse class 10 chemical calculation
Question 8. At STP, mass of 250 ml of a gas is 0.7317g. If the density of hydrogen at STP is 0.08987g •L-1, then find the vapour density of the gas. What will be its molecular mass?
Answer:
Given
At STP, mass of 250 ml of a gas is 0.7317g. If the density of hydrogen at STP is 0.08987g •L-1,
At STP, mass of 250 mL of hydrogen gas = \(\frac{0.08987 \times 250}{1000} \mathrm{~g}\) = 0.0224g
Hence, vapour density of the gas \(\frac{\text { mass of } 250 \mathrm{~mL} \text { of the gas at STP }}{\text { mass of } 250 \mathrm{~mL} \text { hydrogen at STP }}\) = \(\frac{0.7317 \mathrm{~g}}{0.0224 \mathrm{~g}}\) = 32.66.
Molecular mass of the gas = 2 x vapour density = 2 x 32.66 = 65.32 .
Question 9. At 750mm pressure and 25° C 44.8 temperature, 0.15 g of a gas occupies 45.95mL. Find the standard density,vapour density and molecular mass of the gas.
Answer:
Given
At 750mm pressure and 25° C 44.8 temperature, 0.15 g of a gas occupies 45.95mL.
Let, the volume of the gas at STP = V1 mL
p1 = 760mm, T1 = 273 K V2 = 45.95mL, p2 = 750 mm, T2= (25 + 273) K = 298K
By combining of Boyle’s law and Charles’ law \(\frac{P_1 V_1}{T_1}=\frac{P_2 V_2}{T_2}\)
∴ \(V_1=\frac{750 \times 45.95 \times 273}{760 \times 298} \mathrm{~mL}\) = 41.54mL
Thus, at STP, 41.54mL of the gas weighs 0.15g.
Hence, at STP, 1000 mL of the gas weights \(\frac{0.15 \times 1000}{41.54} \mathrm{~g}\) = 3.61g.
Therefore, the standard density of the gas = is 361 g. L-1.
Vapour density = \(\frac{\text { standard density }}{0.089}\) = \(\frac{3.61}{0.089}\) = 40.56.
Molecular mass = 2 x vapour density ^ 2 x 40.56 = 81.12.
Question 10. 44.8 L of oxygen is required to completely convert 24g of a solid element into its oxide at STP. At STP, the gaseous oxide occupies 44.81. Find the mass of the gaseous oxide produced and its vapour density.
Answer:
Given
44.8 L of oxygen is required to completely convert 24g of a solid element into its oxide at STP. At STP, the gaseous oxide occupies 44.81.
At STP, 22.4 L oxygen weighs 32 g.
Hence, 44.8 L oxygen at STP weights \(\frac{32 \times 44.8}{22.4} \mathrm{~g}\) = 64 g.
Mass of gaseous oxide formed = (24 + 64)g = 88g.
At STP, mass of 44.8 L of the gaseous oxide is 88g.
∴ At STP, 22.4 L of the gaseous oxide weighs \(\frac{88 \times 22.4}{44.8} \mathrm{~g}\) = 44g.
Thus, molecular mass of the gaseous oxide = 44g.
Vapour density =44/2 = 22.
Word Problems on Chemical Calculations with Solutions
Question 11. Vapour density of a gas is 8.62. What volume will be occupied by 8.62 g of the gas gat STP?
Answer:
Given
Vapour density of the gas = 8.62.
The molecular mass of the gas = 2 x 8.62 = 17.24 g.
Thus, at STP, 17.24 g of the gas occupies 22.4L
Hence, at STP, 8.62g of the gas occupies
= \(\frac{22.4 \times 8.62}{17.4} \mathrm{~L}\) = 11.1L.
Question 12. Vapour density of a gaseous element is 5 times than that of oxygen. If the molecule of the gas Is triatomic, find its atomic mass.
Answer:
Given
Vapour density of a gaseous element is 5 times than that of oxygen. If the molecule of the gas Is triatomic,
Vapour density of oxygen (D)= 32/2 = 16
∴ Hence, vapour density of the gas = 5×16 = 80.
Molecular mass of the gas = 2 x 80 = 160.
The gas is triatomic, hence its atomic mass = 160/3= 53.33.
Question 13. At standard temperature and pressure, vapour density of sulphur vapour is 9.143 times the vapourdensity of nitrogen gas. Find the molecular formula of sulphur vapour.
Answer:
Given
At standard temperature and pressure, vapour density of sulphur vapour is 9.143 times the vapourdensity of nitrogen gas.
Vapour density of sulphur vapour with respect to nitrogen gas =\(\frac{\text { molecular mass of sulphur vapour }}{\text { molecular mass of nitrogen }}\)
Hence, molecular mass of sulphur vapour = 9.143 Χ 28 = 256.004.
So, atomicity of sulphur vapour = \(\frac{256.004}{32}\) = 8.
∴ Molecular formula of sulphur vapour = S8.
Question 14. Vapour density of a gas with respect to air is 1.528. At 27°C and 750 mmHg pressure, find the mass of 2 L of the gas. (vapour density of air with respect to hydrogen is 14.4)
Answer:
Given
Vapour density of the gas with respect to air = \(\frac{\text { molecular mass of the gas }}{\text { molecular mass of air }}\)
Hence, molecular mass of the gas = 1.528 x 28.8 = 44g.
If the volume of the gas at STP be V, then \(\frac{750 \times 2}{(273+27)}\) = \(\frac{760 \times V}{273}\)
or, \(V=\frac{750 \times 2 \times 273}{300 \times 760} L\) = 1.796L.
At STP, mass of 22.4 L of the gas = 44 g.
∴ At STP, mass of 1.796 L of the gas = \(\frac{44 \times 1.796}{22.4} \mathrm{~g}\) = 3.528g
Wbbse Class 10 Physical Science Solutions
Question 15. log calcium carbonate reacts with excess dilute hydrochloric acid to produce CO2 gas.
Answer:
\(\begin{array}{cc}
\mathrm{CaCO}_3+\mathrm{HCl} & \rightarrow \mathrm{CaCl}_2+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O} \\
(40+12+16 \times 3) \mathrm{g} & (12+16 \times 2) \mathrm{g} \\
=100 \mathrm{~g} & =44 \mathrm{~g}
\end{array}\)
1. From the above equation, it is observed that 44 g CO2 gas is produced from 100 g CaCO3.
Again, 44g CO2 = 1 mol CO2
Thus, we can say , 100g CaCO3 produces 1 mol CO2 10 g CaCO3 produces = 10/100 Χ 1 mol CO2 = 0.1 mol CO2 .
2. At STP, 1 mol CO2 occupies 22.4 L.
∴ At STP, 0.1 mol CO2 occupies = 0.1 X 22.4 L = 2.24 L.
Question 16. What amount of calcium carbonate will react with excess dilute HCL to produce 66g CO2?
Answer: CaCO3 reacts with dilute HCI to form calcium chloride and CO2. The reaction can be represented by the following equation,
Wbbse Class 10 Physical Science Question and Answers
⇒ \(\mathrm{CaCO}_3+\mathrm{HCl} \rightarrow \mathrm{CaCl}_2+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}\)
⇒ \(\begin{array}{cc}
(40+12+16 \times 3) \mathrm{g} & (12+16 \times 2) \mathrm{g} \\
=100 \mathrm{~g} & =44 \mathrm{~g}
\end{array}\)
Wbbse Class 10 Physical Science Solutions
Thus, 44g CO2 is produced from 100 g CaCO3 . 66g CO2 is produced from 66 g = 150g CaCO3 .
Question 17. How much potassium chlorate is required to produce 9.6 g oxygen? [K = 39, Cl = 35.5]
Answer: Oxygen is produced on thermal decomposition of potassium chlorate. The reaction is represented by the following balanced equation,
⇒ \(\mathrm{CaCO}_3+\mathrm{HCl} \rightarrow \mathrm{CaCl}_2+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}\)
⇒ \(\begin{array}{cc} (40+12+16 \times 3) \mathrm{g} & (12+16 \times 2) \mathrm{g} \\
=100 \mathrm{~g} & =44 \mathrm{~g} \end{array}\)
From the above equation it can be said that, to produce 96g oxygen, the amount of potassium chlorate required is 245g.
∴ To produce 9.6 g oxygen, the amount of potassium chlorate required is= \(\frac{245 \times 9.6}{96} \mathrm{~g}\) = 245 x g
Question 18. What amount of Cu turnings and conc. H2SO4 are required to mproduce 64g of SO2 gas?
Answer: The reaction between copper turnings and sulphuric acid can be represented by the following balanced equation,
Cu\(+2 \mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{CuSO}_4+\mathrm{SO}_2+2 \mathrm{H}_2 \mathrm{O}\)
∴ \(\begin{array}{ccc}
63.5 \mathrm{~g} \quad 2(2+32+16 \times 4) \mathrm{g} & (32+16 \times 2) \mathrm{g} \\
=196 \mathrm{~g} & =64 \mathrm{~g}
\end{array}\)
Wbbse Class 10 Physical Science Question and Answers
From the above equation we can conclude that, 63.5g copper and 196 g sulphuric acid is required to produce 64g SO2 gas.
Wbbse Class 10 Physical Science Solutions
Question 19. What amount of ferrous sulphide is required to produce 1.12L H2S at STP on reaction with excess dilute H2SO4?
Answer: The reaction is represented by,
⇒ \(\begin{aligned}
& \mathrm{FeS}+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{FeSO}_4+\mathrm{H}_2 \mathrm{~S} \\
& (56+32) \mathrm{g}=88 \mathrm{~g} \quad 22.4 \mathrm{~L} \text { at STP } \\
&
\end{aligned}\)
Thus, at STP, 22.41 H2S is produced from 88g FeS
∴ At STP 1.12 L H2S is produced from \(\frac{1.12 \times 88}{22.4} \mathrm{~g}\)= 4.4g FeS
Question 20. When steam is passed over red hot iron ferrosoferric oxide is formed and hydrogen gas is evolved. To produce 8 mol of hydrogen how many moles of iron is needed?
Answer:
Given
When steam is passed over red hot iron ferrosoferric oxide is formed and hydrogen gas is evolved.
The above reaction can be represented as
⇒ \(\underset{3 \mathrm{~mol}}{3 \mathrm{Fe}}+4 \mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{Fe}_3 \mathrm{O}_4+\underset{4 \mathrm{~mol}}{4 \mathrm{H}_2}\)
To produce 4 mol of hydrogen number of moles of iron required is 3.
∴ To produce 8 mol of hydrogen number of moles of iron required is =\(\frac{3 \times 8}{4}\) = 6.
Hence, 6 mol of iron will be required.
Wbbse Class 10 Physical Science Question and Answers
Question 21. What amount of Zn-dust is required to produce 7g hydrogen? Zn-dust contains 88% zinc [Zn = 65.5].
Answer: The balanced equation of the reaction is
⇒ \(\underset{65.5 \mathrm{~g}}{\mathrm{Zn}}+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{ZnSO}_4+\underset{1 \times 2 \mathrm{~g}=2 \mathrm{~g}}{\mathrm{H}_2}\)
2g hydrogen is produced from 65.5g Zn dust.
∴ 7g hydrogen is produced from \(\frac{65.5 \times 7}{2} \mathrm{~g} \mathrm{Zn}\) dust = 229.25g Zn dust.
Now, 88g zinc is present in l00g of Zn-dust.
∴ 229.25g zinc is present in \(\frac{100 \times 229.25}{88} \mathrm{~g}\) = 260.51 g of Zn-dust.
Hence, 260.51g of Zn-dust will be required to produce 7g of hydrogen.
Question 22. 15.25g of a mixture containing KClO3 and MnO2 gives off 4.8g oxygen on heating. What amount of MnO2 was present in the mixture as a catalyst? [K = 3S, Ci = 35.5, O = 16]
Answer:
Given
15.25g of a mixture containing KClO3 and MnO2 gives off 4.8g oxygen on heating.
Oxygen is produced on the thermal decomposition of potassium chlorate. The reaction is represented by the following balanced equation,
⇒ \(2 \mathrm{KClO}_3+2 \mathrm{KCl} \rightarrow 3 \mathrm{O}_2\)
⇒ \(\begin{array}{cc}
2(39+35.5+16 \times 3) g & 3(16 \times 2) \mathrm{g} \\
=245 \mathrm{~g} & =96 \mathrm{~g}
\end{array}\)
From the equation it can be said that, to produce 96g of oxygen, the amount of potassium chlorate required is 245g.
∴ To produce 4.8g oxygen, the amount of potassium chlorate required is 4.8g oxygen is produced from 12.25g KCIO3.
The total mass of the mixture = 15.25 g.
Hence, the amount of MnO2 present in the mixture = (15.25 -12.25)g = 3.00g.
Wbbse Class 10 Physical Science Question and Answers
Question 23. When 174g pure MnO2 is added to excess amount of cone. HCI, a gas is evolved. What amount of l2 will be released from Kl by the gas produced in this reaction? [Mn = 55,1 = 127]
Answer:
Given
When 174g pure MnO2 is added to excess amount of cone. HCI, a gas is evolved.
MnO2 reacts with HCI according to the following balanced equation,
4\(\mathrm{HCl}+\mathrm{MnO}_2 \rightarrow \mathrm{MnCl}_2+\mathrm{Cl}_2+2 \mathrm{H}_2 \mathrm{O}\)
55\(+2 \times 16=87 \mathrm{~g} \quad 2 \times 35.5=71 \mathrm{~g}\)
87 g MnO2 produces 71 g chlorine gas.
∴ 174 g MnO2 produces \(\frac{174 \times 71}{87} \mathrm{~g}\) = 142g chlorine gas.
Again, \(\begin{aligned}
& \mathrm{Cl}_2+2 \mathrm{Kl} \rightarrow 2 \mathrm{KCl}+\mathrm{I}_2 \\
& 2 \times 35.5=71 \mathrm{~g} \quad 2 \times 127=254 \mathrm{~g} \\
&
\end{aligned}\)
Thus, 71 g Cl2 gas releases 254g iodine.
Hence, 142g Cl2 gas releases \(\frac{254 \times 142}{71} g\) = 508g iodine.
Question 24. When excess AgNO3 solution is added to 1,873 g of a mixture containing KCI and NaCl, 3.731g AgCI is precipitated. Find the amount of KCI in the mixture. [K = 39,Ag = 108, Cl = 35.5 ]
Answer:
Given
When excess AgNO3 solution is added to 1,873 g of a mixture containing KCI and NaCl, 3.731g AgCI is precipitated.
Let us consider that the mixture contains xg NaCI. Hence, the amount of KCI = (1.873 – x)g.
∴ \(\mathrm{NaCl}+\mathrm{AgNO}_3 \rightarrow \mathrm{NaNO}_3+\mathrm{AgCl}\)
23 \(+35.5=58.5 \mathrm{~g} \quad 108+35.5=143.5 \mathrm{~g}\)
58.5g NaCI precipitates 143.5 g AgCI.
∴ xg NaCI precipitates \(\frac{143.5 \times x}{58.5} \mathrm{~g}\) = 2.453 x g AgCI.
Again, \(\mathrm{KCl}+\mathrm{AgNO}_3 \rightarrow \mathrm{NaNO}_3+\mathrm{AgCl}\)
74.5g KCI precipitates 143.5g AgCI
∴ (1.873 -x)g KCI precipitates \(\frac{143.5(1.873-X)}{74.5} \mathrm{~g}\) = (3.61-1.926x)g AgCI.
According to the problem, 2.453x + 3.61 – 1.926x = 3.731 Or, x = 0.2296 g.
Amount of KCI = (1.873 – 0.2296)g = 1.6434g.
Understanding the Concept of Limiting Reactants
Question 25. When 6.23g of a mixture of magnesium oxide and magnesium carbonate is strongly heated, the mass of the mixture decreases by 2.2g. Find the percent composition of the mixture.
Answer:
Given
When 6.23g of a mixture of magnesium oxide and magnesium carbonate is strongly heated, the mass of the mixture decreases by 2.2g.
On heating MgCO3, it decomposes to MgO and CO2 , but MgO has no effect on the mass. The produced CO2 gas is released from the system and hence the mass decreases.
⇒ \(\underset{24+12+3 \times 16=84 \mathrm{~g}}{\mathrm{MgCO}_3} \stackrel{\Delta}{\longrightarrow} \mathrm{MgO}+\mathrm{CO}_2\)
44 g CO2 is produced from 84g MgCO3.
∴ 2.2g CO2 is produced from \(\frac{84 \times 2.2}{44} g\) = 4.2g MgCO3 .
Thus, the mixture contains 4.2g MgCO3 .
∴Mass percent of MgCO3 in the mixture is = \(\frac{4.2}{6.23} \times 100\) = 67.42%.
Hence, the mass per cent of MgO in the mixture =(100-67.42)% = 32.58 %.
Wbbse Class 10 Physical Science Question and Answers
Question 26. 112g pure iron is converted to ferric chloride and dissolved in water. What volume of H2S gas at STP is required for the complete conversion of ferric chloride to ferrous chloride? [Fe = 56, Cl = 35.5]
Answer:
Given
112g pure iron is converted to ferric chloride and dissolved in water.
The reactions involved can be represented by the following equations:
1. \(\underset{2 \times 56=112 \mathrm{~g}}{2 \mathrm{Fe}+3 \mathrm{Cl}_2} \rightarrow \rightarrow_{2(56+3 \times 35.5)=325 \mathrm{~g}}^{2 \mathrm{FeCl}_3}\)
2. \(\begin{aligned}
& 2 \mathrm{FeCl}_3+\mathrm{H}_2 \mathrm{~S} \rightarrow 2 \mathrm{FeCl}_2+2 \mathrm{HCl}+\mathrm{S} \\
& 2 \times(56+35.5 \times 3) \quad 22.4 \mathrm{~L} \\
& =325 \mathrm{~g} \quad \text { (at STP) } \\
&
\end{aligned}\)
Thus, according to the first equation, 112g of pure iron produces 325g of FeCl3 .
Again, from the second equation it can be concluded that 325g FeCl3 is completely reduced to FeCl2 by 22.4L H2S gas at STP.
Question 27. The mass of 2.24 L of a gas at NtP is 3.17 g. Calculate its molecular mass, vapour density and standard density.
Answer:
Given
The mass of 2.24 L of a gas at NtP is 3.17 g.
At NTP, 2.24 L of the gas has the mass of 3.17 g.
∴ At NTP, 22.4 L of the gas has the mass of \(\frac{3.17 \times 22.4}{2.24} \mathrm{~g}\) = 31.7g
∴ Molecular mass of the gas = 31.7
Vapour density of the gas = 31.7/2 = 15.85
∴ Standard density of the gas = vapour density x 0.089 g. L-1 = 15.85 x 0.089 g. L-1 = 1.41 g. L-1 respectively.
Molecular mass, vapour density and standard density of the gas is 31.7, 15.85 and 1.41 g • L_1 respectively.
Wbbse Class 10 Physical Science Question and Answers
Question 28. What amount of NaOH will be formed by reacting 23g Na with water at room temperature.
Answer: Balanced equation of the corresponding reaction,
⇒ \(2 \mathrm{Na}+2 \mathrm{H}_2 \mathrm{O} \rightarrow 2 \mathrm{NaOH}+\mathrm{H}_2\)
⇒ \((2 \times 23) g=46 g\) \(2(23+16+1) g=80 g\)
According to the equation, 46 g Na produces 80g of NaOH
∴ 23 g Na will produce = \(\frac{80 \times 23}{46}\) = 40g of NaOH.
Question 29. What amount of sulphuric acid is required to neutralise 4 g of NaOH? Or, What amount of sulphuric acid will be needed to neutralise 40g NaOH?
Answer: Balanced chemical equation:
⇒ \(\begin{aligned}
& 2 \mathrm{NaOH}+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{Na}_2 \mathrm{SO}_4+2 \mathrm{H}_2 \mathrm{O} \\
& 2(23+16+1) g(2 \times 1+32+4 \times 16) g \\
& =80 \mathrm{~g} \quad=98 \mathrm{~g} \\
&
\end{aligned}\)
Mass of H2SO4 required to neutralise 4g NaOH = \(\frac{98 \times 4}{80} \mathrm{~g}\) = 4.9 g
∴ 4.9 H2SO4 is needed to neutralise 4g NaOH.
Question 30. CaCl2, CO2 and H2O are formed by the reaction of CaCO3 with diL HCI. To form 55.5g of CaCl2, 22.0g CO2 and 9.0g of H2O from 50.0 g CaCO3, what amount (in g) of HCI will be needed? Calculate the number of moles of the required amount of HCI.
Answer:
Given
CaCl2, CO2 and H2O are formed by the reaction of CaCO3 with diL HCI. To form 55.5g of CaCl2, 22.0g CO2 and 9.0g of H2O from 50.0 g CaCO3
Balanced chemical eqaution:
⇒ \(\begin{gathered}
\mathrm{CaCO}_3+2 \mathrm{HCl} \rightarrow \mathrm{CaCl}_2+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O} \\
(40+12+3 \times 16) \mathrm{g} 2(1+35.5) \mathrm{g}(40+2 \times 35.5) \mathrm{g}(12+2 \times 16) \mathrm{g}(2 \times 1+16) \mathrm{g} \\
=100 \mathrm{~g}=73 \mathrm{~g} \quad=111 \mathrm{~g}=44 \mathrm{~g} \quad=18 \mathrm{~g}
\end{gathered}\)
According to the equation, 100/2 g or 50g of will form 111/2 or 55.5g of CaCl2, 44/2 or 22g of CO2 and 18/2 or 9g of H2O by reacting with 73/2 g or 36.5g HCL.
Wbbse Class 10 Physical Science Solutions
Question 31. What amount of ammonium sulphate will be required to produce 6.8g ammonia by reaction of caustic soda with ammonium sulphate solution?
Answer: Balanced chemical equation:
(NH4)2SO4 + 2NaOH →Na2SO4 + 2NH3 + 2H2O
2 x (14+1 x 4)+32+(16 x 4)g 2 x (14 +1 x3)g
= 132g =34g
Now, to form 34g NH3 amount of (NH4)2SO4 needed = 132g
∴ To form 6.8 g of NH3, amount of (NH4)2SO4 needed = \(\frac{132 \times 6.8}{34} \mathrm{~g}\) = 26.4g
Question 32. If the purity of limestone is 95%, then calculate the amount of CO2 formed by heating 200 g of the said sample. (Assume, no impurity present in the sample produces CO2 on heating)
Answer: Balanced chemical equation:
⇒ \(\begin{array}{cc}
\mathrm{CaCO}_3 \stackrel{\Delta}{\longrightarrow} & \mathrm{CaO}+\mathrm{CO}_2 \\
(40+12+16 \times 3) \mathrm{g} & (12+16 \times 2) \mathrm{g} \\
=100 \mathrm{~g} & =44 \mathrm{~g}
\end{array}\)
Now amount of pure CaCO3 in 200 g of 95% pure lime stone sample = 200 x 95/100g = 190g
∴ According to the equation, the amount of CO2 formed from 100g CaCO3 is 44 g.
Amount of CO2 formed from 190g CaCO3 is \(\frac{44 \times 190}{100} \mathrm{~g}\)
∴ 83.6g CO2 will be formed from 200g of the 95% pure limestone sample.
Question 33. What amount of ferrous sulphide will be needed to react with dilute sulphuric acid to form 1.7g hydrogen sulphide gas.
Answer: Balanced chemical equation:
⇒ \(\mathrm{FeS}+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{FeSO}_4+\mathrm{H}_2 \mathrm{~S}\)
(56 + 32)g = 88g (1 Χ 2 +32)g = 34g
Now, to form 34g H2S, FeS needed = 88g
∴ To form 1.7g H2S, Fes needed = \(\frac{88 \times 1.7}{34} \mathrm{~g}\) = 4.4g.
∴ 1.7 g of H2S will be obtained by the reaction between dil. H2SO4 and 44g FeS.
Question 34. When zinc oxide is heated with carbon, metallic zinc and carbon monoxide are formed. What amount of carbon will be needed to form 31.785g Zn and 14.000g CO from 40.685g zinc oxide? How many moles of carbon monoxide will be formed?
Answer: Balanced chemical equation:
∴ \(\mathrm{ZnO}+\mathrm{C} \rightarrow \mathrm{Zn}+\mathrm{CO}\)
Mass of ZnO = 40.685g
Mass of Zn = 14.000g
Mass of CO = 14.000g
∴ Required amount of carbon = (31.785 +14 – 40.685)g + 5.1g
Number of moles of CO = 14/28 = 0.5
Wbbse Class 10 Physical Science Solutions
Question 35. What amount of H2 will be formed when steam is passed over 21g of red hot Iron? What will be its volume at STP?
Answer: Balanced chemical equation:
3\(\mathrm{Fe}+4 \mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{Fe}_3 \mathrm{O}_4+4 \mathrm{H}_2\)
(3 Χ 56)g = 168g (4 Χ 2)g= 8g
The amount of H2 obtained from 168g red hot Fe is 8g
∴ Amount of H2 obtained from 21g red hot Fe is \(\frac{8 \times 21}{168} g\) = 1g
Again, the volume of 2g H2 at STP is 22.4L
∴ Volume of 1g H2 at STP is \(\frac{22.4}{2} \mathrm{~L}\) = 11.2L
Question 36. Calculate the amount of (1) magnesium chloride and (2) hydrogen (in grams) formed by the reaction between 36g magnesium and an excess amount of dil. HCI.
Answer: Balanced Chemical equation: \(\mathrm{Mg}+2 \mathrm{HCl} \rightarrow \mathrm{MgCl}_2+\mathrm{H}_2\)
24g (24 +2 Χ 35.5)g =95g 2g
1. 24g of Mg forms 95g of MgCl2
∴ 36g of Mg forms \(\frac{95 \times 36}{24} \mathrm{~g}\) = 142.5g of MgCl2
2. Again, 24g of MgCl2 Mg forms 2g of
∴ 142.5 g of and 3g of H2 will be obtained by the reaction between 36g and an excess amount of dil HCL.
Question 37. What amount of KNO3 should be heated to obtain oxygen exactly in the same amount that is obtained by heating 49g KClO3?
Answer: Balanced chemical equation for heating KClO3:
2\(\mathrm{KClO}_3 \rightarrow 2 \mathrm{KCl}+3 \mathrm{O}_2\)
2 Χ 39 + 35.5 + 16 Χ 3)g = 245g 3 Χ 32g + 96g
According to the reaction, 245g KClO3 produced 96g of O2.
∴ 49g KClO3 produces \(\frac{96 \times 49}{245} \mathrm{~g}\) = 19.2 g of O2.
Balanced chemical equation for heating:
⇒ \(\begin{aligned}
& 2 \mathrm{KNO}_3 \rightarrow 2 \mathrm{KNO}_2+\mathrm{O}_2 \\
& 2 \times(39+14+16 \times 3) \mathrm{g} \quad 32 \mathrm{~g} \\
& =202 \mathrm{~g} \\
&
\end{aligned}\)
According to the reaction, 32g O2. is obtained by heating 202g of KClO3.
∴ 19.2 g O2 is obtained by heating \(\frac{202 \times 19.2}{32} \mathrm{~g}\) = 121.2g of KClO3.
∴ 121.2g of KNO3 should be heated to obtain exactly in the same amount that is obtained by heating 49g of KClO3.
Visual Representation of Chemical Reactions and Calculations
Question 38. What is the percentage of phosphorus in calcium phosphate?
Answer:
Molecular mass of Ca3(PO4)2 = 40 Χ 3 +2 (31 +16 Χ 4) = 310
∴ Amount of phosphours in 100g Ca3(PO4)2 = \(\frac{62 \times 100}{310}\) = 20g
∴ percentage of phosphours in Ca3(PO4)2 is 20%
Question 39. Calculate the amount of magnesium oxide formed by the complete combustion of 24g magnesium.
Answer: Balanced chemical equation:
2\(\mathrm{Mg}+\mathrm{O}_2 \rightarrow 2 \mathrm{MgO}\)
2 Χ 24g = 48g 2 Χ (24+16) g = 80g
According to the equation, amount of MgO obtained from 48g Mg is 80g
∴ = \(\left(\frac{80 \times 24}{48}\right) \mathrm{g}\) = 40g
∴ 40g of MgO will be obtained by the complete combustion of 24g Mg.
Question 40. SO2 is produced by the combustion of iron pyrites for the industrial preparation of sulphuric acid. The chemical equation is as follows:4FeS2 + 11O2→2Fe2O3 + 8SO2 . Calculate the amount of FeS2 needed to produce 512g SO2.
Answer:
Given
SO2 is produced by the combustion of iron pyrites for the industrial preparation of sulphuric acid. The chemical equation is as follows:4FeS2 + 11O2→2Fe2O3 + 8SO2 .
Balanced chemical equation:
4\(\mathrm{FeS} 3+11 \mathrm{O}_2 \rightarrow 2 \mathrm{Fe}_2 \mathrm{O}_3+8 \mathrm{O}_2\)
4 Χ (56 + 32 Χ 2)= 480g 8x(32+16×2) = 512g
To produce 512g SO2 , 480g of FeS2 is required.
Question 41. 112g of a metal oxide and a gaseous compound are formed when 200g of a metal carbonate is heated. Vapour density of the gaseous compound is 22.Calculate the number of moles of the gaseous compound formed in the reaction.
Answer:
Given
112g of a metal oxide and a gaseous compound are formed when 200g of a metal carbonate is heated. Vapour density of the gaseous compound is 22.
Mass of the gaseous compound formed in the reaction = (200- 112)g = 88g
Given, vapour density of the gaseous compound = 22
∴ Molecular mass of the gaseous compound = 2 x 22 = 44
∴ Number of moles of the gaseous compound = 88/44 = 2
∴ 2 mol of the gaseous compound will be formed.
Wbbse Class 10 Physical Science Solutions
Question 42. Commercial zinc contains 35% impurity. Calculate the amount of hydrogen gas formed when 50g of such zinc reacts with excess amount of dii H2SO4.
Answer:
Given
Commercial zinc contains 35% impurity.
Percentage of impurity = 35%
∴ Percentage of pure zinc = (100- 35)% = 65%
∴ Amount of pure zinc in 50g of impure sample = 65/100 Χ 50g = 32.5g
Now, balanced chemical equation:
⇒\(\mathrm{Zn}+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{ZnSO}_4+\mathrm{H}_2\)
Now, 65g of pure zinc forms 2g of H2
∴ 32.5g of pure zinc forms \(\frac{2 \times 32.5}{65} \mathrm{~g}\) = 1 g of H2.
∴ 1 g hydrogen gas will be formed
Question 43. The oxygen formed by heating a mixture of 75g potassium chlorate and manganese dioxide reacts completely with 3g hydrogen to form water. Calculate the amount of potassium chlorate and manganese dioxide.
Answer:
Given
The oxygen formed by heating a mixture of 75g potassium chlorate and manganese dioxide reacts completely with 3g hydrogen to form water.
Balanced chemical equation for the formation of water from hydrogen and oxygen is
⇒ \(\begin{gathered}
2 \mathrm{H}_2+\mathrm{O}_2 \rightarrow \mathrm{H}_2 \mathrm{O} \\
(2 \times 2) \mathrm{g}=4 \mathrm{~g}(16 \times 2) \mathrm{g}=32 \mathrm{~g}
\end{gathered}\)
According to the equation, 4g of hydrogen reacts with 32g of O2
∴ 3g of hydrogen reacts with \(\frac{32 \times 3}{4} \mathrm{~g}\) = 24 g of O2.
Now, balanced chemical equation for preparation of oxygen from potassium chlorate is
2\(\mathrm{KClO}_3+\left[\mathrm{MnO}_2\right] \rightarrow 2 \mathrm{KCl}+3 \mathrm{O}_2+\left[\mathrm{MnO}_2\right]\)
2(39+35.5+16×3) = 245g 3 Χ 32 = 96g
According to the equation, to produce 96g oxygen, KClO3 needed
∴ To produce 24g oxygen, KClO3. needed \(\frac{245 \times 24}{96} \mathrm{~g}\) = 61.25g
∴ Amount of KClO3 in the mixture = 61.25 g
∴ Amount of MnO2 in the mixture = (75 – 61.25)g = 13.75g
Question 44. SO2 is oxidised to form SO3 by O2 in presence of catalyst. What amount of SO2 (in g) will be needed to produce 40g SO3
Answer:
Given
SO2 is oxidised to form SO3 by O2 in presence of catalyst.
Balanced chemical equation,
⇒ \(\begin{array}{ccc}
2 \mathrm{SO}_2+\mathrm{O}_2 & \rightarrow & 2 \mathrm{SO}_3 \\
2 \times(32+2 \times 16) \mathrm{g} & & 2 \times(32+3 \times 16) \mathrm{g} \\
=128 \mathrm{~g} & & =160 \mathrm{~g}
\end{array}\)
According to the equation, to produce 160g SO3, SO2 needed = 128g
∴ To produce 40g SO3, SO2 needed = \(\frac{128 \times 40}{160} \mathrm{~g}\) = 32g
∴ 32g of SO2 will be needed.
Question 45. To convert 25.64g of a solid element A into a gaseous oxide, 11.2L of oxygen (at STP) is needed. Volume of the gaseous oxide (at STP) is 2-1.2L What is the vapour density of the gaseous oxide?
Answer: At STP, the mass of 22.4L oxygen = 32g
∴ Mass of 11.2L oxygen = \(\frac{32 \times 11.2}{22.4} \mathrm{~g}\) = 16g
∴ Mass of the gaseous oxide = (25.64 + 16)g = 41.64g
According to the question, 21.2L of the gaseous oxide weight 41.64g
∴ 22.4L of the gaseous oxide weights \(\frac{41.64 \times 22.4}{21.2} \mathrm{~g}\) = 44g
∴ Molecular mass of the gaseous oxide = 44
∴ Vapour density of the gaseous oxide = 44/2 =22
Wbbse Class 10 Physical Science Solutions
Question 46. What amount of HgO should be heated to produce 0.5 mol oxygen gas?
Answer: Balanced chemical equation of the reaction:
2\(\mathrm{HgO} \rightarrow 2 \mathrm{Hg}+\mathrm{O}_2\)
2 Χ (200 +16)g = 432g
Now according to the equation, 1 mol O2 is obtained by heating = 432 Χ 0.5g = 216g of HgO
∴ 0.5 mole O2 is obtained by heating =432 Χ 0.5g = 216g of HgO
∴ 216g HgO should be heated to produce 0.5 mol oxygen gas.
Question 47. What amount of ammonia will be formed by the reaction of 5.35g NH4CI with an excess amount of lime?
Answer: Balanced equation of the reaction:
2\(\mathrm{NH}_4 \mathrm{Cl}+\mathrm{CaO} \rightarrow \mathrm{CaCl}_2+2 \mathrm{NH}_3+\mathrm{H}_2 \mathrm{O}\)
2(14+1×4+35.5)g V = 107g 2(14+3×1)g = 34g
According to the reaction, 107g NH4CI produced 34g NH3
∴ 5.35g NH4CI produces \(\frac{34 \times 5.35}{107} \mathrm{~g}\) = 1.7g NH3
∴ 1.7 g of NH3 will be produced.
Question 48. What amount of Al will be needed (in grams) to produce 558g of Fe by the reduction of Fe2O3 by Al at high temperatures? Calculate the number of moles of Fe2O3 needed in the reaction.
Answer: Balanced chemical equational of the reaction:
⇒ \(\begin{aligned}
\mathrm{Fe}_2 \mathrm{O}_3+ & 2 \mathrm{Al} \rightarrow 2 \mathrm{Fe}+\mathrm{Al}_2 \mathrm{O}_3 \\
(2 \times 27) \mathrm{g} & (2 \times 55.8) \mathrm{g} \\
=54 \mathrm{~g} & =111.6 \mathrm{~g}
\end{aligned}\)
According to the equation of the reaction, to produce 111.6g Fe, Al needed =54g
∴ To produce 558g Fe, Al needed = \(\frac{54 \times 558}{111.6} g\) = 270g
∴ 270g of Al will be required.
Again, according to the equation of the reaction, 54g of Al reacts with 1 mol Fe2O3
∴ 270g of AI will react with \(\frac{1 \times 270}{54}\) mol Fe2O3= 5 mol Fe2O3
∴ 5 mol Fe2O3 will be required for the reaction.
Question 49. How many grams of H2SO4 should react with zinc to form 44.8 litre of H2 gas at NTP?
Answer: Balanced equation of the reaction—\(\mathrm{Zn}+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{ZnSO}_4+\mathrm{H}_2\)
2 Χ 1+ 32+ 16 Χ 4 = 98g 22.4L
To form 22.4L H2, H2SO4 needed =98g
∴ To form 44.8L H2, H2SO4 needed = \(\frac{98 \times 44.8}{22.4} \mathrm{~g}\) = 196g
Question 50. Upon heating with calcium hydroxide, 32.1g ammonium chloride produces 10.2g NH3, 33.3g CaCl2 and 10.8g H2O. What is the amount of calcium hydroxide (in gram) that participates in the reaction? Calculate the number of moles of NH3 and the volume of NH3 at STP, produced in the reaction.
Answer:
Given
Upon heating with calcium hydroxide, 32.1g ammonium chloride produces 10.2g NH3, 33.3g CaCl2 and 10.8g H2O.
According to the law of conservation of mass, mass of NH4CI + mass of Ca(OH)2 = mass of NH3 + mass of CaCl2 + mass of H20
∴ 32.1 g + mass of Ca(OH)2 = 10.2g + 33.3g + 10.8g
∴ Mass of Ca(OH)2 = (10.2 + 33.3 + 10.8)g- 32.lg = 22.2g
∴ 22.2g of Ca(OH)2 participates in the reaction. = 14 +1×3 = 17
Wbbse Class 10 Physical Science Solutions
Question 51. lg limestone, in reaction with an excess amount of HCI, produces 0.4g CO2 gas. Calculate the percentage of purity in the lime stone sample.
Answer:
Given
lg limestone, in reaction with an excess amount of HCI, produces 0.4g CO2 gas.
A balanced chemical equation of the reaction is \(\mathrm{CaCO}_3+2 \mathrm{HCl} \rightarrow \mathrm{CaCl}_2+\mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}\)
(40+12+3Χ 16)g=100g (12+2Χ16)g = 44g
According to the equation, 44g of CO2 is obtained from 100g of CaCO3
∴ 0.4g of CO2 is obtained from \(\frac{100 \times 0.4}{44} \mathrm{~g}\) = 0.909g of CaCO3
∴ 1 g of CaCO3 sample contains 0.909g pure CaCO3
∴ 100g of CaCO3 sample contains = (0.909 x 100)g = 90.9 g of pure CaCO3
Question 52. How many grams of CuO can be reduced completely by hydrogen gas obtained from the reaction of 14g of Fe and dii HCI?
Answer: Balanced equation of the chemical reaction is
⇒ \(\underset{56 \mathrm{~g}}{\mathrm{Fe}}+2 \mathrm{HCl} \rightarrow \mathrm{FeCl}_2+\mathrm{H}_2\)
According to the reaction, 56g Fe produces 2g if H2 in reaction with dil Hcl
∴ 14g Fe produces \(\frac{2 \times 14}{56} g\) = 0.5g H2 in reaction with dil HCl
Now, the balanced chemical equation of the reduction of CuO is
⇒ \(\underset{(63.5+16) \mathrm{g}=79.5 \mathrm{~g}}{\mathrm{CuO}}+\underset{2 \mathrm{~g}}{\mathrm{H}_2} \rightarrow \mathrm{Cu}+\mathrm{H}_2 \mathrm{O}\)
According to the reaction, 2g H2 can reduce = \(\frac{79.5 \times 0.5}{2} \mathrm{~g}\) = 19.875g of CuO
∴ H2 gas obtained from the reaction of 14g Fe and dil. HCL can completely reuce 19.875g of CuO.
Question 53. What amount (in gram) of cupric oxide will be reduced by 3.4g NH3? What will be the volume of the gas produced in this case at NTP?
Answer: Balanced chemical equation of the reaction is—
3\(\mathrm{CuO}+2 \mathrm{NH}_3 \rightarrow 3 \mathrm{Cu}+\mathrm{N}_2+3 \mathrm{H}_2 \mathrm{O}\)
3x(63.5+16)g = 238.5g 2x(14+3)g= 238.5g = 34g 22.4L
∴ 34g NH3can reduce 238.5g CuO
∴ 3.4g NH3 can reduce \(\frac{238.5 \times 3.4}{24} \mathrm{~g}\) = 23.85 g CuO
Again N2 gas is formed during the reaction, according to the reaction
34g NH3 produces 22.4L N2 at NTP
∴ 3.4g NH3 produces \(\frac{22.4 \times 3.4}{34} L\) = 2.24 L N2 at NTP.
Question 54. What amount of Fe2O3 will be needed to produce 22.4l Of oxygen gas according to the following reaction? \(\mathrm{Fe}_2 \mathrm{O}_3 \rightarrow \mathrm{Fe}+\mathrm{O}_2\)
Answer: Balanced chemical equation is
⇒ \(\mathrm{Fe}_2 \mathrm{O}_3 \rightarrow \mathrm{Fe}+\mathrm{O}_2\)
2 Χ (2 Χ 56 + 3 Χ 16)g = 320g 3 Χ 22.4L(at STP)
According to the reaction, 3 Χ 22.4L O2 will be produced by 320g Fe2O3
∴ 22.4L O2 will be produced by \(\frac{320 \times 22.4}{3 \times 22.4} \mathrm{~g}\)
= 106.67g Fe2O3
∴ 106.67g Fe2O3 of will be needed.
Chapter 3 Chemical Calculations Miscellaneous Type Questions Match The Column
Question 1.
| Column A |
Column B |
| 1. Mass-energy equivalence |
1. Vapour density |
| 2. Calculation of relative quantities of reactants and products |
2. Lavoisier |
| 3. Conservation of mass |
3. E=mc2 |
| 4. The quantity whose value is half the molecular mass of a gas |
4. Stoichiometry |
Answer:
1. Mass-energy equivalence: 3. E=mc2
2. Calculation of relative quantities of reactants and products: 4. Stoichiometry
3. Conservation of mass: 2. Lavoisier
4. The quantity whose value is half the molecular mass of a gas: 1. Vapour density
Question 2.
| Column A |
Column B |
| 1. Amount of CO2 produced on heating 1 mol CaCO3 |
1. 32g |
| 2. 0.01 mol C2H4 |
2. 71g |
| 3. 22.4L Cl2(g) at STP |
3. 0.28g |
| 4. Mass of 6.022 × 1023 O2 molecules |
4. 44 g |
Answer:
1. Amount of CO2 produced on heating 1 mol CaCO3: 4. 44 g
2. 0.01 mol C2H4: 3. 0.28g
3. 22.4L Cl2(g) at STP: 2. 71g
4. Mass of 6.022 X 1023 O2 molecules: 1. 32g


![]() = \(\frac{1}{2}\)
= \(\frac{1}{2}\)

![]()
![]()
![]()
![]() =what?
=what?
![]()
![]() = \(\frac{21}{2}\)
= \(\frac{21}{2}\)![]() meters =
meters =![]()
![]()
![]()
![]()
![]()
![]()
![]() metres
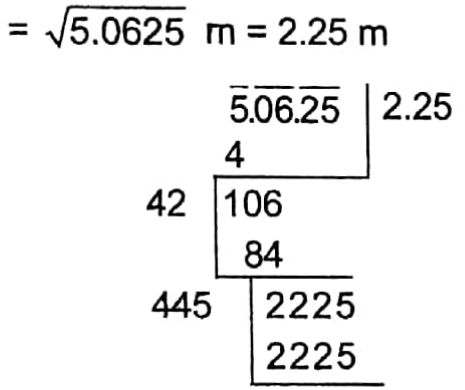
metres![]() metres =\(\frac{5}{2}\) metres = 2 \(\frac{1}{2}\) metres.
metres =\(\frac{5}{2}\) metres = 2 \(\frac{1}{2}\) metres.